Abstract
Introduction
Severe refractory asthma (SRA) occurs in asthma patients failing to achieve disease control despite high-intensity therapy, good adherence, and proper inhalation technique. Lack of disease control in asthma is associated with increased health care costs. Omalizumab, an anti-IgE monoclonal antibody, improves disease control in SRA patients by reducing the number of exacerbations. However, real-world evidence studies are needed to quantify the burden of SRA on health care systems. The aim of this study was to assess the current management of SRA in Italy by analyzing the prevalence of the disease and the economic impact of SRA patients.
Methods
This observational study retrospectively analyzed data concerning adult patients registered in an Italian health care system database with either a prescription of omalizumab or at least a prescription of high-dose systemic corticosteroids from 2013 to 2014.
Results
The overall prevalence of SRA was 0.04%. SRA-related costs were equally distributed among 3 cost drivers (drug treatments, hospital admissions, and outpatient services), amounting to €2,815 per patient per year. In the subgroup of SRA patients treated with omalizumab (n = 55), costs related to hospital admissions and outpatient services decreased, while costs related to drug treatment increased.
Conclusions
The study gives an important contribution to understand SRA management in Italy, by reporting the prevalence and the health care costs of SRA from administrative databases. This real-world evidence could be useful to estimate the impact of the new upcoming biological entities (e.g., omalizumab) in the changing profile of health care cost drivers in SRA management.
Introduction
Asthma is a chronic respiratory disease with high economic and social burden. The disease is heterogeneous and affects around 300 million people worldwide with different phenotypes (1). Although asthma control can be achieved in most patients with available therapies, a fraction of asthmatic patients fails to respond even to high-intensity treatment (high doses of inhaled corticosteroids in combination with long-acting b2-agonists [LABA]) and develops severe refractory asthma (SRA) (2). Different definitions have been given to characterize severe refractory asthma patients throughout the years; currently, it is widely accepted to distinguish between difficult-to-control and refractory disease, with SRA patients being described as patients that adhere to treatment and possess a correct inhalation technique, but do not achieve disease control (3, 4). It is estimated that 5% to 10% of all asthma patients develop severe refractory disease (5, 6), but real-world studies are needed to confirm these numbers.
Therapeutic strategies have recently become available or are emerging on the market (e.g., omalizumab, mepolizumab, benlarizumab, reslizumab) for the treatment of SRA. Omalizumab is the first humanized monoclonal antibody approved as add-on treatment in patients with severe IgE-mediated disease (7). The use of omalizumab has been shown to significantly reduce the number of exacerbations and to improve the quality of life in patients with severe asthma (8, 9). In Italy, it was initially only authorized for in-hospital administration, then, in 2014, it was reclassified for territorial use (from in-hospital administration to outpatient distribution).
Real-world evidence studies are useful to assess the burden of disease and the health care costs. These studies allow treatment effectiveness to be evaluated in real-world patients by overcoming the well-known limits of controlled clinical trials, and are particularly important when analyzing chronic diseases with high economic impact, such as SRA. Indeed, health care costs in asthma increase greatly with disease severity (10). Exacerbations are associated not only with higher asthma-related costs but also with increased total health care costs (11). To date, no study has investigated the burden of SRA in Italy following the approval of omalizumab for the handling of IgE-driven severe asthma. Therefore the objective of this study was to obtain a picture of the current management of SRA in Italy, in terms of the number of patients in treatment and clinical practice of this disease. We analyzed the prevalence of SRA in Italy with related health care-resource utilization and costs from 2013 to 2014. A specific focus on omalizumab was also conducted.
Materials and methods
Study design
This was a real-world, observational study that retrospectively analyzed SRA patient-anonymous data registered in the Accounting and Reporting Console (ARCo) database (a collaboration between CORE and CINECA).
ARCo routinely collects and integrates National Health System (NHS) administrative data for each single patient (i.e., patient demographics, reimbursed drug prescriptions, inpatient hospital discharges, invasive/noninvasive specialist/diagnostic procedures). Demographic information about patients was made anonymous by data holders, according to Italian law regarding the protection of privacy. For each reimbursed drug prescription, the following information were collected: Anatomical Therapeutic Chemical classification (ATC), dose, number of packages, dispensing date, and cost of medicinal products and generics. All hospitalizations and procedures were codified by using ICD9 CM (International Classification of Diseases, Ninth Revision, Clinical Modification).
To conduct this retrospective observational analysis, ethical approval was necessary, since it was based on the collection of anonymous administrative data. It was conducted for institutional purposes, by means of a specific agreement with Italian National Health Facilities (regions and local health units [LHUs]).
Study participants
The study included patients aged ≥18 years affected by SRA. A patient was considered affected by SRA if he/she met at least 1 of the following entry criteria:
A prescription of omalizumab (ATC code R03DX05), a medication indicated to treat severe persistent allergic asthma.
Asthma exemption code (007.493) associated with a prescription of high-dose systemic corticosteroids (25 mg prednisone ATC code H02AB07, methylprednisolone 16 mg H02AB04), identifying asthma exacerbations.
Asthma exemption code (007.493) associated with outpatient service/day hospital with injection of other therapeutic or prophylactic substances (ICD9 CM procedure code 99.29). This last code, for asthma treatment, identified omalizumab administration in the hospital setting.
For each patient, the first date referred to 1 of the above points (drug prescription or outpatient service/day hospital) during the period January 1, 2013 to December 31, 2013 (enrollment period) was considered the index date of the study. Starting from the index date, each patient was followed up for 365 days.
In 2014, omalizumab drug classification changed (from in-hospital administration to outpatient distribution). Starting from 2014, the data on this drug became available in outpatient prescription database for all treated subjects; therefore, we conducted a specific analysis on a further group of patients treated with omalizumab, who were enrolled from January 1, 2014 to March 31, 2014.
Data collection
During the follow-up period, anonymized data regarding drug prescriptions were collected:
high-dose corticosteroids (prednisone 25 mg, methylprednisolone 16 mg);
humanized anti-IgE monoclonal antibody (omalizumab) as a proxy of severe asthma;
inhaled corticosteroids (ICS, ATC code R03BA);
long-acting b2-agonists (LABA, ATC code: R03AC);
adrenergic in combination with corticosteroids (ATC code: R03AK)
Moreover, hospital admissions, related to asthma (registered with the following codes ICD-IX-CM 493, 518.81, and 518.84, and with DRG 096 and 097) or not, and specialist/diagnostic health care services were collected during the follow-up in order to calculate the SRA burden in Italy and the Italian health care utilization with the integrated costs per patient.
For the specific analysis conducted on patients treated with omalizumab in 2014 (following the drug-classification change), the omalizumab prescription pattern was analyzed in terms of cycles (a single prescription was considered as a new cycle of therapy) and in terms of duration of therapy (estimated as days between the first and the last prescription date).
Health care costs were evaluated using the real price paid by the NHS and tariffs for in- and outpatient services. Integrated health care expenditure for patients with SRA were calculated using the costs of medications, hospital admissions, and specialist/diagnostic medical services. The costs of medications were derived according to the dispensation regimen: drugs dispensed to outpatient or drugs dispensed in the hospital setting. For drugs dispensed to outpatients that were reimbursed by the NHS, we used the gross expenditure paid by each LHU, in order to minimize local variations. For the drugs dispensed in the hospital setting, we used the real cost, including value added tax (VAT), paid by the NHS.
Results
Prevalence of SRA
From January to December 2013, anonymous data from 5,627,226 individuals were available in the ARCo database; 4,654,780 were adults (aged 18 years or over). Of these, 1,894 fulfilled the criteria defined to identify SRA patients. Demographics for the SRA cohort are shown in Table I. The mean age of patients was 50 ± 14 years old and 62.2% of the cohort was female.
Characteristics of the population analyzed
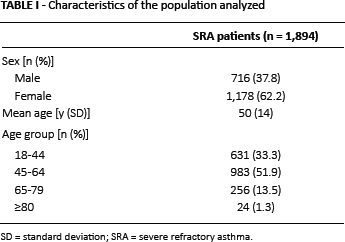
SD = standard deviation; SRA = severe refractory asthma.
Figure 1 shows the prevalence of SRA among an Italian adult population by age group and sex. Overall, SRA prevalence was 0.04%. The highest prevalence estimations were retrieved in female patients aged 45 to 64 years (0.08%); whereas among men, the highest estimation was found in the 45-64 age group (0.05%).
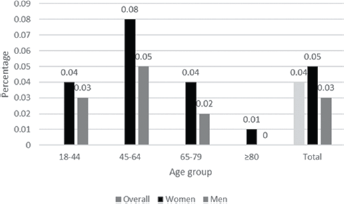
Prevalence of SRA in Italy by age group and sex in 2013-2014.
Health care resource utilization
A summary of the drugs used to treat SRA patients can be found in Figure 2. Almost 80% of patients were treated with prednisone during the follow-up period. Betamethasone 1 mg and methylprednisolone 16 mg accounted for 15.9% and 15.0% of SRA patients, respectively. Omalizumab 150 mg was used in 3.4% of cases, while omalizumab 75 mg was used in 0.8% of SRA patients. The most used drugs for obstructive airway diseases (ATC R03) were salbutamol (40.5%), followed by montelukast (34.9%), and beclometasone/formoterol (28.7%). The mean cost per patient associated with drug treatment in SRA patients was €1,139.
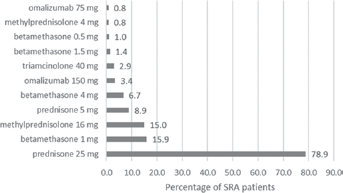
Specific drugs and systemic corticosteroids used by SRA patients in Italy during 2014.
Hospital admissions for asthma occurred in 1.8% of SRA patients. The mean cost per patient for hospital admissions during the follow-up was €900; out of this, €757 was due to asthma-related admissions.
Outpatient services (medical visits and other diagnostic services) had a cost per patient of €777. The most frequent health care service used by SRA patients was “medical visits” (71.9%), followed by “laboratory tests” (66.0%), and “x-ray/scintigraphy exams” (36.0%). Overall, the health care cost (related or not to asthma) of SRA patients was €2,815 per patient per year.
Focus on omalizumab
To investigate the effect of omalizumab in the management of SRA patients, we analyzed health care costs and cost drivers in the subgroup of SRA patients treated with omalizumab. In the SRA cohort (n = 1,894), the costs were almost equally distributed among cost drivers. Drug prescriptions represented 40% of costs, hospital admissions 32%, and specialist/diagnostic health care services 28%; whereas in SRA patients treated with omalizumab (n = 55), the major driver was “drug prescriptions” accounting for 92% of the overall health care cost. Nevertheless, in this subset, costs related to hospital admissions and specialist/diagnostic services reduced to only €449 and €442, respectively, while drug utilization costs increased to €9,989 (Tab. II).
Health care cost drivers in the management of SRA in Italy (comparison between the overall SRA patient cohort [n = 1,894] and the omalizumab-treated SRA patient cohort [n = 55])

SRA = severe refractory asthma.
In addition, the study described the changing in management of patients treated with omalizumab following the re-classification of this drug by the NHS from in-hospital administration to outpatient distribution. Following this change, 83 further patients treated with omalizumab from January to March 2014 were enrolled in the study and were followed up for 9 months. We observed that patients in continuous therapy (n = 72) had a mean of 11.9 omalizumab therapy cycles during the 9-month follow-up (3.8 units per month), which accounted for €8,510. During the follow-up, only 18.1% of patients under omalizumab treatment were tested for IgE.
Discussion
Our study showed that SRA has a prevalence of 0.04% in the Italian population and that each patient costs €2,815 per year to the Italian NHS. This is the first study that assessed the prevalence and burden of SRA in Italy in a real-world setting.
Our analysis found that omalizumab-treated patients had a completely different pattern of health care utilization: reduced costs of hospital admissions and outpatient services, but increased expenses of drug utilization.
Among asthma subjects – representing around 6% of the adult Italian population (12) – not only do SRA patients present the most severe asthma phenotype, but also they are the most difficult to diagnose due to different definitions in the literature, and the complex distinction between difficult-to-control asthma and SRA (5). Such factors might explain the lack of studies assessing the prevalence of this specific cohort in Italy. The PROXIMA study has been designed to assess the prevalence of severe persistent allergic asthma patients treated according to Step 4 of the Global Initiative for Asthma guidelines (13), but the results are not yet available. Our study investigated the number of SRA patients currently treated in Italy by using a large database of real-world evidence (more than 5 million people). We found that SRA affected 0.04% of the Italian population with the highest prevalence among women aged 45 to 64 years (0.08%). Women in this age group represent an important part of the active population, with increased risk of comorbidities. Thus, direct and indirect costs related to severe asthma are expected to be higher in this age group. A recent study analyzed the prevalence of SRA in the Netherlands by using the definition of the Innovative Medicine Initiative consensus and information from Dutch pharmacy databases (14). The analysis showed that the prevalence of SRA is higher in the Netherlands than in Italy (0.1% vs. 0.04%). Of note, the prevalence of SRA determined by both the Italian and the Dutch study was lower than the prevalence reported in the literature for the past years (5). When discussing these differences, Hekking and colleagues (14) pointed out a probable underestimation of difficult-to-control asthma by clinicians that resulted in the misclassification of SRA patients.
SRA is difficult to control and the management of the disease is expected to have a significant economic impact on national health care systems, although few studies have used real-world data to quantify the burden of its management. To our knowledge, our study is the first in Italy to investigate the costs derived from treating SRA using an integrated approach based on data from the real world. Our results showed that health care costs in SRA patients are almost equally distributed between three drivers – (i) drug prescriptions; (ii) hospital admissions; and (iii) specialist/diagnostic services – even if medications are responsible for the largest part of the health care expenditure. Overall, the management of SRA in Italy costs €2,815 per patient per year. These findings are comparable with the results from a 2015 study designed to assess the burden of SRA in the UK (15). O'Neill and colleagues (15) analyzed data from 596 SRA patients from the British Thoracic Society Difficult Asthma Registry and concluded that the major cost driver was medication. Of note, asthma medication expenses (£1,800-£2,200) were similar to nonmedication expenses (£1,300-£2,000), which included outpatient services and hospital admissions. Overall, health care costs among SRA patients in the UK ranged from £2,912 (low-cost scenario) to £4,217 (high-cost scenario). A direct comparison between these values and those obtained in our study is not possible due to the differences between the Italian and the British national health care systems, the drug prices, and the reimbursement strategies (16, 17).
The introduction of omalizumab in the treatment of severe uncontrolled asthma has improved the management of patients that fail to respond to inhaled corticosteroid/LABA treatments (18). We decided to analyze data from the subset of SRA patients treated with omalizumab to assess costs and health care utilization among these patients. Direct comparisons cannot be made between the overall patients affected by SRA and those treated with omalizumab, due to different sample sizes and potential clinical differences (omalizumab-treated patients may represent a particular clinical entity which may, per se, be associated with increased consumption of health care resources). Nevertheless, our subanalysis identified interesting points that should be further investigated. First, SRA patients treated with omalizumab represented a high economic burden with a low utilization of NHS resources. Indeed, these patients were associated with low expenses for hospital admissions and specialist/diagnostic services, but with high medication-related costs. Following changes in omalizumab classification (from in-hospital administration to outpatient distribution), omalizumab-related costs increased. One possible reason could be that clinicians recognized the benefits of this treatment. Although IgE testing is important to assess treatment response and to guide clinicians in their therapeutic approach, our analysis found, surprisingly, that only 18.1% of patients were tested for IgE during follow-up.
We are aware that this study presented some limitations. The main drawback was the missing information on specific diagnosis with related stages of disease in administrative databases. This limitation called for establishing diagnosis proxies that could underestimate or overestimate the disease prevalence. However, until the specific information on diagnosis is collected in Italian administrative databases, this methodological approach remains a unique strategy to conduct studies on Italian real-world data. Concerning the specific identification of an SRA diagnosis, the proxies used in this analysis, to date, have represented the best strategy for selecting patients affected by this peculiar form of asthma disease.
Moreover, due to the lack of information regarding adherence to treatment and inhalation technique, a patient misclassification could have occurred. However, we believe that in Italy severe uncontrolled asthma patients are closely monitored by clinicians and nurses, and that any potential nonclinical reason that may interfere with asthma control is promptly identified and corrected before the prescription of oral corticosteroids. Another limitation is that our study did not consider indirect costs of SRA. Although this did not interfere with the results obtained for the direct burden of the disease, an assessment of indirect expenses would provide a more comprehensive picture of the social weight of SRA. Finally, this study considered prevalent SRA subjects; therefore, it did not take into account the differences in handling specific phases of the disease (initial or long-term).
Conclusions
Our study, based on a large database covering about 10% of the Italian population, gives an important contribution to the understanding of SRA management in Italy and the relevant integrated health care resource utilization. Therefore, it is the first study able to calculate the prevalence and the health care costs of SRA in Italy using real-world data. Our results showed that, for the management of SRA, the Italian health care system spent €2,815 per patient per year, with considerable employment of health care resources. Omalizumab, as well as other new, upcoming biological entities (i.e., mepolizumab, reslizumab, benralizumab), may change the profile of health care cost drivers in SRA by reducing health care utilization. However, studies on a larger number of omalizumab-treated patients are needed to confirm this hypothesis.
Footnotes
Acknowledgment
The authors thank Dr. Carlo Piccinni, researcher at CORE (Collaborative Outcome Research), for his assistance in revising the manuscript.
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
