Abstract
Hypothesis. Withania somnifera is an herb with antioxidant, anti-inflammatory, anticancer, antistress, and adaptogenic properties. Previous studies have shown its antistress effects in animals. Traditional Indian medicine has used it for centuries to alleviate fatigue and improve general well-being. Methods. This is an open-label prospective nonrandomized comparative trial on 100 patients with breast cancer in all stages undergoing either a combination of chemotherapy with oral Withania somnifera or chemotherapy alone. The chemotherapy regimens were either taxotere, adriamycin, and cyclophosphamide or 5-fluorouracil, epirubicin, and cyclophosphamide. Withania somnifera root extract was administered to patients in the study group at a dose of 2 g every 8 hours, throughout the course of chemotherapy. The quality-of-life and fatigue scores were evaluated before, during, and on the last cycles of chemotherapy using the EORTC QLQ-C30 (Version 3), Piper Fatigue Scale (PFS), and Schwartz Cancer Fatigue Scale (SCFS-6). Results. The median age distributions in the study and control arm were 51 years (range = 36-70) and 50.5 years (range = 32-71), respectively. The majority (77%) of patients had stage II and III disease. Patients in the control arm experienced statistically significant higher estimated marginal means of fatigue score compared with the study group (P < .001 PFS, P < .003 SCFS-6). Furthermore, various symptom scales of the EORTC QLQ-C30 were statistically significant in 7 out of 18 symptoms in the intervention group compared with the control group (P < .001). The 24-month overall survival for all stages in study and control group patients were 72% versus 56%, respectively; however, the result was not significant (P = .176), at a median follow-up duration of 26 months. Conclusions. Withania somnifera has potential against cancer-related fatigue, in addition to improving the quality of life. However, further study with a larger sample size in a randomized trial is warranted to validate our findings.
Introduction
Breast cancer is the most common cancer among women and is the leading cause of cancer death. It accounts for 23% (1.38 million) of new cancer cases and 14% of cancer deaths worldwide. 1 In North America, 1 in 8 women develop breast cancer in their life time. We see the same trend in the developing countries, including Malaysia. According to the first National Cancer Registry data reported in 2002, female breast cancer accounts for a third (30.4%) of all cancer cases in Malaysia. 2 The only difference between American and Malaysian breast cancer epidemiology is the stage prevalence. In Malaysia, most of the breast cancers presented at the late stages compared with early stages in the United States.
The primary modality of management is the removal of primary tumor and regional lymph nodes by surgery. Radiotherapy is given to sterilize micrometastasis in loco-regional sites to achieve local control. However, the modern Fisher’s principle gives insight about the systemic nature of breast cancer, which states that “invasive breast cancer is a systemic disease from its beginning.” Therefore, most breast cancer patients are treated using systemic cytotoxic chemotherapy with or without hormone and biological agents. The types of chemotherapy are based on various risk factors described in National Comprehensive Cancer Network guidelines.
Chemotherapy drugs are normally administered in combination. The most common combination regimens are cyclophosphamide, methotrexate, and 5-fluorouracil; cyclophosphamide, doxorubicin, and 5-fluorouracil; adriamycin and cyclophosphamide (AC); cyclophosphamide, epirubicin, and 5-fluorouracil (CEF); AC followed by taxotere; taxotere, adriamycin, and cyclophosphamide (TAC); or a variety of combinations of other cytotoxic drugs. Combination chemotherapy is invariably associated with side effects, namely, alopecia, leukopenia, skin pigmentation, and vomiting. Fatigue is the most disabling and least reported side effect. Cancer treatment–related fatigue (CTRF) is encountered in more than 70% of patients. 3 It is an unexplained tiredness following the administration of chemotherapy, 4 and the fatigue is not relieved by rest. Barbara F. Piper defined fatigue as a multidimensional symptom complex, consisting of behavioral severity, affective meaning, and sensory and cognitive components. 5 CTRF is quantitatively measured using Piper’s Fatigue Scale (PFS) or Schwartz’s Cancer Fatigue Scale (SCFS-6). 6 Fatigue is the main cause of poor compliance to cancer treatment or discontinuance of effective chemotherapy. 7
The conventional treatment for CTRF is physical exercise, correction of hemoglobin level, psychotherapy, cognitive behavioral therapy, and psychostimulants. 8 However, there is no accepted evidence-based guideline for the management of CTRF. Withania somnifera (Syn. Ashwagandha, Dunal, Indian Winter cherry) is a common medicinal herb that is grown ubiquitously throughout Asia, being used in the Indian system of traditional medicine (ayurveda) for more than 3000 years. 9 The Withania somnifera (WS) plant and its root contain numerous medically useful chemical components. 10 The most common are steroidal lactones, the withanolides (eg, withaferin A). The root, however, provides alkaloids along with 18 fatty acids, β-sitesterol, polyphenols, and phytosteroid. 11 In ayurveda, WS is used as extracts of the whole plant or its root.
The extract can be used as a single agent or in combination with other herbs. Very recently, WS has drawn attention in modern medicine. Numerous basic research studies concerning cytotoxicity assays and induction of apoptosis have appeared in the literature. 12 Experimental animal studies have shown adaptogenic and antitumor properties. WS also results in psychological and emotional well-being through its positive influence on brain chemistry. 13 In a clinical trial in pediatric subjects, WS improved memory and enhanced cognitive functions, which are vital components of fatigue. 11 In animal models, WS could regulate neuronal function through the acetylcholine receptor 14 ; WS also has a GABA mimetic effect, thus producing an anxiolytic effect. 15 The extract of WS alleviates reactive depression without causing sedation and optimizes mental functions and psychomotor performance by easing mental stress. 16
WS extract has been used in humans for immunostimulation and as an aphrodisiac agent. Recent studies in cell culture and animal studies showed that WS has restorative, radiosensitizing, antioxidant, hematoprotective, and anticancer properties.17,18 Ashwagandha extract is also active against breast cancer and colorectal cancers cell lines. 19 In another animal study, WS showed protection of the kidney and the bone marrow against cyclophosphamide- and paclitaxel-induced end-organ toxicity.20,21 However, in the existing medical literature, the human use of WS in clinical trials is limited. 9 The common use of WS is for aphrodisiac effect and in the combination therapy for Parkinsonism.12,22 In ayurvedic practice, WS has been tried on all age groups of patients in various dose schedules along with water or milk. So far, there has been no adverse effect observed in therapeutic doses. In this clinical trial, we proposed to use WS extract for the treatment of chemotherapy-induced fatigue and preservation of quality of life (QoL) among breast cancer patients.
Materials and Methods
This is a prospective open-label nonrandomized comparative clinical trial conducted between October 2007 and October 2010. The institution’s research and human ethics committee reviewed and approved the proposed research protocol. Breast cancer patients meeting the selection criteria were entered into the study. Patients eligible for systemic chemotherapy and willing to participate in the clinical trial were assigned alternately into the study or control group in a 1:1 ratio. Thus, the allocation of treatment arm was unbiased rather than according to the investigators’ preference. The inclusion criteria encompass histopathology proof of breast cancer, Eastern Cooperative Oncology Group (ECOG) performance status 0 to 2, and TNM stage I-IV disease. Patients with a previous history of cytotoxic chemotherapy and/or radiotherapy, concomitant psychotropic drugs, uncontrolled systemic illness, or psychiatric disorder were excluded from this trial. The intent of chemotherapy was for both as adjuvant and palliation. All patients were scheduled for intravenous systemic chemotherapy, were willing to voluntarily participate in this trial, and signed the consent form.
Pretreatment Evaluation
Patients screened for the study were documented into a case report form. The patients were evaluated according to the Union for International Cancer Control (UICC) TNM staging system. The examination findings, proof of tissue diagnosis, computed tomography/magnetic resonance imaging scan, and radionuclide bone scan findings were documented for each patient. Before each cycle of chemotherapy, a full blood count and liver and renal function tests were performed. The baseline ECOG performance status, EORTC QLQ-C30, PFS, SCFS-6 score were recorded. Baseline Hospital Anxiety and Depression Scale (HADS) 23 was also documented prior to chemotherapy.
Fatigue Assessment
Fatigue was assessed with 2 standard fatigue assessment tools developed by Barbara F. Piper and Schwartz.5,6 PFS determines 4 defined dimensions of fatigue, whereas SCFS-6 is a straightforward 5-question instrument. EORTC QLQ-C30 also has a fatigue component. The PFS scores were assessed on the first day of the first cycle, the third cycle, and the sixth cycle of chemotherapy, whereas SCFS-6 was evaluated on day 1 of every cycle of chemotherapy (for a total of 6 cycles). EORTC QLQ-C30 was also documented on day 1 of every cycle of chemotherapy. The validated QoL, HADS, and fatigue instruments were verified by one of the coauthors (HCI, a psychiatrist and QoL expert), and a trained research nurse and research assistant collected the data.
Chemotherapy
The indication for combination chemotherapy was for adjuvant or palliative intent. The combination chemotherapy regimens consisted of either CEF (cyclophosphamide 500 mg/m2 + epirubicin 70 mg/m2 + 5-fluorouracil 500 mg/m2) or TAC (taxotere 75 mg/m2 + adriamycin 50 mg/m2 + cyclophosphamide 500 mg/m2). Both chemotherapy regimens were administered intravenously on day 1 and repeated every 3 weeks. Standard antiemetic medication using ondanstron 8 mg or granisetron 3 mg was administrated intravenously. The chemotherapy protocol chart documents the hematological parameters, cytotoxic drugs, antiemetics, dose schedule, and associated adverse effects in NCI common toxicity criteria. For patients on TAC chemotherapy regimen, prophylactic G-CSF (Neupogen, Roche) 300 µg was administered subcutaneously for 5 days, starting 24 hours following day 1 of chemotherapy.
Administration of Withania somnifera
Withania somnifera is supplied as a powder of WS root extract packed in a veggie capsule containing 500 mg of the active ingredients supplied as ashvagandha (Himalaya Drug Co, New Delhi, India). Each patient in the study group was asked to take 4 capsules (2 g) 3 times daily with water throughout the course of chemotherapy (for 6 cycles). The human therapeutic dose used in this trial was based on the experience from traditional use of WS and earlier clinical trials. The liver and renal functions and full blood counts were monitored before administration of each cycle of chemotherapy. Patients who could not swallow 4 capsules at a time or with gastric upset were allowed to take the herb in a more divided doses, but with the same total dose of herb per day.
Statistical Analysis
All patients were treated and followed up in the intent to treat. All patients’ demographic data were recorded in a spreadsheet. The PFS, SCFS-6, and EORTC QLQ-C30 scores were evaluated for differences between study and control group using SPSS (Version 18, IBM) software. We used repeated-measures ANCOVA to compare the effects of treatment versus control on the QoL over time with age as numerical covariate. Age in years was considered as a numerical covariate. The PFS, SCFS-6, and EORTC QLQ-C30 and the interaction between treatment and time was evaluated. The results are derived from the repeated-measures ANCOVA, and P values are considered significant if it is less than .05. The graphs of the estimated marginal mean adjusted for age in years are shown to depict changes of outcome over time between the study and control arms. The level of significance was set at a P value of .05 (2-sided). The overall survival was analyzed using the Kaplan–Meier nonparametric method in the SPSS program.
Results
The study successfully recruited 50 patients in the study arm and 50 in the control arm. The median age of patients was 51 years (range = 36-72) in the study group and 50.5 years (range = 32-71) in the control group. The racial distribution in the study arm was 33 Malays (66%), 13 Chinese (26%), 1 Indian (2%), and 3 Siamese (6%), whereas in the control group the racial distribution was 43 Malays (82%), 6 Chinese (12%), and 1 Indian (2%). The majority (77%) of the patients presented at stage II and III disease (Table 1). Histopathology evaluation and tumor marker study showed 54% versus 56% ER, 48% versus 50% PR, and 26% versus 52% c-erb overexpression in the study and control arms, respectively. The majority of patients except one in the control group (ECOG PS, 2) had performance status of 0-1.
Patient Variables.

Abbreviations: ER estrogen receptor, PR progesterone receptor, ECOG (PS) European cooperative oncology group performance status
Treatment
The patients in the study arm received encapsulated WS at an oral dose of 2 g 3 times daily throughout the course of chemotherapy (for 6 cycles). Surgery was the initial treatment approach in 72% of the study group and 78% in the control group of patients. Radiotherapy was offered to 90% and 88% in the study and control groups, respectively (Table 2). Patients on taxotere-based chemotherapy were offered 300 µg G-CSF (Neupogen, Roche) prophylaxis from day 2 to day 6 of chemotherapy to 17 patients in the study arm and 24 patients in the control arm. Chemotherapy was repeated every 3 weeks. The complete 6 courses of chemotherapy were received by 44 and 41 patients in the study and control groups, respectively (Table 3). The chemotherapy regimens consisted of TAC and CEF as described earlier.
Treatment Parameters.

Abbreviations: MRM, modified radical mastectomy; BCT, breast conservation technique; CEF, cyclophosphamide, epirubicin, and 5-fluorouracil; TAC, taxotere, adriamycin, and 5- fluorouracil; G-CSF, granulocyte colony stimulating factor.
Treatment Outcome.
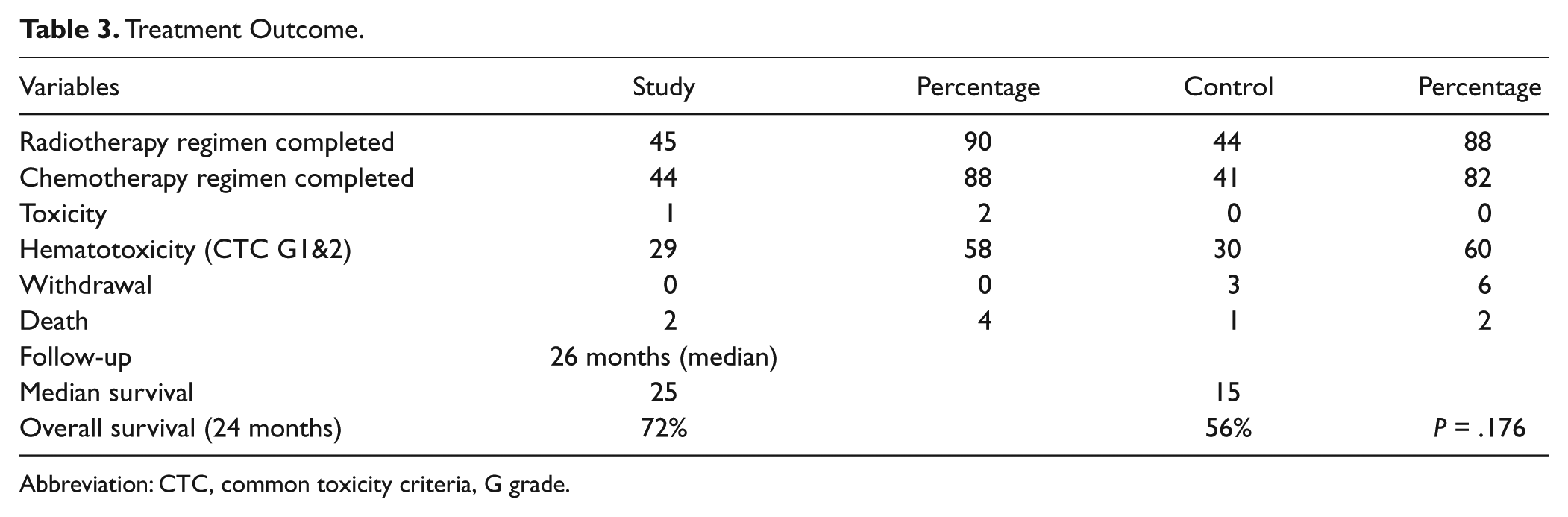
Abbreviation: CTC, common toxicity criteria, G grade.
Fatigue Score
The treatment-induced fatigue was evaluated using PFS, SCFS-6, and the Fatigue domain of the EORTC QLQ-C30 questionnaire. The SCFS fatigue score was determined on every cycle of chemotherapy on day 1; however, PFS score was evaluated in day 1 of the first, third, and sixth cycles of chemotherapy. The median SCFS fatigue score in study arm was 10 to 12 points (interquartile range) and in the control group of patients 11 to 15 points (interquartile range). The marginal means of fatigue on the PFS were 3.16 and 2.08, respectively, in the control and study groups, which was statistically significant (P < .001); the marginal means of fatigue in SCFS-6 was 13.8 versus 11.8, with a P value of .003 (Table 4 and Figures 1 and 2). In the analysis of the EORTC QLQ-C30 Fatigue domain the differences in the fatigue score are as described in Table 4. The EORTC QLQ-C30 in the following symptoms scales was statistically significant: fatigue, physical functioning, role functioning, insomnia, pain, emotional functioning, social functioning, and global health status.
The Comparison of Adjusted Mean Between Control and Study Groups Based on Repeated-Measures ANCOVA for Important Fatigue and Quality of Life Measures.
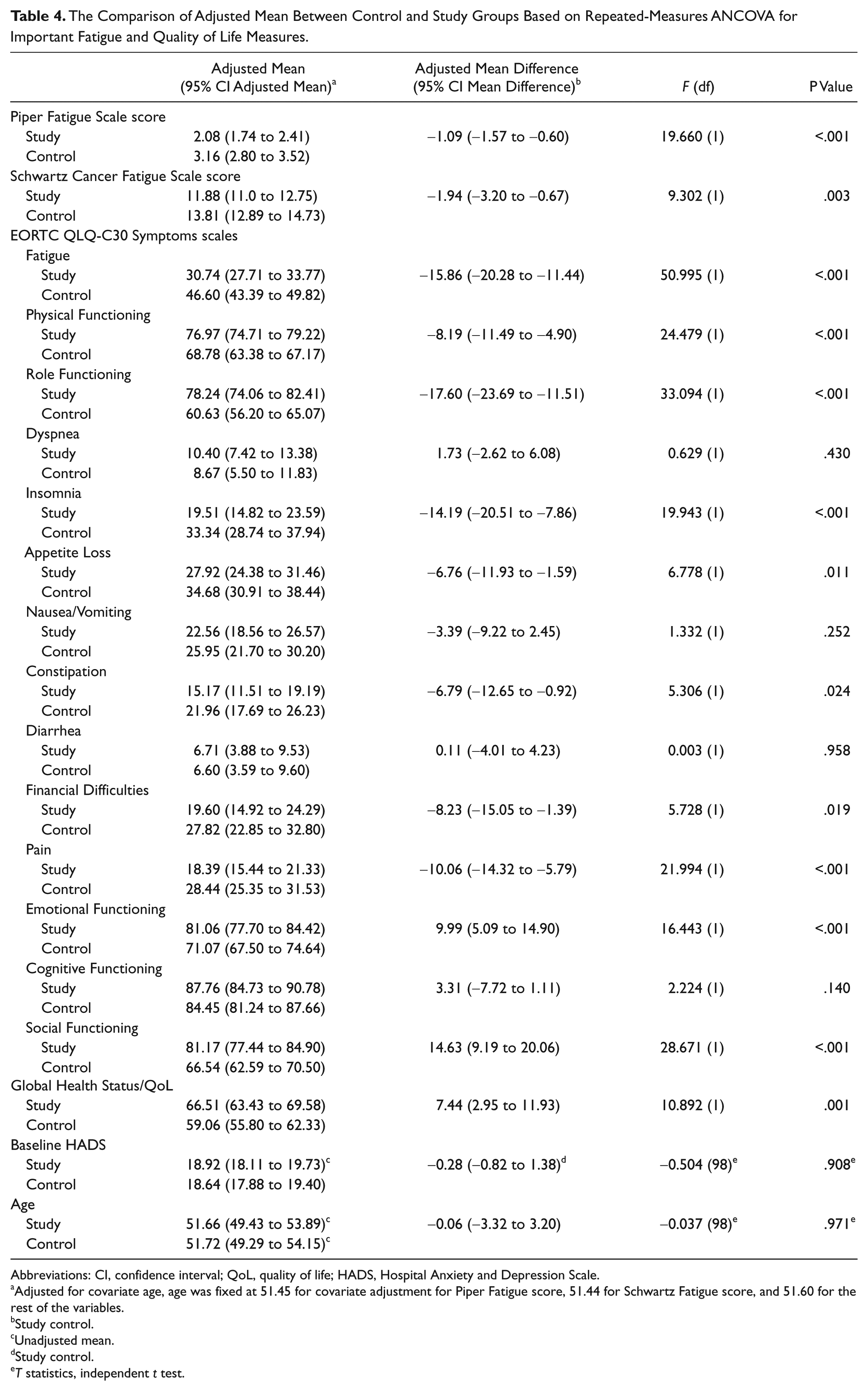
Abbreviations: CI, confidence interval; QoL, quality of life; HADS, Hospital Anxiety and Depression Scale.
Adjusted for covariate age, age was fixed at 51.45 for covariate adjustment for Piper Fatigue score, 51.44 for Schwartz Fatigue score, and 51.60 for the rest of the variables.
Study control.
Unadjusted mean.
Study control.
T statistics, independent t test.

Trend of estimated PFS marginal mean fatigue scores.
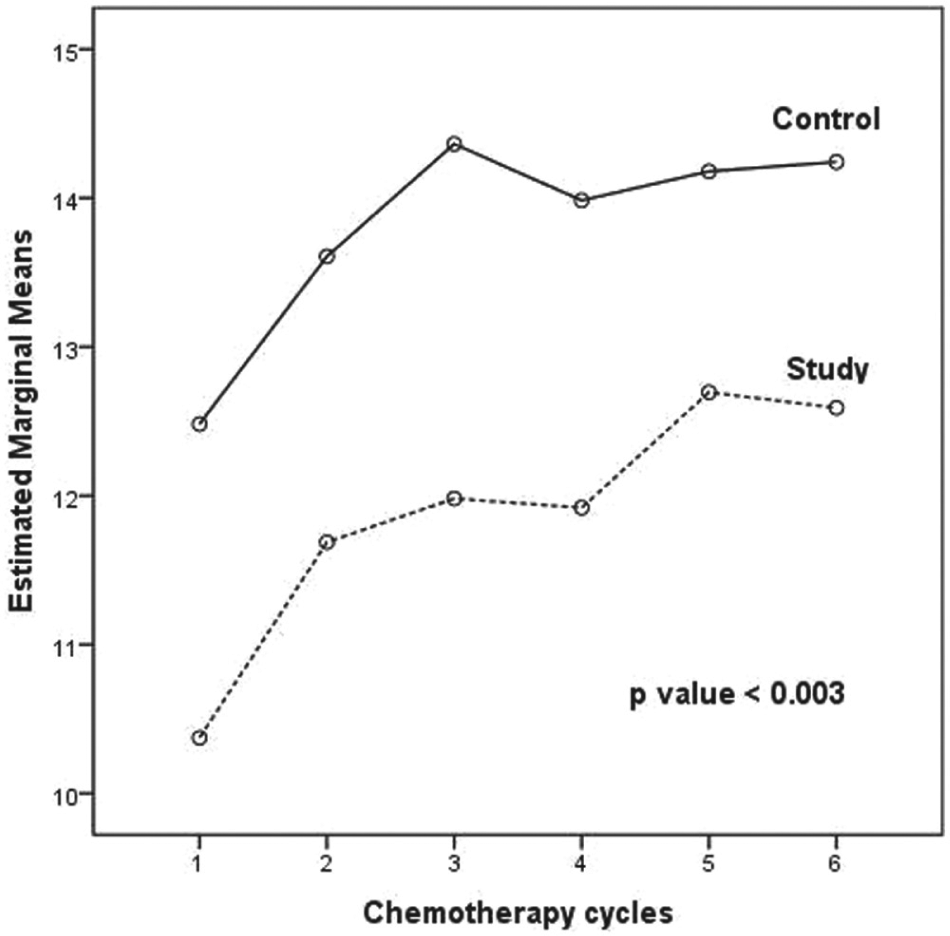
Trend of estimated SCFS-6 marginal mean fatigue score.
The possible psychological disturbances were evaluated using the pretreatment HADS 23 questionnaire. The median baseline HADS score in the study and control arms were estimated at 19 (range = 11-25) and 18 (range = 13-26) points, respectively. The difference in the HADS scores of the study and control arms was nonsignificant (P = .918). The ECOG performance status was 0-1 in 98% of the patient population. The general QoL measures in EORTC QLQ-C30 consisted of 5 functional scales and 9 symptom scale/items. In the repeated-measures ANCOVA method, physical functioning (P < .001), role functioning (P < .001), dyspnea (P = .430), insomnia (P < .001), loss of appetite (P = .011), nausea (P = .252), constipation (P = .024), diarrhea (P = .958), financial difficulties (P = .019), pain (P < .001), emotional functioning (P < .001), cognitive functioning (P = .140), and social functioning (P < .001) components of the EORTC QLC-C30 were favorable in the study group when compared with the control group (Table 4).
Compliance to Treatment
In terms of compliance, 45 versus 44 patients completed radiotherapy and 44 versus 41 completed chemotherapy in the study and the control arms, respectively. This trend was favorable to WS-treated patients though the value was not statistically significant. The oral intake of WS was acceptable to 98% of study patients and chemotherapy was incomplete due to refusal of 3 patients in control arm compared with none in study group. The nonacceptance of WS medication was due to oral intolerance, gastritis, and flatulence. Three patients withdrew from the study in the control arm and none in the study arm. There were 2 deaths in the study arm and 1 in the control arm group of patients. The causes of death were unrelated to cancer or their therapy, including the study agent. The leukocyte counts during the course of chemotherapy were similar in both arms.
We followed-up all patients every 2 to 3 months with clinical and radiological evaluation. The median follow-up duration at the time of analysis was 20 months, with a median overall survival rate of 26 months (range = 3-72 months) and 15 months, respectively, in the study and control arm patients. According to the Kaplan–Meier survival plot, the 24-month overall survival was 72% (95% confidence interval [CI] = 20.81-77.18) versus 56% (95% CI = 14.24-39.75). The above results were not statistically significant (P = .176) but the trend of the plot was in favor of the study arm (Figure 3). A similar trend was also observed when segregated stage by stage; however, we did not include survival as our endpoint due to small sample size in Stage 1 and Stage 4 and because it is out of the scope of our research.

Kaplan–Meier survival plot showing overall survival for all stages.
Discussion
In this study, we found higher fatigue scores among patients in the control group compared with the study groups of patients receiving WS. The fatigue scores were from the PFS, SCFS-6, and the Fatigue domain of EORTC QLQ-C30 questionnaire. The median fatigue score according to the SCFS-6 in the study arm was 10 to 12 points (interquartile range) and 11 to 15 points (interquartile range) in the control arm. Thus, the trend of fatigue was favorable for WS compared with controls.
The estimated marginal mean of SCFS-6 was 11.88 (95% CI = 11.00-12.74) in study arm compared with 13.81 (95% CI = 12.89-14.73) in the control arm. The results were statistically significant (P < .003; Table-4). While evaluating the estimated marginal mean of PFS using ANCOVA, the values were 3.16 (95% CI = 2.80-3.52) in controls compared with 2.06 (95% CI = 1.74-2.46) in the study arm, with P value less than .001. Similarly, in the QoL evaluation the domains of fatigue, physical functioning, role functioning, insomnia, pain, emotional functioning, and social functioning were statistically significant in favor of the intervention group.
WS may be a potential agent for improvement of QoL and amelioration of fatigue. The difference in the fatigue scores may be due to the presence of confounding variables. The other factors that could influence the outcome are anemia, tumor volume, stages of disease, and physical activities of the patient. In this study, the pretreatment evaluation of mean HADS revealed similar values in the control and study arms (18.92 vs 18.64), and the values were not statistically significant (P = .918). The purpose of evaluating the HADS was to determine if the patients were suffering from any psychological distress, which might influence fatigue outcome.
In a series reported earlier, the incidence of CRTF was 14% to 96% in patients undergoing cancer treatment and 19% to 82% among patients after completion of treatment. 8 Several studies have reported worse fatigue among cancer survivors compared with noncancerous control population, as noted in a review. 24 The management of CTRF is evolving. The policies are fundamentally divided into pharmacological and nonpharmacological interventions. The most widely used interventions are psychostimulants: methylphenidate, dextroamphetamine, premoline, modafinil, armodafinil, and caffeine. Despite wide usage of stimulants, there is a paucity of randomized clinical trials. From earlier evidence, methylphenidate alone showed statistically significant improvement in fatigue. 25 The other newer psychostimulants are yet to show any statistical improvement of chronic cancer fatigue. However, the side effects of psychostimulants are manifold in terms of anorexia, insomnia, euphoria, paranoia, and cardiac complications.
Wanchai et al critically reviewed the interventions used in cancer fatigue, and they concluded that exercise, education counseling, sleep therapy, and complementary therapy were feasible as effective options to improve QoL among breast cancer patients suffering from cancer related fatigue. 26 In a preliminary study, Bower et al showed improvement in CRTF in 11 breast cancer patients who participated in the 12-week practice of Iyengar traditional yoga. These results support acceptability of the above therapy and suggested that it may have beneficial effects on persistent posttreatment fatigue. However, considering its limited sample size, the interpretation is questionable and thus warrants a larger sample size for further study. 27 Other nonpharmacological approach includes cognitive behavioral therapy, which showed improvement in amelioration of fatigue in a small group of the patient population. 28
In our study, we used WS with the hope to improve QoL by reducing cancer fatigue. WS contains steroidal alkaloids and steroidal lactones in a class of constituents called withanolides. So far 12 alkaloids, 35 withanolides, and several sitoindosides from this plant have been isolated and studied, namely, withaferin-A and withanolide-D. 12 These ingredients are concentrated in the root of the WS plant.
The idea of using ashwagandha in cancer fatigue came from the antistress effect of WS. 29 Fatigue is related to chronic stress induced by cancer and its psychological manifestations. Chronic stress can result in numerous adverse physiological conditions including cognitive deficit, immunosuppression, sexual dysfunction, gastric ulceration, and change in serum corticosteroid levels. The antistress properties of WS are evident from numerous animal studies. 11 Research data for its clinical benefit in humans are very limited. The reports are mostly from the anecdotal use of WS in various ailments in Indian traditional medicines for thousands of years, and they are not documented in modern medical literature. 9 The first documented use of WS in a human trial was in a double-blind clinical trial comparing effects of milk fortified with WS in 58 children aged 8 to 12 years. The children were subjected to 2 g of ashwagandha per day over a period of 60 days. In this study, subjects treated with WS showed a significant increase in mean corpuscular hemoglobin and serum albumin level. The authors concluded that WS is a growth promoter and has anti-anemic properties. 30 However, there was no difference in hemoglobin levels in both groups of patients on chemotherapy in the present study.
In an interesting animal model study, Davis and Kuttan studied the hematoprotective role of WS among albino mice treated with cyclophosphamide and WS. They observed increased cellularity of the bone marrow among the study group compared with the cyclophosphamide-only treated groups. 31 In another animal study by Gupta et al, pretreatment with oral WS resulted in improvement in white cell count secondary to intravenous paclitaxel to experimental mice. They used paclitaxel at a dose of 1 mg/kg body weight and administered WS 200 mg/kg body weight per day for 4 days. The leukocyte and absolute neutrophil counts were monitored on days 3 and 5. 21 In our study, we monitored our patients’ white blood cell count before every cycle of chemotherapy. However, we did not observe any difference in the leukocyte count in either group of patients. The leukocyte counts of our patients might be affected by those patients who received prophylactic G-CSF in TAC chemotherapy regimen. Furthermore, we did not study the bone marrow cellularity during the course of treatment.
In a study on healthy adults, WS was administered at a dose of 1 g per day for 29 days for mountaineering activities. The psychological and physiological parameters were monitored during their activity. WS improved sleep patterns, responsiveness, alertness, and state of awareness together with physical capabilities. 32 In another recent clinical trial, in healthy adults, 40 subjects were subjected to WS (n = 10), another plant extract of Terminalia arjuna (n = 10), WS plus Terminalia arjuna (10), and placebo capsules (10). In this study, the dose of WS was 500 mg per day for 8 weeks. All subjects were assessed for their physical performance and cardiorespiratory endurance. Both herbal agents improved physical performance, velocity, power, and Vo2 max. So the authors concluded that WS could improve weakness and physical performance. 33 The above-mentioned studies may support the role of WS for the alleviation of CTRF in our patients.
An anticancer property of WS has been extensively studied in in vitro cell culture cytotoxicity assay and on experimental tumors in the animal models. However, evidence of human trials in cancer is nonexistent. To our knowledge, perhaps this is the first clinical trial in the use of WS on breast cancer patients. In an animal study, WS increased the antitumor activity of paclitaxel in a lung cancer model. 34 Withaferin-A, a component of WS, was found to inhibit signal transduction and activator of transcription-3 (STAT3) and induce apoptosis in breast cancer cell culture. 35 In another cell culture assay, WS showed selective killing of cancers by reactive oxygen species–mediated pathways. 17
In our study, we observed similar overall survival figures among WS-treated patients, though the trend is in favor of the study arm. The favorable trend may be by chance or difference in disease load or higher number of HER-2 positive patients in the control arm. In the study and control groups, we had the same number of stage III and IV patients (29 in each arm). More control than study group patients (25 vs 35) received the TAC regimen, which could have favored survival in the control arm. It is also possible that WS enhances the cytotoxicity of chemotherapy mediated through its induction of apoptosis or interference with the estrogen receptor. 36
Use of herbs during chemotherapy is very common (78%) in clinical practice. 37 Interaction of herbs used in complementary and alternative medicine (CAM) with chemotherapy is an important topic of controversy. According to theoretical possibility, there is a strong potential for herb–drug interaction while administering herbs of CAM therapy together with cytotoxic chemotherapy. CAM containing herbs could possibly induce drug metabolizing enzymes (cytochrome-P450) and alter the ATP-binding cassette drug transporter leading to decreased effectiveness of chemotherapy. 38 Herbs could induce unexpected hematotoxicity during cancer chemotherapy and thus decrease compliance to treatment. The best example of herb–drug interaction is in St John’s wort combination with irinotecan and imatinib. Other putative interactions are seen in the use of kava, ginseng, garlic, and echinacea with chemotherapy. On the other hand, Cheng et al reviewed 168 studies on the combination of chemotherapy with CAM therapy, and they found out that the evidence of herb–drug interaction was poor. Thus the authors recommend further trial to establish evidence from clinical trials for the formulation of guidelines to educate oncologists. 39 WS has been studied for herb–chemotherapy interaction in experimental studies. However, animal studies using WS along with cytotoxic drugs paclitaxel and cyclophosphamide did not find it hindered effectiveness of chemotherapy agents; rather, it enhanced cell kill of chemotherapeutic agents. Furthermore, WS could protect bone marrow from chemotherapy-induced hematotoxicity.31,34 Free radical damage is one of the mechanisms of cancer cell kill following exposure to chemotherapy or radiotherapy. In a study, the concurrent use of WS with radiotherapy enhanced radiosensitization. 18 From our study, we did not encounter any higher hematotoxicity or altered liver chemistry despite prolonged course of WS during chemotherapy. We also did not encounter any loss of effectiveness of chemotherapy during treatment. Thus, WS seems to be a safe herb for treatment of cancer patients during chemotherapy.
There are numerous shortcomings that might have affected this study’s outcome. The subjects were mostly advanced stage patients. We used 2 types of chemotherapy regimens with differing tumor load and risk variables, which might have altered the cancer fatigue. In the study arm, 25 patients received TAC chemotherapy regimen compared with 35 patients among controls. The TAC regimen is likely to induce CTRF with the changes in total leukocyte counts. The effect on leucocyte count has been modified due to use of G-CSF treatment to TAC chemotherapy group of patients; hence, the difference in hematotoxicity might not truly reflect the effect of WS.
In conclusion, our preliminary analysis of WS showed improvement in QoL and CTRF, with improvement in trend of overall survival. We were unable to demonstrate hematoprotective effects of WS following cytotoxic chemotherapy compared with controls. The preliminary outcome of this study should be interpreted with caution due to limited sample size and numerous confounding variables. Further study in the future with adequate sample size, stratified randomization for stages of cancer and risk groupings, and identical chemotherapy regimens could convincingly show a difference in the outcome with WS as adjunctive cancer therapy.
Footnotes
Acknowledgements
The authors would like to thank Dr Prahlad S. Patki, Head, Medical Services & Clinical Trials R&D, the Himalaya Drug Company, India, for valuable suggestions; Dr Muhammad Irfan Abdul Jalal for reevaluating research data and statistical calculation; Jimmi Hasanuddin (research assistant) for subject allocation and documentation of data; and Mrs Sujata Biswal for encouragement and preparation of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by USM Research University Grant Number 1001/PSK/8120244 (2009-2011).
