Abstract
Objective:
BTG1 is a member of the TOB/BTG protein family, which is a transducer of ErbB-2 and TOB2. It is known to inhibit tumor genesis, but its role in pancreatic ductal adenocarcinoma (PDAC) is still unknown. The purpose of this study is to investigate the expression of BTG1 protein in PDAC and to determine its prognostic significance.
Methods:
Immunohistochemistry is used to determine the protein expression of the BTG1 gene in 79 surgically resected PDAC. The association of BTG1 expression with all the patients’ clinicopathologic parameters, including survival, was analyzed using statistical software.
Results:
High BTG1 expression was observed in 27.8% (22/79) of the PDAC tissues, which was significantly lower than the 58.2% (46/79) of corresponding normal adjacent noncancerous tissues by immunohistochemical staining (p<0.001).Through the stratified analysis, we found a significant difference of BTG1 expression in peri-neural invasion (p = 0.002), T stage (p = 0.000), N stage (p = 0.018), and tumor, node, and metastasis stage (p = 0.000). Univariate and multivariate Cox analysis revealed that BTG1 expression status was an independent prognostic factor in PDAC (p = 0.027). Moreover, overall survival was better in PDAC cases with higher rather than lower BTG1 expression (p = 0.027).
Conclusions:
This study demonstrated for the first time that lower expression of BTG1 might be involved in the progression of PDAC, suggesting that BTG1 might be a novel prognostic marker and a target for therapy.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of the most lethal cancers. In spite of its incidence rate of 10th in western countries, PDAC is still the fourth leading cause of cancer-related death in the western countries (1). The 5-year overall survival (OS) rate of PDAC worldwide is about 5%, which indicates that its prognosis is very poor (1). Although radical surgical resection of the primary tumor is the only potentially curative option in patients with PDAC, only 15%~20% are suitable for surgery because most PDACs are difficult to detect at an early stage and often they are diagnosed only after regional invasion or distant metastasis (2). Even among patients whose diseases are surgically resectable, at least 80% will subsequently develop local recurrences or distant metastases within 2 years of surgery (3). PDAC is almost resistant to all available radiotherapies and chemotherapies, so we aimed to search for a new molecular target that will contribute to early diagnosis and prognostic judgment of PDAC.
BTG1 (B-cell translocation gene 1) is a member of the TOB/BTG protein family, and contains 6 proteins (BTG1, BTG2/TIS21/PC3, BTG3, BTG4/PC3B, transducer of ErbB-2, and TOB2). All members of this family can inhibit cell proliferation and cell cycle progression, and stimulate cellular differentiation in different kinds of cells (4). Human BTG1, which is localized on chromosome 12q22, was initially discovered in a case of B-cell chronic lymphocytic leukemia and was recognized as a translocation partner of the c-Myc gene – hence it was named the B-cell translocation gene 1 (5, 6). BTG1 expression varies with the progression of cell cycles; it is highest in the G0/G1 phases decreases when cells progress through G1 (4). Previous research has also shown that BTG1 overexpression can reduce vascular endothelial growth factor expression in tumors (6) and contribute to anti-sense Bcl-2-induced cytotoxic effects in apoptotic cells (7, 8).
Previous studies have showed that BTG1 is down-regulated in multiple cancers, such as breast cancer (9), non-small cell lung cancer (NSCLC) (10), gastric cancer (11), ovarian cancer (12), and liver carcinoma (13). These results have consistently shown that BTG1 overexpression decreases proliferation, migration, and invasion, which suggests that BTG1 overexpression may contribute to a better prognosis.
Above all, BTG1 may be a candidate for tumor suppressor genes. However, the relation between BTG1 and clinicopathological features and prognostic factors in PDAC have not been reported. Therefore, we performed immunohistochemistry on 79 PDAC tissues, and then examined the association of the BTG1 expression levels and overall patient survival and clinicopathological features in PDAC.
Materials and methods
Patients and tissue samples
We collected 79 formalin-fixed paraffin-embedded tissue samples of patients with pathologically diagnosed PDAC who had undergone surgical resection at Fuzhou General Hospital of Nanjing Military Command between January 2005 and December 2014. We excluded patients receiving any preoperative anticancer therapies. We only included patients who had survived at least 60 days after surgery, to exclude any bias related to perioperative mortality. The clinicopathologic features of each patient was carefully collected from the clinical database, including sex, age, tumor size, tumor site, histological differentiation, peri-neural invasion (PNI), surgical margin, T stage, N stage, and tumor, node, and metastasis (TNM) stage. We obtained the overall survival data via individual telephone follow-up. These data were updated on September 21, 2015. Median follow-up time was 10.0 months (range: 1.0-114 months). Pathologic tumor staging was performed according to the American Joint Committee on Cancer TNM classification, 7th edition (14).
Our study was approved by the ethical committee of Fuzhou General Hospital of Nanjing Military Command and each patient signed an informed consent form before enrollment into the study to obtain the use of clinical specimens.
Immunohistochemistry
To detect the expression of BTG1 in PDAC, we used formalin-fixed and paraffin-embedded tumor tissue cut into 4 μm thick sections. The immunohistochemical analysis was performed using the Elivision method. The cut sections of tissue were incubated at 37°C overnight. Put simply, the tissue sections were deparaffinized in xylene and rehydrated in graded alcohol, and use citrate buffer (pH 6.0) in a pressure cooker for 2 minutes to retrieve antigen. Endogenous peroxidase blocking was performed at room temperature for 10 minutes with 3% hydrogen peroxide, then washed 3 times with 0.01 mM phosphate-buffered saline (PBS). The sections were then incubated with goat anti-human BTG1 polyclonal antibody (1:100 dilutions, no. sc-18540, Santa Cruz Biotechnology, Inc.) in a wet box at room temperature for 1 hour. After washing twice in 0.01 mM PBS, the secondary antibody (PV polymer HRP anti-goat IgG, ZSGB-BIO) was put into the wet box for incubation for 30 minutes and then washed 3 times with 0.01 mM PBS, followed by DAB staining for 5 minutes. The hematoxylin stain can make DAB staining visual. The final step was dehydration with the coverslip sealed. PBS, as the primary antibody, was used as a negative control.
The immunohistochemical scoring was performed independently by 2 pathologists without knowing patient information. Five representative fields of each section were randomly selected to be counted under a microscope. Cytoplasm labeling with the BTG1 antibody was regarded as a positive cell. The BTG1 expression level was defined by the staining index, which was calculated based on the percentage of positive tumor cells multiplied by the staining intensity. The percentage of positive tumor cells was graded as follows: 0 = 1%-5%; 1 = 5%-25%; 2 = 25%-50%; 3 ≥ 50%. The intensity of staining was classified as: 0 = no staining; 1 = weak staining (light yellow); 2 = intermediate staining (brown); and 3 = strong staining (yellowish-brown). The total points of BTG1 expression were the product of the BTG1 positivity and staining intensity, graded as 0 for low expression of BTG1, + to +++ for high expression of BTG1 (+ = 1-2; ++ = 3-5; +++ = 6-9) (9, 11).
Statistical analysis
All data were analyzed using SPSS22.0 software and p<0.05 was considered statistically significant. The relation between various variables and BTG1 expression in cancer tissue was analyzed by the chi-square test. The comparison of BTG1 expression status with overall survival in different groups of PDAC patients was performed using a log-rank correlation test and a Cox regression test. Clinicopathologic variables with p<0.05 on univariate analysis were selected in the multivariate Cox regression analysis. OS distributions were estimated using the Kaplan–Meier method. The OS was defined as the time from the date of confirmed diagnosis to death, or the date of the last telephone follow-up.
Results
BTG1 protein expression in PDAC tissues and adjacent noncancerous tissues
BTG1 protein was positively detected in the cytoplasm of adjacent noncancerous pancreas tissues where the staining was strong. However, in most of the PDAC tissues, cell staining was weak or negative, especially in the poor-differentiated PDAC (Fig. 1). On the basis of the immunohistochemical scoring, we found significantly lower rates of “high BTG1 expression” in PDAC tissues (27.8%) compared with corresponding normal adjacent noncancerous tissues (58.2%) (p<0.001, Tab. I).
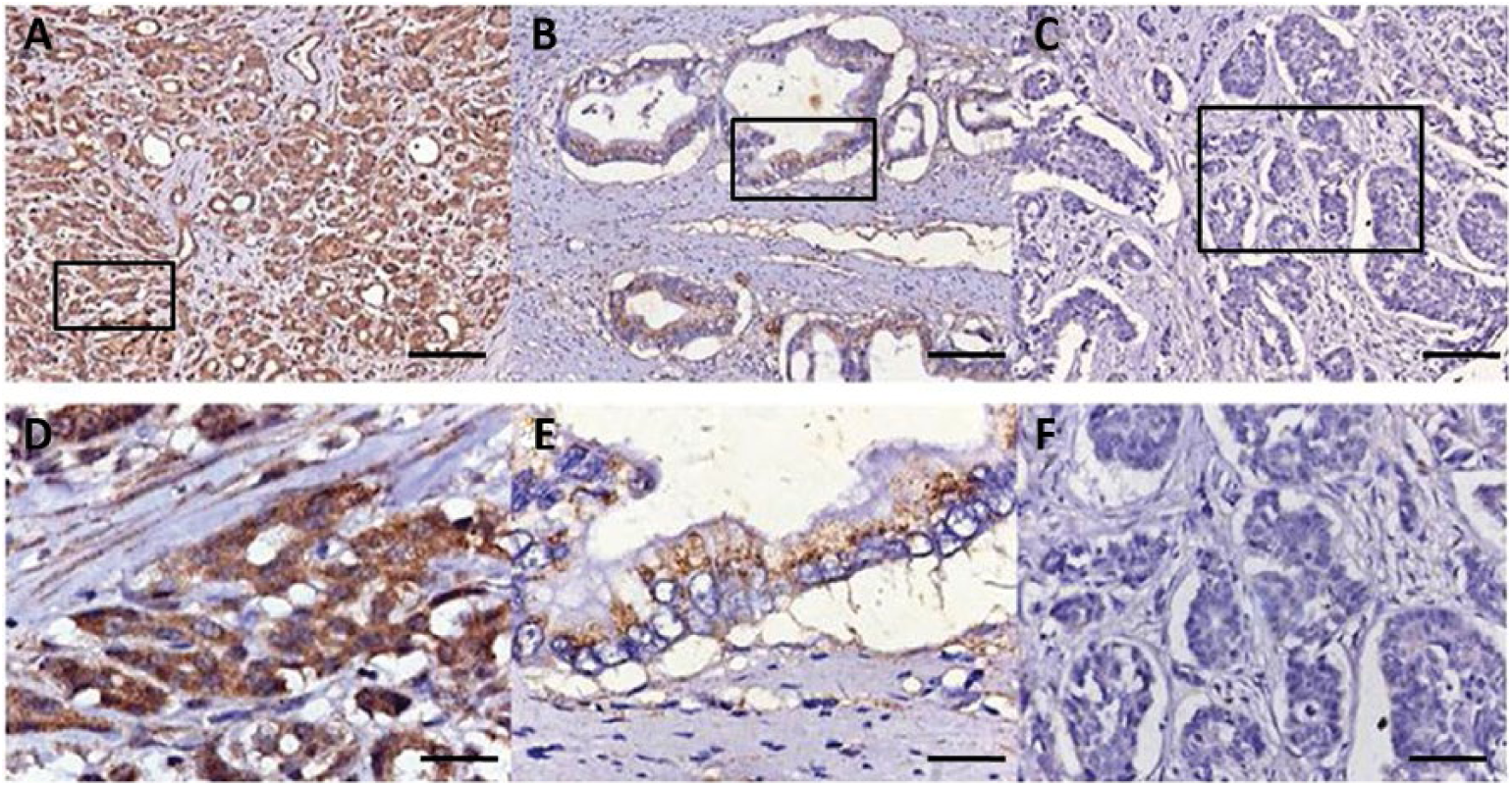
Immunohistochemical labeling for BTG1 protein. (
BTG1 expression in PDAC tissue and in normal tissue
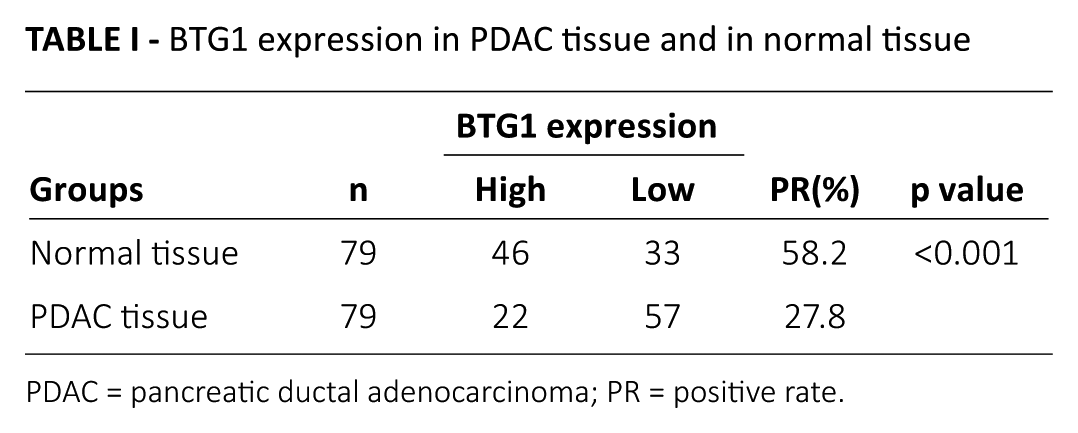
PDAC = pancreatic ductal adenocarcinoma; PR = positive rate.
The relationship between BTG1 expression and clinicopathological parameters of PDAC
We analyzed the BTG1 expression difference according to various variables using the chi-square test, including sex, age, tumor site, tumor size, histological differentiation, PNI, surgical margin, T stage, N stage, and TNM stage. Our results showed that BTG1 expression statistically correlated with PNI (p = 0.002), T stage (p = 0.000), N stage (p = 0.018), and TNM stage (p = 0.000) (Tab. II). However, no significant correlations were found with sex, age, tumor site, tumor size, histological differentiation, and surgical margin (Tab. II).
Summary of relation between BTG1 expression and clinicopathological parameters in patients with PDAC
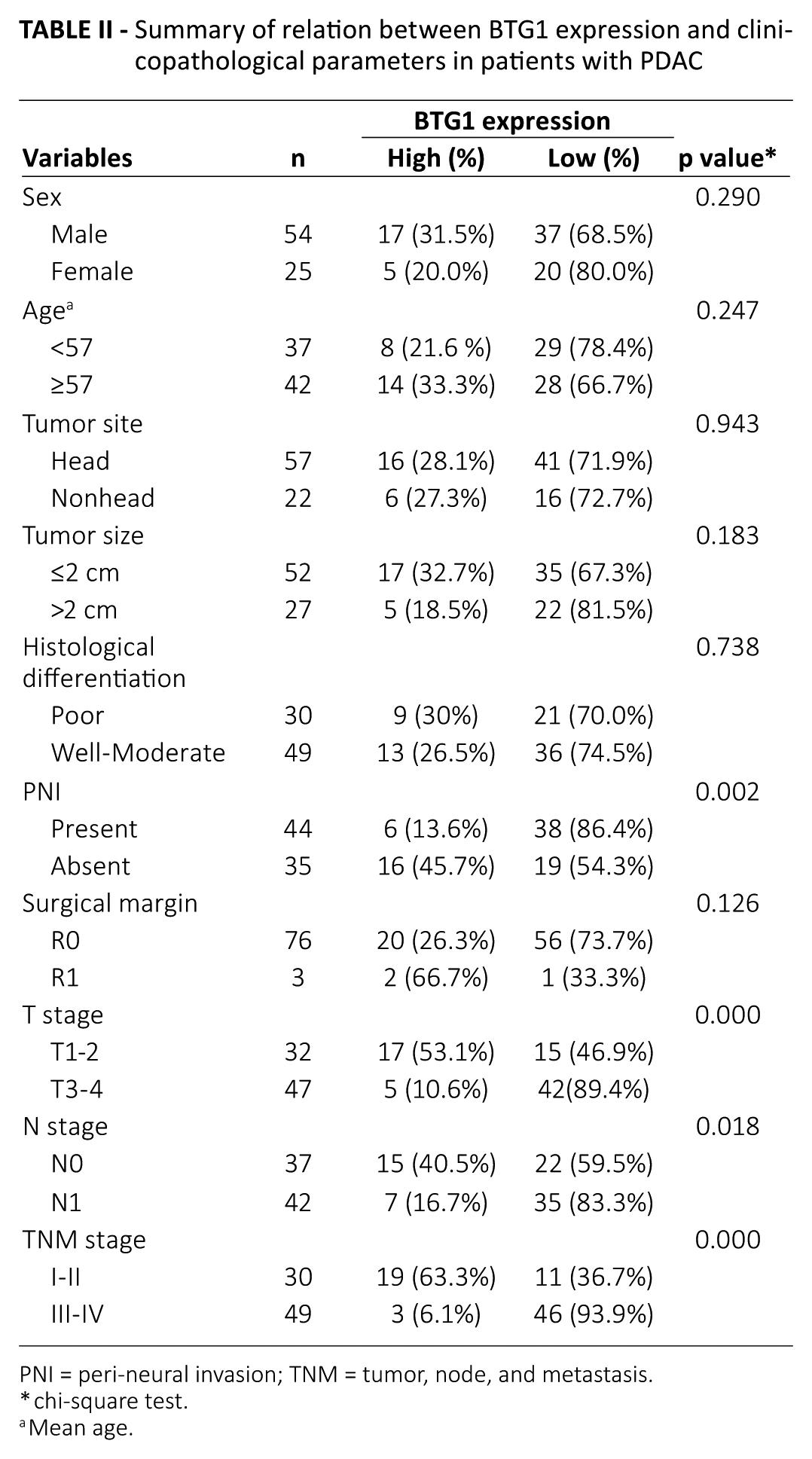
PNI = peri-neural invasion; TNM = tumor, node, and metastasis.
chi-square test.
Mean age.
Relationship between BTG1 expression and prognosis in PDAC patients
We collected survival data of all the patients via telephone follow-up. Only 6 of the 79 patients were still alive at the end of our follow-up. According to different BTG1 expression levels in 79 PDAC tissues, the patients were divided into 2 groups: (i) 22 high BTG1 expression patients; and (ii) 57 low BTG1 expression patients. We performed univariate and multivariate analyses to identify the relative risk of prognostic factors for the 79 PDAC cases (Tab. III). Univariate analysis showed that there are 6 significant prognostic factors correlated with poor prognosis, including histological differentiation (poor differentiation), T stage (T3-4), N stage (N1), peri-neural invasion present, III-IV stage, and low BTG1 expression (Tab. III). The Cox multivariate analysis indicated that histological differentiation (hazard ratio [HR]=2.342; 95% confidence interval [CI], 1.417-3.873; p = 0.001) and BTG1 expression status (HR = 2.331; 95% CI, 1.104-4.924; p = 0.027) were all independent prognostic factors for poor overall survival in patients with PDAC (p<0.05, Tab. III).
Overall survival in patients with PDAC according to multiple variables on univariate and multivariate analysis
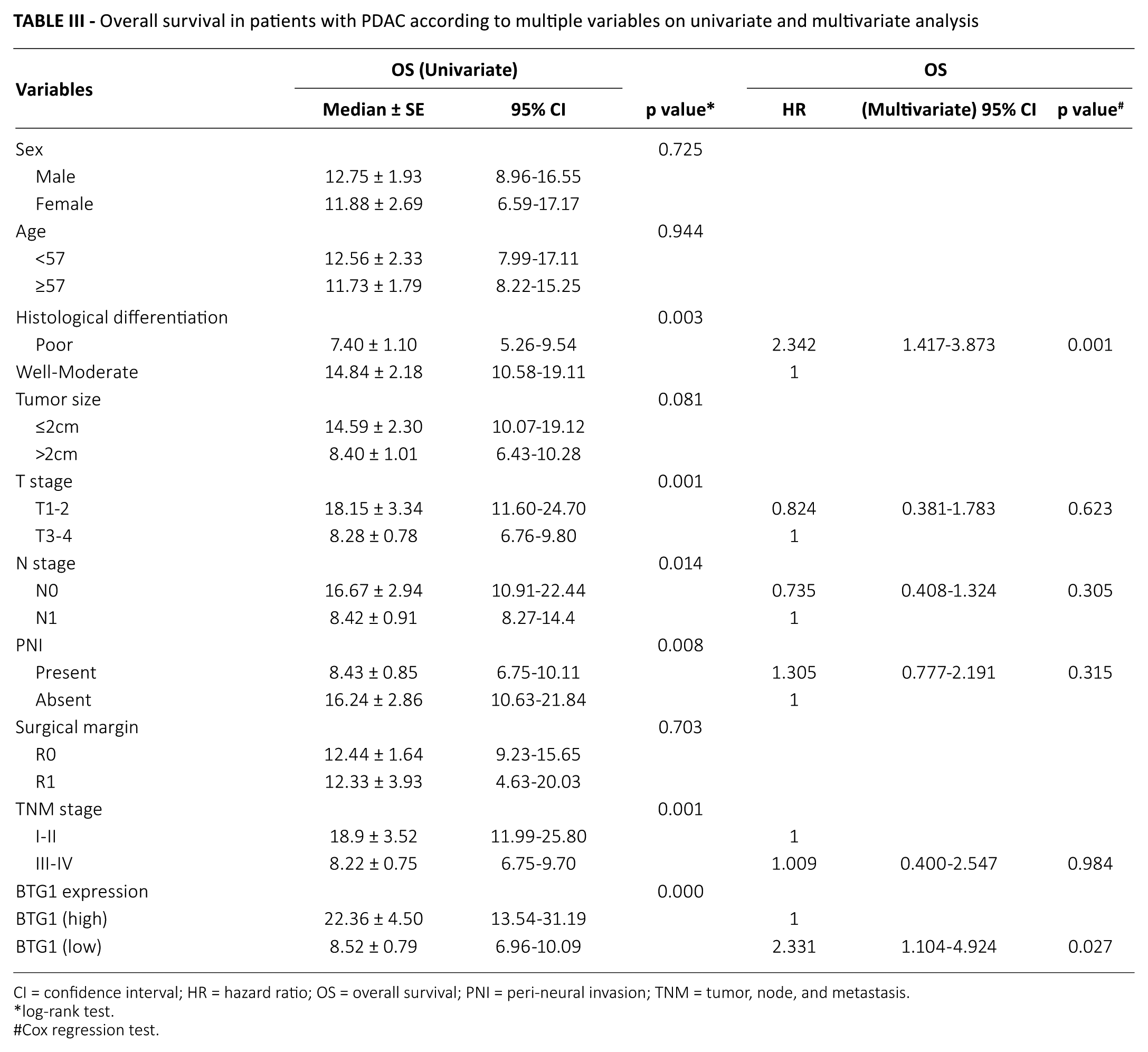
CI = confidence interval; HR = hazard ratio; OS = overall survival; PNI = peri-neural invasion; TNM = tumor, node, and metastasis.
log-rank test.
#Cox regression test.
Survival analysis was performed in all the patients on the basis of their overall survival and corresponding BTG1 expression status. Our results showed that patients with low BTG1 expression in PDAC samples had a significantly lower overall survival than those with high BTG1 expression (p = 0.027, Fig. 2). These results suggested that patients with low BTG1 expression have a worse prognosis than those with high BTG1 expression.
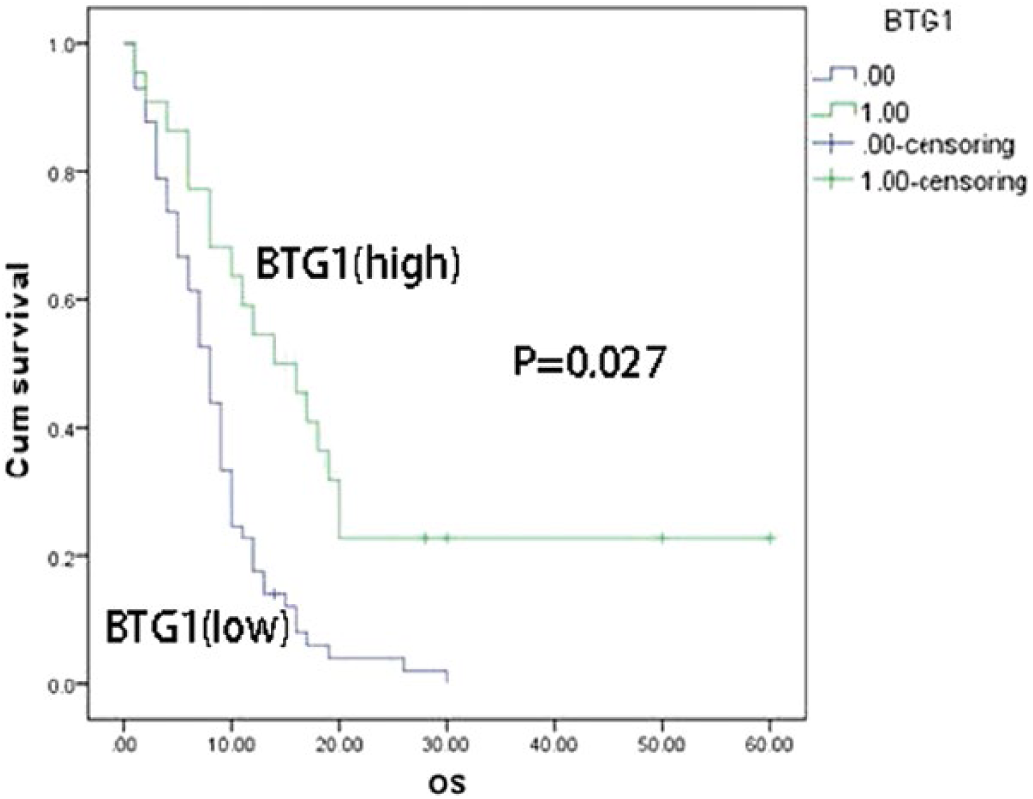
Kaplan–Meier survival curves showing the overall survival according to BTG1 expression status in all the pancreatic ductal adenocarcinoma (PDAC) patients. Overall survival of PDAC patients with low BTG1 expression was significantly shorter than patients with high BTG1 expression.
Discussion
Despite the steady increase in survival for different kinds of cancer in recent decades, the 5-year survival rate of pancreatic cancer is still less than 5% (1).·This is mainly because only 15%~20% of patients have the chance to receive surgical resection at the time of diagnosis and it is almost resistant to all available chemotherapy and radiation therapies (15). For pancreatic cancer, the prediction value of some prognostic indicators that had been known, such as tumor size, histological differentiation, and lymph node metastases, may not be obvious (16, 17). The most common pathological type of pancreatic cancer is PDAC (over 90%), which originates from the exocrine cells (18). A new way to detect and treat PDAC in the early stages is urgently needed. It is not surprising that patients with PDAC are deeply concerned about the prognosis of their disease. TNM staging is known as the preferred prognostic indicator (19). However, cancer patients’ prognoses differ considerably even if they are at the same TNM stage. Therefore, it is important for us to find a significant marker that will contribute to predicting tumor patients’ individual outcomes.
BTG1 expression is barely detectable in brain and muscle tissues, which are fully differentiated, but it will be detectable in cells that can still respond to various signals, especially in G0/G1 transition (6). The expression of BTG1 is highest in the G0/G1 phases of the cell cycle, and is decreased when cells progress through G1 (4), which means BTG1 overexpression can inhibit cell proliferation and cell cycle progression, increase cell apoptosis, and reduce vascular endothelial growth factor expression in tumors (6).
According to previous studies, we can draw some useful conclusions about BTG1. BTG1 overexpression was considered as a marker for favorable prognosis in breast, NSCLC, gastric cancer, ovarian carcinoma, hepatocellular carcinoma, nasopharyngeal cancer, esophageal squamous cancer, and thyroid cancer (9-13, 19). Zheng et al (11) indicated that BTG1 expression is associated with the worse prognosis in gastric cancer patients by inhibiting proliferation, and enhancing autophagy and apoptosis. They also suggested that lower BTG1 expression might promote gastric tumorigenesis partly due to its promoter methylation (11). Scheijen et al (20) also indicated that BTG1 is a tumor suppressor gene in leukemia; and when deleted, it strongly enhances the IKZF1-deleted B-cell precursor acute lymphoblastic leukemia relapse risk. Our results are consistent with recent research, showing that BTG1 may be a tumor suppressor gene.
Our study is the first in which BTG1 expression has been investigated in PDAC tissues using immunohistochemistry. The results showed that BTG1 expression levels were significantly lower in PDAC tissues than in normal adjacent noncancerous tissues (p<0.001, Tab. I). We found that BTG1 expression levels correlated with the PNI (p = 0.002), T stage (p = 0.000), N stage (p = 0.018), and TNM stage (p = 0.000). According to univariate and multivariate analysis, lower BTG1 expression (p = 0.027) and poor differentiation (p = 0.001) may predict decreased overall survival. We concluded that tumoral BTG1 loss may contribute to the development and progression of PDAC.
Our study revealed that the overall survival of patients with PDAC is significantly better in high BTG1 expression groups than in low BTG1 expression groups. Consequently, combining BTG1 expression status with clinical stage may help clinicians to decide on the most appropriate treatment plans and to predict prognosis.
Conclusion
This study demonstrated for the first time that lower expression of BTG1 correlated with poor survival in PDAC patients, which suggests that BTG1 expression may serve as an available prognostic biomarker in the future. However, there are still some defects in our study: more large population-based studies and long-term follow-up are still required to support our findings. A potential signal mechanism regarding the down-regulation of BTG1 in PDAC is still needed in the future, and we will conduct further studies to investigate this.
Footnotes
Disclosures
Financial support: This work was funded by grants from the National Natural Science Foundation of China (No.81502360) and Natural Science Foundation of Fujian Province (No.2016J01576 and No.2016J01586 and No.2017J05121).
Conflict of interest: None of the authors has financial interest related to this study to disclose.
