Abstract
The use of hydraulic calcium silicate cements (HCSCs) is essential for vital pulp therapies (VPT). However, their high cost restricts access in low- and middle-income countries, preventing the widespread benefits of VPT. This study aimed to evaluate four previously developed and characterized low-cost HCSC prototypes by examining their biological properties, specifically the rat connective tissue response after implantation and antimicrobial activity against five strains of interest. Additionally, their compressive strength, bond strength, and microhardness, which are critical mechanical properties of materials used in VPT, were assessed. All HCSCs caused an inflammatory reaction, which decreased over time in all cases, with most reactions categorized as mild. When comparing the number of inflammatory cells at each time point, no significant differences were observed between the HCSCs and those compared to MTA Angelus. Regarding the fibrous capsule, its thickness gradually decreased, and all capsules ultimately had a thickness with no significant difference compared to those formed in the empty control group. No significant differences in antimicrobial activity were seen among the four prototypes, as they exhibited similar performance against the five tested strains. However, some prototypes showed significantly better performance compared to MTA Angelus. Concerning mechanical properties, most prototypes exhibited substantially higher compressive strength than MTA Angelus, with a gradual increase over time—though this increase was not always significant. Additionally, no prototype demonstrated significant differences in bond strength compared to each other or MTA Angelus. Microhardness also increased over time, with significant differences observed when comparing prototypes to MTA Angelus at each time point. These findings, along with previously reported data on their microstructure, composition, and physical properties, support the potential clinical use of these prototypes. However, further research is needed to evaluate their effectiveness in clinical settings.
Keywords
Introduction
The history, formulation, and significance of hydraulic calcium silicate cements (HCSCs), along with their role in revolutionizing endodontics, have been previously discussed, as well as their importance in vital pulp therapies (VPT), including their limitations. 1
To this day, a wide variety of HCSCs remain accessible on the market, some of which have overcome their initial significant limitations by modifying their formulations, as it is well known that even minor chemical differences2,3 or the inclusion of different additives,4–8 as well as variations in the size and shape of particles, could affect their physical, mechanical, and biological properties. However, the most critical limitation that remains unresolved is their high cost. This issue may be less significant in developed countries with accessible healthcare. Unfortunately, because of the high cost of these materials, treatments like VPT are not routinely available in public primary healthcare clinics in low- and middle-income countries, which significantly impacts patient health outcomes and quality of life. 9
Consequently, the Multidisciplinary Dentistry Research Laboratory at the Faculty of Medicine, Universidad Autónoma de Querétaro, Mexico, has initiated a multi-year project to design, produce, and evaluate various experimental HCSCs. These prototypes aim to achieve ideal physical, mechanical, and biological properties while maintaining low production costs. To date, four promising HCSC prototypes have been characterized in terms of their microstructure, composition, primary physical properties, and in vitro bioactivity. However, their mechanical and biological features still require description.
For many HCSC applications, mechanical properties are not deemed essential; however, for VPT, they are considered an important feature. 10 Such therapies require that the cement used for pulp capping remain in place despite dislodging forces from operative procedures. Additionally, it is essential that the cement can withstand masticatory forces.11,12 Compressive strength, bond strength, and microhardness are regarded as the primary mechanical properties of HCSCs and are common test parameters used to evaluate dental materials.12,13 Furthermore, biocompatibility is crucial; this term describes a material’s overall ability to interact safely and appropriately with a host. Therefore, a material should only be considered biocompatible if it overcomes multiple biological tests. Nevertheless, tissue reaction tests play a fundamental role in determining the biological effects induced by an HCSC. Subcutaneous implantation in animals is the most suitable, accessible, and common method for evaluating the local tissue response to these materials. It offers an accurate assessment of the type and severity of the local reaction triggered by the materials. 14 Furthermore, as a relevant biological property, the antimicrobial activity of a material is crucial for its use in VPT, since microorganisms are the main etiological factor in pulpitis. 15 Therefore, controlling them is crucial during these therapies, where pulp healing largely depends on the absence of irritating agents from bacterial metabolic products, and working under the principles of asepsis and antisepsis is not enough. 16 Consequently, it is essential to use materials that can inhibit and prevent bacterial growth. 17 For all these, this investigation aimed to characterize the four HCSC prototypes to determine their biological properties: rat connective tissue response after implantation and antimicrobial activity against five microbial strains of interest, as well as to evaluate their compressive strength, bond strength, and microhardness, which are critical mechanical properties for materials used in VPT. These properties were compared with two well-known commercial materials, MTA Angelus (Angelus, Londrina, Brazil) and a zinc-oxide eugenol-based material (IRM; Dentsply Sirona, Charlotte, NC, USA). The null hypothesis was that there are no differences in these properties between the four HCSCs and MTA Angelus.
Methods
Four previously reported HCSC prototypes (w175, z175, w175z25, and w10z10) were prepared. 1 Their constituents are presented in Table 1. MTA Angelus and IRM served as control materials for most experiments. These control materials were prepared according to the manufacturer’s instructions, while the HCSC prototypes were manually prepared using distilled water at a water-to-powder ratio of 0.35 to achieve a putty-like consistency. This study was approved by the Institutional Ethics Committee for Investigation of the Faculty of Medicine at the Universidad Autónoma de Querétaro, with reference numbers: EO-010/2024-5 (for the use of human extracted teeth) and BIOM-017/2024 (for the use of laboratory animals).
Powder ratios of the HCSC prototypes and their assigned names.

CS-base: Calcium-silicate powder (CPC-30R-B, Cemex, Mexico) subjected to a 60°C thermal treatment for 24 h. m-CaWO4: Calcium tungstate (Cat. 248665, Sigma Aldrich) ball-milled for 20 h. m-ZrO2: Zirconium oxide (Cat. 230693, Sigma Aldrich) ball-milled for 20 h.
Animal and histological procedures and analyses
A total of twenty male Wistar rats, aged 3 months and weighing between 250 and 280 g, were incorporated into the study. These subjects were housed within a temperature-controlled environment maintained at 22 ± 1°C with 55% humidity, adhering to a 12-hour light/dark cycle, and were provided with water and food ad libitum. All procedures involving the animals were conducted following the official regulations for the use and care of laboratory animals in Mexico. 18 Additionally, the experimental protocol followed the rules and guidelines established by the U.S. National Institutes of Health. Measures were implemented to minimize pain and discomfort and to reduce the number of animals utilized. The subjects were randomly assigned to four groups, each comprising five animals. Anesthesia was administered via intraperitoneal injection of ketamine (80 mg/kg) combined with xylazine hydrochloride (8 mg/kg). Following shaving and disinfection of the dorsal region, three 1 cm incisions from head to tail were made using a #15 Bard-Parker blade (BD, Franklin Lakes, NJ, USA). Subsequently, two pockets were created within the subcutaneous tissue on either side of each incision. A total of 120 sterile polyethylene tubes, each measuring 3.0 mm in length and 1.6 mm in diameter, were filled with freshly prepared materials (n = 100 tubes: w175-n = 20, z175-n = 20, w175z25-n = 20, w10z10-n = 20 and MTA Angelus-n = 20) or left empty, serving as controls (n = 20 tubes). These tubes were immediately implanted into the prepared tissue pockets, with each animal receiving six implants one of each HCSC and the empty as control. The skin was then sealed with cyanoacrylate, and the animals were housed individually in cages, and maintained as previously described. At intervals of 3, 7, 14, and 28 days post-implantation, five rats were randomly selected and euthanized using pentobarbital (Sedalphorte 100 mg/kg). The polyethylene tubes, along with the surrounding tissues, were carefully excised and fixed in 10% buffered formalin. Subsequently, the tissues were subjected to dehydration using a graded ethanol series, treated with xylene, and subsequently embedded in paraffin. Longitudinal sections, 6 µm in thickness, were then stained with hematoxylin and eosin (HE). Images of the tissue proximal to the opening of the implanted tube were captured utilizing a digital camera (Axiocam 105, Carl Zeiss AG, Oberkochen, Germany) affixed to a light microscope (Axiolab 5, Carl Zeiss AG, Oberkochen, Germany) at magnifications of 100× for capsule thickness assessment and 400× for cell counting. ImageJ software (available at https://imagej.nih.gov/ij/) was employed for analysis in both instances. The count of inflammatory cells was performed on each implanted tissue, analyzing three non-serial HE-stained sections. In each image, two quadrangular fields measuring 70 × 70 µm, delineated adjacent to the open end of the tubes, were examined, and inflammatory cells were enumerated. This process resulted in determining the number of inflammatory cells per approximately 10,000 μm2. To assess capsule thickness, three measurements were obtained at the center and the edges of each capsule. The mean values of capsule thickness per specimen and per group were computed thereafter. Both the quantification of inflammatory cells and capsule thickness measurements were conducted by a single calibrated and blinded examiner. Additionally, the images at 100× magnification facilitated the identification and documentation of edema, necrosis, and dystrophic calcifications. These features were recorded as dichotomous variables, indicating either absence or presence.
Antimicrobial activity
The agar diffusion test was employed to assess the antimicrobial activity of the prototypes and control cements against five reference microbial strains: Streptococcus mutans ATCC 25175, Streptococcus mutans ATCC 35668, Staphylococcus aureus ATCC 29213, Enterococcus faecalis ATCC 29212, and Candida albicans ATCC 10231.
Each strain was inoculated into sterile tryptic soy broth (Difco, Detroit, MI, USA) to produce a suspension of approximately 0.5 McFarland, which corresponds to 108 colony-forming units/mL. Sterile cotton swabs were employed to distribute suspensions onto five Mueller–Hinton agar (Difco, Detroit, MI, USA) plates per strain, resulting in a total of twenty-five plates, with the process repeated twice independently. Freshly prepared materials were placed into six preformed wells, 5 mm in diameter, within the agar plate (w175, z175, w175z25, w10z10, MTA Angelus, and IRM). The test materials were pre-diffused for 2 h at room temperature. All plates were subsequently incubated at 37°C and examined after 24 h. Images of each plate were captured, and the ImageJ software was utilized to measure the inhibition zones.
Compressive strength
The compressive strength assessment was conducted in accordance with ISO 9917-1. 19 A total of 240 cylindrical specimens—comprising 40 for each of the HCSC (w175, z175, w175z25, w10z10, MTA Angelus) and the control (IRM)—were fabricated utilizing molds with dimensions of 12.0 mm ± 0.1 in height and 6.0 mm ± 0.1 in diameter. Each material was freshly prepared and subsequently compacted within the molds. The filled molds were pressed between two glass slides, and finger pressure was applied to extrude excess material and achieve a smooth, uniform surface. Following the initial setting phase, the specimens were carefully removed from the molds and preserved at 37°C with a humidity of 95% ± 5%. At intervals of 1, 7, 14, and 21 days post-mixing, each subgroup (n = 10) underwent a compressive strength assessment utilizing a computer-controlled universal testing machine (UTM; CMS Metrology, WDW-5Y, Querétaro, Mexico). Each specimen was aligned parallel to its longitudinal axis between the compressive plates, and a compressive force was applied at a crosshead speed of 1.0 mm/min. The peak load necessary to induce fracture in each specimen was recorded, and the compressive strength was derived in megapascals employing the formula: Compressive strength = 4P/πD2, where P denotes the maximum load in Newtons and D signifies the diameter in millimeters.
Bond strength test—Push‑out
A total of 240 mature human extracted uniradicular premolars, possessing approximately similar buccolingual diameters and lengths, were selected for this study. A low-speed diamond disc was employed to section the coronal and apical segments, thereby creating a middle segment measuring 2 mm in length. Each segment was affixed in an acrylic resin block, with the root canal space enlarged to a diameter of 1.5 mm. Subsequently, the specimens—comprising root segments embedded in acrylic resin—were immersed in a 2.5% sodium hypochlorite solution for 15 min, followed by a 1-min rinse in distilled water. They were then immersed in 17% EDTA for 3 min, rinsed again in distilled water for 1 min, and finally dried using gauze and paper points. The specimens were randomly allocated into six groups, each consisting of 40. Freshly prepared HCSCs (n = 200) and IRM (n = 40), used as control, were packed into the canal space and stored at 37°C under 95 ± 5% humidity. Bond strength testing was conducted at 1 h, 3, and 7 days post-mixing. Each subgroup (n = 10) was subjected to a test using a UTM (CMS Metrology, WDW-5Y, Querétaro, Mexico). Each specimen was loaded with a 1.5 mm diameter cylindrical stainless steel plug at a crosshead speed of 1.0 mm/min. The maximum failure load, recorded in Newtons, was converted into megapascals. Bond strength was calculated by dividing the maximum load (N) by the surface area (mm2). The surface area was determined based on the area of the tested slice section, calculated as follows: where π = 3.14 and the height corresponds to the thickness of the specimen slice in millimeters.
Microhardness test
A total of 192 disc-shaped specimens (comprising 32 for each of the HCSC (n = 160) and IRM (n = 32) were employed in this study. These specimens were fabricated using molds measuring 3.0 mm ± 0.1 mm in height and 10.0 mm ± 0.1 mm in diameter. Each material was prepared freshly, inserted, and compacted within the molds. The molds were then compressed between two glass slides, and finger pressure was applied to extrude excess material and produce a smooth, uniform surface. Following the initial setting, the specimens were demolded and stored at 37°C under conditions of 95% ± 5% relative humidity. At intervals of 1, 7, 14, and 21 days post-mixing, each group (n = 8) was subjected to a microhardness assessment using a microhardness tester (CMS Metrology, CHV-1, Queretaro, Mexico). A force of 2.9 Newtons was applied with a diamond indenter for 15 seconds. All hardness measurements were documented as Vickers hardness numbers (VHN). The VHN (kgf/mm2) was calculated using the following equation: VHN = 1.854 (L/d)2, where L represents the applied load (kgf) and d denotes the mean diagonal length (mm). This value was obtained from three indentations at different zones on one side of each specimen.
Statistical analysis
For quantitative data, its normality was evaluated using the Kolmogorov–Smirnov test. Depending on the data distribution, either ANOVA with Tukey post hoc or Kruskal–Wallis with Dunn post hoc were applied. For qualitative data, the Fisher test was applied. All analyses were conducted with GraphPad Prism version 10.5.0 (GraphPad Software, San Diego, CA, USA). Statistical significance was defined as p < 0.05.
Results
The intensity of the inflammatory reaction and the thickness of the fibrous capsule were evaluated (Figure 1). The former was assessed by quantifying the number of inflammatory cells per 10,000 µm2 at 400× magnification. The density of these cells decreased over time in the presence of the implant. When comparing the number of cells induced by each HCSC (including de MTA Angelus) at each time point, no differences (p > 0.05) were observed among them. Only in two instances differences (p < 0.05) were noted when compared to the empty control at 3 days (z175 prototype) and 28 days (w10z10 prototype) (Figure 2(a)). The reduction in cell numbers was significant (p < 0.05) for most implants by the 28th day (Table 2).

Representative light micrographs displaying tissue sections proximal to the HCSCs and the control (empty tube) at each time point. Varying densities of inflammatory cells are observed, with all groups exhibiting fully mature, thin capsules by 28 days.

(a) Number of inflammatory cells per 10,000 μm2 in tissue sections proximal to the HCSCs and the control (empty tube). The Kruskal–Wallis test followed by Tukey post hoc showed significant differences at 3 and 28 days between two HCSCs and the control (empty tube); a comprehensive comparison across time points is detailed in Table 2. (b) Capsule thickness, analyzed using ANOVA followed by Dunn post hoc, indicating that “a” represents significant differences between all the HCSCs and the control and “b” indicates a significant difference between MTA Angelus and the control (empty tube); notably, at 28 days, all HCSC prototypes showed no significant difference compared to the control (significances not displayed in the graph), with detailed intra-period comparison provided in Table 2.
Multiple comparisons of inflammatory cell counts and capsule thickness for each HCSC and the empty control across different exposure times.

NC: number of inflammatory cells; CT: capsule thickness.
The Dunn test was used to analyze the number of inflammatory cells, whereas the Tukey test was employed for comparison of capsule thickness.
p < 0.05. **p < 0.01. ***p < 0.001. ****p < 0.0001.
Regarding the thickness of the fibrous capsule, measurements were conducted even during the early stages when the capsule had not yet fully matured. It was observed that the thickness of the capsule gradually diminished (p < 0.05) over time, attaining complete maturity at 28 days in all instances (Table 2). All HCSCs elicited a similar response during the initial two time points; however, notable differences emerged in the subsequent two periods. Markedly, MTA Angelus exhibited an opposite trend, forming one of the thinnest capsules at 14 days (p < 0.05) yet the thickest (p < 0.05) at 28 days. At this time point, the capsules of all HCSC prototypes did not differ (p > 0.05) in thickness compared to the capsule induced by the empty tube (Figure 2(b)).
No edema nor necrosis was observed in any case; however, dystrophic calcification was noted (Figure 3), beginning on the 7th day in some cases and present in all cases by 28 days (Table 3), except in the empty tube control group, where dystrophic calcification was never observed.

Representative light micrographs illustrating tissue sections proximal to the HCSCs at each designated time point.
Occurrence of dystrophic calcification in tissues exposed to each HCSC and control.
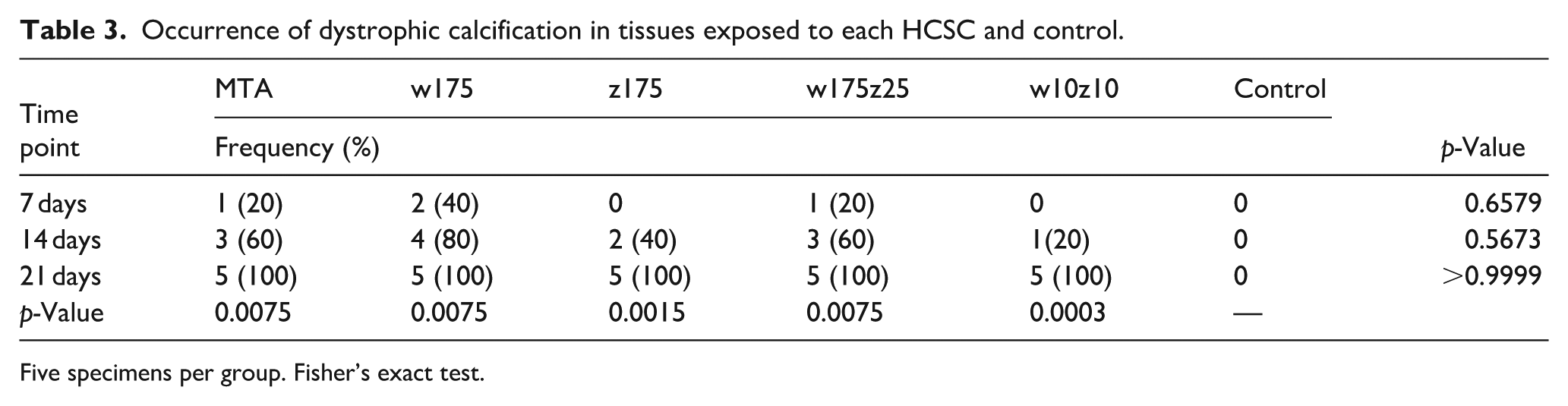
Five specimens per group. Fisher’s exact test.
Concerning antimicrobial activity, all HCSCs inhibited four out of five strains. Their efficacy was dependent on the microorganism (Table 4). No differences (p > 0.05) were observed among the four HCSC prototypes; they exhibited comparable performance against the five strains. However, at least one prototype consistently outperformed (p < 0.05) MTA Angelus, with w175 being the most effective, followed by w175z25. As expected, IRM exhibited the highest activity, producing the largest inhibition zones (p < 0.05). The growth of E. faecalis was unaffected by the HCSCs but was minimally inhibited by IRM.
Antimicrobial inhibition zones (mm) for each HCSC and IRM.

S. mutans-68 refers to S. mutans ATCC-35668, and S. mutans-75 to S. mutans ATCC-25175. The Kruskal–Wallis test was used.
Different superscript letters denote significant differences between values when Dunn’s multiple comparison test was applied.
Regarding mechanical properties, Figure 4(a) illustrates the compressive strength of each HCSC and IRM. It is evident that, at all four experimental time points, the majority of prototypes exhibited higher compressive strength compared to MTA Angelus (p < 0.05). Although not indicated in the figure, significant differences (p < 0.05) were also present when compared to IRM. Overall, no differences (p > 0.05) among the four prototypes were observed at each time point. When examining the compressive strength of each HCSC over time (Table 5), no changes (p > 0.05) were detected between consecutive time points (1 to 7, 7 to 14, or 14 to 21 days). For most materials (MTA Angelus, w175, w175z25), an increase (p < 0.05) was noted by the second time point (1–14 days), while others (z175, w10z10) showed an increase (p < 0.05) by the third time point (1–21 days). In Figure 4(b), it is observed that microhardness generally increased over time, predominantly in an insignificant (p > 0.05) manner (Table 5); however, significant differences (p < 0.05) were detected between MTA Angelus and the prototypes, as well as among some of the prototypes themselves at each experimental interval. Furthermore, Table 6 displays the push-out bond strength values. Although a gradual increase over time was noted, this was primarily not significant (p > 0.05). Notably, only the w175 and w175z25 prototypes exhibited a significant (p < 0.05) increase from 1 h to 7 days. IRM was the only material demonstrating differences (p < 0.05) when compared to MTA Angelus.
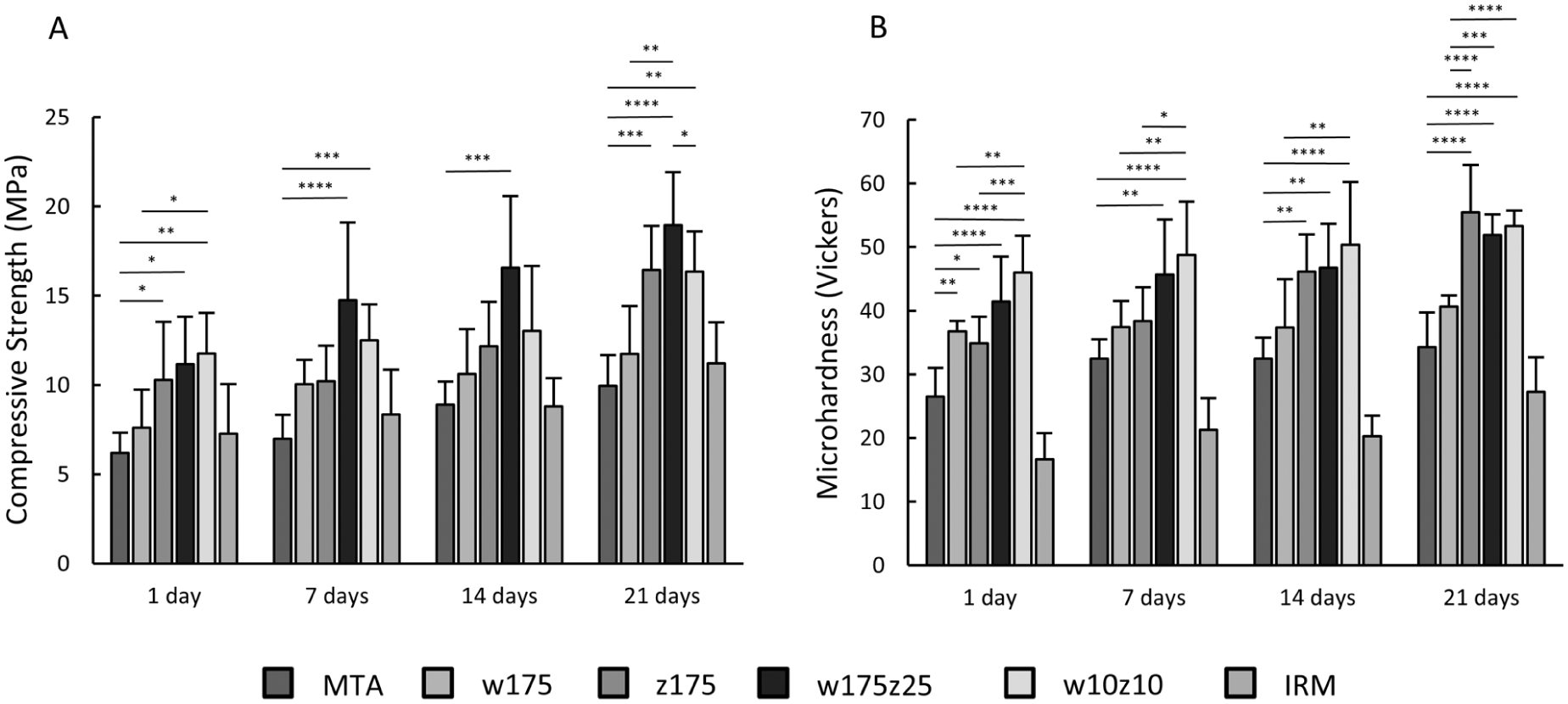
(a) Compressive strength (MPa) of the HCSCs and the IRM. A Kruskal–Wallis test followed by a Dunn post hoc test was applied, revealing significant differences both among the HCSCs themselves and between the HCSCs and the IRM (differences with the IRM are not indicated in the graph); a comprehensive comparison across time points is detailed in Table 5. (b) Microhardness (Vickers) of the HCSCs and the IRM. An ANOVA test followed by Tukey post hoc showed significant differences among the HCSCs and between each HCSC and the IRM (differences with IRM are not indicated in the graph); a detailed intra-period comparison is also provided in Table 5.
Multiple comparisons of compressive strength and microhardness of each HCSC and IRM at different time points.

CS: compressive strength; MH: microhardness.
To analyze the compressive strength, the Dunn test was employed, whereas the Tukey test was utilized for microhardness comparison.
p < 0.05. **p < 0.01. ***p < 0.001. ****p < 0.0001.
Push-out bond strength (MPa) of the HCSC and IRM.

The Kruskal–Wallis test was employed, and the Dunn’s multiple comparison tests revealed significant differences when comparing the 1-hour and 7-day measurements of the w175 and w175z25 prototypes, as well as MTA Angelus and IRM at 7 days.
Discussion
In Mexico and internationally, VPT presents a promising approach to mitigating tooth loss within populations that have limited access to root canal procedures, particularly those dependent on public healthcare systems. 9 Nonetheless, a considerable obstacle persists: the elevated cost of commercial HCSCs constrains their utilization in public dental services. To address this, we leveraged existing knowledge regarding HCSCs to develop low-cost prototypes. These prototypes emphasize optimal physical, mechanical, and biological characteristics by employing affordable, laboratory-grade ingredients within the context of a public university. All our prototypes have a mean production cost that is at least 150 times lower than that of the most widely used MTA in our country, namely, MTA Angelus, a well-known commercial product.
The majority of outcomes from this investigation suggest a tissue response and antimicrobial efficacy comparable to MTA Angelus, coupled with excellent mechanical characteristics. These findings lead us to reject the null hypothesis, given that certain prototypes surpassed MTA Angelus, while others fulfilled the minimum requirements for VPT applications.
The in vivo model employed in this investigation, despite its limitations, is one of the most comprehensive and clinically pertinent models for examining tissue response. 20 This model is capable of assessing not only tissue characteristics associated with irritant or inflammatory potential but also the duration of these effects on tissues. 21 To evaluate the tissue response, we adhered to well-established criteria,22,23 but adopted a quantitative approach by documenting the number of inflammatory cells and capsule thickness instead of categorizing them. Furthermore, binary variables for edema, necrosis, and calcification were incorporated, as reported in other studies.24,25 Overall, the tissue inflammatory response was moderate and diminished over time, ultimately converging with the control (empty tube), with virtually no inflammation remaining after 28 days. It is well documented that these materials generally induce a moderate to severe cellular response during the initial days, characterized by abundant inflammatory cells in the surrounding tissues. This initial response is associated with the surgical procedures employed for implantation, as well as the early reaction to the materials’ high pH, and the elevated temperature during setting, which ultimately translates into cytokine production. 26 Most studies rely on a subjective grading system to evaluate the severity of inflammation, yet this approach can be unreliable due to variations in cell counting influencing the assigned grade. To improve accuracy within this investigation, cells were enumerated within specific, standardized areas, facilitating more consistent comparisons across groups. Nevertheless, when these results are contrasted with others employing categorical assessments, a count exceeding 75 cells signifies a severe inflammatory response. In the present findings, only prototypes z175 and w10z10 surpassed 75 cells, and this occurred solely at the initial time point. z175 was the only prototype to exhibit a statistically significant difference from the control (empty tube). In almost all cases and across the first three experimental time points, responses were predominantly moderate, involving no more than 50 cells. By 28 days, all groups demonstrated a decline in inflammatory cells, often to fewer than 25, indicating negligible inflammation. It is well established that the nature and progression of the inflammatory infiltrate are directly linked to the host’s reaction to substances released by the materials. Immune cells produce and release various cytokines and growth factors, which actively contribute to the complex and coordinated cascade of cellular and molecular events that regulate the inflammatory process.27,28
Since 1977, the formation and gradual thinning of a fibrous capsule have been recognized as indicators of biocompatibility.23,29,30 This process has been progressively documented in this investigation, and by 28 days, all HCSCs were surrounded by a well-defined capsule with minimal inflammatory response, consistent with previous studies. 24 The capsule thickness exhibited a significant decrease across all groups over time. Overall, no substantial differences were observed between the prototypes; however, some significant variations, particularly with MTA Angelus, were identified. Notably, at 28 days, there were no significant differences in fibrous capsule thickness between the prototypes and the control group (empty tube). Given that polyethylene tubes are regarded as inert, the initial inflammatory response may be attributable to traumatic injury incurred during experimental procedures. 22 By the 28th day, this response was nearly resolved; therefore, the absence of significant differences among the prototypes suggests that all materials are well tolerated by the surrounding tissues.
Among the quantitative histological variables examined in this study, the qualitative variables of edema or necrosis were not observed; this aligns with previous reports for MTA Angelus. 24 Nevertheless, all tissues exposed to HCSCs showed the formation of dystrophic calcifications. Multiple studies22,31,32 have documented this phenomenon following subcutaneous implantation, noting that these calcifications appear irregular in this tissue but resemble dentin when implanted in the pulp. 32 All the HCSCs exhibited these structures to varying extents, primarily differing in the time point at which they appeared. By 14 days, a significant proportion of tissue samples from all HCSC groups showed these structures, and by 28 days, they were present in all samples. This finding is consistent with previous studies,22,24 in which these structures became observable after 14–15 days and were visible in all tissues by the 28th day. This development of calcic structures in subcutaneous studies has been associated with the osteoinductive capacity of the material, 33 and coincides with our previously reported data of in vitro bioactivity detected in all HCSCs. 1
Microorganisms are recognized to play a vital role in the development and progression of pulp disease. The high success rates of VPT may partly be attributed to the absence of microorganisms and their products within the pulp, which underscores the relevance of using antimicrobial materials during these procedures. In this study, five relevant strains associated with initial and secondary pulp lesions34,35 were selected, and the agar diffusion test was utilized, given its status as a standard method for assessing antimicrobial activity. Furthermore, efforts were undertaken to standardize testing conditions to reduce the influence of confounding factors. The results demonstrated that all HCSCs inhibited the growth of fungi and bacteria, as anticipated, due to their antibacterial activity primarily deriving from tricalcium silicate and dicalcium silicate, common constituents of all these materials. These components hydrate rapidly upon contact with water, forming an alkaline calcium silicate gel within a few hours of mixing, thereby creating a high alkalinity environment that is hostile to microbial proliferation.36,37 The prototypes exhibited superior antibacterial activity compared to MTA Angelus, potentially owing to enhanced diffusion properties attributable to slightly finer particles observed in both powder and set forms via scanning electron microscopy. 1 Notably, the w175 and w175z25 prototypes, which contain higher amounts of calcium tungstate, demonstrated the most pronounced effects. This may be due to the fact that calcium tungstate facilitates a higher calcium release than the zirconium oxide present in the other prototypes. This is supported by our previous research, 1 which found that higher proportions of calcium tungstate in the prototypes resulted in slightly higher pH levels, potentially enhancing their antimicrobial efficacy. However, none of the HCSCs inhibited the growth of E. faecalis despite the elevated alkaline pH. This alkalinity was insufficient to alter the microenvironment and suppress the proliferation of this bacterium, the underlying cause of which remains unidentified. Nonetheless, it is consistent with previous studies,38,39 including the initial report on the antimicrobial properties of the first commercially available MTA (ProRoot), exhibiting no activity against E. faecalis. 17 Conversely, inhibition of C. albicans, S. aureus, and S. mutans was observed, concordant with other research findings.40,41
Meanwhile, compressive strength is an essential mechanical property of HCSCs, and evaluating it provides insights into their setting process 10 and correlation with clinical performance. In this study, the compressive strength of all tested materials increased significantly from 1 to 21 days, which was expected and is consistent with other research. 42 This increase is attributable to the ongoing setting process, resulting in enhanced strength and stability over time. All prototypes demonstrated greater resistance to compressive forces than MTA Angelus, likely due to its shorter setting times, which are associated with differences in particle size, the presence of various additives, and their quantities, as previously documented. 1 In turn, marginal adaptation and sealing of HCSCs are essential for clinical success, as most endodontic failures result from leakage of microorganisms or their irritants. Both issues can be assessed through bond strength evaluations, reflecting the integrity of the dentin-cement interface. 43 To assess bond strength, the push-out bond strength test is employed as a practical and reliable method to measure the extent of material adaptation to the surrounding dentin. 13 Although various methods exist to evaluate adhesion, the push-out test offers efficient and dependable results. 44 In the current study, the push-out bond strength of all HCSCs increased significantly over time, yet remained consistently similar among them and when compared with MTA Angelus. This observation was anticipated, given their very similar composition. It is well established that, generally, all HCSCs interact with dentine to promote the incorporation of intratubular calcium and silicon and the formation of tag-like structures.45,46 It has been reported that apatite nucleation at the interface enhances sealing by reducing voids 47 and increases dislocation resistance. All prototypes exhibited higher values compared to MTA Angelus, which can be attributed to their finer particle size, thereby improving cement infiltration into dentinal tubules and influencing dislocation resistance. 48 However, for most of them, the differences were not significant, except for prototypes with elevated levels of calcium tungstate (w175, w175z25), which showed significantly higher values. This may be due to calcium tungstate’s contribution to increased calcium release, thereby promoting greater biomineralization. 49 As previously demonstrated, all are considered active biomaterials that produce calcium phosphate and apatite-like precipitates at the cement-dentin interface and within the dentinal tubules. This process leads to the formation of tag-like structures and an interfacial hybrid layer responsible for both chemical and mechanical bonding. 50
The Vickers’ hardness test is frequently employed to evaluate surface microhardness. It helps determine the material’s strength and the degree of hydration it has undergone during the setting process.51,52 A reduction in microhardness may result in increased solubility and diminished sealing efficacy. 53 The findings of the study demonstrated a notable increase over time across all the HCSCs. In numerous cases, the prototypes outperformed MTA, suggesting that, despite previous reports of shorter setting times, these rapid setting times do not significantly affect the setting process.
This study involved conducting various biological and mechanical assessments comparing the prototypes to the MTA Angelus, which remains regarded as the gold standard material for numerous clinical procedures, as substantiated by extensive evidence supporting its efficacy. Although the in vitro and in vivo (animal) outcomes of this and prior studies appear promising and indicate the potential application of all tested prototypes in VPT, it is important to acknowledge that each of these models has inherent limitations. Therefore, a comprehensive clinical evaluation is necessary to determine their performance within real-world conditions accurately.
Conclusion
All HCSC prototypes demonstrated the capacity to elicit an appropriate tissue response and exhibited antimicrobial and mechanical properties similar to or exceeding those of MTA Angelus, such as compressive strength, bond strength, and microhardness. These promising findings suggest their potential for use in VPT. However, further clinical studies are necessary for definitive application.
Footnotes
Acknowledgements
The authors wish to thank “Proyecto Ciencia de Frontera” by the Mexican Secretariat of Science, Humanities, Technology, and Innovation (SECIHTI).
Author contributions
Conceptualization, RADP, MVG, LFEC, RGST; methodology, ELJ, OCL, MLR, RMPS; formal analysis, RADP, MLR, RMPS, LFEC; investigation, MVG, RGST; writing—original draft preparation, ELJ, RADP; writing—review and editing, OCLM MLR, RMPS, MVG, LFEC, RGST; supervision, RADP; project administration, ELJ, RADP, RMPS; funding acquisition, RADP, MVG, RGST. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Mexican Secretariat of Science, Humanities, Technology, and Innovation (SECIHTI) through the program “Frontiers of Science 2023 – Ciencia de Frontera 2023,” Grant Number “CF-2023-G-29.”
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon reasonable request.
