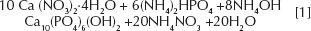Abstract
Purpose
We investigated the effects of alumina addition on microstructure and compressive strength of a porous silicate substituted hydroxyapatite (Si-HA).
Methods
Hydroxyapatite (HA) was synthesized under precipitation conditions and 10 %Wt. of sol–gel derived CaO. P2O5.SiO2 based bioglass (BG) powder was added to HA. Polyurethane foam was used to form a high porous structure with integral porosity of 70%. Phase analysis was performed using XRD and FTIR and the microstructure was studied using SEM.
Results
The results confirmed that the Si-HA was the only formed phase before Al2O3 addition while after addition the presence of silicon-incorporated HA and alumina without any other phases was proved using these analyses.
Conclusions
The porous structures of Si-HA and Al2O3 were synthesized using the replication technique. The compressive strength of porous bioceramics increased with increasing Al2O3 content up to 30 wt % (ANOVA, p<.05).
Introduction
Many porous bioceramics have been prepared for tissue engineering. Porous calcium phosphates such as hydroxyapatite (HA) and tricalcium phosphate are currently applied as scaffolds for bone tissue engineering and implants for the regeneration of damaged and diseased tissues (1-4). Concerning the fabrication of porous ceramic bodies, several techniques, such as the use of polymeric sponge (4) and organic additives (5–7) and moulding processes (8), have been investigated. For example, Li et al (9) combined a foaming method and a dual-phase mixing method (involving HA slurry and polymethyl methacrylate (PMMA) resin) to prepare macroporous HA. Among a variety of processes for fabrication of porous materials, the replication technique, also called the polymer sponge method, produces porous ceramic structures that are most similar to those of spongy bone (10, 11). Porous HA has received increasing attention as a bone substitute material to promote the ability to bond chemically with living bone tissues owing to its similar chemical composition and crystal structure as apatite in the human skeletal system (12). Recently, bioactive glass (BG) materials, with a better biological response than the calcium phosphate based materials, have been investigated (13, 14). The presence of silica in many biocompatible and bioactive materials has generally been shown to improve the bioactivity (11, 15). Studies show that the created or already present silanol groups provide good nucleation sites for HA (11). The silicon substitutions induce complex structures at the unit-cell level and play a role in influencing the dissolution rate and bioactivity of bone mineral (16). Porous silicate substituted hydroxyapatites Ca10(PO4)6-x(SiO4)x(OH)2-x 0>x>2 are highly biocompatible and osteoconductive, but their mechanical properties such as fracture toughness and compressive strength are very poor. Previous attempts have been made to improve the strength of bioceramics by incorporation of Al2O3 as a second phase (17, 18). Alumina has been widely used for orthopedic and dental applications because of its superior load bearing capability and biostability (17, 18). Thus, it can be considered a suitable reinforcement materials for bioceramics. The combination of Al2O3 is considered for producing composite samples because of the fact that this composite can introduce some interesting features like microcracks at grain boundary, residual stress development, etc., which can modify the behavior of mechanical properties of porous bioceramics. In this study, the effect of alumina addition on properties of porous Si-HA has been investigated.
Materials and Methods
Materials
In this investigation analytical grade of Ca (NO3)2·4H2O (Merck, Germany), (NH4)2HPO4 (Merck, Germany), alumina (Merck, Germany), and tetraethoxysilane (TEOS; Si(OC2H5)4, Merck, Germany), tetraethoxysilane triethylphosphate (TEP; OP(OC2H5)3, Merck, Germany) were used as starting materials. Polyurethane foam (45ppi) was purchased from Safoam - Iran.
Synthesis and preparation of samples
HA powder was prepared according to the following reaction:
The 0.29M (NH4)2HPO4 aqueous solution was added drop-wise to the 0.24M Ca (NO3)2 solution under precipitation conditions (19). The precipitate was separated using the centrifugation method at the rotation speed of 3000 rpm. The resulting powder was dried at 80°C for 24 h and then calcined at 900°C for 1 h. CaO.P2O5.SiO2 based BG was prepared by the sol–gel method with the ratio of SiO2: CaO: P2O5 = 60: 36: 4 (in mol %). The sol–gel derived bioactive glass was prepared by means of the hydrolysis and condensation of a mixed solution of tetraethoxysilane (TEOS), triethylphosphate (TEP), and calcium nitrate tetra-hydrate (Ca(NO3)2·4H2O). HCl solution was used as a catalyst for the hydrolysis and condensation reactions. The formed sol was then sealed in a beaker and aged in an oven at 70°C for two days. The dried gel after crushing was further calcined at 700°C for 1 h, followed by ball milling to obtain the BG powder (5). The composition of powder mixture is indicated in Table I.
COMPOSITION OF POWDERS (WT%) AND THEORETICAL DENSITY AND POROSITY OF EACH SAMPLES

The porous samples were fabricated by polyurethane foam and ceramic slurry (10). The polyurethane foam template was replicated with the slurry by a repeated dipping and drying process. After drying in air, the coated substrates were heated up to 1200°C in a schedule which minimized disruption during pyrolysis and enabled the bioceramic to attain high density.
Results
The phase composition of the synthesis starting materials and sintered samples was analyzed using a Siemens D5000 X-ray diffractometer, with Cu K radiation at 30 mA, 40 kV. The XRD patterns of samples with various content of Al2O3 and HA are presented in Figure 1.
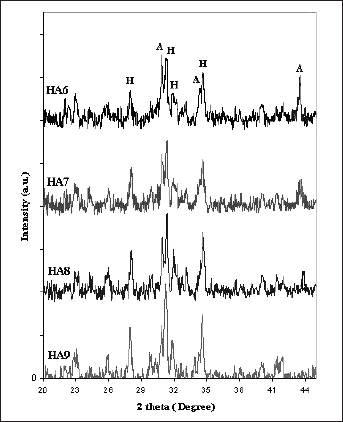
X-ray diffraction patterns of sintered samples with various Al2O3. H: HA, A: Al2O3.
The calculated theoretical densities (dt) of each compound used are illustrated in Table I. The theoretical density (dt) was determined using the following equation:
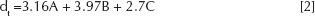
where A, B, and C are the percentage weight rates and 3.16, 3.97, 2.7 g/cm3 are the theoretical densities of HA, Al2O3, and BG respectively. The density of the porous samples (dp) was determined from the mass and dimensions of the sintered bodies.
The porosity (P) was then calculated by:

The microstructure of the porous samples was observed by the Stereoscan 360-Leica Cambridge scanning electron microscope (SEM). The morphology of the porous samples can be seen in Figure 2.
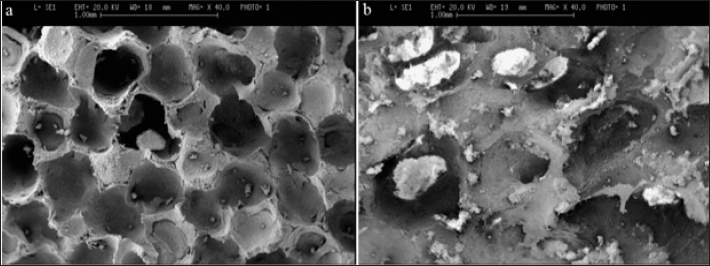
SEM micrographics of (a):HA9 and (b): HA6 composites sintered for 3 h at 1200 °C.
The compressive strength was measured using a Zwick/Roell 2005 at a loading rate of 0.01 mm/s, and six specimens were fractured at each test condition (n=6). The compressive strength and modulus of elasticity were determined from mechanical test recordings. A one-way analysis of variance (ANOVA) was used to compare the means of the different groups, and statistical significance was accepted at the .05 confidence level. The compressive strengths of the porous samples are shown in Figure 3.
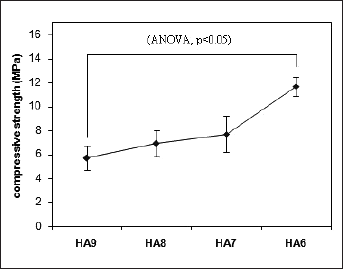
Compressive strength of porous samples with various percentage of Al2O3.
Discussion
Phase and composition
According to XRD patterns adding 10%wt of BG in HA9 resulted in no secondary phases which were formed for the HA. Both Ca and P ions in HA structure can be incorporated with various ions. Because of their ionic sizes, it is generally accepted that silicon substitutes P sites and exists as silicate ion. The substitution of silicon of the BG into the HA lattice did not affect the phase composition, therefore no changes took place in the symmetry of the HA crystallographic unit cell. XRD analyses of HA and HA9 strongly support the incorporation of Si into HA lattice, with the charge balance maintained through the loss of hydroxyl ions, as per the following equation Si-HA: (Ca2+)10(PO43–)6-x(SiO44–) x(OH–)2–x. Si-substituted calcium phosphates have shown an increase in biological activity (11). Si substitution in calcium phosphates facilitates the precipitation of a biologically equivalent HA on the implant surface, as well as directing the effects of Si released to the extracellular media or presenting at the material surface. Si promotes biomimetic precipitation by increasing the solubility of the material by means of the creation of crystalline defects with substitution of PO43- and associated charge compensation mechanism, by generating a more electronegative surface with the exchange of SiO44- for PO43- and by creating a nanocrystalline material (20).
Morphology
As bone substituent, a three-dimensionally porous structure with pore-size range of 100–500 μm and 50% to 70% porosity is required (10). Pore sizes in fabricated samples were typically distributed in the range of 200–300 μm. All samples exhibited porosity of ∼70%, as determined by measurement of their mass and dimensions. These samples had a high porous structure, consisting of open pore channels. This morphologic feature is expected to provide large surface area and sufficient space for blood circulation and cell ingrowth (1). Figure 2 shows SEM images of composites with 0 and 30 wt% of Al2O3 after sintering. The micrograph relative to composites sintered with 30 wt% Al2O3 (HA6) reveals a significant intragranular porosity (Fig. 2-b). From the energy dispersive X-ray analysis (EDAX) on the SEM of HA6 composites in Figure 2-b, it can be found that the light phase is Al2O3, which has been segregated at the grain boundaries of the grey colored matrix phase.
Mechanical properties
The compressive strength of the HA9 was 5.7 ± 1.2 MPa when it was sintered at 1200 °C. After addition of Al2O3 to the HA9 powder and sintering under the same conditions, the compressive strengths increased. The mechanical properties increased steadily with increasing of Al2O3 content up to 30 wt % (ANOVA, P<.05). Using Al2O3 as reinforcing agents of Si-HA led to chemical reactions between them. Furthermore, the addition of Al2O3 is effective to improve the low fracture strength and high resistance to crack propagation of HA.
Conclusion
This work has successfully synthesized highly porous implants (200–300 μm) for bone engineering, using the replication technique. When HA and bioglass sintered under an optimal condition, the nearly full densification and the fine crystals of Si substituted HA. The alumina additive in the composites remained Al2O3 after sintering and XRD analysis of the samples reveals the presence of silicon-incorporated hydroxyapatite and alumina without any other phases. The manufactured porous samples had a compressive strength of about 6-12 MPa at a porosity of ∼ 70%. Under the processing conditions used, Al2O3 increased significantly the compressive strength of composites. Therefore, the goal of an ideal porous implant that provides sufficient mechanical support temporarily is achievable with the present hydroxyapatite-bioglass-alumina.
Footnotes
Financial support: This study was supported by the Babol University of Technology and Amirkabir University of Technology.
Conflict of interest: None.