Abstract
Solution thermal method was used to synthesize CuInSe2 powders from metal chlorides and SeO2 at 160∼200°C. The phases of product samples were analyzed by x-ray diffraction (XRD), the size and morphology of the products were observed by JSM6380LA scanning electron microscope (SEM). The aim is to explore the best technology to obtain the pure CuInSe2 powders. Experimental results show that, CuInSe2 phase can be formed with metal chlorides by reacting for 20 h at 160°C, 180°C and 200°C and has XRD peaks corresponding to (112), (204) and (312) crystal planes, respectively. It consists of irregular spherical grains with about 0.2∼0.5 μm in diameter, which agglomerate larger particles with average particle size about 1∼3 μm. CuInSe2 phase was formed more easily at the temperature higher than 180°C and shows better crystallinity.
Introduction
The CuInSe2 materials with suitable band gap and excellent absorption coefficient and reliable stability have become one of the most interesting issues in photovoltaic materials research (1). The conventional methods for preparing CuInSe2 mostly require vacuum environment, which leads to complex processes in preparation and equipment with high cost. As the result, it is hard to produce CuInSe2 materials on a large scale (2). It is also a hot spot for preparing absorbing layer CuInSe2 film from CuInSe2 powder in a certain way (3-4-5). Preparation methods for CuInSe2 powders are mainly divided into solid phase synthesis methods such as mechanical grinding and thermal sintering, and liquid phase synthesis methods such as hydrothermal method or solvent thermal method (6, 7), thermal injection method, microwave assisted synthesis method, etc. (8-9-10). In this work CuInSe2 powders were prepared by solution thermal coreduction method under the conditions of different reaction temperatures, reaction time and raw materials. The effects of different conditions on the phase, crystallinity, size and morphology of CuInSe2 powders were investigated. Cu2ZnSnSe4 and ZnSe were successfully synthesized by our group, which provided a good reference for this work (11, 12).
Experimental details
For preparing CuInSe2 product powder, CuCl2·2H2O, InCl3 and SeO2 with a stoichiometric ratio of 1:1:2 were added into a stainless steel autoclave with a teflon liner of 20 mL capacity. The autoclave was filled with absolute ethyl alcohol up to 80% of the total volume. After ultrasonic agitation for about 30 min, 2 mL hydrazine hydrate (N2H4·H2O) was poured into the reactants. The autoclave was sealed and heated at different temperatures (140∼200°C) for 10∼48 h in an electric furnace. After heating, it was cooled down to room temperature naturally. The black products were collected by filtration, washed with deionized water and absolute ethanol, and then dried at 70°C.
The phases of product powder samples were analyzed by x-ray diffraction (XRD) on a model of Bruker D8 Advance XRD system with Ni-filtered Cu-Kα (λ = 1.5059 Å). The composition, sizes and morphology of the products were characterized by JSM6380LA scanning electron microscope (SEM).
Results and discussion
The effects of different reaction temperatures on CuInSe2 films
Figure 1 shows the XRD patterns of product powders prepared by reacting for 20 h at 160°C, 180°C and 200°C. According to the standard PDF card No.65-2740, it can be known that CuInSe2 phase can be formed with XRD peaks corresponding to (112), (204) and (312) crystal planes, respectively. When the reacting time is 20 h, impurity phases of Cu2Se and In(OH)3 appeared in the products at 160°C and 180°C, respectively. SEM image and EDS results of product powders prepared by reacting for 20 h at 160°C are shown in Figure 2. It indicates that the product powder consists of irregular spherical grains with about 0.2 μm in diameter, which agglomerate larger particles with average particle size about 1∼3 μm. The EDS results can prove that the product powder contains three elements of Cu, In, and Se which may be reduced from raw materials in experiments.
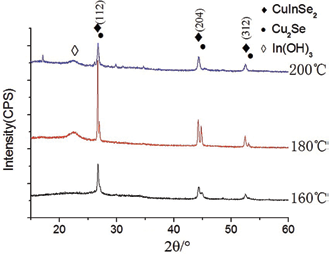
XRD patterns of product powders prepared by reacting for 20 h.
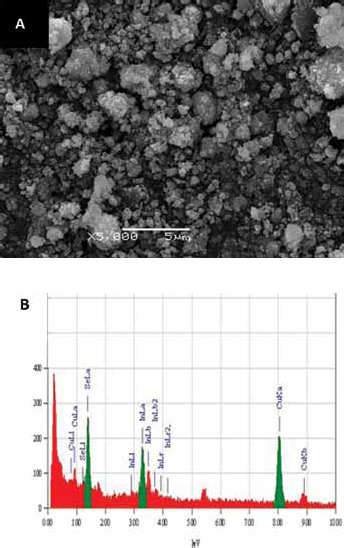
SEM and EDS results of product powders prepared by reacting for 20 h at 160°C
The CuInSe2 films prepared with longer reaction time
Figure 3 shows XRD patterns of product powders prepared by reacting for 48 h at 140°C, 160°C, 180°C and 200°C. It can be seen that, when the reaction temperatures are 180°C and 200°C, the phase of the product powders obtained was CuInSe2 without obvious impurities. Its strong XRD peaks also correspond to (112), (204) and (312) crystal planes, repectively. When the temperature is below 160°C, the XRD peaks intensity for CuInSe2 become very low, the impurities such as CuSe and Se began to appear in the product powders obtained at 140°C and 160°C. So it can be considered that CuInSe2 phase was formed more easily at the temperature higher than 180°C.
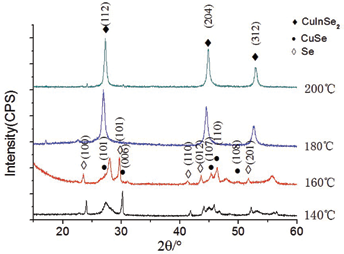
XRD patterns of product powders prepared by reacting for 48 h.
Comparing Figure 1 with Figure 3, it can be seen that there was impurity phase CuSe in the product powder reacted for 20 h, while the sample obtained with longer time 48 h shows better crystallinity with less impurity phase.
Figure 4 shows the SEM images and EDS results of product powders prepared by reacting for 48 h 180°C and 200°C. Figure 4a shows the sample of 180°C has very serious agglomeration phenomenon and larger particle size about 1.5∼2.0 μm. However, Figure 4b indicates that the product sample obtained at 200°C with good dispersion and is composed of spherical particles with diameters about 0.3∼0.5 μm. The EDS results proved that for the product powders prepared by reacting for 48 h contains Cu, In, and Se elements which can be reduced in experiments.
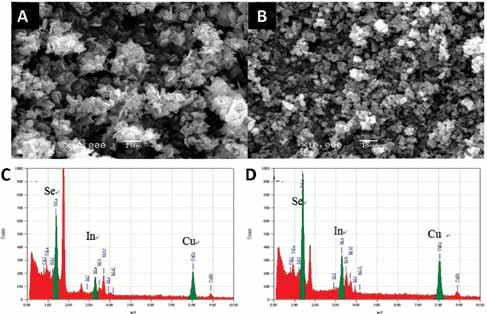
SEM images of product powders prepared by reacting for 48 h. SEM images: (a) 180°C +48 h, (b) 200°C +48 h; EDS results: (C) 180°C +48 h, (d) 200°C +48 h.
Discussion for the reaction mechanism
The reaction mechanism is proposed as follows: When the CuCl2·2H2O, InCl3, SeO2 and the reducing agent are put into the autoclave and heated, the reactive Cu, In, and Se atoms are easily reduced from raw materials by reducing agent N2H4·H2O, and can easily combine to be CuInSe2 molecules. The possible reacting processes are as follows:
CuCl2·2H2O + N2H4·H2O → Cu + 2HCl + N2↑ + 3H2O + H2↑
SeO2 + H2O → H2SeO3
H2SeO3 + N2H4·H2O → Se + N2↑ + 4H2O
2InCl3·4H2O + 2N2H4·H2O → 2In + 6HCl + 2N2↑ + 10H2O + H2↑
Cu + 2Se + In → CuInSe2
Conclusions
The CuInSe2 powders were synthesized by solution thermal coreduction method from metal chlorides and SeO2 with hydrazine hydrate as reducing agent at 160∼200°C. CuInSe2 phase can be formed with metal chlorides by reacting for 20 h at 160°C, 180°C and 200°C. The XRD peaks correspond to (112), (204) and (312) crystal planes, respectively. The product powders consist of irregular spherical grains with about 0.2∼0.5 μm in diameter, which agglomerate larger particles with average particle size about 1∼3 μm. CuInSe2 phase was formed more easily at the temperature higher than 180°C and shows better crystallinity with less impurity phase.
Footnotes
Financial support: This work was financially supported by the National Natural Science Foundation of China (No.51272140).
Conflict of interest: None of the authors has financial interest related to this study to disclose.
