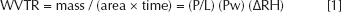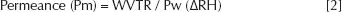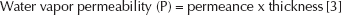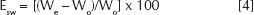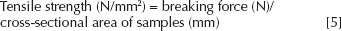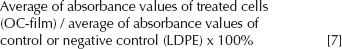Abstract
Background
The physical and biological characteristics of oligochitosan (O-C) film, including its barrier and mechanical properties, in vitro cytotoxicity and in vivo biocompatibility, were studied to assess its potential use as a wound dressing.
Methods
Membrane films were prepared from water-soluble O-C solution blended with various concentrations of glycerol to modify the physical properties of the films. In vitro and in vivo biocompatibility evaluations were performed using primary human skin fibroblast cultures and subcutaneous implantation in a rat model, respectively.
Results
Addition of glycerol significantly influenced the barrier and mechanical properties of the films. Water absorption capacity was in the range of 80%-160%, whereas water vapor transmission rate varied from 1,180 to 1,618 g/m2 per day. Both properties increased with increasing glycerol concentration. Tensile strength decreased while elongation at break increased with the addition of glycerol. O-C films were found to be noncytotoxic to human fibroblast cultures and histological examination proved that films are biocompatible.
Conclusion
These results indicate that the membrane film from O-C has potential application as a wound-dressing material.
Keywords
Introduction
Chitosan (poly(1,4-β-D-glucopyranosamine)) is the N-deacetylated polysaccharide from chitin which has valuable properties for biomedical application (1-3). Chitosan is well known for its natural properties to accelerate the healing of wounds in humans (4). Furthermore, chitosan confers considerable antibacterial activity against a broad spectrum of bacteria (5). Owing to these advantages, chitosan has been applied as wound dressing, gauzes and medical sutures (6).
The use of oligochitosan (O-C) for biomedical applications has been reported previously in the literature (7-13). More recently, O-C has been used for the development of in situ–forming hydrogel for hemorrhage control (7). In vitro evaluation of cytocompatibility on human dermal fibroblasts of O-C sheet and paste for wound dressing has also been carried out (8). Antibacterial wound-healing gels from O-C salts were developed and evaluated in both in vitro and in vivo models (11).
In wound management, protection of the skin defect from further infections and dehydration in the intervening period between hospitalization and grafting is necessary. Protection can be accomplished by covering the wound with wound dressing (4, 14-16). The wound surface should be kept just damp enough for fast healing, but eliminating the accumulation of fluid between the wound and the dressing to minimize risk of infection (17). Bacterial invasion can make the wound unsuitable for skin grafting due to the growth of a bacteria population.
While chitosan has many desirable properties, its mechanical strength is poor, and to enhance its mechanical strength, it is often blended with other polymers. In a study by Kuo and Lin (18), a chitin-chitosan scaffold was created and modified with the addition of hydroxyapatite. The scaffold consisted of a chitin and chitosan blend to create faster cellular attachments. Hydroxyapatite is often used to accelerate the degradation rate of a scaffold because of its salt-like properties. Furthermore, the genipin cross-linking agent was used to enhance the mechanical strength. Blending of chitosan with plasticizers can be a convenient and effective method to enhance mechanical and barrier properties for practical utilization (19, 20). Polyethylene glycol 200, glycerol, propylene glycol and sorbitol have been used as plasticizers in edible film production (21). A film wound dressing has to be soft and flexible to enable the film to adhere to the surface wound and contour. To improve the flexibility of the film, it is necessary to alter the tensile strength and elongation at break of the material without compromising its barrier properties. In this work, we have attempted to use glycerol as a modifier to improve the physical properties of O-C film intended for use as wound dressing. To the best of our knowledge, the use of glycerol to improve the barrier properties, tensile strength and elongation at break of O-C films for wound-dressing applications has not been previously reported.
Generally, a film wound dressing should have the following features: (i) reasonable tensile strength, which could withstand the stresses exerted by different parts of the body having varying contours; (ii) rapidly and uniformly adherent and conforming to wound bed topography and contour to prevent air and fluid pocket formation; (iii) offering an absolute barrier to bacterial ingress; (iv) preferably permeable to water vapor to the extent that a moist exudate under the dressing is maintained, but excess fluid absorption and evaporation leading to desiccation of the wound bed are prevented; and (v) must be compatible with body tissues, nontoxic, nonantigenic and nonallergenic (22).
Chitosan dissolved in dilute organic acids may limit the application in the medical and pharmaceutical fields. Therefore in the present study, O-C membrane films were prepared by casting solutions of O-C blended with glycerol at various concentrations. The mechanical properties such as tensile strength and elongation at break were investigated. The water vapor transmission rate and water absorption were studied to examine the barrier properties of the O-C films. In vitro and in vivo biocompatibility studies were conducted to determine the suitably of the O-C membrane film as wound dressing.
Materials and Methods
Materials
Chitosanase and all other reagents were purchased from SIGMA Chemicals Co. Technical grade chitosan was purchased from a local commercial supplier.
Preparation of O-C
Five liters of a 5% solution of chitosan was prepared in 0.017 M acetic acid solution. Acetic acid (100 mL) was added into the solution, stirred and followed by 100 mL water. Ten units of chitosanase enzyme was added to the solution and stirred for 6 hours at 50°C-55°C in a jacketed glass vessel. At the end of the 6 hours, the temperature of the solution was raised to 100°C for 1 hour to deactivate the enzyme. The O-C solution was then stored in a chiller for further use.
Preparation of Films
Film-forming solution was prepared by dissolving 1 mL of O-C (5%) in 99 mL of distilled water and stirred until the film solution was homogenized. O-C films were prepared by casting the solution of O-C onto glass plate and evaporating the water in an incubator. In the preparation of modified films, different compositions of plasticizer were added to the O-C solution. For each film solution, 10, 30 and 50 mL of 1% glycerol was added, respectively. Then each of these solutions was stirred for 5-10 minutes before pouring onto a glass plate. They were dried in an incubator at 40°C.
Thickness measurement
A Mitutoyo Digimatic Micrometer was used to measure film thickness to an accuracy of 0.001 mm. Five measurements were made at several different locations on the film, and the average value was calculated.
Water Vapor Transmission Rate (WVTR)
Water vapor transmission rate were determined according to the ASTM method E96-90, Procedure D. An evaporimeter was constructed in a desiccator to prevent variations owing to ambient conditions. The desiccator was put in an incubator at 35°C, with a digital hygrometer, with a continuous % relative humidity (RH), temperature and dew point display. The permeability cup was filled with 10 g of deionized distilled water, and the test film was monitored by measurement of loss of weight of the cup. An open cup was used as the control. The RH in the evaporimeter after equilibration was approximately 40%.
The WVTR of a material was calculated according to the following equation:
P is the water vapor permeability, L is the membrane thickness, (P/L) is the water vapor transmission (or permeance, Pm), Pw is the saturated water vapor pressure at the experimental temperature and the Δ (RH) is the difference of relative humidity between inside atmosphere and outside atmosphere of the membrane sealed cup. Permeance value and the water vapor permeability of the material were calculated using the following equations:
Water Absorption
The water absorption capacity of the O-C films was estimated by any increase in the weight of films after exposure to saturated water vapor for about 1 week. A known weight of the film was placed in a jar filled with distilled water for the required period of time. The wet weight of the film was determined by first blotting the film with filter paper to remove absorbed water on the surface, and then weighing it immediately on an electronic balance. The percentage water adsorption of O-C films was then calculated from the following formula:
Esw is the percentage water absorption of O-C films at equilibrium. We, denotes the weight of the O-C films after water absorption, and Wo is the initial weight of the chitosan derivatives films.
Mechanical Property Measurements
The mechanical properties of chitosan derivatives film were evaluated using a texture analyzer equipped with 500-kg load cell. A film strip 10×50 mm and free from air bubbles or physical imperfections was held between 2 clamps positioned at a distance of 30 mm. During measurement, the film was pulled by the top clamp at a rate of 30 mm/min to a distance of 5 cm before returning to the starting point. The force and elongation were measured when the film broke. Measurements were run 4 times for each film. The tensile strength and elongation at break were calculated as below:
Cytotoxicity in vitro via direct contact method
Skin samples were obtained from consenting patients who had undergone elective surgery at the Hospital Universiti Sains Malaysia, Kubang Kerian, Kelantan, after the study was approved by the human ethics committee of Universiti Sains Malaysia. The epidermal layer was lifted from the dermal layer after incubation in a Dispase solution (2.4 U/mL). The remaining dermal layer was minced into smaller pieces and digested using collagenase type-I (200 cu/mL of Dulbecco's modified Eagle's medium [DMEM]) for 12 hours at 37°C to extract the fibroblasts. The dissociated fibroblasts were washed with Dulbecco's Phosphate-Buffered Solution (DPBS), and the cell pellets were sieved through a 70-μm strainer before being centrifuged at 200 g for 10 minutes. The fibroblasts were seeded at 2×105 viable cells/mL in DMEM supplemented with 10% fetal bovine serum (Gibco, Invitrogen) and 1% penicillin-streptomycin (Gibco, Invitrogen).
Direct contact cell culture evaluation of materials was employed to assess the cytotoxicity of the O-C film. Fibroblasts (6×104 cells/mL) in DMEM with 10% fetal bovine serum (FBS) were seeded in each well of 24-well culture plates (Nunc, Denmark). Plates were incubated at 37°C in humidified 5% CO2 atmosphere until cultured cells reached 70% confluence. The culture medium was aspirated, and test material pieces (O-C film, 0.5 cm2) were placed carefully on the cell layer in a minimum culture medium. To evaluate the effect of O-C film on fibroblast cells, cultures were incubated with or without test materials for 24, 48 and 72 hours. High-density polyethylene was used as negative control. Morphology of cells was assessed using inverted light microscope, and quantitative evaluation of cytotoxicity was performed using tetrazolium salt (MTT) reduction assay.
MTT assay
After the desired time exposure, 1 mL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (5 mg/mL in phosphate-buffered saline [PBS]) was added to each well of a 24-well plate. After incubation for 4 hours at 37°C in the dark, medium and MTT was removed from each well. Formazan crystal was dissolved by adding 1 mL of DMSO. The color intensity was measured by enzyme-linked immunosorbent assay (ELISA) reader. Absorbance was read at 570 nm. High-density polyethylene (HDPE) was used as negative control, and Triton X-100 was used as positive control. Medium without cells was used as blank. The percentages of viability were calculated as below:
Film implantation in vivo
Creation of subcutaneous pockets
The rats were subjected to an implantation period of 4, 7, 14, 21 and 28 days. Immediately before operation, the rat was anesthetized with an intramuscular injection of ketamine hydrochloride 35.0 mg/kg and xylazine hydrochloride 5.0 mg/kg in the gluteal area. When fully anesthetized, the shaved area was cleaned 3 times with povidone iodine. Paravertebrally, at both sides of the spinal column, 2 longitudinal incisions were made through full-thickness incisions of the skin. Subsequently, lateral to the incisions, a subcutaneous pocket was created by blunt dissection with scissors. The implant was inserted into these pockets. The contralateral pocket served as control site. Finally, the wounds were carefully closed intracutaneously with Ethilon 3-0.
At 4 days postimplantation, the rats were euthanized, and the implant and the control with surrounding tissue were retrieved. The remaining implants and control site were retrieved at 7, 14, 21 and 28 days postimplantation. Implants from all rats were randomly chosen, sectioned and analyzed by histological examination.
Microscopic examination
Immediately after retrieval, specimens were fixed in a 10% buffered formalin solution. Subsequently, the tissue blocks were dehydrated in a series of ethanol (from 10% (v/v) to 100%) and embedded in paraffin blocks. After polymerization, non-decalcified thin (10-μm) sections were prepared in a transversal direction to the axis of the implant using a modified sawing microtome technique. Six histological sections were made for each tissue block. These sections were stained with hematoxylin and eosin (H&E) and examined using a light microscope. The histological evaluation was done in 6 randomly determined fields along the implant–tissue interface.
Statistical analysis
The results were expressed as means ± standard deviation (SD). The Wilcoxon signed-rank test was used to assess the significance of differences between different experimental conditions. Differences were regarded as significant at a P value ≤0.05.
Results and Discussion
Film thickness
The films prepared from O-C blended with glycerol were clear, smooth and pliable. The thicknesses of the films prepared with and without the addition of glycerol are shown in Table I. The thickness increased with increasing amount of glycerol concentration in the films, in agreement with the observations made by Caner et al (19) and Miranda et al (20). In the case of the addition of glycerol to O-C films, the behavior observed could be due to the size and solubility of the molecule; consequently a great amount of water was trapped in the matrix, making the films swollen (23).
THICKNESS OF MODIFIED O-C FILMS
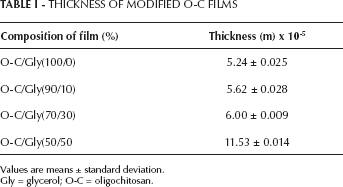
Values are means ± standard deviation.
Gly = glycerol; O-C = oligochitosan.
WVTR
One of the important factors for wound dressing is an optimal WVTR. An ideal wound dressing material should control the evaporative water loss from a wound surface. The rate for normal skin is 204 g/m2 per day, while for injured skin, it can range from 279 g/m2 per day for a first degree burn to 5,138 g/m2 per day for a granulating wound. It was recommended that a rate of 2,500 g/m2 per day, which is the mid-range of loss rates, would provide an adequate level of moisture without risking wound dehydration (24). If the WVTR of a material is lower than that of normal skin, tissue becomes dried out, while exudate between wound and the covering results in an infection. Therefore, materials for wound dressing should have a higher WVTR value than normal skin (25).
The O-C film blended with 50% of glycerol gave the highest WVTR (Tab. II). The rate of water transfer for O-C film was lower than the recommended value of 2,500 g/m2 per day. The permeance values for O-C film ranged from 5.49×10−9 kg/Pa.m2.s to 8.46×10−9 kg/Pa.m2.s. The highest permeance value was observed in the O-C film blended with 50% glycerol. The permeance of human skin is known to be 1.1111×10−9 kg/Pa.m2.s (26).
WATER BARRIER PROPERTIES OF MODIFIED OLIGOCHITOSAN (O-C) FILMS
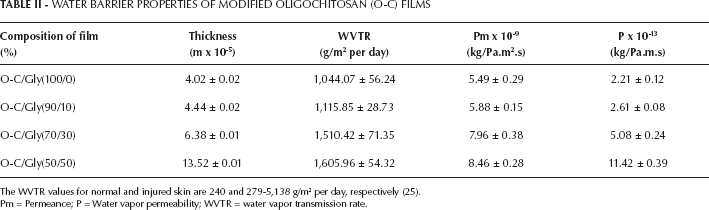
The WVTR values for normal and injured skin are 240 and 279-5,138 g/m2 per day, respectively (25).
Pm = Permeance; P = Water vapor permeability; WVTR = water vapor transmission rate.
In addition, water vapor permeability can also be employed to evaluate the specific properties of O-C film. The water vapor permeability of O-C films ranged from 2.21×10−13 kg/Pa.m.s to 11.42×10−13 kg/Pa.m.s. The highest water vapor permeability was obtained from O-C film with 50% glycerol content. Increasing the glycerol concentration resulted in increased values of WVTR, permeance and water vapor permeability of the films.
Water absorption
The exudate absorption ability of the O-C films can be determined by measuring the water absorption capacity. Modifying O-C films with a plasticizer resulted in higher water absorption capacity (Fig. 1). O-C films blended with a 50% concentration of glycerol showed the highest water uptake. Wound fluid should be absorbed in a balanced manner, and moisture should be kept under control during wound healing (27, 28).
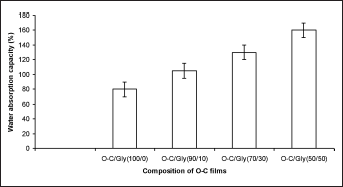
Water absorption capacity of oligochitosan (O-C) and O-C/glycerol blends.
Mechanical properties
It is suggested that films suitable for wound dressing should possess sufficient strength and yet be flexible. The highest tensile strength was obtained from unmodified O-C film, which corroborates the observations by Miranda et al (20), whereby tensile strength of chitosan films decreased with increasing plasticizer concentration (Fig. 2).
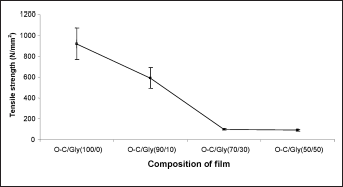
Tensile strength of oligochitosan (O-C) and the O-C/glycerol blend films.
The O-C film blended with 10% glycerol gave the highest value of elongation at break, being 4 times higher compared with unmodified O-C film (Fig. 3). Miranda et al (20) reported that the elongation of chitosan films increased with increasing plasticizer concentration. Blending with glycerol improves the elongation at break of modified O-C films, making the films soft and pliable.
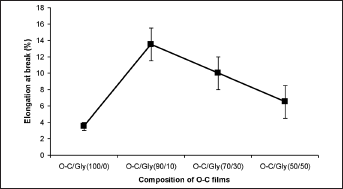
Elongation at break of oligochitosan (O-C) films with and without the addition of glycerol.
Cytotoxicity in vitro
O-C film did not induce cytotoxic effects in fibroblasts in vitro, as they showed more than 90% relative cell viability compared with negative control in a direct-contact test (Fig. 4). According to Tice et al (29) and Lim et al (30), viability of cultured cell above 70% after treatment with biomaterials can be acceptable as the bottom line in the cytocompatibility assay.
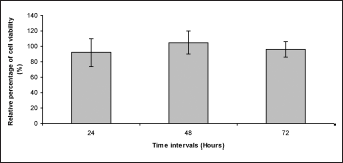
Viability of cultured fibroblasts in the presence of test materials (direct contact) at different time points. MTT reduction capacity values are expressed as the relative percentage of control values (incubation of cultured cells with high-density polyethylene [HDPE]).
Microscopic observation demonstrated that all cells exhibited a spindle shape as in the negative control, suggesting that O-C films contributed comparable cell viability to the negative control (Fig. 5). Time exposures did not significantly affect the growth of fibroblasts treated with O-C (P>0.05), which corroborates the findings by Lim et al (15).
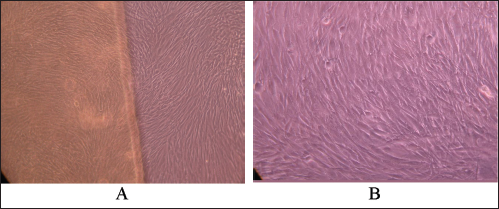
Cultured fibroblasts treated with oligochitosan (O-C) films at 72 hours.
In vivo biocompatibility evaluation
The inflammatory reactions seen in all of the implants were fairly organized, from severe inflammation to a mild phase in the final stage of implantation (Tab. III). Occasionally, tissue granulation was seen in all of the implants especially in the early period of implantation. Multinucleated giant cells were also present in all of the implants especially toward the end of the implantation period in O-C film. Angiogenesis was rather intense during the first 3 weeks for all implants. However, this process markedly decreased toward the end of the inflammatory phase. Fibroblasts increased with longer implantation period for all of the implants and gradually decreased toward the end. However, collagen depositions were abundant, especially 28 days postimplantation. The implants of O-C film had undergone mild to moderate degradation. This material was still abundantly present at 28 days postimplantation.
HISTOLOGICAL FINDINGS FOR OLIGOCHITOSAN (O-C) FILM POSTIMPLANT, AT DAYS 4, 7, 14, 21 AND 28
Conclusions
In this study, membrane films from O-C blended with glycerol were fabricated and evaluated for their barrier and mechanical properties. The membrane films exhibited high water absorption capacity and high water vapor transmission rates. The films thus have the ability to absorb exudate and control evaporative loss of water from a wound surface. Addition of glycerol results in lower tensile strength but higher elongation at break values. O-C films were biocompatible as assayed in the in vitro and in vivo experiments. These results indicate that the membrane films from O-C may have potential to be used as wound-dressing materials.