Abstract
Background
Short-term memory (STM) decline in breast cancer patients resulting from chemotherapy was evaluated by means of blood biomarkers, a questionnaire, and a computerized STM test.
Methods
This study was conducted from January 2013 to June 2013, recruiting 90 subjects: 30 breast cancer patients beginning the 3rd of 4th cycles of docetaxel and cyclophosphamide chemotherapy, 30 recovered patients (who completed 4 cycles of docetaxel for a minimum of 6 months), and 30 healthy subjects (disease-free females). The levels of hemoglobin, red and white blood cells, and cortisol in serum, and a computerized STM test were analyzed to estimate the effects of chemotherapy on STM. A questionnaire was given to all subjects to assess quality of life.
Results
Statistically significant differences were observed for the blood parameters (hemoglobin, red and white blood cells, and cortisol levels) between healthy and on-treatment subjects (respectively 13.47±0.96 g/dL vs 5.37±0.38 g/dL, 4.58±0.41 1012/L vs 2.07±0.13 1012/L, and 6.15±1.03 109/L vs 0.86±0.41 109/L). Scores of the STM test were significantly lower for patients compared to healthy subjects. As indicated by the results of the questionnaire, breast cancer patients had a higher tendency to forget than healthy controls (X2=3.15; p<0.0001) and recovered subjects (X2=3.15; p<0.0001).
Conclusion
We found depleted levels of hemoglobin, red and white blood cells as a result of chemotherapy, and elevated levels of stress correlated with poor performances in the computerized STM test. A higher cortisol level might be an important precursor of STM deterioration. Monitoring cortisol would be beneficial for evaluating the quality of life of breast cancer patients on chemotherapy.
Introduction
Human working memory is comprised of long-term memory (LTM) and short-term memory (STM) (1, 2). The LTM is important for human interactions, relations, study, and work; it is stored endlessly and deeply inside the human mind (3–5). Healthy humans have normal functioning memory, which through reflection or recalling of specific information allows for information kept deep inside the memory box of the human mind to be seamlessly brought forth (4, 5). The STM keeps information for a short period of time in the memory box of the mind (1, 2). Unlike LTM, STM marks small pieces of information and stores that information in the upper levels of the memory box for swift extraction (4, 6). The STM involves tracking information such as appointment times, location of items, and phone numbers and is vital for work and everyday life. (4) The STM can be affected by both the external and internal environment. Restlessness and insomnia may cause minor disruptions to STM functions, while critical illness requiring treatment will lead to STM decline and disruptions to daily life (7–9), and will bring about brief interruptions and frustrations.
Breast cancer and the respective treatment is one example of a critical illness that can affect STM functions in patients (10, 11). Breast cancer is the leading cause of female death in both developing and developed countries. Patients diagnosed with breast cancer face with different treatment options, of which surgery for removal of the tumor(s) and chemotherapy are common considerations (12). Chemotherapy works by eradicating not only cancerous cells but also rapidly dividing healthy cells. In this instance, chemotherapeutic regimens will temporarily paralyze the immune system (13). Besides the common side effects, such as fatigue, nausea, and vomiting (14–17), patients also often complain of deteriorated STM.
Post-chemotherapy cognitive impairment (PCCI)–-also referred to as chemotherapy-induced cognitive dysfunction–-describes a decline in cognition as a result of chemotherapy treatment for cancer (12, 18). This situation was made known by the large number of breast cancer survivors who commented on their changes in memory, fluency, and other cognitive abilities that impeded their ability to function compared to the period prior to receiving treatment (19). A decline in cognition is accompanied by anxiety, depression, and deterioration in memory functions, in particular STM (19–22). Several studies reported that stress is correlated with increases in cortisol, and that such increases affect the brain and memory functioning (23–27). Furthermore, higher levels of cortisol will allow more toxic effects of chemotherapy to penetrate into the brain through the blood-brain barrier (23), leading to dysfunction in memory and cognitive performances. It has been reported that gender influenced the association of hemoglobin to cognition (28): women are more affected by changes in hemoglobin levels compared to men. Although the mechanism is not clearly understood, the association between hemoglobin and cognition has been widely accepted (28–30). The mechanisms linking the lower hemoglobin levels with worse cognition in breast cancer patients might be due to impaired red blood cell production, and/or increased red blood cell destruction. Lower erythropoietin levels may increase the risk of neuronal degeneration in certain cognitive pathways (20).
In this study, the levels of cortisol and hemoglobin were observed. A questionnaire and a computerized version of the STM test were given and used to evaluate the STM functions in breast cancer patients in comparison to healthy individuals and recovered breast cancer survivors.
Materials and Methods
Subject recruitment
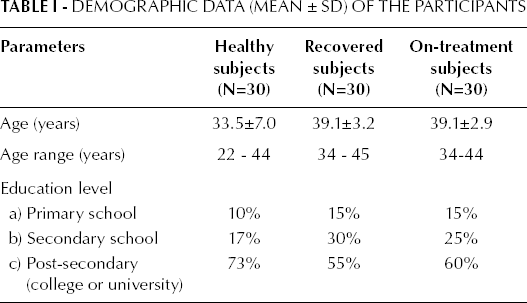
This study was conducted from January 2013 to June 2013 under the study protocol UW 12-046 approved by the Hong Kong University/Health Authority Hong Kong West Cluster Institutional Review Board (HKU/HA HKW IRB) and Hong Kong Institute of Education Research Ethics Review (FRHDC:12/13-ER016). A total of 90 subjects were recruited, comprising 3 groups: 30 patients on chemotherapy (aged 34-44), 30 post-chemotherapy treatment patients (aged 34-45), and 30 healthy subjects (aged 22-44). All subjects were recruited from UNIMED Medical Institute. Subjects' demographics are presented in Table I. All subjects consented to participate prior to the start of the study.
Demographic Data (Mean ± SD) of the Participants
Subjects' inclusion and exclusion criteria
For inclusion in the chemotherapy study group, subjects had to meet the following criteria: (i) be female breast cancer patients with pathological diagnosis of invasive ductal carcinoma; (ii) be aged between 30 and 50 years; (iii) be at the start of cycle 3 (of a total of 4 cycles) of docetaxel (75 mg/m2) and cyclophosphamide (600 mg/m2) chemotherapy treatment.
For inclusion in the group of post-treatment patients, subjects had to meet the following criteria: (i) be female breast cancer patients; (ii) have been pathologically diagnosed with invasive ductal carcinoma; (iii) have completed 4 cycles of chemotherapy treatment (docetaxel [75 mg/m2] and cyclophosphamide [600 mg/m2]) for at least 6 months; (iv) be aged between 30 and 50 years.
For inclusion into the healthy controls' group, subjects had to meet the following criteria: (i) be females aged 20 to 50 years; (ii) be free of a cancer history. Subjects included in the healthy group were selected from a pool of female subjects undergoing health screening.
For all 3 groups the exclusion criteria were: (i) diagnosis of systemic diseases (diabetes, hypertension, cardiac vascular disease, etc.); (ii) being regular drinkers or having a past history of heavy episodic alcohol intake; (iii) being regular smokers or have a past history of smoking; (iv) having mental disorders or psychiatric illnesses.
Complete blood count and hemoglobin measurement
All subjects had peripheral venous blood samples collected by direct venipuncture from the arm vein: 3 mL of blood were drawn in EDTA tubes and clot blood tubes (BD Vacutainer, NJ, USA) for complete blood count by an automated blood system (SYSMEX, XS-800i, Japan), and for cortisol test by an automated multi-analyte analyzer (COBAS INTEGRA 400, Roche Germany). The normal ranges of blood parameters were derived from the Oxford Handbook of Clinical Medicine (31).
Measurement of cortisol in serum
All subjects in this study had blood drawn in the morning, prior to taking medications, for a cortisol test. Serum cortisol levels were measured by the auto-analyzer COBAS 400. The normal range for cortisol was determined for the time range 7-10 am: 171-536 nmol/L (32).
Questionnaire design and adaptation
Questionnaire
The Cognitive Failures Questionnaire, referenced from the study by Broadbent et al (33), is a self-reported assessment in perception, memory, and motor function in the previous 6 months. The questionnaire, which originally consisted of 25 questions formulated in English, was later fully translated into Chinese and was revised till the final version consisting of 20 questions. Some questions were revised to update older terms with current vocabulary: “typewriter” and keyboard” were replaced respectively by “computer” and “keypad”. All questions pertained to events that would occur in everyday life, such as “Do you have trouble remembering whether you have turned off the light, or closed a window in your home?” or “Do you have trouble remembering a task that you intended to do?” or “Do you find yourself holding your phone but forgetting who you intended to call?”. The questionnaire's goals are of determining: (i) whether STM is affected; and (ii) the frequency that subjects forget common instances in their everyday lives. This allows making a comparison among healthy controls, the chemotherapy study group and the post-chemotherapy treatment group. To ensure consistent answers across all subjects and groups, the 5 available responses–-never (N), occasionally (O), sometimes (S), frequently (F), and always (A)–-for the questionnaire were defined as: never = not at all; occasionally = occurrence within 4 weeks after the completion of cycle 2 through cycle 4 of chemotherapy; sometimes = monthly occurrence; frequently = weekly occurrence; and always = everyday occurrence. Each group of subjects were asked to reply to the questions according to a specific timeframe: breast cancer patients were to fill in the questions with respect to 4 weeks after completion of cycle 2 to cycle 4 of chemotherapy (docetaxel and cyclophosphamide); healthy subjects and post-treatment subjects were to reply to the questions with respect to the last 6 months.
Computerized STM test
Source of memory test, content, and requirements of the test
All subjects were asked to complete a 25-trial computerized test of STM (Coglab). The STM test, including forward digit span and word span, was obtained from a set of classic experiments and concepts in cognitive psychology (34). Subjects are presented a list of 5 stimuli-items on screen for 1 second and asked to recall the list in the order presented as to examine the word length and phonological similarity effects on span performance. The 5 stimuli included numbers, letters that sound different, letters that sound alike, short words, and long words. The answer was regarded as “correct” if the items were recalled in the presented order. Any mistakes (recalling too many items, recalling too few items, or recalling items in the wrong order) were counted as “incorrect”. No amendments of the inputted answers were allowed.
Data analyses
All calculations were performed using SPSS 17 for windows (SPSS Inc., U.S.A.). A test of means (one-way analysis of variance [ANOVA] and chi-square test) was applied to compare the blood parameters of hemoglobin, red and white blood cells, and cortisol among the 3 groups. Comparison of the responses of the questionnaires among the groups was tested by the chi-square test, as were also the correct answers obtained form the computerized STM test. A p value of <0.05 was considered statistically significant.
Results
Blood parameters (hemoglobin, red and white blood cells) for indication of fatigue and cell nutrition
For healthy subjects the mean hemoglobin level was 13.47±0. 96 g/dL, while red and white blood cell counts were, respectively, 4.58±0.41 10 12 /L and 6.15±1.03 10 9 /L (Tab. II), all within the normal range. In recovered subjects the mean hemoglobin level was 12.68±0.89 g/dL, while red and white blood cell counts were, respectively, 4.22±0.29 10 12 /L and 4.49±0.42 10 9 /L, slightly below the results for the healthy group, but still within the normal range. For on-treatment subjects, the mean hemoglobin level was below the normal range (5.37±0.38 g/dL), as were red and white blood cell counts, (2.07±0.13 10 12 /L and 0.86±0.41 10 9 /L, respectively) (Tab. II). Significant differences were observed for the levels of hemoglobin, red and white blood cells (p<0.05) between the healthy subjects and the group of on-treatment patients. Numerical differences were observed for blood parameters between recovered and on-treatment subjects, albeit the absence of statistical significance.
Values of Tested Blood Parameters (Hemoglobin, Red Blood Cells, White Blood Cells, and Cortisol) for the Healthy Subjects' Group (N=30), Recovered Patients' Group (N=30), and On-Treatment Patients' Group (N=30)
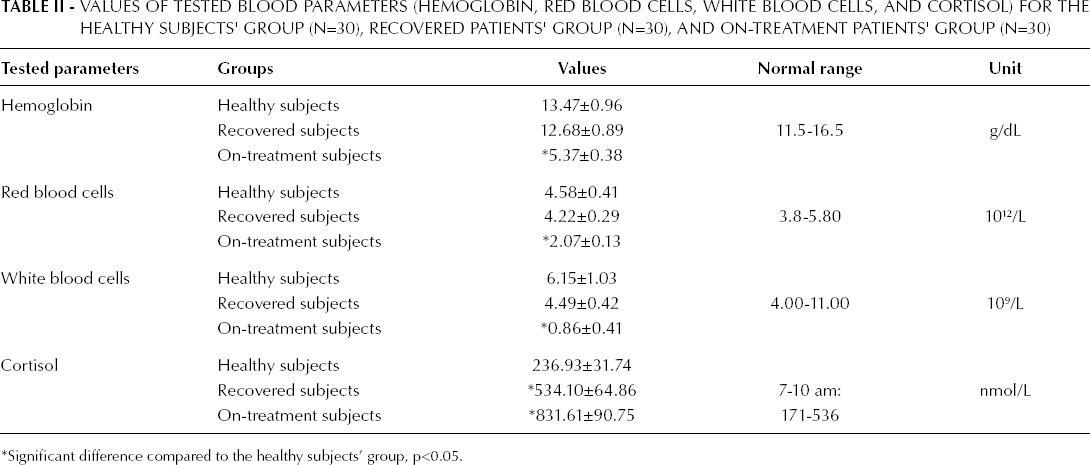
Significant difference compared to the healthy subjects' group, p<0.05.
Cortisol levels as indication of stress
In healthy subjects the cortisol levels were all within the normal range (236.93±31.74 nmol/L). For recovered subjects the cortisol levels were, on average, right within the normal range (534.10±64.86 nmol/L), although higher than healthy subjects (Tab. II). The levels of cortisol in the on-treatment group were well above the normal levels, reaching 831.61±90.75 nmol/L (Tab. II). The ANOVA test comparing the cortisol levels between the healthy and on-treatment subjects' group, as well as the recovered and on-treatment group revealed a computable significant difference (p<0.05).
Responses to the questionnaire and impacts on daily life
A comparison of the responses to the questionnaire, computed by means of a chi-square test, between the healthy and the on-treatment group indicated a highly significant difference (X 2 =3.15; p<0.0001). Amongst all responses to the questionnaire, the response “never” was most frequently selected by the healthy subjects' group. The frequency at which the “never” reply was selected by the control group far exceeded the on-treatment group, corresponding to a total of 307 (76.8%) compared to 66 (16.5%) responses. For the on-treatment group, the reply “occasionally” was the most selected amongst all 5 available responses, corresponding to 148 (37%) compared to 71 (17.8%) responses for the group of healthy controls (Tab. III).
Distribution of Questionnaire Replies and Chi-Square Test Results Among the Three Groups
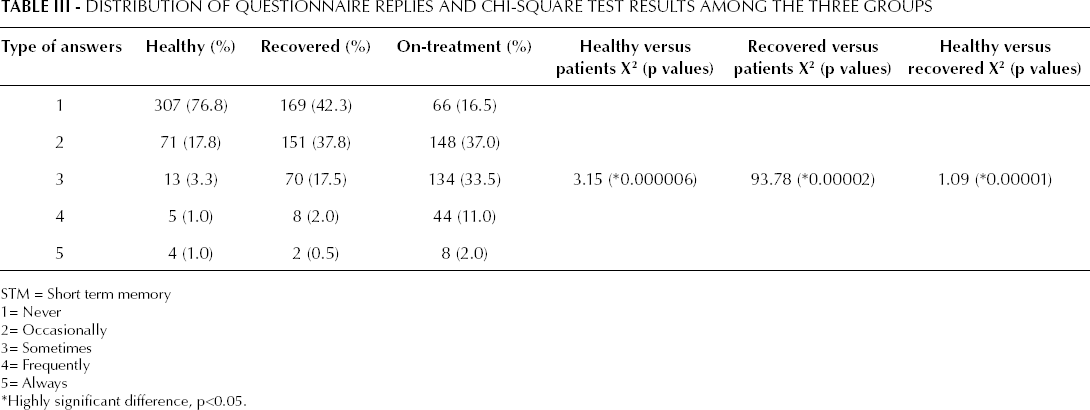
STM = Short term memory
1= Never
2= Occasionally
3= Sometimes
4= Frequently
5= Always
Highly significant difference, p<0.05.
A comparison between the recovered and the on-treatment groups also revealed a statistically significant difference in the distribution of responses for the questionnaire (X 2 =93.78; p<0.0001). For the recovered subjects' group, the most selected response was “never”, corresponding to a total of 169 (42.3%) compared to 66 (16.5%) (Tab. III). The most selected response by the on-treatment patients' group in comparison to the recovery group was “occasionally” (148 [37%] vs 151 [37.8%] selected responses).
The chi-square test further confirmed a statistically significant difference in the distribution of the questionnaire responses of the healthy subjects' group in comparison to the recovered subjects' group (X 2 =1.09; p<0.0001). The response “never” was the most selected response by both the healthy subjects' group and recovered subjects' group, accounting for, respectively, 307 and 169 responses (Tab. III). Amongst all available responses “occasionally” was the second most selected in both the abovementioned groups, accounting for 71 responses among the healthy subjects, and 151 among the recovered subjects' group.
Effects of the 5 stimuli on STM
The comparison between the frequency of correct answers to the STM test, for the category “numbers”, between the healthy control group and the on-treatment patients' revealed a highly significant difference (X 2 =34.69; p<0.0001, Tab. IV). The percentage of correctly answered responses for the category “numbers” was 98% and 66%, respectively for the healthy and on-treatment groups. Similarly, a highly significant difference was also observed for the stimuli “numbers” when comparing recovered and on-treatment patients (X 2 =24.50; p<0.0001, Tab. IV). In the computerized STM test for the stimuli “numbers” the total percentage of correct responses was 94% and 66%, respectively for the recovered and on-treatment subjects’ group.
The Percentage of Correct Results for the Short-Term Memory Test Among the Three Groups
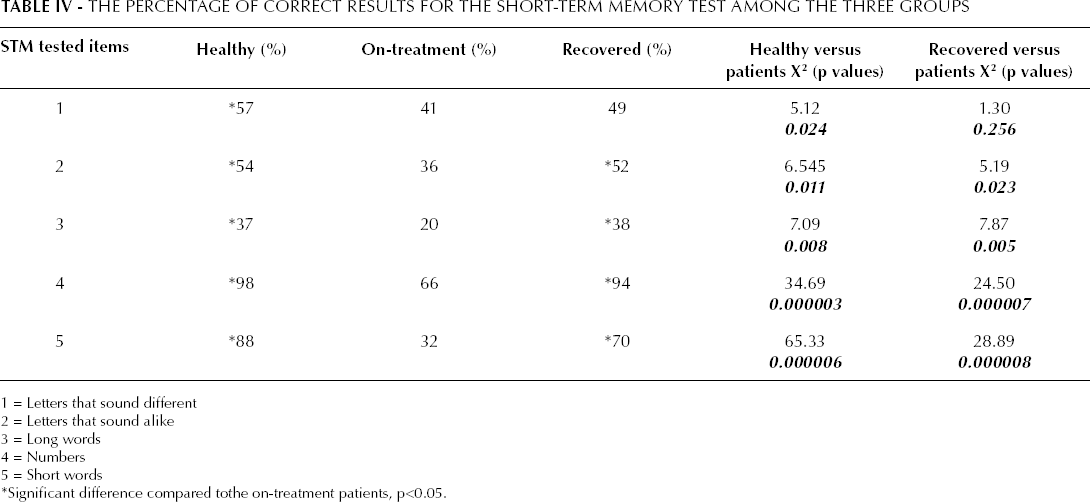
1 = Letters that sound different
2 = Letters that sound alike
3 = Long words
4 = Numbers
5 = Short words
Significant difference compared tothe on-treatment patients, p<0.05.
The difference in the percentage of correctly answered questions for the stimuli “short words” between the healthy and on-treatment patient groups was found to be extremely significant (X 2 =65.23; p<0.0001, Tab. IV). The percentage of correctly answered questions corresponded to 88% and 32%, respectively for the healthy and on-treatment groups. Similarly, the results for the tested “short words” stimuli for the recovered and on-treatment groups exhibited a high statistically significant difference (X 2 =28.89; p<0.0001, Tab. IV). The percentage of correct responses for the “short words” stimuli was 70% for recovered subjects and 32% for on-treatment patients.
Similarly, statistically significant differences for the stimuli “letters that sound alike” (X 2 =6.545; p=0.011) and “long words” (X 2 =7.09; p=0.008) were found between the healthy and on-treatment groups, as well as between recovered and on-treatment patients (X 2 =5.19; p=0.023 for “letters that sound alike” and X 2 =7.87; p=0.005 for “long words”) (Tab. IV). A comparison of the stimuli “letters that sound different” revealed a statistically significant difference only between the healthy and on-treatment groups (X 2 =5.12; p=0.024). Comparisons between the healthy and recovered subjects for all 5 tested stimuli showed a lack of statistically significant differences in the results of the computerized STM test (Tab. IV).
Discussion
In recent years, the advancements in treatment and screening for breast cancer patients have led to increased survival rates (35). With the increased number of survivors, reports of cognitive dysfunctions have emerged, although reports vary between patients of different age groups, educational levels, and menopause status. The common domains of impaired cognitive functions include disruptions to STM, poor concentration, and sluggish motor functions (36, 37). Similar reports have also been made by breast cancer patients during chemotherapy treatment. Both breast cancer patients and survivors have indicated difficulties in learning and recalling new information, an important aspect of STM function. This phenomenon has led researchers to study the effects of chemotherapy on cognitive impairment in cancer patients and survivors, known as PCCI (12, 18).
With respect to the effects of chemotherapy on breast cancer patients, a number of clinical trials have been conducted, beginning in 1995. After much research, it is still not completely clear how chemotherapy leads to cognitive impairment, but the causes are believed to be multifactorial with common risk factors (35). A higher-dose chemotherapy regimen or the use of a combination of chemotherapy regimen has been predicted to lead to more toxic effects and cause more severe effects on cognitive function than standard-dose chemotherapy or the use of a single chemotherapy agent. Higher doses of chemotherapy will also penetrate the blood-brain barrier more easily than standard-dose chemotherapy (36–38). The common risk factors of STM disruptions in breast cancer patients receiving chemotherapy are: low levels of hemoglobin as well as red and white blood cells, and high cortisol levels (11).
The results of the study questionnaire and computerized STM test administered to the healthy subjects, recovered patients, and breast cancer patients undergoing treatment, confirmed the correlation between the effects of chemotherapy and a decline in STM memory. In response to the questionnaire, breast cancer patients on chemotherapy, who are characterized by low hemoglobin and red blood cells' counts, responded that they “occasionally”, “sometimes”, or “frequently” forgot everyday occurring events as the 3 most selected replies. Questions were framed with the STM function in mind, such as “Do you tend to read something and forget it quickly, so you have to read it again?”; “Do you find yourself holding your phone but forgetting who you intended to call?”. By vast contrast, healthy individuals responding to the questionnaire most often selected “never” as their response, corresponding to 76% of all answers. Also breast cancer survivors most often chose “never” in response to the questionnaire (42.3%), while this response only corresponded to 16.5% of all responses for on-treatment patients.
The questionnaire results imply that STM functions of breast cancer patients treated with repeated chemotherapy are significantly worse than healthy individuals and breast cancer survivors. Thus, repeated chemotherapy treatment for patients will be combined with low levels of hemoglobin and red blood cells, and with depleted STM performance (29). As demonstrated in this study, statistically significant differences were observed in the hemoglobin levels, and the red and white blood cells' counts between the on-treatment group and the other 2 groups. The study conducted by Jacobsen et al concluded that patients with hemoglobin levels below or equal to 12 g/dL showed more fatigue and negative changes in cognitive tasks (29). The patients receiving chemotherapy in this study were characterized by mean levels of hemoglobin below 6 g/dL. The patterns of the questionnaire results, namely that patients “occasionally”, “sometimes”, or “frequently” forgot everyday occurring events can be attributed to STM disruptions of chemotherapy treatment-depleted hemoglobin (29). Levels of hemoglobin above 12 g/dL were not demonstrated to affect memory functions.
The levels of hemoglobin, red and white blood cells in patients under the influence of chemotherapy treatment were below the reference range and were lower than those of both the healthy and post-treatment groups. The absence of statistically significant differences between on-treatment patients and healthy subjects in terms of blood tests' results can be explained by the fact that the effects of chemotherapy may not be permanent. In the months following the completion of treatment, the recovery process begins: the body can then effectively deliver oxygen to tissues and vital organs. However, during treatment, breast cancer patients will experience depleted levels of hemoglobin, red and white blood cells' counts, but these side effects will be reversible after the end of the treatment.
For a majority of breast cancer patients, the traumatic experience of facing a major physical change will cause posttraumatic stress disorder and psychological problems. The most prevalent problems reported include depression, stress, and anxiety (39). The amount of stress in patients is detectable by elevated levels of cortisol in the body, most accurate if measured from 7 am to 10 am in the morning (40, 41). In line with current research, breast cancer patients receiving chemotherapy treatment in this study had significantly higher levels of cortisol compared to the healthy individuals, an indication that breast cancer patients experience massive amounts of stress during the treatment period rather than healthy individuals. Measurement of cortisol in breast cancer survivors (post-chemotherapy subjects) compared to the healthy individuals of this study showed that the level of stress in both groups was within the normal range, despite a higher level of cortisol in survivors. The higher level of stress in survivors, while still within the normal range, might be attributed to the fear of disease recurrence, the need to receive treatment, the possibility of death (13), and the financial burden of cancer treatment.
A few explanations may serve to shed light on the low STM test scores of breast cancer patients compared to healthy subjects and recovered patients. A low blood circulation decreases nutrient uptake and oxygen available for vital organs, especially the human brain. If an insufficient level of oxygen and nutrients is supplied to the brain, the neurotoxic effects of chemotherapy will likely lead to more cell death. An increased strength or a combination of chemotherapeutic agents administered to patients will also lead to higher neurotoxic effects. A higher-dose chemotherapy will penetrate more easily the blood-brain barrier (36–38), thus leading to stronger effects of STM impairment, in addition to the consequences on other important domains of cognitive functions.
The contrasting results of the STM tests between the on-treatment group and the healthy and post-treatment groups can also be explained by observing the elevated levels of cortisol in patients. Research has demonstrated that increased levels of cortisol affect brain anatomy (24, 25, 42) and are associated with detrimental memory functioning (26, 27). In the computerized test of STM, subjects are made aware of the results after completing each trial, for a total of 25 trials. In answering the questions incorrectly, patients may feel anxious and insecure. These subjects feel more anxiety and stress during the test than healthy individuals and breast survivor counterparts of this study (23). Repeatedly answering the trials incorrectly will only cause the level of stress, anxiety, and frustration to increase in patients. Frustration may result in the patients' inability to recall or remember the tested items correctly, thus leading the subjects to continuously answer the trials incorrectly. This will further deter patients from being able to concentrate on the trial, as the attention and focus may be placed on the previous incorrectly responded trial. Our results for the patients on cycle 3 of 4 cycles of chemotherapy are also supported by Wieneke and Dienst's study (43); the authors compared a group of patients to healthy subjects and showed that the cognitive performance of 75% of the patients was below that of the healthy group. Clinical trials conducted by Tannock et al (19) and Brezden et al (44) also indicated that cognitive deficits, STM function, and the ability to remain focused or organized will also be disrupted as a result of chemotherapy treatment for breast cancer patients.
Impairment in STM memory, although subtle, can significantly affect the daily life of breast cancer patients (12, 21, 29, 35). A thorough analysis of the questionnaire and responses of breast cancer patients will give a better understanding of the areas affected. Collectively, breast cancer patients in our study selected “occasionally”, “sometimes”, and “frequently” as the 3 top responses of the questionnaire. These answers were responses to questions pertaining events that occur every day, not simply daily life but also social events. Forgetting 1 or 2 of the mentioned items may seem quite minor in nature, but for the studied patients' group these events occurred simultaneously. Disruption of STM on everyday occurrences at home, an environment in which patients should feel most comfortable, will affect the quality of their daily life.
The results of this study showed that normal STM functions are affected by the toxicity of chemotherapy, the psychological factors connected with the diagnosis of breast disease, and the physiological factors affecting breast cancer patients. Depleted levels of hemoglobin, and low red and white blood cells' counts, combined with high levels of stress during the course of treatment led to significant STM impairment in breast cancer patients. STM decline, although subtle, can significantly affect the quality of daily life of these patients. The application of tests for cortisol, hemoglobin, and STM as predictors of STM and quality of life deterioration could be further elaborated as a longitudinal study and be beneficial for evaluating the quality of life of breast cancer patients on chemotherapy or breast cancer survivors.
Footnotes
Acknowledgments
We would like to thank Dr. Michael Yip and his team for their contributions in analyzing the psychological aspects of this research.
