Abstract
Purpose
The aim of this study was to evaluate the cytotoxic effects of 6 different orthodontic bracket types on human gingival fibroblasts (HGFs) using the xCELLigence system.
Methods
The orthodontic brackets used in this study were gold-plated steel (Apollo Gold), titanium (Rematitan), stainless steel (Equilibrium 2), lucid ice (Inspire ICE), metal-reinforced ceramic (Clarity) and composite (OrthoFlex). Gingival fibroblasts were isolated from human gingival connective tissue of systemically healthy individuals. Tested brackets were incubated in DMEM culture medium for 72 hours according to ISO 10993-5 standards. Gingival fibroblasts were maintained with Dulbecco modified Eagle medium containing 10% fetal bovine serum. The xCELLigence system was used to evaluate cell survival. The statistical analysis used was ANOVA and Tukey-Kramer multiple comparison tests.
Results
When the data were evaluated in the 30th hour, Apollo Gold showed significant decreases in cell index (P<0.001). It also showed statistically significant decreases (P<0.001) in the 65th hour, but Clarity and Inspire ICE showed significant increases in cell indices (P<0.001, P<0.01). In the 114th hour, Clarity and Equilibrium 2 showed statistically significant increases in cell indices (P<0.001). Inspire ICE and Rematitan demonstrated significant increases (P<0.05). There were significant decreases in cell index of Apollo Gold (P<0.001).
Conclusions
The tested brackets are suitable for clinical application, but further studies using different test methods are needed for gold-plated brackets.
Introduction
In a standard orthodontic treatment, every orthodontist uses bands, brackets and arch wires. They are produced from alloys containing nickel, cobalt and chromium. The different types of orthodontic appliances contain 15%-54% nickel, 20%-30% chromium and 40%-60% cobalt (1). Since they can be in the mouth for 2 years or more, biodegradation of metals occurs due to varying oral temperatures, microbiologic and enzymatic properties. The appliances used are exposed to corrosion in the oral cavity because they are immersed in the patient's saliva, resulting from several aggressive physical-chemical events, such as high concentrations of oxygen and chloride (2). Additional factors influencing corrosion are the presence of plaque and the daily dietary intake (3). Therefore, data on the biocompatibility of these alloys are of great importance.
Stainless steel brackets can easily be formed by casting and milling. The metal injection molding (MIM) technique was introduced as a new method of production in recent years (4). Some other brackets are built by fiber-reinforced composite or polycarbonate (5). Plastic brackets have been used for more than 30 years in an attempt to ensure esthetics (6). Over the last several decades, manufacturers have put miniaturized brackets and arch wires on the market, and developed tooth-colored ceramic brackets, followed by metal-lined ceramic brackets. These metal-lined ceramic brackets have been produced to bring esthetics into equality with function, as stainless steel has replaced gold materials (6). Regarding patients with an allergy to nickel and other specific substances, gold and titanium brackets can be an alternative. Nevertheless, the tendency of these metallic brackets to corrode, with consequent release of metal ions, is well described (7).
There are 2 major aspects of corrosion: First of all, corrosive processes cause the ruin of the surface by a loss of metal ions. Secondly, adverse biologic effects of the ion release are the major problem in the oral cavity. The recent literature shows great awareness regarding attributes of dental alloys such as cytotoxicity, carcinogenicity, mutagenicity and allergenicity (3, 8, 9). Biocompatibility is strictly connected to ionic release, and therefore the public may express concern about possible leakage of metal ions from an orthodontic appliance. The stainless steel currently used in orthodontic clinics is of type 302 or 304, both of which contain 8%-10% nickel (10). Nickel increases the strength, ductility and resistance to general, crevice and erosion corrosion. The most remarkable corrosion products are iron, chromium and especially nickel from stainless steel, and titanium and nickel from nickel-titanium alloy (10).
Several studies examining the concentration of metal ions in oral mucosa cells, and cellular viability and apoptosis, have been conducted in recent years (6, 8, 10-12). The xCELLigence system was newly introduced, following the predecessor impedance-based Real-Time Cell Electronic Sensing (RT-CES®) system. Urcan et al (13) used the xCELLigence impedance analysis system to investigate cytotoxicity of the most common monomers/comonomers in dental resin composites on human gingival fibroblasts (HGFs).
Previous studies have investigated the release of these metals from orthodontic appliances both in vitro (1) and in vivo (14), but less data are available about adverse biologic effects of these metals. Furthermore, these are not sufficient to assess the biocompatibility of orthodontic brackets that are in the mouth for several years. The purpose of this study was to evaluate the cytotoxic effects on HGFs of 6 different orthodontic bracket types, using the xCELLigence system.
Materials and Methods
The orthodontic brackets used in this study were gold-plated steel (Apollo Gold brackets; G&H Wire Company, Hanover, Germany), titanium (Rematitan brackets; Dentaurum, Ispringen, Germany), stainless steel (Equilibrium 2; Dentaurum, Ispringen, Germany), lucid ice (Inspire ICE; Ormco, Glendora, CA, USA), metal-reinforced ceramic (Clarity; 3M Unitek, Monrovia, CA, USA) and composite (OrthoFlex; Ortho Technology, The Hague, The Netherlands) (Tab. I).
ORTHODONTIC BRACKETS USED IN THIS STUDY
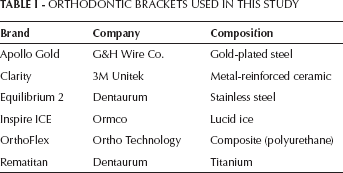
Two sets of orthodontic brackets (5-5 upper and lower) were used for each group for cytotoxicity testing. The samples were immersed in 7 mL of culture medium for 24 hours at 37oC to extract residual monomer or cytotoxic substances. The culture medium–containing material extracts were sterile filtered for use on the cell cultures. The test procedures were regulated by ISO-standard 10993-5 (15).
Cell culture
Gingival fibroblasts were isolated from human gingival connective tissue of systemically healthy individuals. The HGFs were obtained from samples of gingiva removed for crown-lengthening purposes. All patients gave informed consent before providing the samples. The procedures used in the present study were in accordance with the guidelines of the Helsinki Declaration on human experimentation. This protocol was approved by the Ethics Committee of the Selcuk University, Faculty of Dentistry. Briefly, gingival tissues were cut into small pieces, rinsed with biopsy media, placed in tissue culture dishes and incubated in biopsy medium in a humidified atmosphere of 95% air and 5% CO2 at 37oC overnight. The following day, biopsy medium was replaced with culture medium (Dulbecco's modified Eagle's medium [DMEM] with 10% fetal bovine serum [FBS], 100 units/mL penicillin and 100 μg/mL streptomycin). After reaching confluence, cells were passaged with 0.25% trypsin and 0.1% ethylenediaminetetraacetic acid (EDTA). HGFs between the fourth and sixth passages were used for all experiments.
Preparation of materials
Brackets were incubated in DMEM culture medium for 72 hours according to ISO 10993-5 standards (surface area to volume ratio of the specimen to cell culture medium; 3 cm2/mL). Gingival fibroblasts were maintained with DMEM containing 10% FBS. The xCELLigence system (Roche Applied Science and ACEA Biosciences) was used to evaluate cell survival, according to the instructions of the supplier. After seeding 200 μL of the cell suspensions into the wells (20,000 cells/well) of the E-Plate 96, gingival fibroblasts were treated with bioactive components released by tested brackets (1/1 and 1/2 dilutions) and were monitored every 15 minutes during a 117-hour period.
Morphology of gingival fibroblasts
After treatment with bioactive components of brackets, morphology of gingival fibroblasts was evaluated using inverted microscopy (TS100 Nikon Eclipse; Nikon, Tokyo, Japan) (magnification ×10).
Statistical analysis
All calculations were performed using the RTCA-integrated software of the xCELLigence system. The RTCA software performs a curve-fitting of selected “sigmoidal dose–response equation” to the experimental data points. Data are represented as means (mmol/L)±SEM (n=5).
For proliferation experiments and gene expressions, the statistical analysis used was 1-way analysis of variance (ANOVA), followed by Tukey-Kramer multiple comparison test. A value of P<0.05 was considered to be statistically significant.
Results
Monitoring of cytotoxicity in real time using xCELLigence system
The optimal concentration for cell proliferation and viability measurements was first determined. Then, 40,000, 20,000, 10,000 and 5,000 cells/well were seeded in the E-Plate 96, and the impedance was determined (Fig. 1).
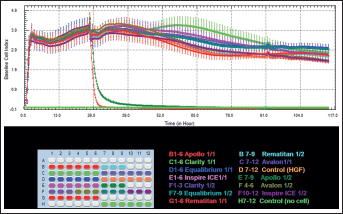
Dynamic monitoring of cell adhesion and proliferation. After seeding 200 μL of the cell suspensions into the wells (20,000 cells/well) of the E-Plate 96, gingival fibroblasts were treated with bioactive components released by the tested brackets (1/1 and 1/2 dilutions) and were monitored every 15 minutes during a 117-hour period.
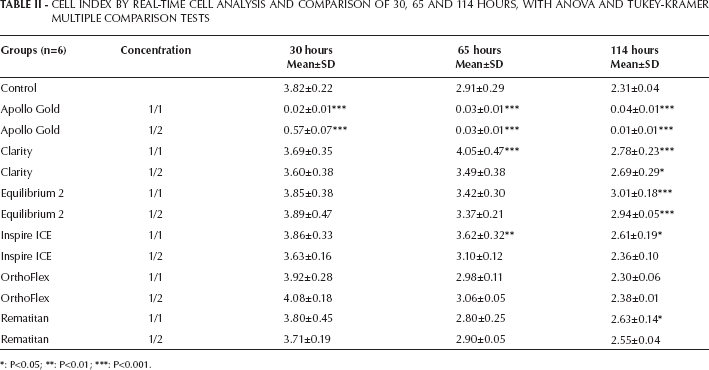
According to ANOVA results, when the E-Plates were left to elute for 30, 65 and 114 hours, there were significant differences between the cell indices of the control and study groups for 1/1 and 1/2 dilutions (Tab. II and Fig. 1).
CELL INDEX BY REAL-TIME CELL ANALYSIS AND COMPARISON OF 30, 65 AND 114 HOURS, WITH ANOVA AND TUKEY-KRAMER MULTIPLE COMPARISON TESTS
P<0.05
P<0.01
P<0.001.
When the data were evaluated in the 30th hour, 1/1 and 1/2 dilutions of Apollo Gold brackets (mean 0.02±0.01 and 0.57±0.07, respectively) showed statistically significant decreases in cell index (P<0.001) compared with that of the control group. However, there were no significant differences in other study groups (P>0.05).
Apollo Gold brackets (mean 0.03±0.01) also showed statistically significant decreases (P<0.001) in both 1/1 and 1/2 dilutions in the 65th hour, but Clarity (mean 4.05 0.47) and Inspire ICE (mean 3.62±0.32) showed significant increases in cell indices in 1/1 dilutions (P<0.001; P<0.01, respectively).
In the 114th hour, Clarity (mean 2.78±0.23 and 2.69±0.29, respectively) and Equilibrium 2 (mean 0.03± 0.01 and 2.94±0.05, respectively) showed statistically significant increases in cell indices (P<0.001) in both 1/1 and 1/2 dilutions. In addition, Inspire ICE (mean 2.61±0.19) and Rematitan (mean 2.63±0.14) demonstrated significant increases (P<0.05) in 1/1 dilution. Again, there were statistically significant decreases in cell indices of Apollo Gold brackets (mean 0.04±0.01 and 0.01±0.01, respectively) in both 1/1 and 1/2 dilutions in this period (P<0.001).
Morphologic assessment
Figures 2 and 3 show the normal, altered and dead cells of the cell cultures containing the materials tested.
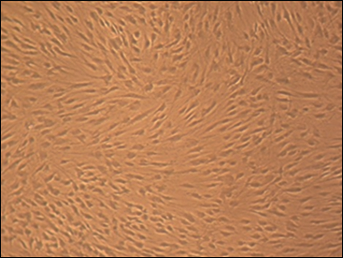
Cultured human gingival fibroblast (HGF) cells for control group (magnification × 10).
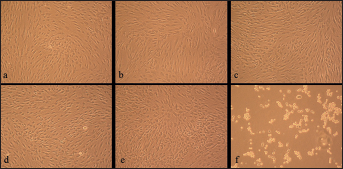
Cultured human gingival fibroblast (HGF) cells for
We observed regular, dense HGF cells and numerous mitoses in the control culture. They were elongated and spindle-shaped cells (Fig. 2). In comparison with the control culture, the HGF culture with Clarity and Equilibrium 2 was less dense. We determined normal cells and only a few rounded or dead fibroblasts. Their spindle shape was not deformed (Fig. 3a, b).
In the Rematitan, OrthoFlex and Inspire ICE cell cultures, there were retraction fibers and rounded cells. This appearance suggested the process of degeneration and cell death (Fig. 3c-e).
However, all HGF cells exposed to media containing Apollo Gold brackets were significantly retracted and rounded; many of them were floating. These images gave an impression of cells dying (Fig. 3f).
Discussion
Brackets used in orthodontic treatment provide the means to transfer force to the teeth using the arch wire. Biodegradation of the orthodontic brackets can occur in the warm and moist conditions of the mouth. So, this is an ideal medium for releasing metals ions that may induce adverse effects in oral mucosa and gingiva or alveolar bone.
Using cell cultures for testing of dental materials has some advantages over controversial animal experiments, because it is relatively easy to perform, reproducible and cost effective, and can be controlled accurately (3, 8, 16-18). In the human physiological situation, oral fibroblasts are highly exposed to exogenous substances in the oral cavity (19). Therefore, the aim of this study was to investigate the dynamic monitoring of cell proliferation of HGFs during the 30th, 65th and 114th hours.
Some researchers have conducted thermal and chemical analyses to evaluate decomposing of the orthodontic products just like in oral conditions (6). Amini et al (10) used atomic absorption spectrophotometry with a graphite furnace to determine the metal content of oral mucosa cells in patients with and without fixed orthodontic appliances. In some studies (8, 12), corrosive processes were measured by inductively coupled mass spectrometry (ICP-MS). In the present study, we conducted our experiments through a relatively new and different method, the real-time xCELLigence impedance analysis system, to investigate the cytotoxicity of the 6 orthodontic bracket types, and continuously monitor the cell growth, proliferation and viability. What is more, this saves expensive reagents used in conventional cell analysis. In this system, the kinetic control of cellular status during the entire experimental runs reveals continuous information about cell growth, morphologic changes and cell death. The real-time cell analysis system allows for the calculation of time-dependent physiological values, which can be more informative than the MTT-based viability tests (3, 16, 17, 20, 21).
A unit-less parameter termed the cell index is derived to represent cell status based on the measured relative change in electrical impedance that occurs in the presence and absence of cells in the wells (22). All of the factors that increase the number of attached cells on the electrodes – e.g., attachment from solution or cell proliferation – lead to a higher cell index value. However, cell death or toxicity induces cell detachment leading to a decreased cell index value (13).
We observed a lack of continuity of cell index at hour 26 and a little peak at hour 91. Despite the same cell numbers, dimensional alterations of the attached cells on the electrodes will cause a change in the cell index. Since the E-Plate was removed from the incubator during the experiment, plate temperature decreased to room temperature and the shape of the cells could have been affected by their sensitivity to temperature fluctuations. In our previous studies, similar trends in cell index in the untreated cells were detected. This was an expected result for this experiment as well (23-25).
Apollo Gold brackets have no cell proliferation at any time points. These results suggested that the gold-plating procedure could not prevent the cytotoxic or allergic effects to the adjacent tissues, and gold-plated steel brackets remarkably decreased the cell proliferation of HGFs. This could have resulted from severe corrosion. We can speculate that these corrosive effects rely on the manufacturing process, such as surface finishing. Contrary to the results in our study, Mockers et al (20) declared that metallic brackets including stainless steel, titanium and gold-plated steel were similar in terms of cytotoxicity, to the polycarbonate and ceramic-based materials. Schuster et al (26) evaluated allergic potentials of orthodontic alloys. They concluded that to minimize the possibility of allergic contact reactions, corrosion-resistant materials should be used as a matter of principle. For this purpose, welding is preferable to soldering (27), and the recycling of the brackets and the bands is obsolete in all events. Gold plating or other coatings (titanium nitride) of brackets, bands and arch wires should be avoided in general since they stimulate the development of corrosion following a brief period of protection (26).
Rematitan brackets demonstrated increased cell indices, especially in 1/1 dilution and at 114th hours in comparison with the control HGFs. We concluded that Rematitan did not inhibit cell proliferation in this setting. Titanium brackets consist of titanium or a titanium alloy (Ti-6Al-4V), and the Ti-6Al-4V alloy with a friction coefficient of 0.28 might have different frictional variants from the commercially pure titanium with a coefficient of 0.34; whereas, from a corrosion perspective, brackets formed from 2 components might be more susceptible to galvanic corrosion (28). Harzer et al (29) tested how the surfaces of titanium brackets react to the corrosive influence of acidic fluoride-containing toothpaste during orthodontic treatment. Following their research by light microscopy and scanning microscopy 7.5 to 17 months after bonding, they concluded that plaque accumulation on titanium brackets was high because of the very rough surface. The researchers suggested that since the changes were so minor, titanium brackets could safely be used for up to 18 months. In agreement with this study, Fernández-Miñano et al (30) stated that titanium brackets and tubes were biocompatible to the oral mucosa cells. These considerations are in accordance with our finding of the low cytotoxic effect of Rematitan.
Equilibrium 2 showed statistically significant cell proliferations in both 1/1 and 1/2 dilutions in only 114th hours compared with control HGFs. According to our study results, HGF cell proliferation increased relative to time. This result means Equilibrium 2 has no cytotoxic potential. Nickel is a strong immunologic sensitizer, although nickel sensitivity has been reported to be lower in patients who have received orthodontic treatment (31); perhaps they develop immunologic tolerance over the long period of treatment (32). According to recent studies, due to the genotoxic effects of nickel, care should be taken to minimize potential exposure to this element and its compounds (11).
However, nonnickel and low-nickel stainless steels were introduced in orthodontics as alternatives to conventional 316 and 318 stainless steel types. These steels contain substantially less nickel relative to conventional types and the same or even higher hardness relative to the types of steel used for bracket manufacturing (27). Sfondrini et al (33) investigated 3 kinds of orthodontic brackets: new conventional stainless steel, recycled stainless steel and nickel-free brackets. They indicated that while reconditioned brackets released the most nickel, the nickel-free brackets released the least 0.03±0.06 μg per gram in their in vitro study using an atomic absorption spectrophotometer and an inductively coupled plasma atomic emission spectrometer. Moreover, the highest nickel release was recorded in the 2 experiments performed at pH 4.2; it was lower at pH 6.5 and 7.6.
Most studies in the literature used static conditions, but more metal release could be observed in clinical conditions because of the composition and fluidity of the saliva in the mouth, and also because oxide layers are removed by tooth brushing (29). Moreover, in the clinical setting, brackets are mechanically activated to enable movement of the teeth. Thus, movements of wires, friction in the brackets and deformation of brackets might result in various types of corrosion (34). Heat treatments of the alloys under laboratory conditions have been shown to markedly increase the release of metal ions by 15-60 times (35). Simulated function conditions (dynamic) compared with static conditions (36), and some mouth washes (37) have been shown to increase the release of nickel ions from nickel alloys (10). These conditions would cause more cytotoxic effects to the adjacent tissues.
Inspire ICE, a monocrystalline bracket, indicated significant cell proliferation in 1/1 dilution at the 65th and 114th hours. There was a significant increase in cell index of HGFs in comparison with the control group. This condition may have resulted from the test sensitivity. Clarity, a polycrystalline bracket, showed a significant sharp increase of cell proliferation in 1/1 dilution at 65th hours, and it caused a marked increase of cell index in 1/1 dilution at 114th hours. These results indicated a statistically significant increase of HGFs compared with the control group.
Although ceramic brackets are more attractive than their metallic counterparts, they have several undesirable effects, such as higher incidences of bracket fracture attributed to the lack of grain boundaries for the inhibition of crack growth, excessive wear and failure to deliver sufficient torque because of their low modulus (38). Currently, ceramic brackets have superior mechanical properties, increased transparency, decreased reactivity with the oral environment, and an inert biologic character (27). Consequently, Clarity and Inspire ICE could be regarded as biocompatible in the oral environment according to our results.
Clinically, every orthodontist should focus on using minimal cytotoxic materials to reduce the adverse effects. Schuster et al (26) recommended that based on careful attention to patients' case histories, the orthodontist should use ceramic, plastic or titanium brackets for fixed appliance therapy in patients known to have a nickel allergy. However, it is a matter of dispute whether plastic brackets should be used, because of the potential action of various polymers at subtoxic levels (27). Pereira et al (39) examined the effects of 2 metal and 2 ceramic brackets on the epithelial cells of the buccal mucosa in 21 individuals of both sexes. With the use of liquid-based exfoliative cytology, morphometric and morphologic changes in buccal mucosa cells adjacent to these brackets were determined. They concluded that buccal mucosa cells adjacent to the metal brackets showed greater changes than those adjacent to the ceramic brackets, and buccal mucosa cells adjacent to the metal and ceramic brackets had a tendency to return to their initial morphology after removal of the brackets. We also assessed morphologic changes of HGFs after exposure to tested brackets. In contrast to their results, we detected nearly the same morphologic alterations between Clarity and Equilibrium 2 in our in vitro study.
Thus, the results of initial cytotoxicity tests cannot be immediately extrapolated to clinical conditions. They will explain only some biologic behavior of dental materials and their components (17). Cytotoxicity testing allows a comparison among available products and information for choosing a material with optimal characteristics. However, the results of the present study remain part of an open question, and further studies using different test methods are needed.
Conclusions
Within the evaluated experimental period, the following conclusions could be drawn:
Apollo Cold brackets remarkably decreased the cell proliferation of HGFs.
Rematitan mildly increased cell proliferations in the last time interval.
Equilibrium 2 has no cytotoxic potential.
Clarity and Inspire ICE could be regarded as biocompatible in the oral environment.
OrthoFlex did not show cytotoxic effects on HCFs.
The tested brackets are suitable for clinical application, but further studies using different test methods are needed for gold-plated brackets.
Footnotes
Acknowledgements
The authors thank Uysal Medical, Benlioğlu Dental, Medifarm and Medikodental Company for providing the brackets tested in this study.
