Abstract
Despite being already known that p27 can regulate cell proliferation, cell motility and apoptosis, the role of p27 expression in oral squamous cell carcinoma (OSCC) remains controversial. The purpose of this study was to comprehensively evaluate, with a meta-analysis, the clinicopathological and prognostic role of p27 expression in OSCC. A meta-analysis of eligible studies was performed to assess the effects of p27 expression on clinicopathological parameters and overall survival (OS) in patients with OSCC, using pooled relative risks with 95% confidence intervals. Heterogeneity and publication bias were also assessed. Fourteen studies involving a total of 1,010 patients met the inclusion criteria. Low p27 expression was significantly associated with advanced TNM stage (p<0.001), worse histology (p=0.025), and lymph node metastasis (p<0.001), but not with tumor size (p=0.181). The pooled RR of 0.743 (p=0.002) suggested that low p27 expression has a poor prognosis in patients with OSCC. A significant heterogeneity among studies was detected for lymph node status (χ2=34.60, I2=68.2%, p<0.001) and OS (χ2=14.86, I2=39.4%, p=0.095). We did not detect a significant publication bias in this meta-analysis. Our meta-analysis suggests that p27 expression status might be useful as a predictive biomarker in clinical practice, and might potently predict OS in OSCC patients.
Introduction
Oral squamous cell carcinoma (OSCC) accounts for about 90% of all malignant oral lesions, and is the most prevalent malignant tumor of oral structures, with approximately 41,380 new cases diagnosed annually in the United States (1). Despite the improved application of multimodal therapy, the survival rate of patients frequently suffering from locoregional and distant recurrences is still poor and highlights the need for reliable prognostic markers concerning early diagnosis and treatment alternatives (2).
The p27 protein was identified as an inhibitor of the G1 cyclin-dependent kinases (CDKs), its role being to negatively regulate the progression from G1 into S-phase, by binding to and regulating the activity of CKDs (3). Diverse post-translational modifications determine the physiological role of p27. Phosphorylation modulates p27 bindings to cyclin-CDK complexes, whose localization and ubiquitin-mediated proteolysis can drive cell migration and cell proliferation. A study in a knockout mouse model showed that heterozygous p27+/- mice can develop pituitary tumors late in life and have accelerated onset rate and increased number of tumors when exposed to carcinogens (4). Moreover, low expression of p27 has been identified in a wide variety of solid human tumors, including OSCC (5–8). With regard to the associations between p27 expression and clinicopathological parameters in OSCC patients, the results are still conflicting and inconclusive.
In the current study, we conducted a systematic review of the published literature and used a meta-analysis to comprehensively and quantitatively synthesize the results concerning the clinicopathological and prognostic role of p27 in OSCC.
Materials and Methods
Publication search
The electronic databases of PubMed, Embase, and Web of science, as well as the Wanfang data and the China Academic Journal Network Publishing Database were searched for all articles relating to p27 and OSCC (last update: June 2012). Relevant references were also manually screened for articles potentially missed in the initial search. The search strategy included the following key words, variably combined: (i) oral cavity and cancer OR oral squamous cell carinoma OR neoplasm OR carcinoma; (ii) p27 OR p27Kip1. Investigators of eligible studies were contacted for supplementing additional data relevant to our meta-analysis.
Study inclusion/exclusion criteria
Studies were considered eligible if they met all of the following inclusion criteria: (i) investigated the relationship between p27 and clinicopathological parameters or prognosis; (ii) p27 protein expression was evaluated by immunohistochemistry (IHC); (iii) the study was published either in English or Chinese in the periodical literature; (iv) the study provided sufficient data to allow comparison of survival results among articles. The following were the exclusion criteria: (i) articles were review articles, case studies or letters; (ii) studies were conducted on oral cancer cell lines and animal models; (iii) articles were lacking part of relevant data or contained inappropriately presented data. For studies published by the same authors or study group, the newest or most informative single article was selected.
Data extraction
Data were independently extracted by 2 investigators (LG and WG) by means of a predefined form. Potential disagreements were resolved by discussion. The following information was extracted from the eligible studies: first author, year of publication, number of patients, antibody used for IHC, cutoff value for p27 positivity, clinicopathological parameters, and survival results. Differences in data extrapolation were checked by other 2 authors.
Statistical analysis
The impact of p27 expression on clinicopathological parameters and overall survival (OS) was estimated for each study by considering the relative risk (RR), together with its 95% confidence interval (CI). According to their clinical parameters, we combined patients according to the following tumor characteristics: T1 and T2; T3 and T4; stage I and II; stage III and IV; good and moderate tumor differentiation. Data and figures from the original articles were extracted according to the methods described by Parmar et al (9). For the Kaplan-Meier survival curves, 2 authors independently analyzed the curves to reduce the inaccuracy in the extracted OS estimates.
Heterogeneity of the included studies was checked by the chi-squared Q-test. A p-value >0.1 for the Q-test indicates a lack of heterogeneity among studies. When I 2 was <50% the studies with an acceptable heterogeneity were considered, and the fixed-effects model was used for meta-analysis. Otherwise, the random-effects model was adopted (10).
Begg's funnel plot and Egger's regression test were applied to assess potential publication biases (11). When p was >0.05 the study was considered free of publication bias. All statistical analyses were performed using the Statistical Analysis System software (STATA SE 12.0) and Revman version 4.2 review manager software.
Results
Study characteristics
A total of 14 studies (8, 12–24), published from 1998 to 2012, met the inclusion criteria for this meta-analysis, which thus included a total population of 1,010 patients, comprising between 30 and 206 patients per study. All studies were based on data of retrospective analysis. Positive p27 expression varied from 5% to 50% in the enrolled studies. A correlation between p27 expression and clinicopathological parameters was found in 12 studies, of which 10 found p27 to have prognostic value.
Although the expression of p27 was measured by IHC in all publications, the specific IHC protocol varied widely among studies. The most frequently used antibodies were bought from Transduction Laboratories. Different antibody dilutions were employed, ranging between the values of 1:1,200 and 1:20. The cutoff values for positive p27 staining were also different in the studies here considered (ranging between 5% and 50%). The main characteristics of the 14 eligible studies are showed in Table I.
Main Characteristics and Results of the Eligible Studies

IHC, immunohistochemistry; T, tumor size; LN, lymph node status; TNM, TNM stage; ND: no data, NS: non-significant; Negative, negative relationship between p27 expression and survival results.
Association of p27 with clinicopathological parameters
Four studies assessed the relationship between tumor size (T1/T2 vs T3/T4) and p27 expression (Fig. 1). The test of heterogeneity was non-significant (χ 2 =4.64, I 2 =35.3%, p=0.200) and, therefore, a fixed-effect model was used in meta-analysis. The combined RR was 1.244 (95% CI: 0.90-1.71, Z=1.34, p=0.181), suggesting that expression of p27 was not associated with tumor size. In 8 studies we observed that p27 expression in patients with stage III/IV tumors was much lower than those with stage I/II tumors. The combined RR 1.575 (95% CI: 1.35-1.84, Z=5.76, P<0.001) was calculated using a fixed-effect model, with a lack of heterogeneity (χ 2 =8.96, I 2 =21.9%, p=0.256) (Fig. 2). Eleven studies reported the association of p27 expression with histological differentiation. Since there was no significant heterogeneity among the studies (χ 2 =5.99, I 2 =0.0%, p=0.741), the combined RR with a fixed-effect model was 1.354 (95% CI: 1.04-1.77, Z=2.23 p=0.025) for the parameters of good and moderate differentiation vs poor differentiation. This indicates that a low p27 expression is significantly associated with worse OSCC histology (Fig. 3). When analyzing the lymph-node status in OSCC, our analysis revealed that p27 expression was significantly associated with lymph-node status in 12 studies. The combined RR, calculated using a random-effects model was 0.594, and was statistically significant (95% CI: 0.45-0.79, Z=3.52, p<0.001), with a significant heterogeneity (χ 2 =34.60, I 2 =68.2%, p<0.001) (Fig. 4). The detailed RR values and 95% CIs for each clinicopathological factor are shown in Table II.
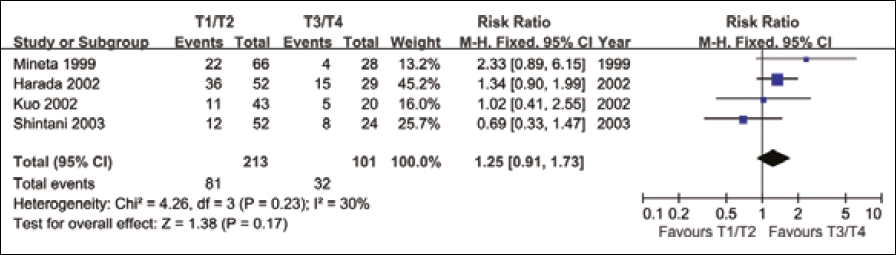
Meta-analysis on the relation between p27 expression and tumor size.
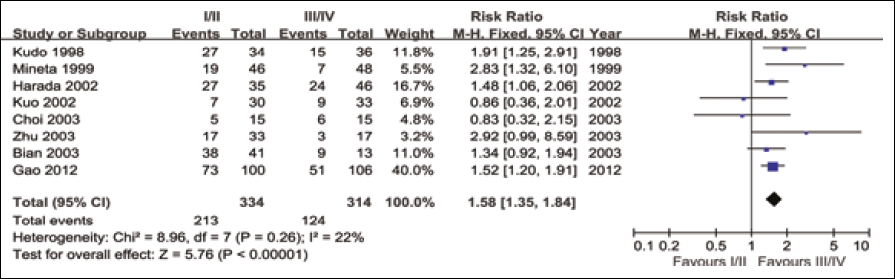
Meta-analysis on the relation between p27 expression and TNM stage.
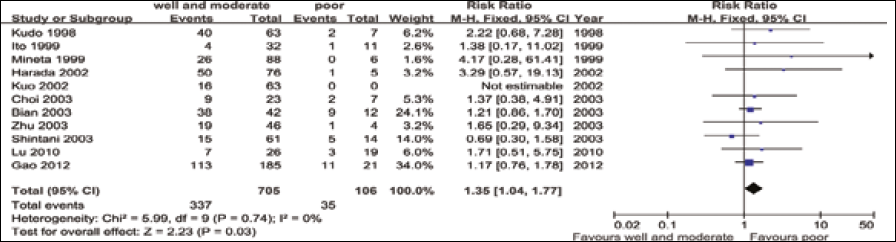
Meta-analysis on the relation between p27 expression and histologic grade.
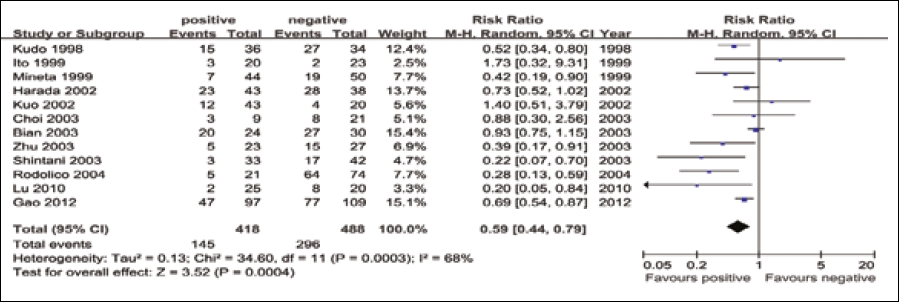
Meta-analysis on the relation between p27 expression and lymph node status.
Meta-Analysis: RR Value and 95% CI of Clinicopathological Factors
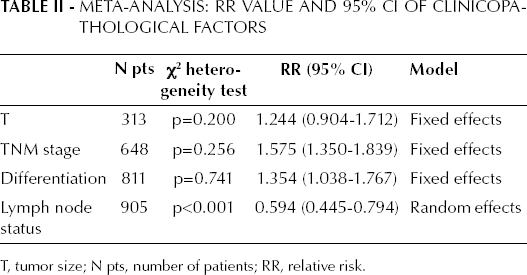
T, tumor size; N pts, number of patients; RR, relative risk.
p27 expression and OS in OSCC
We then considered 10 studies, including 768 patients, as to evaluate the relation of p27 expression and OS. For some studies not reporting the RR and 95% CI, the RR was calculated as described in the section “materials and methods”. A significant heterogeneity was detected across these studies (χ 2 =14.86, I 2 =39.4%, p=0.095), and thus a random-effects model was applied for calculating the RR, which was 0.743 (95% CI: 0.62-0.89, Z=3.17, p=0.002), suggesting that a low expression of p27 was significantly correlated with poorer survival for OSCC patients (Fig. 5).
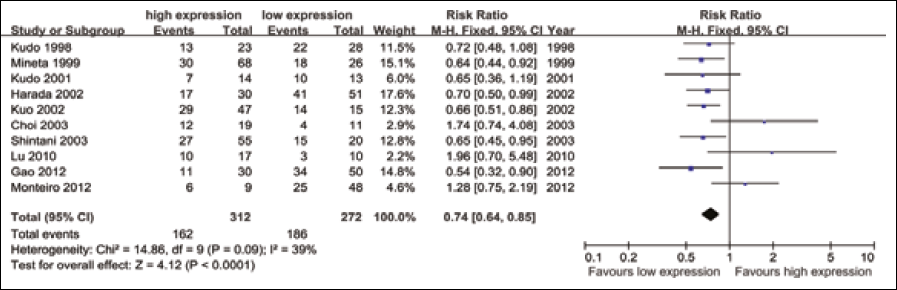
Meta-analysis on the relation between p27 expression and OS.
Publication bias
Both Begg's funnel plot and Egger's tests were applied to assess the existence of a publication bias. Begg's funnel plot did not reveal any evidence of obvious asymmetry in our studies. The z-scores and p-values were respectively: 0.34 and 0.734 for tumor size; -0.12 and 1.000 for TNM stage; 1.25 and 0.210 for histologic grade; 0.21 and 0.837 for lymph node status; and 1.43 and 0.152 for OS (Fig. 6). Similarly, the results of the Egger's test were not suggestive of any evidence of publication bias (p>0.05, data not shown).
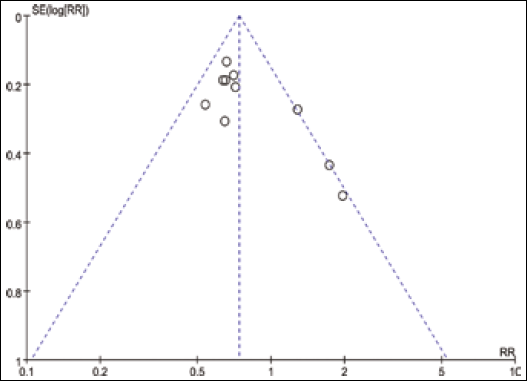
Begg's funnel plot analysis to detect publication bias for OS.
Discussion
Meta-analysis is a quantitative approach in which individual study findings on the same topic are statistically integrated and analyzed. Recently, it has been used for the evaluation of cancer prognostic markers (25). To clarify the clinical role of p27 expression in OSCC, in the present meta-analysis we included 14 eligible studies, altogether comprising 1,010 patients. The studies compared clinicopathological parameters and OS in OSCC patients according to the expression levels of p27 in the primary tumor.
Although mutations in p27 are only rarely found in human tumors, decreased levels of the p27 protein have been associated with tumor size, TNM stage, histology, or lymph node status among patients with head and neck cancer (5), lung cancer (6), or breast carcinoma (7). Our results showed that a low p27 expression, as detected by IHC, was confirmed in patients with OSCC, and that the lack of p27 expression was highly associated with advanced TNM stages, worse histology, and lymph node metastasis, but not with tumor size. The biological mechanism regulating the activity of p27 is still inconsistent. Tomoda et al reported that p27 is a critical component of the cell-cycle machinery (26, 27). Nuclear exportation and subsequent degradation of p27 are induced by interacting directly with the ectopically expressed Jab1, via ubiquitin-mediated proteolysis in a cell cycle-dependent manner; this mechanism promotes cell proliferation. On the other hand, accumulation of cytoplasmic p27 results in increased cell motility and migration through its interaction with RhoA, a GTPase that regulates cell motility by reorganizing actin filaments (28, 29). Besides, Andre et al (30) suggested that in early breast cancer patients treated with anthracycline-based chemotherapy the p27 nuclear/cytoplasmic ratio was predictive of therapeutical efficacy.
In the recent years, the studies conducted on the prognostic value of p27 for OS in patients with OSCC have lead to inconsistent results. Studies have increasingly pointed at a subcellular localization of p27, through cytoplasmic translocation, in human tumors that are associated with poor survival rates (31, 32). The results of our meta-analysis indicate that a reduced p27 expression seems to be an independent prognostic factor for OSCC in terms of OS (Fig. 5). This observation is in agreement with recent meta-analysis conducted in lung cancer (33) and breast cancer (34), and is also supported by findings from previous studies (8, 12, 14–16, 24).
Significant heterogeneity of results among the studies was found for the associations of p27 with lymph node status and OS. All these studies used IHC for assessing the p27 levels in OSCC patients. IHC results, however, depend on various factors, such as the choice of the primary antibody and its dilution, the cutoff value applied, and the protocols used, which are all arbitrary choices made by the investigators. The antibody concentration is an important factor influencing the result of the staining: in the studies here considered the dilution of the antibody ranged from 1:1,200 to 1:20. Besides, the cutoff points defining the levels of p27 expression varied from 5% to 50% among the studies, a factor that may have contributed to the heterogeneity of the results. We thus performed a further analysis within a subgroup of studies which used cutoff values >10%. In this case the heterogeneity of lymph node status was reduced, and the heterogeneity of OS was eliminated, thus we assumed that 10% may be the most appropriate cutoff value in defining the positive expression of p27 in OSCC patients. Furthermore, we could not fully neglect the differences in terms of length of follow-up, and the inconsistencies in clinical and pathological features.
Although we did not detect a significant publication bias among the studies by applying the Begg's and Egger's tests (p>0.05), other limitations of this analysis should be acknowledged. First, this meta-analysis was restricted to studies published in English and Chinese, which probably led to a publication bias. Secondly, studies reporting negative results are less frequently published or, similarly, were excluded from our meta-analysis due to the lack of detailed data. Finally, if the authors did not provide sufficient data for direct calculation of survival rates, we extracted the data ourselves from the survival curves using several time points during the follow-up period.
In conclusion, our meta-analysis suggests that expression of p27 is associated with histologic grade, TNM stage, and lymph node status in OSCC patients. Besides, low p27 expression was confirmed to be a poor prognostic factor for OS in patients with OSCC, an observation that can help screening earlier high-risk patients and may guide clinical individual therapy. However, this conclusion should be interpreted cautiously when introduced into clinical practice. Therefore, we believe that the clinicopathological and prognostic roles of p27 in OSCC should be confirmed, in the future, by an adequately designed prospective study, including appropriate multivariate analysis.
