Abstract
Purpose
S100P has been shown to participate in processes of various human malignancies. In this study, we analyzed the tissue expression of S100P in gastric cancer and evaluated its significance.
Methods
We determined the S100P expression in 156 gastric cancer patients by quantitative RT-PCR. Tumor characteristics and overall survival (OS) for each patient were examined. In vitro experiments were conducted to examine whether ectopic expression of S100P modifies the proliferation and drug resistance of gastric cancer cells.
Results
Higher expression of S100P occurred in human gastric cancer tissues in comparison with normal controls. Highly expressed S100P in gastric cancer was correlated with TNM stage and prognosis. The 5-year survival rate was significantly lower in patients with high levels of S100P expression than in patients with low levels of expression. Ectopic expression of S100P was associated with an increase in tumor cell proliferation and drug resistance.
Conclusion
The expression of S100P in human gastric cancer tissues was upregulated in comparison with normal controls. By establishing an association between S100P expression and shortened OS, increase in proliferation and drug resistance, this study indicates that S100P may be a useful prognostic marker for gastric cancer patients.
Introduction
Gastric cancer remains a common cause of death worldwide; specifically, it is the second most common cause of death from cancer in Asia (1). Despite rapid progress in the understanding and clinical management of this cancer, the outcome of gastric cancer patients has not improved profoundly. In Europe, the 10-year survival rates of patients diagnosed with gastric cancer range from 7.7% to 23.0% (2). Prognostic biomarkers are needed for clinical care in gastric cancer patients.
S100 proteins constitute a family of intracellular calcium-binding proteins characterized by EF-hand motifs (calcium-binding motifs composed of 2 helixes E and F joined by a loop) (3). Growing evidence shows that aberrant expression of S100 proteins occurs in many tumors and is often associated with tumor progression. Thus, these proteins are potentially important tumor biomarkers and therapeutic targets (4). S100 calcium-binding protein P (S100P), a member of the S100 protein family, was first isolated from human placenta (5, 6). Expression of S100P has been found in a number of tumors including pancreatic cancer cell lines and lung cancer tissues (7, 8), and it was correlated with immortalization of human breast epithelial cells in vitro and progression of breast cancer in vivo (9, 10). S100P expression was associated with the grade of tumors, and it promoted tumor invasion and metastasis (10, 11). Expression of S100P was shown in colorectal cancer tissues compared with lower expression in normal mucosa (12–14), and S100P stimulated cancer cell growth and migration in vitro (13, 14). S100 proteins are useful in the diagnosis of metastatic melanoma in cytology material (15). It is unclear whether S100P might be correlated with the prognosis of gastric cancer patients. In this study, we aimed to detect the expression of S100P in gastric cancer, and more importantly, to demonstrate whether S100P could be used as a biomarker to predict prognosis and drug resistance.
Materials and Methods
Clinical and pathological data
This study was approved by the Institutional Review Board of the General Hospital of the People's Liberation Army and conformed to the provisions of the Declaration of Helsinki. A consecutive series of 156 patients undergoing operation for gastric cancer (from January 1999 to January 2005) were enrolled in a 5-year follow-up till January 2010. The survival time was calculated from the date of surgery to the follow-up deadline or date of death, which was mainly due to carcinoma recurrence or metastasis. Eighty-two noncancerous gastric tissues from 82 patients were obtained from gastrectomies of gastric cancer margins more than 5 cm from the lesion. All participants gave their written informed consent according to the stipulations of the Institutional Review Board of our hospital. Clinical parameters were obtained from the medical records of the 156 patients. Pathological stage was assessed by the tumor-node-metastasis (TNM) classification. Routine chemotherapy was given to those patients with advanced-stage disease after surgery, but no radiation treatment was administered to any patients included in our study.
Tissue samples
All tumors were histologically examined by 2 independent pathologists to confirm the diagnosis of gastric cancer, and then the pathological stages (I-IV) were established. Both normal and tumor tissue samples were obtained immediately after surgery, snap-frozen in liquid nitrogen, and stored at -80°C until further detection.
Patient follow-up
Clinical follow-up after diagnosis and surgery was performed every 3 months during the first year, every 6 months during the second year, and then yearly until relapse. The follow-up was based on clinical and biochemical tests as well as computed tomography scans. In addition, an ultrasonic examination was conducted in the case of impaired liver function. Overall survival (OS) and disease-free survival (DFS) were the study endpoints. OS was defined as the period from time of diagnosis until death. DFS was defined as the interval between diagnosis and first recurrence.
RNA extraction and quantitative real-time RT-PCR
RNA was extracted from 30 mg tissue samples from gastric cancer and normal control samples using TRIzol® (Life Technologies). RNA was quantified by determining absorbance at 260 nm spectrophotometrically. For the first-strand cDNA synthesis, 500 ng of RNA was reverse-transcribed, using a Thermo Scientific RT-PCR kit according to the manufacturer's instructions. Random hexamers were used as primers for cDNA synthesis. Real-time RT-PCR was performed using primers specific for S100P and the housekeeping gene TATA-box binding protein (TBP), which was used to normalize S100P expression. The primer sets for S100P and TBP were designed using Primer Express version 2.0 (Applied Biosystems). The following primers were used: S100P: forward, 5′-AAGGGGGAGCTC AAGGTGCTGA-3′, and reverse, 5′-ATCTGTGACATCTCCAGGGCATC-3′; TBP: forward, 5′-CACGAACCACGGCACTGATT-3′ and reverse, 5′-TTT TCTTGCTGCCAGTCTGGAC-3′. RT-PCR was performed on a 7900HT Fast Real-Time PCR System (Applied Biosystems) using SYBR Green and all reactions were performed in triplicates. Thermal cycling for both genes was subjected to 35 cycles of PCR (denaturing at 94°C for 10 seconds, annealing at 58°C for 5 seconds and elongation at 72°C for 5 seconds). At the end of the PCR cycles, melting curve analyses were conducted, followed by sequencing, to validate the generation of the specific PCR product expected. S100P expression levels were calculated relative to TBP using the 2–ΔΔCt method (16).
Proliferation and drug resistance of gastric cancer cells
The gastric cancer cell line SGC7901 cells were obtained from the American Type Culture Collection and maintained in Dulbecco's Modified Eagle Medium (DMEM). Cells were seeded in triplicate and transiently transfected with a pc-DNA plasmid encoding S100P or the empty vector (both from Santa Cruz), using Lipofectamine 2000 (Invitrogen) according to the manufacturer's instructions. At 24, 48, 72 and 96 hours post-transfection, different fractions were collected for further detection.
Cell proliferation was assessed by 3 different methods. Firstly, cell density was evaluated by means of a cell-counter apparatus (Digital Bio). Secondly, 2×10 4 SGC7901 cells were seeded in triplicates in 96-well plates and subjected to MTT assay (Cayman Chemical). At 24, 48 and 72 hours after transfection, MTT reagent was added and absorbance was detected at 570 nm (Thermo Scientific). The real-time cell electronic sensing (RT-CES) system was used for analysis as determined by cell index as previously reported (17). Cells were transfected when attached (15 hours after seeding), and impedance changes were shown 12 hours after transfection (27 hours after the beginning of the process).
To investigate drug resistance, SGC7901 cells were treated with cisplatin (100 μmol/L) for 36 hours. Subsequently, floating and adherent cells were collected and tested for viability by flow cytometry using the Annexin V-FITC Kit (BD Pharmingen) where cells were resuspended in 1 × binding buffer at a concentration of 1×10 6 cells/mL. The cell suspension (200 μL) was transferred to a 5-mL polypropylene tube, and propidium iodide (5 μL) and Annexin V-fluorescein isothiocyanate (FITC) was added simultaneously. Cells were incubated at room temperature for 20 minutes in the dark.
Statistical analysis
All statistical analyses were performed using SPSS version 16.0 software. Fisher's exact test, the chi-square test and Mann-Whitney's U test were used where appropriate to analyze the correlation of S100P expression with clinicopathological parameters. The Spearman correlation coefficient was used for correlation analysis between S100P mRNA expression and tumor stage. The 5-year DFS and 5-year OS of patients were calculated using the Kaplan-Meier method and differences were compared by the log-rank test. Backward stepwise multivariate Cox proportional hazards analysis was performed to determine the influence of S100P expression on DFS and OS. The hazard ratios (HRs) estimated from the Cox models were reported as relative risks with corresponding 95% confidence intervals (CIs). P values <0.05 were considered statistically significant.
Results
Association of S100P mRNA levels with tumor stage
Pathological stage is a well-known prognostic factor that has clearly shown practical use in gastric cancer. Our current study showed that S100P expression was significantly higher in gastric cancer tissue than in normal tissue (Fig. 1A, p<0.001). Moreover, Spearman's correlation showed that S100P was highly correlated with pathological stage (r=0.623, p<0.001) (Fig. 1B).
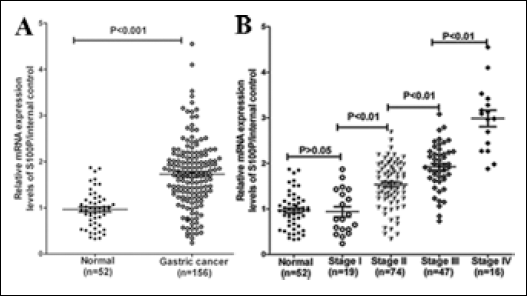
S100P mRNA levels in healthy controls and gastric cancer patients. (A) Normal controls vs gastric cancer patients. (B). S100P mRNA levels in gastric cancer with different clinical stages. Expression levels for mRNA were normalized to internal control (TBP) and shown as the relative quantity. The Mann-Whitney U test was conducted to examine the difference of mRNA expression between normal controls and/or gastric cancer patients at different clinical stages.
Correlation between expression of S100P and prognosis
Until October 2010, patients had been followed for a median of 69 months (range, 3-104 months). During this period, 46 recurrences (30.3%) were recorded and 49 patients (31.4%) died. The details of recurrences and deaths are described in Table I.
Number of Recurrences and Deaths for each Variable
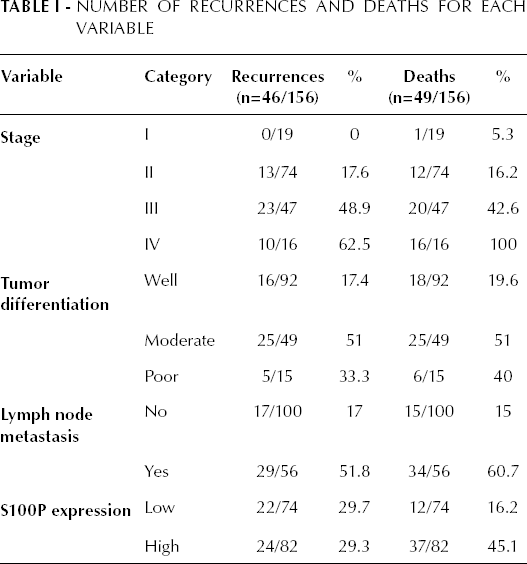
Statistical analysis showed that the 5-year OS rate was 59% (95% CI, 37.9-71.4). Tumor stage correlated in the Kaplan-Meier and univariate analyses with OS (p<0.01). Patients at stage IV had a 5-year OS of 18% (95% CI, 0-50.6); patients at stage III, 30.4% (95% CI, 9.1-50.6); patients at stage II, 70.7% (95% CI, 60.4-89); and those at stage I, 83.7% (95% CI, 65.7-100). S100P expression levels were calculated relative to TBP using the 2–ΔΔCt method. The median expression of this ratio (0.658) in the cancerous gastric tissues was used to define a cutoff value to subdivide tumor tissues into high or low expression. The Kaplan-Meier survival analysis revealed an association between OS and S100P expression when its levels were divided into low and high levels by the median (p=0.018; Fig. 2).
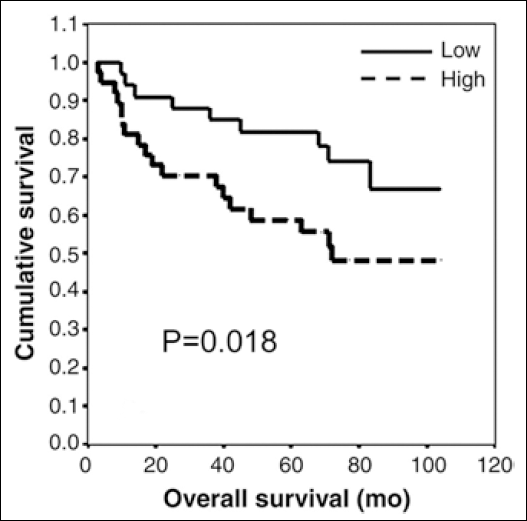
Impact of S100P mRNA levels on OS (3-104 months) in gastric cancer by Kaplan-Meier curves. S100P expression levels were calculated relative to TBP using the 2–ΔΔCt method. The median expression of this ratio (0.658) in the cancerous gastric tissues was used to define a cutoff value to subdivide tumor tissues into high or low expression.
Patients with low S100P expression had a 5-year OS rate of 66.7% (95% CI, 47.1-88.6), whereas patients with a high expression level had a rate of 47.9% (95% CI, 30.3-64.2). No statistical associations were observed between S100P levels and DFS.
Ectopic expression of S100P increases proliferation and drug resistance
We transfected SGC7901 cells transiently with a vector containing S100P or the corresponding empty vector. At 72 hours post-transfection, a significant increase in the cell number was observed in those cells ectopically expressing S100P (Fig. 3A). The MTT cell-proliferation assay was also consistent with such increase (Fig. 3B). Furthermore, a profound difference in the initiation and rate of cell proliferation measured by the cell index and the slope of the curves in the RT-CES system was observed between both cell groups (Fig. 3C). The ectopic expression of S100P did not affect the cell viability in both cell types, with cells overexpressing the isoform and the control variant in the range of 95% to 98%. Cells expressing the S100P vector showed 30% higher viability after cisplatin exposure than those transfected with the mock vector (Fig. 4). Cisplatin did not modify the endogenous levels of S100P.
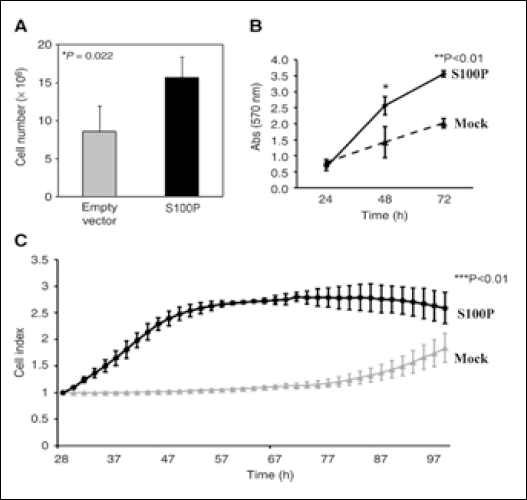
Ectopic expression of S100P increases the proliferation of SGC7901 cells 72 hours post-transfection. A) Significant increase in the number of cells. Experiments were done in triplicates and counted in a cell-counter apparatus. B) MTT assay showed that ectopic expression of S100P led to an increase in the cell-proliferation rate compared with the mock SGC7901 cells. C) Significant difference in the initiation and rate of proliferation measured by the cell index and the scope of the curves in the RT-CES system.
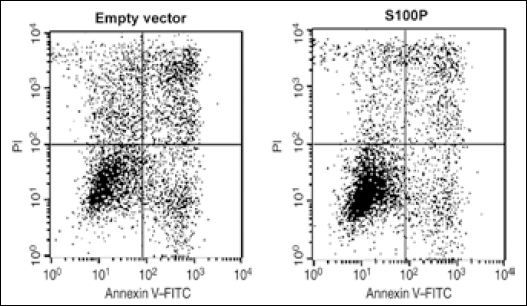
Ectopic expression of S100P induces resistance to cisplatin in gastric cancer cells. SGC7901 cells were exposed to 100 μmol/L cisplatin for 36 hours and checked for viability by flow cytometry. Cells containing the S100P-expressing vector showed a 30% increase in cell viability. Results are representative of 3 independent experiments.
Discussion
Although great improvements in the pursuit of cancer biomarkers have been achieved over the past decade, most biomarkers are not used in clinical care. Novel and reliable biomarkers are required to predict the prognosis of gastric cancer. S100P has been detected in a variety of tissues and proven to play an important role in the regulation of cell functions (12). It has become of special interest because of its ability to promote tumor invasion and metastasis (7, 10, 11). In this study, we examined S100P for its potential clinical value for gastric cancer. The results revealed that S100P was correlated with gastric cancer stage. Importantly, our data also demonstrated that higher tissue S100P was associated with shorter survival and drug resistance in gastric cancer.
In a study of 303 breast cancer patients followed for up to 20 years, survival was significantly worse in patients with S100P-positive cancer than those with S100P-negative cancer (18). It was reported that S100P expression was associated with metastasis and poor prognosis in early-stage non-small cell lung cancer (19) and ovarian cancer patients (20). These previous findings and our data suggested that the S100P protein could be a potential biomarker for prognosis in a variety of cancers. Our data showed that the 5-year survival rate of patients with high levels of S100P expression was significantly lower than that of patients with low levels of S100P expression.
S100P could promote the proliferation, migration and invasion of pancreatic cancer cells via a receptor for activated glycation end products (21); it also stimulated colon cancer cell proliferation and migration in vitro (13, 14), and stimulated lung cancer cells in an autocrine manner (22). These findings may, at least partially, provide molecular insight into the relationship of S100P and prognosis in various cancers. In our study, we found ectopic expression of S100P to be associated with an increase in tumor cell proliferation. Nevertheless, further studies focusing on the molecular mechanisms of S100P functions in gastric cancer should be conducted.
In previous studies, the expression of S100P was found to be associated with tumor stage in pancreatic cancer (7), prostate cancer (11) and lung cancer (23). Consistent with this, we found that S100P expression was associated with gastric cancer stage.
S100P overexpression was associated with resistance to 5-fluorouracil in pancreatic cancer cells and to irinotecan in prostate cancer (21, 24). In addition, a correlation between S100P overexpresion and resistance to cyclophosphamide, etoposide, methotrexate and mitoxantrone was observed (25). In agreement with this, our results suggested that drug resistance/sensitivity was altered by S100P, but the mechanism of chemotherapy resistance is complex and merits further investigation.
As the above data support the oncogenic role of S100P, the mechanisms and targets underlying these functions are currently of great interest. In this article, we present original data with regard to the impact of S100P on the outcome of gastric cancer patients. In addition, we found that S100P is a prognostic marker of OS. Further experiments are needed to identify specific targets of S100P that carry out oncogenic functions and could be used as clinical markers of relapse.
