Abstract
Background:
Rapid corrosion rates are a major impediment to the use of magnesium alloys in bone tissue engineering despite their good mechanical properties and biodegradability. Zinc is a promising alloy element, and it is an effective grain refiner for magnesium. β-Ca3(PO4)2 (β-TCP) is widely used for bone regeneration because of its good biocompatibility, and it also has a similar chemical and crystal structure to human bone.
Methods:
In this research, the magnesium alloy was reinforced by adding 3%Zn (wt.%) and 5%β-TCP (wt.%) particles in order to improve the corrosion resistance and biocompatibility. Furthermore, the biomaterial was prepared through powder metallurgy technology using NH4HCO3 as space-holding particles to construct porous Mg–3%Zn/5%β-TCP scaffolds.
Results:
The results revealed that the magnesium-zinc phase and calcium phosphate phase were uniformly distributed in the α-magnesium matrix. Mechanical and corrosion tests indicated that the scaffolds had mechanical strengths similar to that of human bone, and their corrosion resistance decreased with an increase in the porosity. The scaffolds had cytotoxicity grades of 0–1 against MG63 cells, SaoS2 cells, and HK-2 cells, which suggested that they were appropriate for cellular applications. In addition, the scaffolds demonstrated excellent biocompatibility when tested in rabbits.
Conclusions:
These results indicate that porous Mg–3%Zn/5%β-TCP scaffolds are promising biodegradable implants for bone tissue engineering.
Keywords
Introduction
Recently, studies on magnesium and its alloys as medical implants have found that magnesium alloys have several outstanding advantages compared with other metal implants. First, the elastic moduli of the magnesium alloys match well with that of the human bone. By eliminating the stress shielding effect, the implant and body can achieve ideal mechanical compatibility. 1 In addition, as a biodegradable implant, magnesium alloys do not need to be removed through a second surgery after the bone heals. 2 Furthermore, Mg has excellent biocompatibility and is an essential element in the human body. The daily dose of Mg for humans is 300–400 mg, and it is involved in many physiological reactions. Its corrosion products are nontoxic and can be discharged from the body through urine.3–5 Finally, porous Mg-based implants can provide a three-dimensional space for cell growth, facilitate the exchange of metabolites and nutrients, promote tissue regeneration and reconstruction, and accelerate the process of bone healing. 6 However, the degradation rates of magnesium alloys in a physiological solution containing Cl- ions occur rapidly, which affects the integrity of the mechanical properties during the growth of the new bone. In addition, the high pH values around Mg-based implants can deplete the cell growth and differentiation.7,8 Therefore, it is necessary to study the corrosion behavior and biocompatibility of magnesium-based implants for bone tissue engineering.
In previous studies, much focus has been directed toward the corrosion resistance of different magnesium alloys as biodegradable materials in the orthopedic field. Alloying is an effective way of improving the mechanical properties and corrosion resistance of magnesium alloys. By incorporating rare earth elements and aluminum in the magnesium matrix, the corrosion resistance is obviously improved, but they are harmful to humans.9–11 Based on the adverse effects of these elements on the human body, our research aims at developing a new alloying element that is nontoxic and can improve the corrosion resistance of the alloy.
Zinc (Zn) is an essential element in the human body, which plays an important role in many physiological processes.12,13 Moreover, Zn is a promising alloy element, and it is an effective grain refiner for magnesium. 14 Cai et al. reported that the content of Zn in the Mg–Zn binary alloy increased from 1% to 5%, resulting in the alloy having significantly refined grains, thereby improving the mechanical strength and corrosion resistance. 15 He et al. found that the Mg–3Zn–0.8Zr alloy has good mechanical properties and corrosion resistance. 16 In our research, the Mg–3%Zn (wt.%) alloy was chosen as the matrix material because of its good corrosion resistance, high strength, and nontoxic elemental composition.
β-Ca3(PO4)2 (β-TCP) is widely used for bone regeneration because of its good biocompatibility, and it also has a similar chemical and crystal structure to human bone. 17 When implanted into the human body, the degraded Ca and P elements can be absorbed into the living system and form new bone.18–20 Our previous work demonstrated that β-TCP can be used as an effective reinforcement in the Mg–Zn matrix when the amount of β-TCP added was over 5 wt.%, and the composites exhibited improved corrosion resistance and biocompatibility. 19 In this study, 5% β-TCP (wt.%) particles were added in order to reinforce the corrosion resistance and biocompatibility of the Mg–3% Zn alloy.
Powder metallurgy technology is a promising technique used in the development of open-cell structures using space-holder particles.21,22 The pore size, distribution, and porosity of the scaffold mainly depends on the content of the space-holder particles, and the ideal primary porosity (before the degradation) of Mg–Zn scaffolds should be lower than 20%. 23 In our work, the Mg–3%Zn alloy was strengthened by adding 5% β-TCP particles. The biomaterial was prepared by PM technology using NH4HCO3 as space-holding particles to construct porous Mg–3%Zn/5%β-TCP scaffolds, and the effect of the porosity on the mechanical strength and corrosion resistance of the scaffolds was studied and discussed in the range of 6–18%. More importantly, the biocompatibility of scaffolds was examined. In summary, the aim of our study was to develop a biodegradable and nontoxic biomaterial for medical implants.
Methods
Scaffold preparation
The β-Ca3(PO4)2 powder was prepared from commercial CaHPO4 (Merck) and CaCO3 powders (G-CLONE) through the calcination reaction. The CaHPO4 and CaCO3 powders were weighed in a molar ratio of 2:1, and distilled water was added in a mass ratio of 1:1.5. They were mixed fully by milling at 40 r/m for 60 h and dried at 80°C. Then, the dry powder was heated at 950–960°C for 4 h in a muffle furnace (PT-M1700-8LX), and the β-Ca3(PO4)2 powder was obtained after cooling. The reaction took place as follows (19):
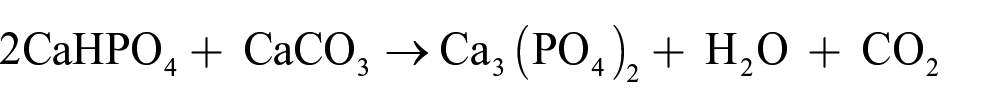
Pure Mg (purity ⩾99.5%, particle size ⩽75 µm) and Zn (purity ⩾99.8%, particle size ⩽45 µm) powders and β-Ca3(PO4)2 powders (average particle diameter of 9.0 µm determined by a Micro-Plus laser particle size analyzer) were used as raw materials. Ammonium bicarbonate particles (NH4HCO3; purity ⩾99.9%, particle diameter 75–250 µm) were used as the space-holder particles. The composition of the experimental composite was Mg-3% Zn (wt.%)/5% β-Ca3(PO4)2 (wt.%) powders with 0%, 5%, and 10% (vol.%) NH4HCO3 particles added to prepare different porosities according to our previous study. 23 To prevent powder oxidation during mixing, powders and particles were mixed in an argon-filled vacuum tank under a standard atmosphere. After mixing for 2 h, the mixtures were cold compressed to billets (the diameter of the billets was 20 mm and the height was 20 mm). Then, they were transferred to the vacuum sintering furnace, and the sintering process consisted of three steps. First, the billet samples were heated at 130°C for 2 h in a vacuum furnace, and the ammonium bicarbonate particles were removed. Afterward, the billets were heated at 550°C for 2 h under a highly pure argon atmosphere. Finally, the sintered samples were cooled to room temperature under a highly pure argon atmosphere.19,23 Porosities of the prepared composite scaffolds were measured using Archimedes Principle, similar to a published method. The dimensions of the sample were measured using the vernier caliper to produce a total volume (V). The dry weight (mdry) was measured with an analytical balance. The sample was then dipped into deionized water and suspended from an analytical balance to obtain wet weight (mwet). All weights were in grams and ρ was the density of absolute ethanol (0.8 g/cm3). Thus, the porosity (θ) of sample was calculated based on the following formula: 24

As a result, the porosities of the samples were found to be E1 (6%), E2 (13%), and E3 (18%), respectively.
Microstructural analysis
Microstructural and elemental analyses were conducted using scanning electron microscopy (SEM; Quanta-200) and energy-dispersive X-ray spectrometry (EDS; Quanta-200). The phase of the corroded products was detected using X-ray diffractometry (XRD) (DMAX-2500X). 23 The wavelength used was CuKα. The machine was equipped with a graphite monochromator; it was operated with a tube voltage of 40 kV, with a scanning range 5° to 85°. Continuous scanning was performed at a scanning speed of 8°/min.
Mechanical characterization
The mechanical properties of the porous Mg–3%Zn/5%β-TCP scaffolds were measured by compression tests performed on an MTS-810 testing machine at room temperature with a loading rate of 1.0 mm/min according to the ASTM E9-2009 standard. 23 The average scaffold size for compression tests was φ20 mm × 20 mm. Hardness tests were conducted using a Brinell hardness tester HBE-300 with a load of 250 N.
The immersion test
The immersion test was carried out in Hanks’ solution containing 0.14 g/L CaCl2, 0.35 g/L NaHCO3, 0.40 g/L KCl, 8.00 g/L NaCl, 0.06 g/L KH2PO4, 0.1 g/L MgSO4·7H2O, 0.1 g/L MgCl2·6H2O, and 0.048 g/L Na2HPO4. The ratio of the volume of the solution to the scaffold surface area was determined to be 20 mL/cm2. 23 The original pH value of Hanks’ solution was adjusted to 7.4 to simulate the environment of the human body. The sizes of scaffolds for immersion tests were φ20 mm × 20 mm. Three kinds of porous (6%, 13%, and 18%) scaffolds were immersed in Hanks’ solution at 37 ± 0.2°C for 168 h. The sample surfaces were ground with 1000 grit SiC sandpaper and cleaned with ethanol. 23 SiO2 balls with diameters of 2 mm were placed at the bottom of the container to prevent the sample surfaces from contacting the bottom of the container. The pH value of Hanks’ solution was measured by a PHSCAN10 model pH sensor. After the immersion test, the scaffolds were washed using distilled water and dried without removing the corroded product. Weight changes were measured with an electronic balance. Hydrogen bubbles generated from the immersed scaffolds were transferred to a measuring cylinder, and the hydrogen content was measured at different heights of Hanks’ solution. 24 Five scaffolds were verified for each group.
Cytotoxicity test
The cytotoxicity of the material was determined by an indirect contact test according to ISO10993-5:1999 standard. The experimental scaffolds were immersed in Dulbecco’s modified Eagle’s medium (Gibco) containing 10% fetal bovine serum (Gibco) for 24 h, and the material extract (100% concentration) was obtained. The material extract was diluted to 50% and 10% concentrations with DMEM medium containing 10% fetal bovine serum. MG63 cells, SaoS2 cells, and HK-2 cells were cultured in DMEM as well as 100%, 50%, and 10% concentrations of the material extract, respectively. DMEM acted as negative control. MTT was dissolved in phosphate buffered saline at a concentration of 5 mg/mL. After adding 10 μL of MTT solution, cells were cultured for 4 h. One hundred milliliters of Formazan solution was added to each cell group, and its optical density was measured by a spectrophotometer. The calculation of relative growth rate (RGR) was based on the formula below: 19
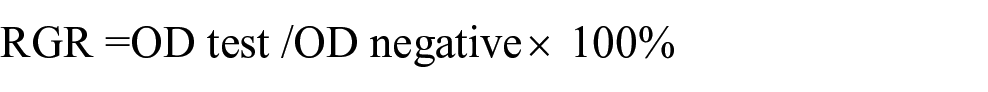
Hoechst staining
Four groups of HK-2 cells were fixed with 40 g/L formaldehyde and stained with 2 µg/mL Hoechst solution after 5 days of incubation. After incubation in the incubator for 10 min, the morphology of the cells was observed under a fluorescence microscope to verify the apoptosis of cells.
In vivo biocompatibility test
All animal experiments were carried out in accordance with the animal welfare legislation of the People’s Republic of China and approved by the Ethics Committee of the Second Xiangya Hospital. Eight adult New Zealand male rabbits with a body weight of 2.5–3.0 kg were randomly divided into two groups. Mg–3%Zn/5%β-TCP scaffolds were implanted into the dorsal muscles of the implanted scaffold group rabbits. The scaffold size processed from the experimental scaffold was φ2 mm × 6 mm, and the porosity was 6%. The control group rabbits were not implanted with scaffolds. All rabbits were euthanized 12 weeks after surgery. Histomorphological examination of the dorsal muscle tissue around the implanted scaffold as well as tissues from the heart, liver, and kidneys of rabbits was performed by hematoxylin-eosin staining (HE staining) to detect whether the degradation of the scaffolds was harmful to these important organs.14,19
Results and discussion
Morphologies of porous Mg–3%Zn/5%β-TCP scaffolds and XRD analysis before the immersion test
The microstructure and XRD analysis of Mg–3%Zn/5%β-TCP scaffolds are shown in Figure 1. The ratio of interconnected and noninterconnected pores and pore size increased gradually with an increase in the porosity (Figure 1(a–c)). Further observations of pores and the distribution of pore sizes on the surface of samples (E1, E2, and E3) have been supplied in the Supplemental Files. The pore distribution of the sample is more uniform with an increase in the porosity. The XRD patterns of the scaffolds revealed the presence of Mg phase and MgO phase (Figure 1(d)). There was no Mg–Zn phase or Ca3(PO4)2 phase observed, which was likely due to their low contents. In addition, no ammonium bicarbonate phase was observed, indicating that it was completely decomposed during the sintering process. Thus, further accurate assessment of the elemental distribution by EDS was employed to ascertain the chemical composition of the scaffold.
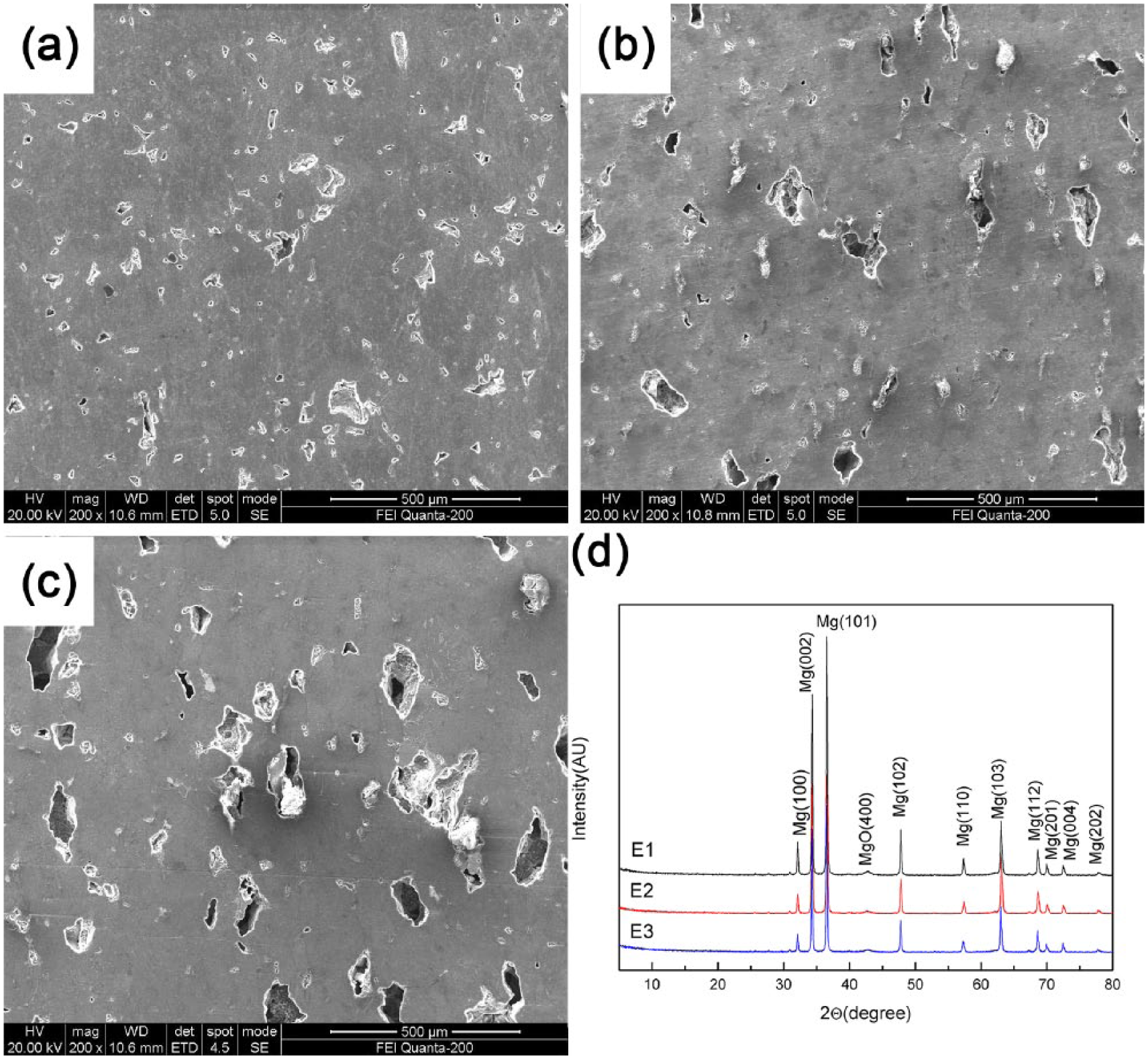
SEM micrograph (a) E1, (b) E2, (c) E3; (d) XRD analysis before the immersion test.
EDS analysis before the immersion test
The EDS analysis of scaffolds (Figure 2) indicated that the chemical composition of the grain area (point “a”) was Mg matrix. A white agglomerate compound (point “b”), whose energy spectrum exhibited a high content of calcium, phosphorus, and oxygen, suggested an uneven distribution of Ca3(PO4)2. After enlarging the grain boundary, it was found that there was higher content of magnesium and zinc in point “c.” This probably resulted from the diffusion and reaction of magnesium and zinc powder causing the formation of Mg–Zn phases. Moreover, the EDS analysis at point “d” demonstrated a relatively high content of O, indicating the presence of MgO. This was due to a small oxidation reaction.
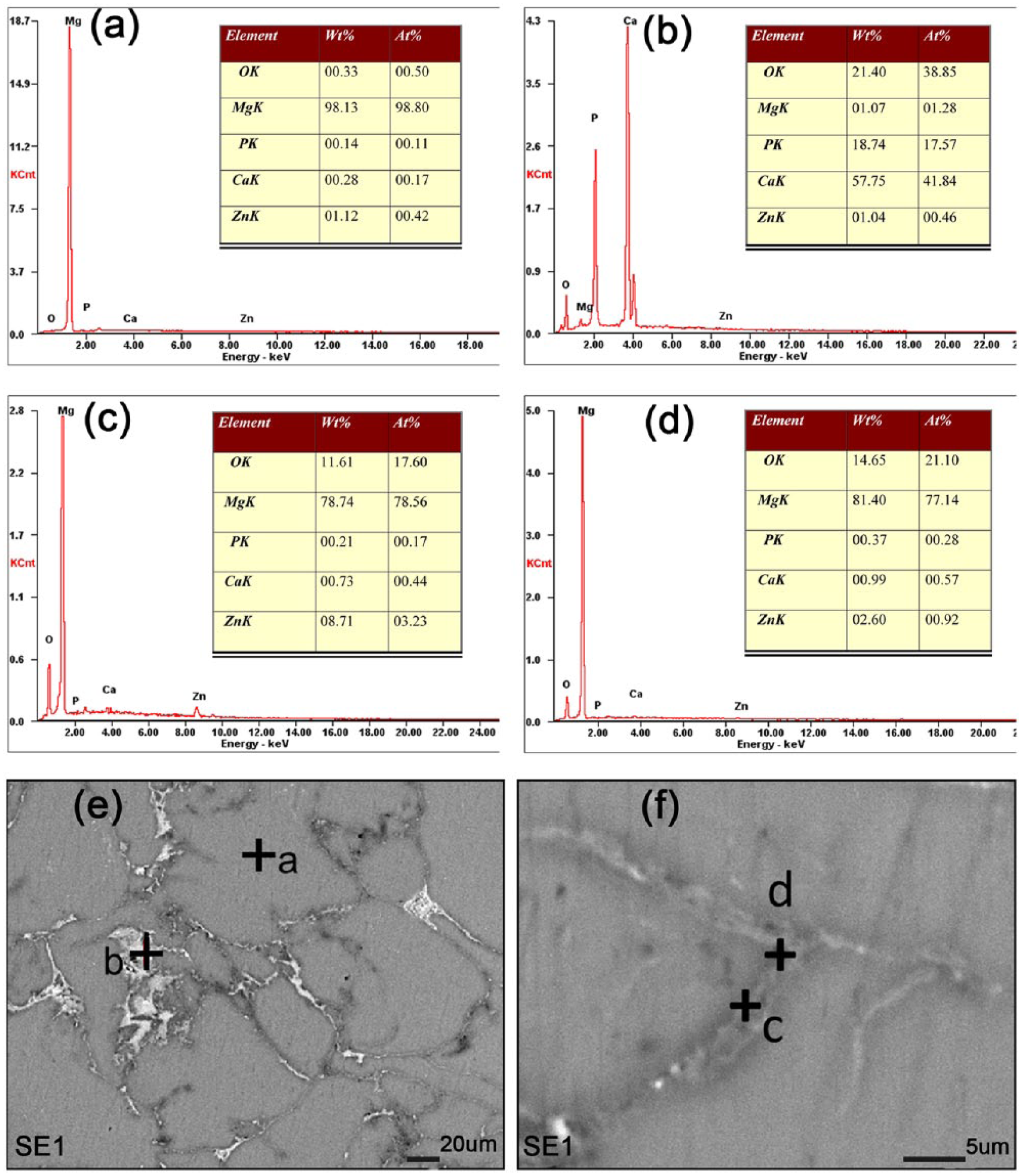
EDS analysis before the immersion test.
Mechanical properties and corrosion resistance of porous Mg–3%Zn/5%β-TCP scaffolds
The Brinell hardness and compressive strength of the scaffolds were found to be close to those of human bone and decreased with an increase in porosity (Table 1). This was probably due to the susceptibility of the edge of the pore wall to the concentration of stress during the test process, resulting in the origin of fracture. Furthermore, the more pores that are produced, the more pore walls, and thus, the more the concentration of stress that can easily induce fracture. The Young’s moduli of the scaffolds were relative to that of human bone and suitable for the elimination of the stress shielding effect caused by the severe mismatch of the human bone and implants. Therefore, the scaffolds have suitable mechanical properties and can be used for bone tissue engineering.
Mechanical properties and corrosion resistance of the Mg–3%Zn/5%β-TCP scaffolds.
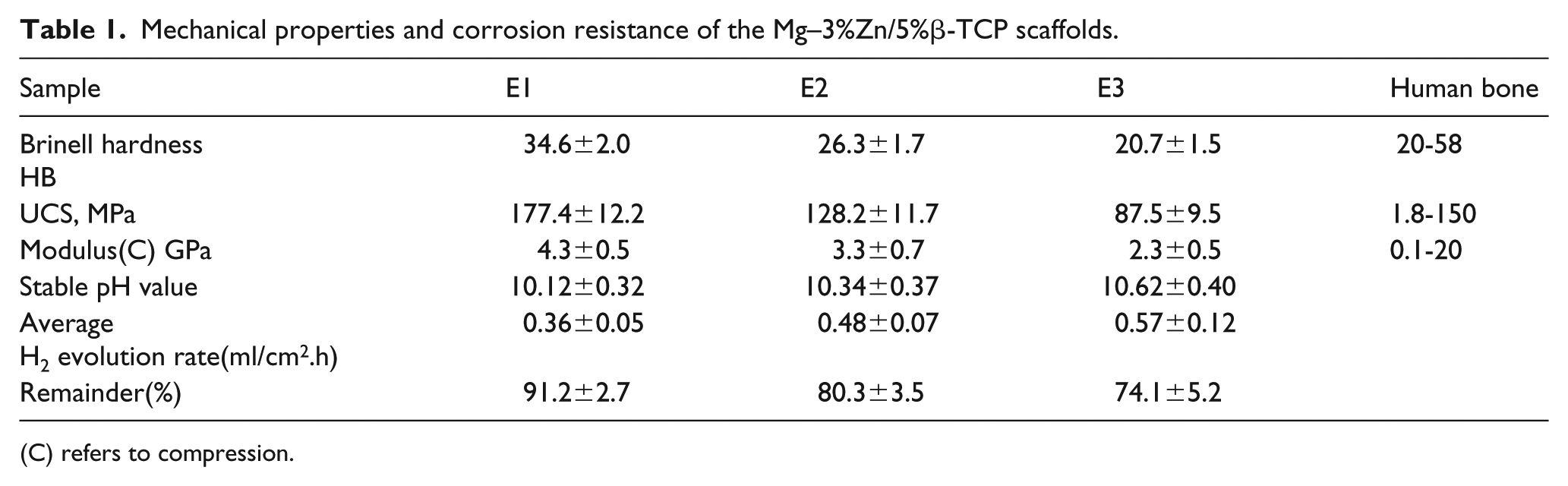
(C) refers to compression.
The weight loss, pH values of the solutions as they change, and the hydrogen gas collection values have been widely used to measure the corrosion rates of Mg alloys. The pH values increased significantly during the initial 24 h phase, and afterward, the increase was slower until the peak values stabilized. The stability of pH values at 120 h was E1<E2<E3. A lower pH value growth rate represents a lower corrosion rate. 25 Zhang et al. pointed out that an excessive pH value can cause cell death and inflammation of bone tissue. 26 Therefore, E1, with lower stable pH values, is more suitable for the cytotoxicity test and in vivo biocompatibility test.
Table 1 shows the average H2 evolution rate during 168 h of immersion testing. At the beginning of the test, a precipitation layer of corrosion products rapidly coated the surface of the scaffold, which reduced the release of gas bubbles. With the prolonged immersion time, more hydrogen gas gathered under the precipitated layer, which resulted in the flaking off of the corrosion product and the release of hydrogen gas. The hydrogen production rate of E1 was lower than those of E2 and E3, which indicated a better corrosion resistance.
The remaining weight of the scaffolds varied with the immersion time in the Hanks’ solution with a maximum time of 168 h. The weight change was determined as follows:

where R is the remainder, m0 is the initial weight, and m1 is the weight with different immersion times. Higher R values mean better integrity and a lower loss of strength for scaffolds. 3 After 168 h, the R values of E1 dropped lower than those of E2 and E3. The main reason for this phenomenon was that the increase in the porosity of the scaffold increased the contact area between the scaffold and Hanks’ solution, and then the pores transferred the solution more deeply into the scaffold matrix, promoting further corrosion. 23 In addition, Cl- ions could accumulate in the pores, rapidly increasing their content. Therefore, a large number of pores promoted and accelerated the occurrence of pitting corrosion and galvanic corrosion, leading to the destruction of the scaffold surfaces. Consequently, the in vitro degradation test results showed that E1 had the highest corrosion resistance and the porosity had a significant effect on the corrosion resistance of the scaffolds.
XRD analysis and EDS analysis after the immersion test
XRD patterns of Mg–3%Zn/5%β-TCP scaffolds after the immersion test for 24 h were obtained (Figure 3(a)), and the Mg, MgO and Mg(OH)2 peaks could be observed. The main corrosion product was Mg(OH)2. With the increase in porosity, the Mg(OH)2 peaks became more conspicuous. There was no noticeable formation of phases containing calcium phosphate products or Zn, likely due to their low contents. According to the EDS analysis of the scaffolds after 6 h of immersion testing (Figure 3(b)), the chemical composition of the grain area (point “a”) was identified as the Mg matrix. EDS analysis of point “b” exhibited a large amount of Mg and O, indicating the formation of Mg(OH)2. Based on its location, we could determine that the initial corrosion occurs at the pore. EDS analysis of point “c” beside the pore demonstrated higher amounts of Ca, P, and O, implying that there was a distribution of Ca3(PO4)2. This result further confirmed that Ca3(PO4)2 had a protective effect of inhibiting further degradation.
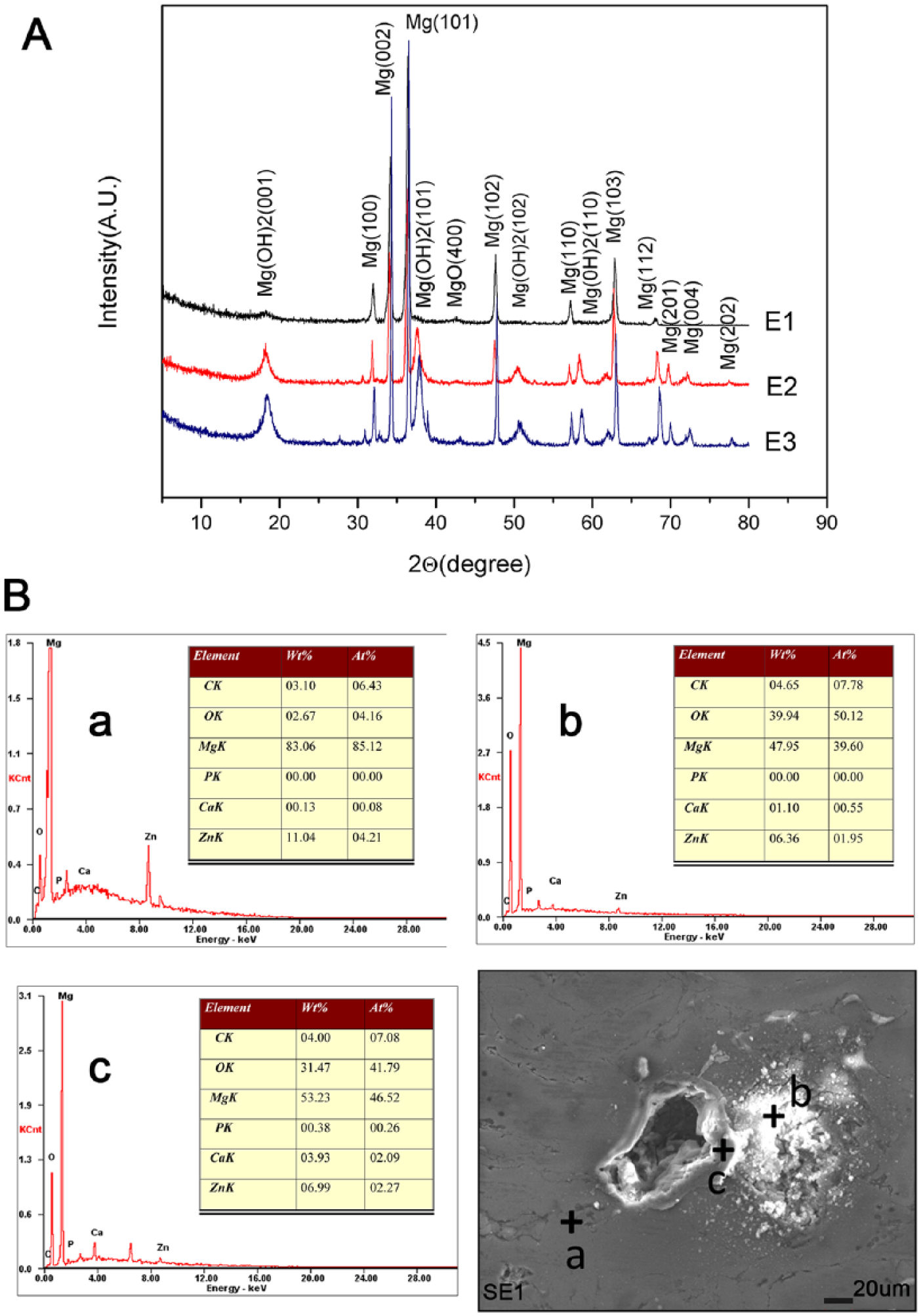
(a) XRD analysis after 24 h of the immersion test. (b) EDS analysis after 6 h of the immersion test.
Cytotoxicity test
Based on the above results, the scaffolds with a porosity of 6% have the best mechanical strength and in vitro corrosion resistance. Therefore, they were used as the model for the cytotoxicity test to assess the cytotoxicity of the materials. In this study, MG63 cells, SaoS2 cells, and HK-2 cells were cultured in DMEM as well as 100%, 50%, and 10% concentrations of the material extracts, respectively. All MG63 cells, SaoS2 cells, and HK-2 cells had normal morphologies and were capable of adhering, surviving, and proliferating in the cell culture system after incubation for 5 days in different concentrations of extracts, similar to the negative control group. The RGRs of the three kinds of cells after 1, 3, and 5 days of incubation (Figure 4(a)) suggested that there were no significant differences between the RGRs of the different concentration material extract groups and the negative control group (
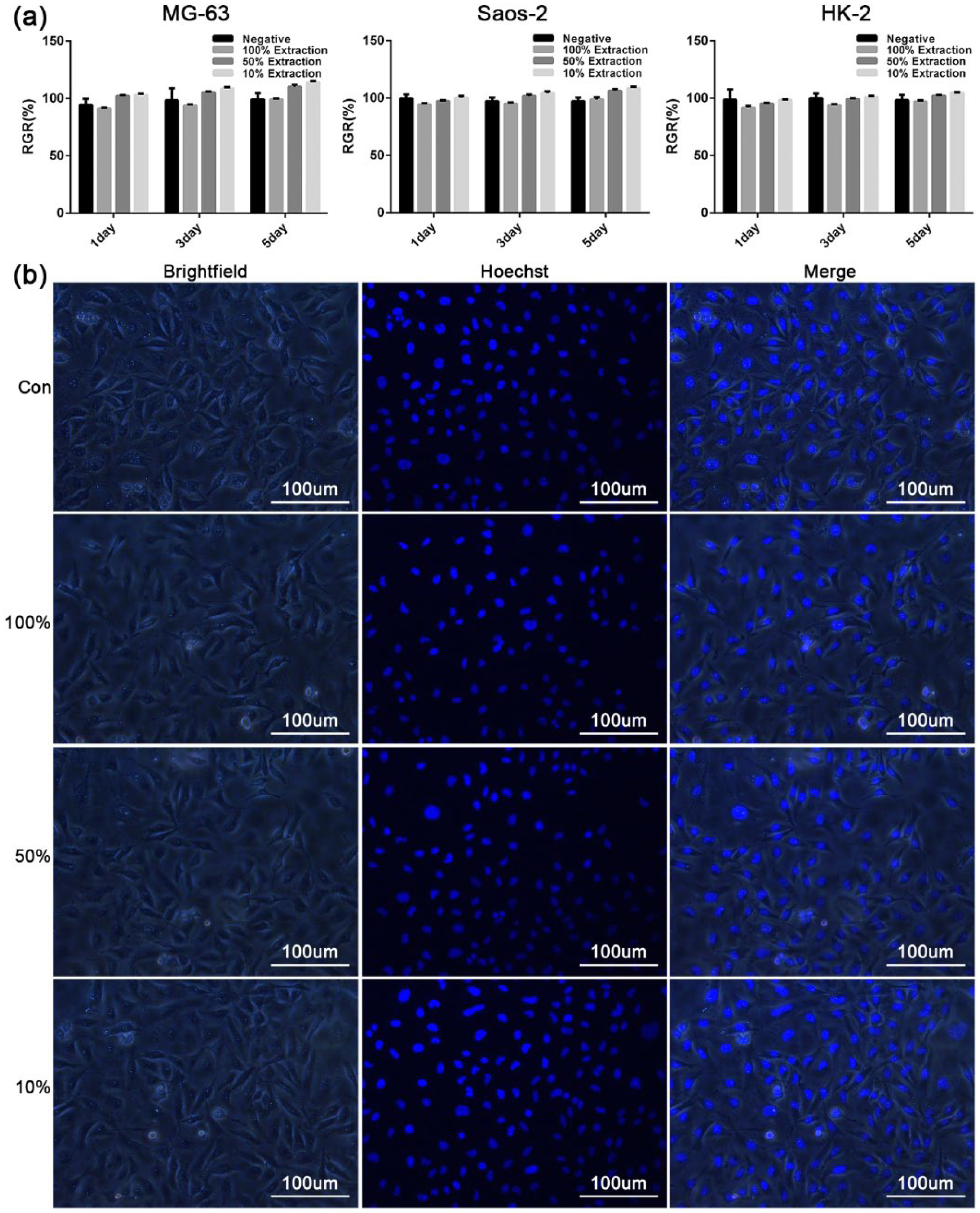
(a) RGRs of MG63 cells, SaoS2 cells, and HK-2 cells after 1, 3, and 5 days of incubation (
In vivo biocompatibility test
The HE-stained sections of heart, liver, kidney, and dorsal muscle tissue around the implanted scaffold of the implanted scaffold group rabbits indicated that all tissues were normal and there was no infiltration of inflammatory cells (Figure 5(b)). There were no significant differences between the implanted scaffold group (Figure 5(b)) and the negative control group (Figure 5(a)). These results suggested that the degradation products of scaffolds could be excreted through the normal metabolic process, and they did not cause local tissue inflammation or harm the important organs. In summary, the scaffold has good biocompatibility and is a good candidate for implant materials.
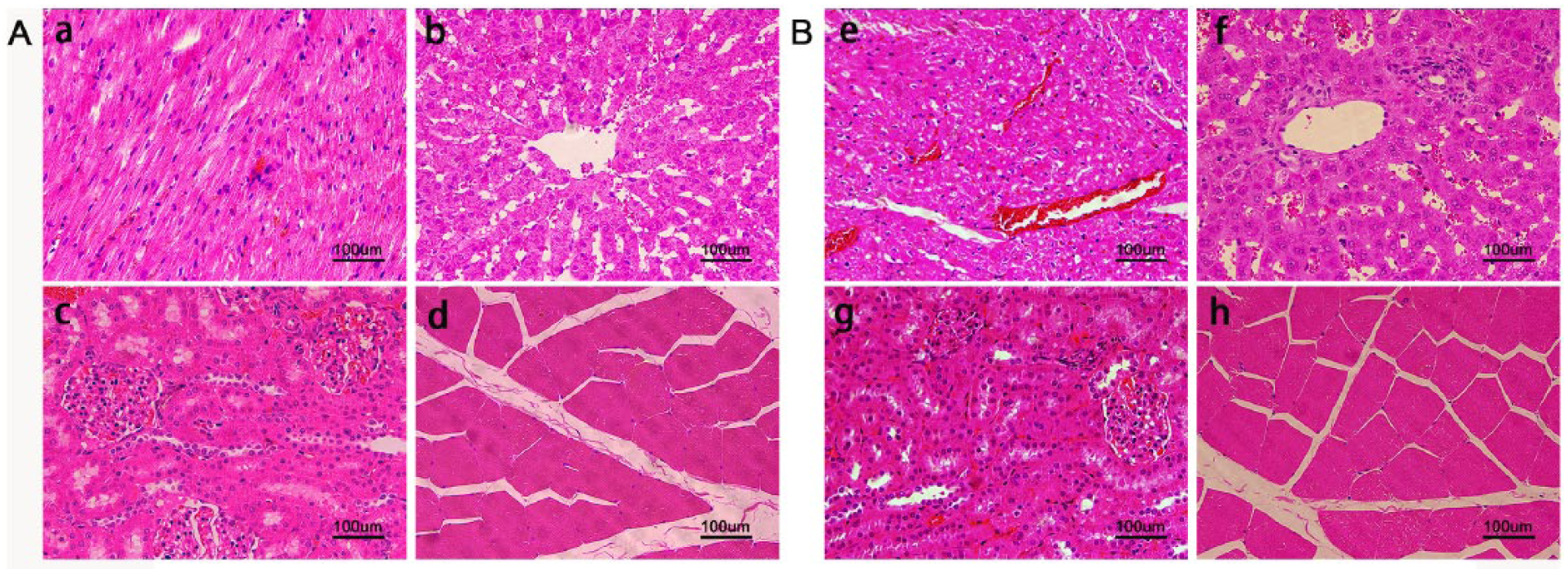
HE staining pathological sections of 12 weeks after implantation: A (negative control group): (a) heart, (b) liver, (c) kidney, (d) the dorsal muscle tissue. B (the implanted scaffold group): (e) heart, (f) liver, (g) kidney, (h) the dorsal muscle tissue around the implanted scaffold.
Conclusions
In summary, the microstructure of porous Mg–3%Zn/5%β-TCP scaffolds showed uniformly distributed Mg–Zn and β-TCP phases, and polygonal pores were formed in the α-magnesium matrix. Mechanical and corrosion tests showed that the scaffolds had mechanical strengths similar to that of human bone, and their corrosion resistance decreased with the increase in porosity. The scaffolds had cytotoxicity grades of 0–1 against MG63 cells, SaoS2 cells, and HK-2 cells, which suggested that they were appropriate for cellular applications. Finally, in vivo biocompatibility tests indicated that the scaffolds had good biocompatibility in important organ tissues, such as the heart, liver, kidney, and muscle of rabbits. Consequently, these results suggest that porous Mg–3%Zn/5%β-TCP scaffolds are promising biodegradable orthopedic implants, such as plates and screws for bone tissue engineering.
Supplemental Material
Supplemental_Flies – Supplemental material for Research on corrosion behavior and biocompatibility of a porous Mg–3%Zn/5%β-Ca3(PO4)2 composite scaffold for bone tissue engineering
Supplemental material, Supplemental_Flies for Research on corrosion behavior and biocompatibility of a porous Mg–3%Zn/5%β-Ca3(PO4)2 composite scaffold for bone tissue engineering by Mingying Tang, Yang Yan, Jianan OuYang, Kun Yu, Congcong Liu, Xiaohua Zhou, Zhenting Wang, Youwen Deng and Cijun Shuai in Journal of Applied Biomaterials & Functional Materials
Footnotes
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the following funds: (1) The National Natural Science Foundation of China (No. 81472058); (2) Hunan Science and Technology Innovation Plan (2018SK2105); (3) Hunan Science and Technology Innovation Plan (kq1606001); (4) Fundamental Research Funds for the Central Universities of Central South University (1053320184279).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
