Abstract
BACKGROUND:
Rap1GAP, a member of the family of GTPase-activating proteins, is reported to be involved in cancer development and progression.
OBJECTIVE:
The study aimed to investigate the expression and prognostic value of Rap1GAP in gastric cancer patients.
METHODS:
Real-time quantitative polymerase chain reaction and western blotting were performed to examine Rap1GAP expression in tumorous and matched adjacent non-tumorous gastric tissues. Immunohistochemical staining was used to analyze Rap1GAP expression in 456 gastric cancer tissues. The correlation between Rap1GAP expression level and clinicopathological features as well as gastric cancer prognosis was analyzed.
RESULTS:
Rap1GAP expression was remarkably decreased in tumor tissues at mRNA (
CONCLUSION:
Rap1GAP may play a significant role in gastric cancer progression and act as a valuable prognostic marker for gastric cancer.
Introduction
Gastric cancer is one of the leading causes of cancer-associated mortality in the world. The advancement in the diagnosis and treatment strategies has reduced the incidence and mortality rate of gastric cancer in recent years; however, gastric cancer still remains as the leading cause of cancer-related mortality in developing countries [1]. As most gastric cancer patients have advanced or metastatic diseases at diagnosis, their overall clinical outcome remains unsatisfactory, with a 5-year survival rate of less than 30% [2, 3]. The TNM clinicopathological staging system is the most commonly used tool by clinicians for gastric cancer prognosis. However, gastric cancer patients with identical TNM stage often exhibit varying clinical outcomes, suggesting that there are additional factors affect long-term outcomes [4]. Gastric cancer is a biologically heterogeneous disease that involves various molecular and genetic alterations in the stomach epithelial cells [5, 6]. Several new genes related to gastric cancer have been recently identified that can be used for diagnosis or targeted therapies [7, 8]. Therefore, it is essential to understand the molecular variables that affect gastric cancer for the development of appropriate approaches for its diagnosis and treatment.
Rap1 is a Ras family of small GTPase protein that is involved in cancer growth, invasion and metastasis. Rap1 is activated by guanine-nucleotide-exchange factors (GEFs), which replace GDP by GTP, and inactivated by GTPase-activating proteins (GAPs), which enhance Rap1 to hydrolyze GTP into GDP. Rap1 can be inactivated by Rap1GAP that induces GTPase activity [9]. Rap1GAP belongs to the family of GAPs for Rap1/2 GTPases, which include the splice variants Rap1GAPII, SPA-1, and E6TP1 [10, 11, 12, 13]. The loss of the function or expression of Rap1GAP results in an increase in GTP-bound Rap1 level. Active Rap1 induces the mitogen-activated protein kinase (MAPK)-dependent signaling cascade, which has been implicated in the proliferation, differentiation, invasion and metastasis of cancers [14, 15].
Recent studies have demonstrated that Rap1GAP may function as a tumor suppressor and is shown to be downregulated in several cancers such as kidney cancer [16], oropharyngeal squamous cell carcinoma [17], pancreatic cancer [18], papillary thyroid cancer [19], melanoma [20], and acute myeloid leukemia [21]. In squamous cell carcinoma, Rap1GAP promotes invasion through the upregulation of matrix metalloproteinase 9 (MMP9) secretion [22]. In thyroid cells, the increase in the expression of Rap1GAP results in the impairment of p70S6 kinase activity and inhibition of cell proliferation [23]. In myeloid leukemia cell lines, Rap1GAP increases leukemia cell invasion by the induction of MMP9 expression. However, the expression of Rap1GAP and its clinical prognostic relevance is not understood in gastric cancer [21]. In this study, we investigate the expression of Rap1GAP in gastric cancer and evaluate its prognostic significance by correlating its expression with clinicopathologic features and survival in 456 gastric cancer patients.
Materials and methods
Ethics statement
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Sun Yat-sen University Cancer Center. All subjects provided informed consent for inclusion before their participation in the study.
Human tissue samples
A total of 24 paired tumor and adjacent non-tumorous gastric tissues were collected from gastric cancer patients undergoing gastrectomy at the department of gastric and pancreatic surgery in Sun Yat-sen University Cancer Center from 2013 to 2014 (Table 1). All patients had no history of preoperative radiotherapy or chemotherapy. After surgical resection, fresh tissues were immediately frozen in liquid nitrogen and stored at
Clinical characteristics of the 24 gastric cancer patients
Clinical characteristics of the 24 gastric cancer patients
Another 456 paraffin-embedded primary gastric adenocarcinoma samples were collected from 2003 to 2006 at the department of gastric and pancreatic surgery in Sun Yat-sen University Cancer Center (Table 2). All patients, enrolled in our study, had no history of preoperative radiotherapy or chemotherapy. Post-operative TNM stage was classified according to the seventh edition of the American Joint Committee on Cancer.
Correlation between RAP1GAP expression and clinicopathological variables in gastric cancer
The patients had a periodic follow-up every 3 month for the first 2 years, every 6 months for the next 3 years, and annually for an additional 5 years or until patient death, whichever occurred first. The follow-up data from gastric cancer patients in this study were available and complete. Overall survival was defined as the time from the day of surgery to the day of death or the last follow-up.
Real-time quantitative polymerase chain reaction (RT-PCR)
Total RNA was extracted by TRIzol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instruction. RNA was reverse transcribed to cDNA using PrimeScript RT reagent Kit (Takara, Dalian, China) in 20
Western blot analysis
Total proteins were extracted from 24 paired fresh frozen gastric cancer specimens and corresponding adjacent normal gastric specimens using radioimmunoprecipitation assay (RIPA) lysis buffer and protease inhibitor (Beyotime, Nantong, China). Protein concentrations were determined using the bicinchoninic acid assay kit (Pierce, Rockford, IL, USA). Approximately, 30
The mRNA and protein expression of Rap1GAP in human gastric cancer tissues and matched adjacent non-tumorous tissues. (A) The relative Rap1GAP mRNA expression was significantly decreased in gastric cancer tissues (T) compared with the matched adjacent non-tumorous tissues (ANT) (
Immunohistochemical analysis of Rap1GAP protein expression in primary gastric cancer surgical specimens. (A) Immunostaining of the adjacent non-tumorous (ANT) area. (B) Well-differentiated gastric cancer. (C) Moderately differentiated gastric cancer. (D) Poorly differentiated gastric cancer. Images in the upper panel: 200 
Paraffin-embedded tissues were serially sectioned to a thickness of 4
Immunostained sections were analyzed by two pathologists with no knowledge about the clinicopathological parameters of the gastric cancer patients. The total Rap1GAP immunostaining score was calculated with the percentage of positively stained tumor cells and staining intensity. The percent positivity was scored as “0” (
A
Results
Rap1GAP mRNA and protein expression levels decreased in gastric cancer
The mRNA level of Rap1GAP was determined by real-time quantitative PCR in 24 paired human gastric cancer tissues and matched adjacent non-tumorous (ANT) tissues. The expression level Rap1GAP mRNA was significantly lower in 19 (19/24, 79%) gastric cancer tissues as compared with matched ANT tissues (Fig. 1A,
Rap1GAP expression and its relationship with clinicopathological features of gastric cancer
To evaluate the association between Rap1GAP protein expression level and clinicopathological parameters, immunohistochemical staining was performed for 456 paraffin-embedded gastric cancer specimens. As shown in Table 2, 53.3% (243/456) samples showed low Rap1GAP expression (
Correlation between Rap1GAP expression and gastric cancer survival
Kaplan-Meier survival analysis was performed to evaluate the prognostic values of Rap1GAP in gastric cancer patients. The results showed that gastric cancer patients with low Rap1GAP expression had significantly shorter overall survival time as compared with patients with high Rap1GAP expression (Fig. 3A,
Rap1GAP expression and its prognostic value in different histologic grade of gastric cancer. (A) Overall survival of all patients with low or high Rap1GAP expression. (B) Overall survival of well differentiated gastric cancer patients with low or high Rap1GAP expression. (C) Overall survival of moderately differentiated gastric cancer patients with low or high Rap1GAP expression. (D) Overall survival of poorly differentiated gastric cancer patients with low or high Rap1GAP expression.
To explore the independent prognostic factor for overall survival of gastric cancer patients, univariate and multivariate Cox regression analyses were performed. Univariate Cox regression analysis showed that Rap1GAP expression, tumor size, histologic grade, T classification, N classification, and M classification were significantly related with overall survival of gastric cancer patients (Table 3). Multivariate Cox regression analysis indicated that Rap1GAP expression was an independent predictor for overall survival (Table 3), suggesting that Rap1GAP expression may be useful for the prediction of the overall survival of gastric cancer patients.
Univariate and multivariate analysis of overall survival in gastric cancer patients
HR, Hazard ratio; CI, confidence interval.
Rap1GAP expression and its prognostic value at different clinical stages of gastric cancer. (A) Overall survival of stage I gastric cancer patients with low or high Rap1GAP expression. (B) Overall survival of stage II gastric cancer patients with low or high Rap1GAP expression. (C) Overall survival of stage III gastric cancer patients with low or high Rap1GAP expression. (D) Overall survival of stage IV gastric cancer patients with low or high Rap1GAP expression.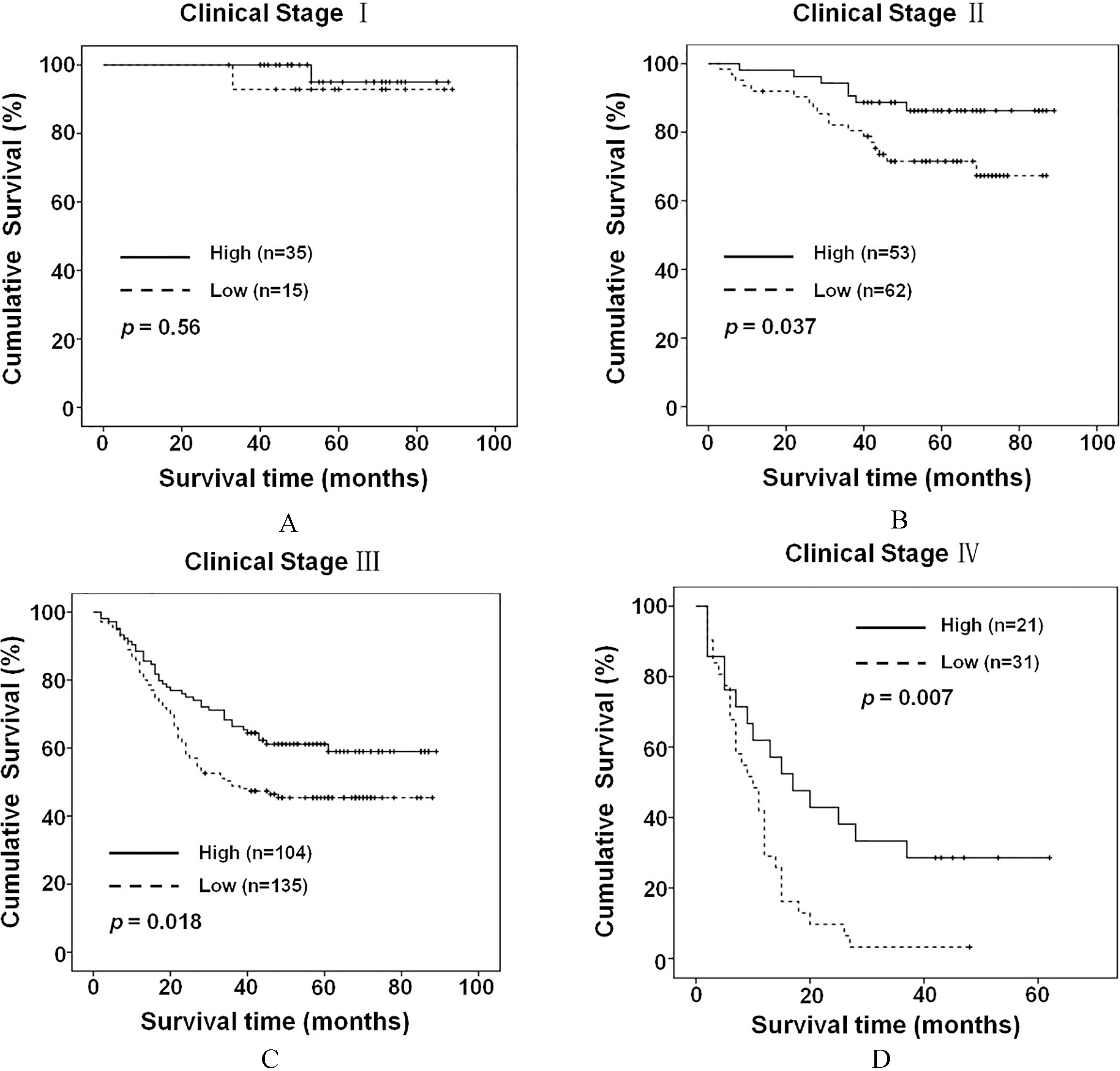
Despite the advancements in the diagnosis and treatment of advanced gastric cancer, the prognosis of gastric cancer patients is still poor due to the low rate of diagnosis during its early stages. The discovery of new biomarkers and their applications, in conjunction with traditional cancer diagnosis, staging, and prognosis, may help to improve early diagnosis and treatment of gastric cancer [5]. In recent years, great advancements have been made in the discovery of molecular biomarkers and development of therapeutic agents based on the target genes in several types of solid tumors. A few molecules have been identified and tested for their clinical relevance in gastric cancer development. For instance, HER2 overexpression in gastric cancer has been correlated with poor outcome and more aggressive disease as well as shorter survival [24, 25]. Moreover, PFKFB3 was overexpressed in gastric cancer patients and shown to contribute to the progression of gastric cancer and poor prognosis [26]. It is beneficial to search and identify novel molecular biomarkers for early gastric cancer diagnosis and disease monitoring.
In the present study, we investigated Rap1GAP expression at mRNA and protein levels in gastric cancer tissues and their matched ANT tissues using real-time quantitative PCR and western blot analyses. As reported in a previous study on thyroid cancer [19], Rap1GAP mRNA and protein levels were significantly decreased in gastric cancer tissue samples as compared with the matched ANT tissue samples. These results suggest that Rap1GAP may play an important role in the development of gastric cancer. Moreover, the relatively large samples of gastric cancer patients (456 cases) allowed us to observe that the low expression of Rap1GAP was significantly correlated with larger tumor size, more poorly histologic grade, and greater tumor progression (T3
The results of Kaplan-Meier survival analysis revealed the significant correlation between Rap1GAP expression and overall survival in all patients as well as in those with well differentiated, moderately differentiated and poorly differentiated gastric cancer. Moreover, significant correlation was also observed between Rap1GAP expression and overall survival of patients with clinical stage II, III and IV cancer. However, we did not find a significant correlation between Rap1GAP expression and overall survival in early clinical stages of gastric cancer (clinical stage I,
Recent findings suggest that Rap1GAP is frequently inactivated and downregulated in several tumor types; however, its precise mechanism may vary in different cell types. The mechanisms of gene silencing and inactivation of tumor suppressor genes include inactivating mutations, loss of heterozygosity (LOH) and epigenetic alterations. The gene rap1GAP has been mapped to chromosome 1p35-36, a region with high frequency of LOH observed in several types of human cancers, including chronic myelocytic leukemia [27], gliomas [28] and oral squamous carcinoma [29]. In pancreatic cancer, 33% frequency of LOH was observed in rap1GAP gene region and loss of Rap1GAP function was shown to promote growth [18]. The high frequency of LOH in rap1GAP gene region is also common in thyroid cancer, particularly in most invasive tumors [30]. In addition, Rap1GAP downregulation may be mediated through the hypermethylation of CpG islands in the promoter region of the gene. It has been reported that Rap1GAP is frequently downregulated in malignant melanoma and thyroid cancer via promoter hypermethylation [20, 30]. As epigenetic gene silencing through promoter methylation and changes in chromatin structure is reversible [31], which suggest that Rap1GAP may serve as a potential target for cancer therapy using demethylating agents. Furthermore, mutations (C257R and Y609C) in rap1GAP gene have been identified in breast cancer [32] that may affect the function of Rap1GAP.
In conclusion, our results demonstrate that Rap1GAP expression is downregulated in gastric cancer at both mRNA and protein level and that the low expression of Rap1GAP is correlated with poor prognosis in a large number of clinical specimens. Our data suggest that Rap1GAP may function as a valuable prognostic biomarker for gastric cancer, although further research is required to clarify the molecular mechanism underlying Rap1GAP function in the pathogenesis of gastric cancer.
Footnotes
Acknowledgments
This work was supported by research grants from National Natural Science Foundation of China (No. 81402560) and Science and Technology Planning Project of Guangdong Province (2017A020215029).
Conflict of interest
The authors declare that they have no conflict of interest for this study.
