Abstract
Purpose
The distribution and relationship of hydroxyapatite debris, nanometric organic and metal wear particles and metal ions on periimplant interface membranes following aseptic and septic arthroplastic loosening were investigated.
Methods
Scanning electron microscopy and X-ray spectroscopic analysis were used to analyze debris and ion distribution.
Results
Hydroxyapatite debris appeared with different morphology in a particular distribution among several membranes. These differences may reflect the occurrence of different friction forces taking place between prosthesis and bone interface in the several types of prostheses studied. Metal wear particles were detected in greater numbers in membranes from noncemented prostheses compared with those from cemented ones. In contrast, more organic particles were present in membrane from cemented prosthesis. No differences were observed between aseptic and septic membranes.
Conclusion
Our findings support the need to evaluate the occurrence of friction forces that periprosthetic bone debris production may induce to exacerbate cellular reactivity. Furthermore, cellular engulfment of debris and the high level of different ions released indicate the occurrence of a toxic environment that may induce failure of any reparative pathways.
Introduction
Arthroplasty clinical failure is linked to periprosthetic osteolysis and is widely associated with inflammatory response elicited by orthopedic biomaterial wear debris (1, 2). Several authors have observed particulate debris to induce inflammatory cytokine release by macrophages and osteolysis in in vitro and in vivo experimental models (1, 3-5). Indeed the severity of osteolysis was highly related to the number of particles observed (6-8). Furthermore, inflammatory cellular response is highly dependent on the composition, size and shape of particles (2). All of the various components of the prosthesis, such as polyethylene, metal, ceramic and bone cement generate particle debris as a consequence of the occurrence of frictional forces (9). Moreover, the type of prosthesis in terms of material and structure is crucial for wear debris development and subsequent osteolysis. In conclusion, wear debris represent the most important cause of periprosthetic osteolysis, although some authors have pointed also to the involvement of other factors such as fluid pressure and adherent endotoxins (10, 11).
Differences in the chemical composition of debris have generated different hypothesis on modalities of osteolysis and arthroplastic failure. Cement debris is known to induce cytokine production by macrophages, that may be responsible for the observed cell death and implant failure (1). Instead metallic debris were imputed to induce mainly a direct cytotoxic action on cells (12, 13), although an inflammatory effect on resident macrophages was also evidenced (14). In particular, the occurrence of metal nanoparticles and ions was claimed to be the most potential candidate for arthroplastic failure by metal wear debris (15).
Recently the occurrence of metal sensitivity reaction, following arthroplastic substitution, has been pointed out. Metal sensitivity reaction is characterized by a predominantly lymphocytic response but not necessarily high wear. This reaction termed aseptic lymphocytic vasculitis associated lesion (ALVAL) (16, 17), is thought to be a reaction to metal ions, with development of metal–protein complexes (haptens) stimulating an immune reaction in which unusual lymphocytic infiltrates were found. These findings were similar to those found in association with type IV hypersensitivity (18). In this context, it may be important to consider that the mechanical stress occurring in a prosthetic joint may lead to a release of bone particulate as hydroxyapatite debris, which may overburden scavenger cellular systems inducing a more rapid evolution of arthroplastic loosening.
In the present work, using scanning electron microscopy (SEM) and X-ray spectroscopic analysis, we investigated hydroxyapatite particles, prosthetic wear and metal and organic debris distribution, focussing our attention on wear debris of submicron and nanometric dimensions; the occurrence of ion pollutants and ion aggregates were also evaluated. The analysis was performed on histological sections of periprosthetic membrane obtained following arthroplastic loosening. To assess the influence of immune response within the periprosthetic membrane environment, aseptic and septic arthroplastic loosening was also studied.
Materials and Methods
Specimen collection
Twenty-eight specimens of bone implant interface membrane from cementless and cemented total hip replacements were taken at the time of first revision surgery for septic (cemented, 3 male and 3 female, n=6; cementless, 2 male and 4 female, n=6) and aseptic (cemented, 3 male and 5 female, n=8; cementless, 4 male and 4 female, n=8) loosening. Specimens were selected to obtained an homogeneous population in terms of age of patients and duration of implant. Age of patients (male and female) was a mean (±SD) of 68 ± 6 years; mean implant duration, 8 ± 2 years.
The cementless prostheses had Ti6Al4V-treated cups coupled to titanium alloy SCL stems (SCL, Lima, Italy); cemented prosthesis had standard ultra-high-molecular-weight polyethylene (UHMWPE) cemented cups coupled to polymethylmethacrylate (PMMA)-cemented Charnley stems (Co-Cr alloy stems; Charnley, DePuy Inc., Warsaw, IN, USA).
All enrolled patients had radiographic and clinical evidence of prosthetic loosening, and antiinflammatory medication had been discontinued 10 days before revision surgery. Patients with systemic or organ pathology were excluded. Tissue samples were taken from zone II, according to DeLee/Charnley's radiographic classification (19), corresponding to the back of the component. No tissue was taken from areas showing aggressive lysis. Surgical removal was performed between 1993 and 1995, samples were immediately processed for fixation and paraffin embedding, than stocked in paraffin until use.
Specimens of hip joint capsule of 5 patients (male and female: 65 ± 7 years old) were used as control group in ion detection analysis.
Tissue processing
Each specimen was washed in cold phosphate-buffered saline (PBS) and fixed in 10% buffered formalin for 48 hours at 4°C. Samples were then dehydrated and embedded in a semisynthetic paraffin (Sherwood Medical Co., St. Louis, MO, USA). Whole paraffin blocks were cut serially with a microtome (Leica-Jung, Germany) in consecutive 5-μm-thick sections (1 section per slide).
Three couples of consecutive sections for each sample were chosen for analysis, the first section of each couple was stained with hematoxylin and eosin to observe the general microanatomy, the second one was processed for the morphological and structural analysis of particles and ions by SEM and Fourier transform infrared (FTIR) spectroscopy.
Tissue cell identification
Tissue cells were identified as elements presenting a spongy body (a well-known consequence of fixation and embedded protocols) (20). Leukocytes were identified by a well-defined spheroid cell body and superficial microvilli. Erythrocytes were identified by their discoid biconcave architecture.
Particulate detection
Sections were deparaffinized by xylene washing (2 × 3 minutes), than hydrated by washing in descending series of alcohol (Ethanol solution 100, 95, 70 and 50, water; 1 minute each) and air-dried at room temperature. The dried section was attached to a standard SEM stub, sputter coated with 20 nm of gold (representing the morphological limit of SEM analysis resolution), and observed with a Quanta200 FEI Philips SEM. Images were transferred to a computer-based, digital image analysis system (QWin Image Analysis, Leica).
Hydroxyapatite particles or organic (UHMWPE and PMMA) and metallic wear debris (submicron particles having diameter <0.26 μm, and nanoparticles) were counted and analyzed. The morphology and area of individual particles were also evaluated. Identification of the particles' chemical composition was assessed by an X-ray microanalysis EDX instrument (EDX Genesis; EDAX, Mahwah, NJ, USA) instrument attached to SEM.
Ion detection
Ion distribution and evaluation in the membrane were done by X-ray microanalysis with an EDX instrument attached to SEM.
For each field of view, the blank image was merged with the detected image of each specific element analyzed, and the specific signal associated with particulate debris observed under SEM resolution (blank image) was used to identify the particulate, but deleted from spot calculation and therefore from ion quantification. Free ion analysis was focussed on elements composing the different metallic alloys of the prosthesis, such as Cr, Co and Mo for cemented prosthesis, and Al, Ti and V, for noncemented prosthesis. Ions occurring as contaminants derived from industrial catalyzer in the forming alloy were also detected. In particular, ions such as Fe, Zn, Al and Ti were researched in membranes from cemented prosthesis; ions such as Fe and Zn were researched in membranes from noncemented prosthesis. Background ions were evaluated to better visualize tissue dishomogeneity (vessels' lumen or tissue borders): in particular, the presence of C and O ions for the tissue background and Si ions for the glass-slide background were detected.
To distinguish the occurrence of ions analyzed in tissue catalytic enzymes, or their occasional occurrence as alimentary or environmental contaminants, the X-ray microanalysis was performed on control specimens of hip joint capsule. Their quantification was used as background value and subtracted from the experimental group free ion quantification. In particular, to distinguish the Fe present in hemoglobin, Fe signals occurring in erythrocytes were not detected.
Data analysis
Quantitative analyses of particles and ions were performed on 5 randomly chosen fields of view of 150 μm2 for each slide of the 28 samples under study (3 slides for each sample). For each slide, the number of particles was expressed as the sum of particles detected in several fields of analysis. The borders of all particles observed in each field of view were delineated, and the area of single particles evaluated. Then for those particles for which the minor diameter to major diameter ratio was ≤1 and ≥0.5 (indicated in the text as “d/D+”), the average size (i.e., the ideal diameter if the particle was a perfect sphere) was calculated. This parameter was chosen in presenting particles' morphometric data to make the comparison easier with data published by other authors.
In each field of view (150 μm2), ion data quantification was expressed as a percentage of the respective element on the total ions evaluated, because this is the reporting data on which our EDX instrument is set by manufacturer. Moreover, we have setted analysis for detection of all ion species under examination for each field of view in each of the samples analyzed.
The Kolmogorov-Smirnov statistical test was used to assess the normal distribution of data. The average of the different parametric data investigated was calculated from single-sample data, and the mean ± SEM for each experimental group was then obtained. Parametric statistical analysis of variance (ANOVA) was used, followed by post hoc Newman-Keuls multiple range test, to identify any differences between the averages. The minimum level of significance was set at a p value <0.05. All statistical procedures were performed with the statistical software Prism 2.01 (GraphPad Software Inc., San Diego, CA, USA).
Results
Tissue morphology under SEM visualization
SEM histological visualization showed a tissue composed of compact longitudinal bundles, while the presence of elements showing a spongy matrix could be observed lying between bundles. Moreover, morphologically well-defined lymphocytes were observed to invade the tissue surface, especially in septic membranes (Fig. 1A).

Microphotograph panel of periprosthetic membranes following SEM analysis.
Membrane fibroblast cells were identified as spongy bodies showing a well-elongated profile, strictly associated with fibres bundles (Fig. 1B). Macrophage cells were considered those elements showing a roundish cell spongy body, larger than a fibroblast cell body (Fig. 1C); both cell types showed shrinkage from surrounding tissue, a normal occurrence in tissues undergoing to process of dehydration and hydration related to paraffin protocols (Fig. 1C).
Bacterial invasion was seen in septic membrane as the occurrence of many little oval bodies on the membrane tissue (Fig. 1D); frequent matrix spongy elements engulfing these oval bodies were observed, which may correspond to macrophage phagocytic action (Fig. 1E). Vascular profiles were observed and identified by the presence of morphologically well-defined erythrocytes (Fig. 1F). Thin, spongy matrix elements, strictly bordering vascular profiles, were interpreted as endothelial cells (Fig. 1F).
Particles analysis
Particles different in nature and larger than 0.26 μm in average size were observed in the periprosthetic membranes analyzed, but they were not subject to analytical attention, because their analysis in the same prosthetic membrane was focused and detailed previously (12).
For each field of view, no more than 3 or 4 particles were observed. The smallest particle debris observed following SEM analysis was a metal particle (in size = 57 nm).
Hydroxyapatite particles were detected in several membrane types. These particles appeared as single or agglomerate granular or lamellar bodies diffuse on the tissue; all hydroxyapatite particles were detected as d/D+. Different hydroxyapatite particles showed a particular distribution among several membrane types analyzed; results are reported in Figure 2A and B. Agglomerate (large) particles were detected in membranes from aseptic noncemented prosthesis (AnC), aseptic cemented prosthesis (AC), septic noncemented prosthesis (SnC), but not in membranes from septic cemented prosthesis (SC). In particular, lamellar-type agglomerates were observed in AnC and AC (Fig. 3A), instead granular-type agglomerates were observed only in SnC (Fig. 3B). No quantitative differences were detected in large hydroxyapatite particle numbers (Fig. 2A), the average size of granular agglomerate was similar to that of lamellar agglomerate (Fig. 2B).
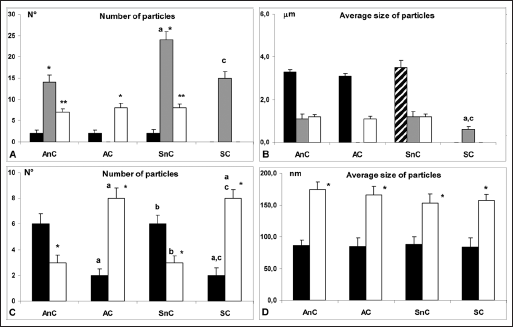
Histogram panel of quantitative analysis (
Small single granular hydroxyapatite particles were detected in all of the examined membranes, with the exception of AC membranes; the greatest number of particles was observed in SnC membranes, while the smallest average size was observed in SC membrane (Fig. 2A, B).
Small single lamellar particles (thickness = 26.3 ± 4.2 nm) were observed in all of the examined membranes with the exception of SC membranes; no differences in number and average size (referred to superficial plane) were detected (Fig. 2A, B).
Wear metal and organic particles of average size smaller than 260 nm were analyzed in several membrane types of the present study; results are reported in Figure 2C, D.
Metal particles detected showed a roundish shape, while organic particles showed ovoid shape; all metal and organic particles were detected d/D+ (Fig. 3D). Quantitative analysis evidenced that metal particles were more numerous in membranes from noncemented prosthesis (Ti6Al4V alloy as confirmed by EDX analysis) than from cemented ones (CoCr alloy as confirmed by EDX analysis); the contrary was observed for organic particles, while no differences were observed between aseptic versus septic membranes. Average size of organic particles was higher than metal ones in all membranes analyzed (P<0.05); no differences were detected comparing organic or metal particles average size between several membranes analyzed (Fig. 2C, D).

Microphotograph panel of periprosthetic membranes following SEM analysis.
Sometimes organic or metal wear particles, as indicated by EDX analysis, were observed inside macrophage spongy bodies indicating an active engulfing process of the particulate (Fig. 3C).
Ion detection and quantification
Analysis of ion presence and distribution is illustrated in Figures 4 and 5. In membranes from cemented prosthesis (AC and SC membranes), the presence of Cr, Co and Mo, representing specific ions in the alloy stem of these prosthesis, was detected. Quantitatively the presence of Cr, Co and Mo was higher in AC membrane than SC membrane (Fig. 4). The presence of a evaluable amount of contaminant ions such as Al, Ti and Fe was detected in membranes from cemented prostheses; in particular, in SC membranes compared with AC membranes, a significantly greater amount of Al and Fe and a lesser amount of Ti were detected (Fig. 4).
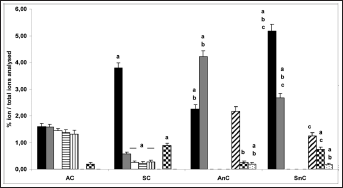
Histogram of ions analysis performed by X-ray microanalysis with EDX instrument attached to SEM. Black bars: Al ions; gray bars: Ti ions; white bars: Cr ions; horizontal line bars: Co ions; vertical line bars: Mo ions; diagonal line bars: V ions; square bars: Fe ions; small dot bars: Zn ions. a = P<0.05 vs. AC; b = P<0.05 vs. SC; c = P<0.05 vs. AnC. AC = aseptic cemented prosthesis; AnC = aseptic noncemented prosthesis; SC = septic cemented prosthesis; SnC = septic noncemented prosthesis.
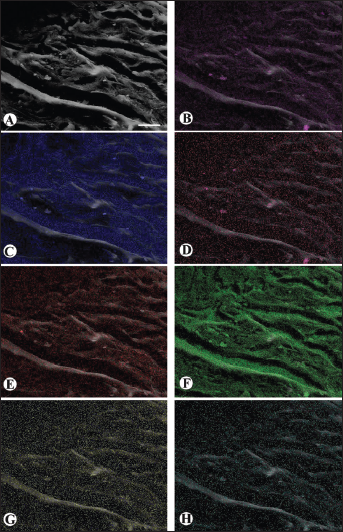
Microphotograph panel of aseptic noncemented prosthesis (AnC) membrane
following EDX ion analysis.
In membranes derived from noncemented prostheses (AnC and SnC membranes), a large presence of Al, Ti and V, the ions in the prosthesis alloy, was detected. In particular a greater amount of Al was observed in SnC membranes compared with AnC membranes, while a greater amount of Ti and V was observed in AnC membranes (Figs. 4 and 5). The occurrence of a evaluable amount of alloy contaminant ions such as Fe and Zn was observed in these membranes; in particular Fe ions were more represented in SnC membrane than in AnC ones, while Zn ions were detected at similar levels in both types of noncemented membranes (Figs. 4 and 5).
The comparative analysis of Fe and Zn alloy ion contaminants showed that in aseptic membranes (AnC and AC membranes), Fe ions were found in a similar lower level to septic ones, while the amount of Zn ions detected in SnC and AnC membranes was greater than in SC and AC membranes (Fig. 4).
Following EDX analysis for Al, Fe and Ox elements, in SC and SnC membranes only, specific light spot clusters were detected. These spot clusters were identified as AlFeOx ion aggregates (Fig. 5B, D, F).
Discussion
Micrometric wear debris are regularly detected for pathological evaluation in histological processing slides in which several solution baths are used (21-23). No evidence has been observed regarding the alteration of particle quantification by means the deparaffinization protocol application. Indeed xylene is an apolar solvent, and solvation properties are not expected of it. Otherwise ethanol has coagulative properties on biological tissue, and this is expected to favor the permanence of ions and particles. Furthermore, the short time of the steps of the deparaffinization protocol may be helpful into limining the washing out of particles and ions.
The direct visualization of deparaffined periprosthetic membrane tissue using SEM microscope and EDX analysis is a new investigative approach that allows us to perform an accurate qualitative and quantitative detection and localization of wear particles and metal ions within histological elements.
SEM analysis is a high-resolution morphological investigation leading to the visualization of nanoparticles. The need to cover samples with a 20-nm-thick gold coating to make the tissue resistant to high-voltage electron beams represents the limit for the detected size of particles.
The ion analysis was performed by EDX instruments attached to SEM, its functionality is based on the interaction of a narrow electron beam on tissue elements. The instrument is able to detect the elements occurring on a sample and recognize their chemical nature. No limit exists for the discovery of elements on the sample because the electronic beam penetrates the layer of sample, and no elements can hide from this analysis, including the sample support. The gold coating is detected as a covering layer and does not affect the identification of other elements. EDX analysis detects each element both as free ions and as elements chemically linked in a molecule or larger structure. In the case of free ions, the EDX report shows an isolated spot signal on the tissue blank image; in the case of molecules or larger structures, the EDX report shows collected spot signals. Moreover, by EDX analysis, particles smaller than 20 nm (not visible by morphological SEM analysis) can be detected as positive signals (spot cluster) and investigated for their composition (not morphology). Furthermore, particles larger than 20 nm (visible by morphological SEM analysis) can be analyzed for their chemical composition.
Spongy matrix structures lying between compact longitudinal bundles, observed following SEM tissue visualization, are compatible with cellular elements immersed in their extracellular matrix, as observed following light microscope tissue visualization. Therefore, occasional metal and organic particles found inside spongy matrix were interpreted as phagocytic materials.
Hydroxyapatite particles were observed on the membranes studied. The distribution of hydroxyapatite particles different in shape and number between membranes from cemented and noncemented prostheses may be a reflection of different friction forces occurring between the prosthesis surface and periprosthetic bone, between the 2 types of prosthesis. Nevertheless, the different distribution of hydroxyapatite particle types between septic and aseptic membranes led us to hypothesize that in the septic environment, activation of osteoclastic function and/or macrophage scavenger function may account for the differences detected. Indeed the engulfment of a single granular particle was observed by supposed macrophage cells. We cannot exclude the possibility that the occurrence of the septic condition leads to an alteration of the fluid hemodynamic perfusion of the membrane tissue influencing the distribution of hydroxyapatite particles.
The nature of wear particles is an important factor in determining cellular activation or inflammatory response. It is well known that Ti6Al4V particles elicit the production of very high levels of inflammatory cytokines, while CrCo debris are less stimulatory than Ti6Al4V debris, but more stimulatory than UHMWPE debris (14). In the present work, as well as in our previous work (12), in cemented prosthesis, we found a small amount of CrCo alloy debris, but the occurrence of a large number of organic debris may produce a particle pool more reactive than that obtained from Ti6Al4V prostheses, even if a high production of metal debris occurs in that kind of prosthesis.
The morphology of wear particles has been supposed to represent an important variable in stimulating cellular response (22). In our work, the morphology of the wear particulate detected was rather similar; these findings may highlight the occurrence of constant shearing forces in producing particulate. Any further speculation about this parameter was difficult in the present work.
Nanometric dimensions of wear debris have received particular attention as being responsible for several tissue reaction phenomena (15). In our previous work (12), the analysis of wear particle debris performed by light microscopy had its limits for the particle dimensions observed. In the present work, using SEM, nanometric particles were detected, although their quantity was not elevated, although a large number of nanoparticles are supposed to be produced in metal–metal arthroplastic joint substitution (15). Nevertheless, the occurrence of metal wear and organic wear nanoparticles claimed attention also in cemented prostheses. Both septic and aseptic environments did not influence the production of nanometric wear particles, as their quantity and typology was rather linked to prosthesis composition.
In the our previous work (12), performed on the same membranes, we observed by immunohistochemical analysis a large number of histiocytes in aseptic membranes. Histiocytes (also known as foreign body giant cells) are cells occurring as fusions of macrophage cells in response to the presence of undigested particles. The large presence of this cell type is difficult to explain as a consequence of the observed micrometric wear particles, while it can be explained by the presence of submicrometric and nanometric particles. However, in septic membranes, only a small number of histiocytes was observed, while a large number of macrophages were detected.
It has been reported that the production of inflammatory factors (such as TNF and IL-10) following bacterial invasion, exerts some protective effect on wear debris-induced osteolysis (2). These finding may be related to the high level of macrophage occurrence in septic membranes.
The occurrence of ions released in tissue is often associated with serious toxicity: the local and systemic toxicity induced by Cr and Co ions was assumed (24, 25). Ions may induce both toxicity and activation of inflammatory pathways by bone cells, macrophage and perhaps membrane fibroblast cells, contributing to a faster failure of any mechanism of repair (26). In fact in the analyzed membranes, we have already observed apoptotic and necrotic cell death, and hypothesized the occurrence of a toxic local environment (12). The ion detection analysis widely confirmed our hypothesis.
An interesting finding of our analysis was the detection of contaminant ions such as Fe, Al and Zn ions, derived from catalytic processes. They represent chemical reactive species which favor the formation of the observed ion aggregates, and they may contribute to increasing the toxicity level of oxidative stress. These ions are also directly involved in tissue damage: Fe ions are well known to mediate a series of chemical reactions that may induce oxidation or form different reactive radical elements; moreover, recent findings revealed an involvement of Zn ions in complex cell toxicity pathways (27).
Conclusions
Arthroplastic substitution creates a reactive complex environment in which numerous and different debris and ion pollutant are generated.
The submicrometric and nanometric wear debris represent an underestimated debris population that could cause high levels of cellular reactivity and consequent periprosthetic bone osteolysis.
In the present study, no significant differences in the distribution of particulate between aseptic and septic membrane were observed. On the other hand, a peculiar response of macrophages in the septic environment was shown (2). From this point of view, it could be of interest to investigate the response of macrophages and other cells present in the periprosthetic environment, interacting with both nanometric particles and sepsis. Furthermore, the findings regarding hydroxyapatite particles suggest the need to evaluate the occurrence of friction forces, which by producing periprosthetic bone debris may induce exacerbated cellular reactivity. Moreover the high level of ion pollutants led us to hypothesize the occurrence of a highly toxic periprosthetic environment that could alter tissue mechanisms of repair.
