Abstract
Purpose:
Bioprinting is an alternative method for constructing tissues/organs for transplantation. This study investigated the cross-linker influence and post-printing modification using oligochitosan and chitosan for stability improvement.
Methods:
Oligochitosan was tested as a novel cross-linker to replace Ca2+ for pectin-based bio-ink. Oligochitosan (2 kD) and different molecular weight of chitosan were used to modify the bioprinted scaffold. Fourier transform infrared (FTIR) spectroscopy and scanning electron microscopy (SEM) were used to characterize the scaffolds.
Results:
Oligochitosan failed to serve as a viable cross-linker. Successful post-printing modification was confirmed by FTIR and SEM analyses.
Conclusion:
Regarding post-modification, chitosan-treated scaffolds showed enhanced stability compared to untreated scaffolds. In particular, scaffolds modified with 150 kD chitosan exhibited the highest stability.
Introduction
According to the United States Department of Health and Human Services, over 116,000 people are on the organ transplant waiting list, and each day 20 people die while waiting for a transplant. 1 Bioprinting of artificial tissues and organs provides a method for stemming this discrepancy between organ donors and those waiting for a transplant.2,3 These tissues and organs can be engineered using a bio-ink made of biopolymers for scaffolding incorporated with the patient’s own cells.4,5
During a previous study, a novel pectin-based bio-ink and corresponding printing method was developed. 6 Pectin shows similar properties to alginate, a common polysaccharide used for constructing scaffolds, but is preferable to alginate, which may lead to allergic reactions. 7 Pectin is a natural biopolymer found in citrus rinds and is composed of galacturonic acid with carboxyl groups. Because of these negatively-charged carboxyl groups, pectin can be cross-linked with divalent cations to create hydrogel scaffolds. Ca2+ has commonly been used for such cross-linking purposes.8,9 Another potential cross-linker is oligochitosan, which was previously discovered.10,11 Calcium ions can be toxic at elevated levels within the body, 12 while oligochitosan has been demonstrated to show excellent biocompatibility.13,14 In addition to cross-linking, post-printing modification is necessary to further stabilize scaffolds. One such modification is treating the scaffold with oligochitosan/chitosan. Chitosan is the only naturally-occurring positively-charged biopolymer with good biocompatibility and biodegradability.15,16 Modification using polycationic polymers is critical for tissue engineering applications, since a positively-charged structure is preferred for cell adhesion.17–19
Materials and methods
Materials
Low-methoxyl pectin and Pluronic® F-127 were obtained from WillPowder (Miami Beach, FL) and Sigma-Aldrich (St. Louis, MO), separately. Pharmaceutical grade (>90% deacetylation) oligochitosan (2–3 kD) and chitosan (50,150, and 1000 kD) were purchased from Zhejiang Golden-Shell Pharmaceutical Co. (Zhejiang, China). A BioBot1 bioprinter (Philadelphia, PA) was used for bioprinting scaffolds. Dulbecco’s Modified Eagle’s Media (DMEM) was obtained from American Type Culture Collection (Manassas, VA).
Scaffold bioprinting process
An STL file of an object was first opened by the Repetier-Host software. Object size and position adjustments were made to optimize printing parameters. The object was then sliced with Slicr software and converted into a G-Code file. The G-Code file was then imported to the BioBot software that controlled the bioprinter.
The bio-ink used was composed of 3% (w/v) pectin, 20% (w/v) Pluronic® F-127, and a red food coloring dye, which was used to aid visualization. 6 The bio-ink was extruded through a 27 gauge blunt needle tip to begin the bioprinting process with a strand diameter of approximately 0.4 mm. A printing pressure of 35 psi was used with an axis movement velocity of 0.5 mm/s.
The bio-ink was extruded via a single strand onto a petri dish on top of an AmScope microscope temperature control stage slide warmer at 37°C (TCS-100; AmScope, Irvine, CA) to induce gelation of the Pluronic® F-127 in the first few layers of the scaffold. 6 Warm (~37°C) 150 mM CaCl2 was added around the base of the scaffold during the printing process to cross-link the pectin, further gelling the structure and providing a more permanent foundation for the remaining layers.
Post-printing modification
Once printing was complete, scaffolds were immersed in 37°C 150 mM CaCl2 for 5 min. The CaCl2 was then removed. The scaffolds were further treated with 2 kD, 50 kD, 150 kD, and 1000 kD chitosan solution, respectively, at a concentration of 0.25% (w/v) for an additional 5 min.
Scaffold characterization
For scanning electron microscopy (SEM) imaging, scaffolds were vacuum-dried and then mounted onto an aluminum stub. Samples were then examined under a Hitachi S-4800 ultrahigh resolution cold cathode field emission scanning electron microscope (FE-SEM). Dried scaffolds were also used for Fourier transform infrared (FTIR) analysis. FTIR spectra of the samples were recorded at room temperature using a NicoletTM 6700 spectrometer (Thermo Fisher Scientific, Waltham, MA).
Stability testing
Scaffolds were immersed in DMEM and incubated at 37°C, with their integrity and weight being monitored at different time intervals for a period of three days.
Results and discussion
Bioprinted pectin/Pluronic F-127 scaffolds were not stable if no cross-linking occurred (Figure 1(a)). Cylinder-shaped scaffolds were used to demonstrate the cross-linking effectiveness, though other geometries could be printed using the extrusion-based method (Figure 1(b)). The initial gelation was due to heating of the Pluronic F-127 from the heat stage on which the petri dish was placed.6,13 However, once the scaffold was removed from this heat the scaffold lost its defined shape, becoming a viscous liquid rather than a solid gel. Permanent hydrogel scaffolds can be created using divalent cations that serve as cross-linkers for the negatively-charged pectin molecules. Calcium chloride was added around the edges of the scaffold mid-print, as shown in Scheme 1, creating more stability than only heat-induced gelation of Pluronic F-127. Heat-induced gelation of Pluronic F-127 only occurred in the first few layers of the printed scaffold; the rest of the scaffold was gelled with calcium. 6 Using oligochitosan as a novel cross-linker was found to be ineffective, unfortunately, as scaffolds quickly reverted to viscous liquid state (Figure 1(a)). It is thought that oligochitosan did not cross-link the pectin in the same manner as Ca2+.11,20 Pectin cross-linking with Ca2+ results in an “egg-box” like structure caused by intermolecular chelate binding, while pectin cross-linking with oligochitosan results in a polyelectrolyte complex. 11
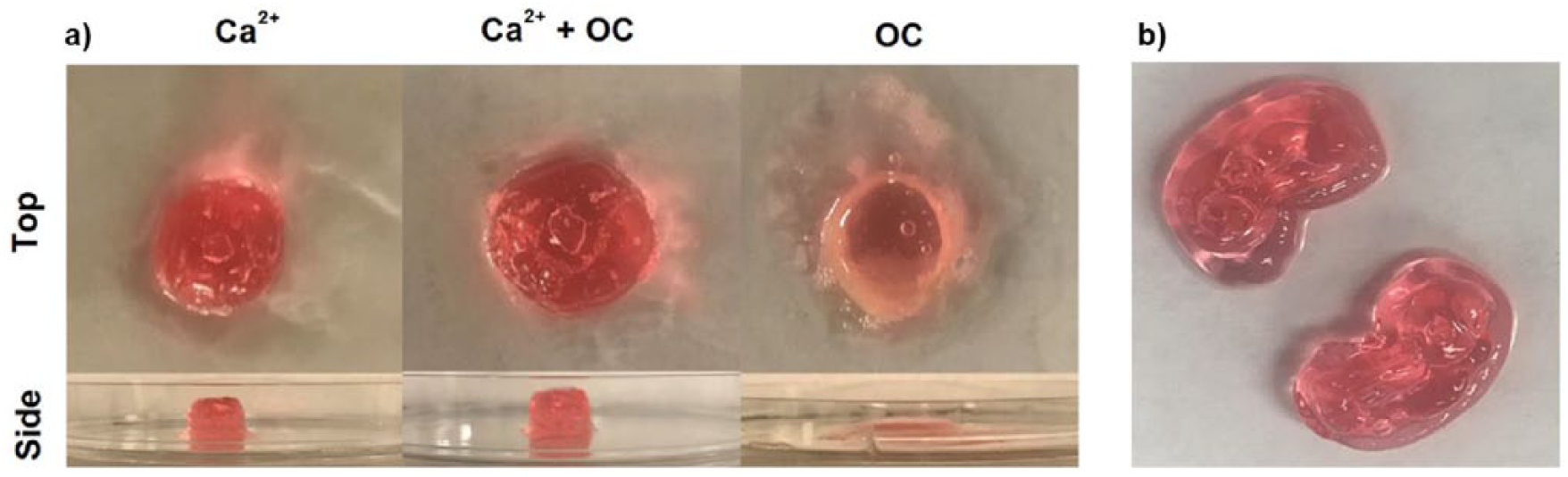
(a) Top and side view of pectin/Pluronic F-127 extrusion-based bioprinted scaffolds: cross-linked with calcium (left panel), cross-linked with calcium and further treated with oligochitosan (middle panel), and cross-linked with oligochitosan (right panel). (b) Top view of Pectin/Pluronic F-127 bioprinted kidney-shaped scaffolds cross-linked with calcium.
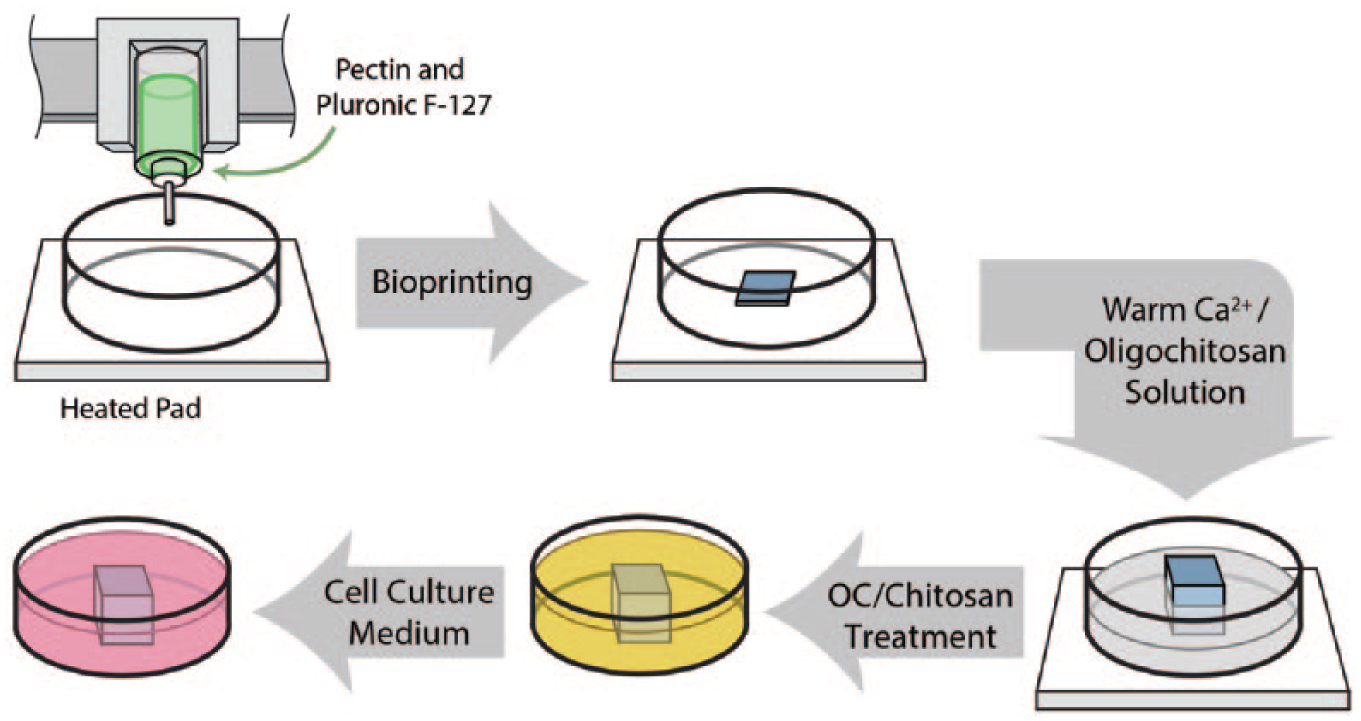
A schematic illustration of the bioprinting process and subsequent post-printing modification.
As shown in Figure 2, all post-printing modified samples showed similar FTIR spectra due to the strong hydrogen interaction of pectin and chitosan both intramolecularly and intermolecularly. For all samples, the carboxylic acid groups from pectin were ionized to carboxylate groups in the presence of Ca2+ or chitosan. The signals at 1600 cm−1 and 1413 cm−1 represent the carboxylate group. However, compared with the control sample, a signal at 1620 cm−1 is much more pronounced in the samples with oligochitosan or chitosan. This is caused by the protonated amine (NH3+) group from chitosan due to the neutralization of carboxylic acid (pectin) by amine (chitosan). In addition, signals at 1680 cm-1 represent the free amines (NH2), which come from chitosan. FTIR results confirmed pectin was further cross-linked by adding chitosan, leading to higher scaffold stability.10,15
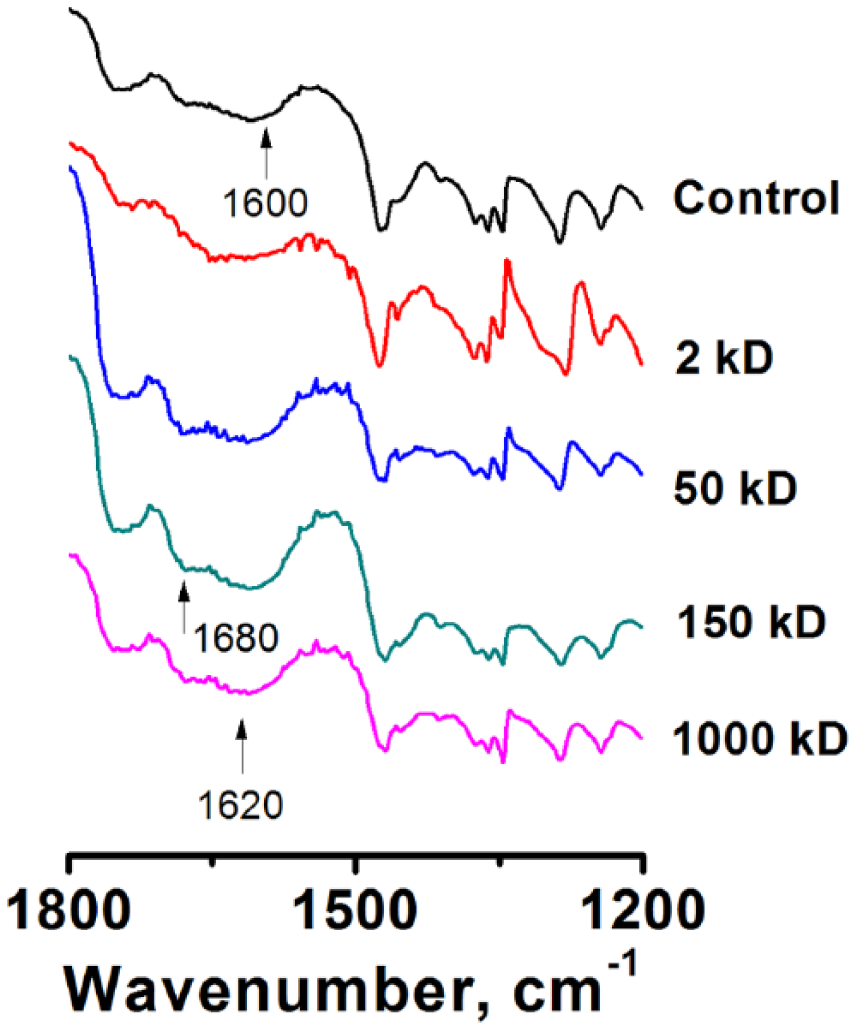
Fourier transform infrared (FTIR) spectra of bioprinted scaffolds.
The SEM images shown in Figure 3 provide additional insight into the surface and internal morphology of the samples discussed above. It is clearly seen that the 150 kD scaffolds have a less layered structure as compared to the other samples both internally and at the surface—there are relatively large, uniform, monolithic areas. This morphology can explain the increased stability by providing higher resistance to the structural deterioration.
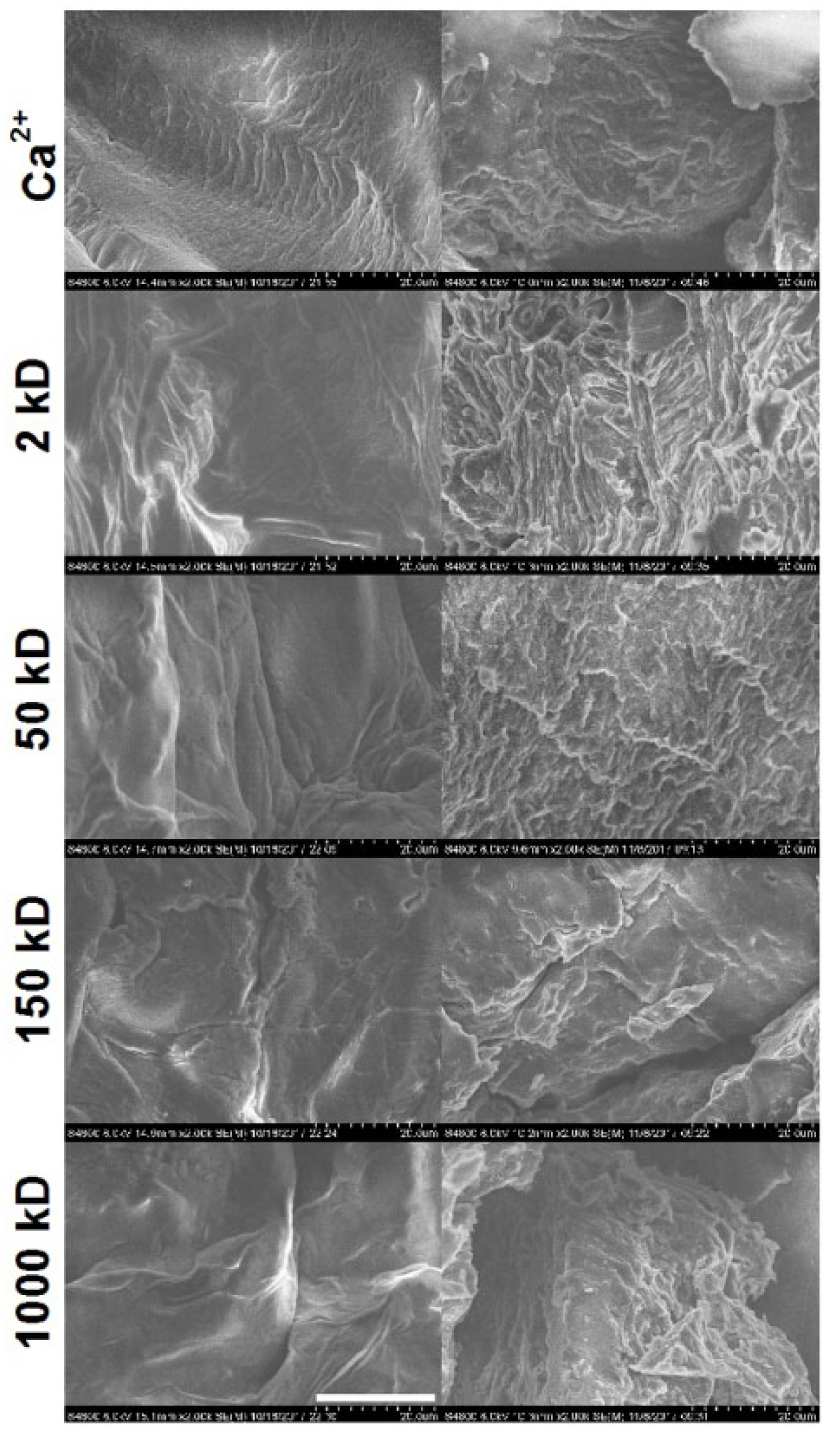
Scanning electron microscopy (SEM) images of bioprinted scaffolds cross-linked with Ca2+ and then further treated with 2 kD oligochitosan, 50 kD chitosan, 150 kD chitosan, or 1000 kD chitosan. Left panels depict surface structure, while right panels show internal structure. Scale bar: 20 µm.
Calcium cross-linking and subsequent chitosan modification of scaffolds was found to be more effective than only Calcium cross-linking (Figure 4). All scaffolds showed a substantial increase in mass in the first few hours of incubation in DMEM, which can be attributed to liquid uptake by the hydrogel. Sodium bicarbonate and phosphate ions in DMEM can also negatively affect scaffold stability by “grabbing” calcium ions away from the pectin. 21
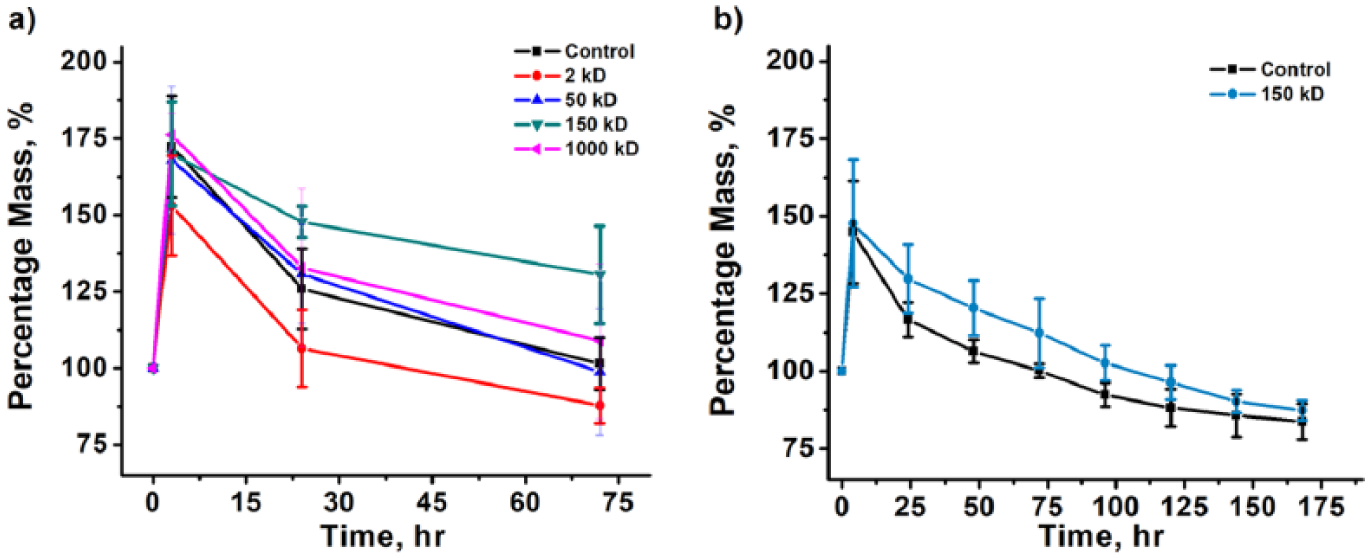
(a) Stability testing of bioprinted scaffolds cross-linked with calcium and further treated with oligochitosan or various molecular weights of chitosan based on percentage of initial scaffold mass. Scaffolds incubated in DMEM at 37°C for three days. (b) Prolonged stability testing of bioprinted scaffolds incubated in DMEM at 37°C for seven days. Scaffolds tested were cross-linked with calcium and either modified with 150 kD chitosan or unmodified.
Among chitosan-coated scaffolds, 150 kD chitosan was the most effective, while 50 kD and 1000 kD were rather ineffective (Figure 4(a)). Oligochitosan-treated scaffolds were extremely brittle after a period of only three hours, falling apart while being weighed (data not shown). It is thought that oligochitosan became competitive with Ca2+ for carboxylic acid group binding, which led to deconstruction of the scaffold. 11 Chitosan-coated and calcium-treated scaffolds maintained their shape through 24 hours. However, after this period 50 kD-coated scaffolds fell apart more easily than other scaffolds. After the incubation period of three days, 150 kD-modified, 1000 kD-modified, and Ca2+ scaffolds had maintained their shape and remained above their initial mass, a measure of stability. The 150 kD scaffolds resisted breakage substantially more than other scaffolds. Furthermore, 150 kD-modified scaffolds were more stable than unmodified Ca2+-treated scaffolds after incubation in DMEM for seven days (Figure 4(b)). Most likely, 50 kD chitosan was too small to effectively complement the Calcium cross-linking, and, instead, competed for binding sites. On the other hand, 1000 kD chitosan was too large to penetrate deep enough to effectively stabilize the scaffolds. Thus, post-printing coating with chitosan, specifically 150 kD chitosan, shows great promise for creating stable pectin-based scaffolds and further implantation of cells to engineer artificial tissues and organs.
Conclusion
Bioprinted pectin-based scaffolds were found to be most stable when cross-liked with Ca2+ and then modified with 150 kD chitosan. Oligochitosan was determined to be an ineffective cross-linker and modifier for the scaffolds. Scaffolds modified with chitosan remained relatively stable compared to unmodified scaffolds after incubation in DMEM during the testing period, showing great promise for potential inclusion of cells to engineer artificial tissues and organs.
Footnotes
Acknowledgements
The authors would like to graciously thank Mason Degeneffe and Devon McCune for their technical assistance and Mark Hoelzer for the schematic diagram.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the National Science Foundation (Grant Number EEC-1460183) and a Milwaukee School of Engineering Summer Faculty Development Grant.
