Abstract
Aim
This study aimed to assess the molecular subtypes of breast cancer for patients attending a dedicated breast care center and examine the association with clinicopathological features, treatment and survival outcomes.
Methods
Demographic, clinicopathological and treatment details were collected from women with primary breast cancer. Immunohistochemical subtypes were also collected. The association between breast cancer subtypes and clinicopathological features was assessed using the chi-square or Fisher's exact test. Survival outcomes were compared among subtypes with the log-rank test.
Results
Immunohistochemical subtypes were not associated with tumor size, lymphovascular invasion or lymph node involvement but differed by histological grade (p=0.014) and nuclear grade of tumors (p=0.001). The 5-year overall survival estimates for luminal A, luminal B, HER-2-positive and triple-negative tumors were 100%, 96.2%, 71.4% and 92.3% respectively. Compared to luminal A tumors (93.4%), luminal B (80.8%), HER-2-positive (71.4%) and triple-negative (76.9%) tumors exhibited a reduced disease-free survival (DFS). Patients with ER-positive tumors had a higher DFS than their ER-negative counterparts (p=0.036). Patients with tumors expressing a low Ki-67 level had a more favorable prognosis (p=0.02).
Conclusions
The most prevalent luminal A subtype is associated with relatively better prognosis, whereas HER-2-positive and triple-negative tumors are prone to early relapse with diminished survival.
Introduction
Breast cancer, the most common female cancer worldwide, remains a major health burden in spite of advances in diagnosis and treatment (1). Currently, about 40% of patients suffer a disease recurrence, of whom the majority die (2). Although tumor metastasis remains the most common cause of cancer-related death in breast cancer patients, only a minority has clinical signs of metastasis at the time of diagnosis. There is evidence that cancerous cells can remain clinically undetectable for months or years (3), indicating that the long-term risk of metastatic relapse is the most difficult challenge facing breast cancer patients.
Both early detection and adjuvant systemic therapies for patients with early-stage breast cancer improve survival (4). However, the administration of systemic therapy remains largely empirical (5). It is important to identify prognostic factors to select candidates for adjuvant therapy based on their tumor relapse risks. Classical prognosis evaluation using histopathological information alone does not allow assessment of the clinical course of breast cancer and identification of patients with a high risk of recurrence (6, 7).
Human breast cancer is a heterogeneous malignancy comprising tumors with distinct characteristics and biological behaviors. Increasing insights into its molecular pathogenesis have led to new treatment approaches, with increasing movement towards personalized treatment, which has the potential to increase efficacy and decrease toxicity (8).
The emergence of prognostic and predictive markers has helped clinicians to make better informed decisions regarding therapeutic regimens and selection of the most effective adjuvant therapy for each individual (9). More recently, gene expression profiling has allowed molecular classification of breast tumors into distinct subtypes with unique clinical outcomes (10). Subsequent clinical studies have shown that immunohistochemical (IHC) analysis can be a useful surrogate of gene expression profiling using a limited panel of molecular markers (11). In this regard, attention has been paid to molecular subtypes that have been shown to have significant predictive value for the therapeutic response of breast cancer.
Materials and Methods
This study of a case series of women with operable breast cancer was conducted in the UNIMED Medical Institute, a comprehensive center for breast disease in Hong Kong. Data collected from the case reports included demographic characteristics, histopathological features of tumors, and molecular biomarker analysis of the tissue samples.
Study population and breast cancer identification
Inclusion criteria were primary cases of noninvasive and invasive female breast cancer diagnosed between 1 January 2006 and 31 December 2007, confirmed by medical record review. Subjects completed a questionnaire on established breast cancer risk factors: age at menarche, age at first pregnancy, number of deliveries, number of pregnancies, menopausal status, oral contraceptive use, hormone replacement therapy and breastfeeding.
Clinical and pathological information from women who underwent surgery for primary breast cancer included age, age at diagnosis, pathological tumor size, histological type, pathological stage, lymph node status and number of metastatic axillary lymph nodes, as well as immunohistological determination of ER and PR expression, HER-2 status and the proliferation marker Ki-67. Clinical management of patients including surgical treatment and adjuvant therapy was also recorded. Patients with missing information on molecular phenotype were excluded. Cases with complete tumor marker data were categorized into distinct subtypes based on ER/PR/HER-2 status. Missing information on variables such as demographic or histopathological characteristics was excluded from the analysis.
Immunohistochemical staining
ER, PR and HER-2 results were obtained from the histopathological report. For the purpose of this study, nuclei of tumors exhibiting staining for ER or PR at either a low (1+) or higher level (2+ and 3+) were considered positive for expression, ER- and PR-negative tumors being defined as those exhibiting complete absence of staining. HER-2 positivity was defined as tumor cells showing strong membrane staining (3+) whereas tumors exhibiting 0, 1+ or 2+ were considered HER-2 negative.
Definition of breast cancer molecular subtypes
IHC markers can be used as a surrogate for molecular classification of breast cancers (12). A simple categorization based on the expression of ER, PR and HER-2 was used to classify cases into 4 main subtypes: luminal A (ER and/or PR+, HER-2–); luminal B (ER and/or PR+, HER-2+); HER-2 (ER-, PR-, HER-2+); triple negative (ER-, PR-, HER-2–).
Calculation of mortality and breast cancer recurrences
Study participants were followed up until death or June 2011, whichever was earlier. Knowledge of deaths was obtained from family members. Breast cancer was assumed to have recurred if a second cancer in the liver, bone or brain was reported, as these are the most common sites of breast cancer metastasis. Local recurrence was assumed to have occurred if there was subsequent cancer development in the same breast or opposite breast.
Statistical analysis
Statistical tests were performed with the SPSS statistical software version 16.0. Patient demographics and tumor characteristics were portrayed using basic descriptive statistics. Differences among breast cancer subtypes were evaluated using 1-way analysis of variance (ANOVA) for continuous variables such as patient age, age at menarche, age at menopause and age at first pregnancy. Comparisons between breast cancer subtypes were adjusted using the Bonferroni correction. Pearson's chi-square test was conducted to assess the relationship between breast cancer subtypes and the clinicopathological variables of prognostic significance. Fisher's exact test was used when expected cell counts were less than 5, using the Monte Carlo method. The Kruskal-Wallis test was used for comparison of breast cancer subtypes with ordinal variables. The association of therapeutic options and tumor markers was evaluated using chi-square tests.
Survival analysis was performed for the subtypes of patients followed up after the incidence period of January 2006 through December 2007. Overall survival (OS) was calculated from the date of diagnosis to the date of death from breast cancer or the follow-up cutoff, whichever came first. Disease-free survival (DFS) was calculated from the date of diagnosis to the date of first metastatic recurrence or the follow-up cutoff. For estimation of OS and DFS, survival curves were calculated using the Kaplan-Meier method. The log-rank test was used to examine the statistical significance of the difference observed between IHC subtypes. All statistical tests were 2-sided and p values less than 0.05 were considered statistically significant.
Results
Analysis was performed on 107 of the 116 cases with noninvasive and invasive breast cancer, 9 being excluded due to missing tumor markers. The mean age at diagnosis was 50.3±10.4 years, with a median of 48 years (range 27-78 years). Analysis of the age distribution revealed that the 40-49 age group accounted for the highest proportion of breast cancer (n=47, 43.9%), followed by the 50-59 (n=26, 24.3%) and the 60-69 age groups (n=14, 13.1%).
The use of IHC markers allowed all cases to be assigned a molecular classification category. The most common IHC subtype was luminal A (57%), whilst luminal B, HER-2, and triple negative accounted for 24.3%, 6.5% and 12.1% respectively. The distribution of tumor markers is shown in Table I.
Summary Of Tumor Markers For Breast Cancer Subtypes
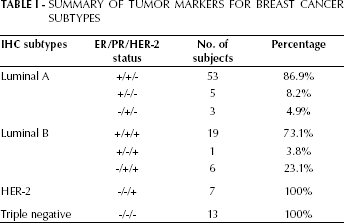
Characteristics of breast cancer patients
There was no significant difference in the frequency of any established breast cancer risk or protective factor between molecular subtypes. The age-specific incidence of breast cancer subtypes revealed that luminal B was the most common subtype in patients aged <40 years (n=5, 19.2%) compared with luminal A (n=7, 11.5%) and triple-negative (n=1, 7.7%) subtypes. The HER-2 subtype was more frequent in the 50-59 years age group (n=5, 71.4%). In this study, women with triple-negative tumors were younger at diagnosis (49.2±6.9 years; 95% CI 44.98–53.33) than those with other subtypes (Fig. 1).
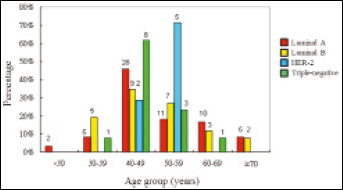
Age-specific incidence for breast cancer subtypes.
Overall, 81.3% of breast cancers were hormone receptor positive (ER and/or PR positive), with the majority presenting in premenopausal women (73.1%). Although not reaching significance, early menarche and late menopause were observed more frequently in patients with luminal A and luminal B tumors than in those with HER-2 and triple-negative tumors.
Clinicopathological characteristics of breast cancer subtypes
The associations of breast cancer subtypes with prognostic variables are shown in Table II. Although not reaching significance, tumors with a diameter <2 cm were most commonly found in the luminal A subtype group whereas large tumors (diameter >5 cm) were observed more frequently in the triple-negative subtype. The expression of Ki-67 differed significantly within the breast cancer subtypes, with luminal A showing the highest proportion of tumors with a low Ki-67 expression, whilst the proliferation index was high in HER-2 and triple-negative subtypes. Additionally, comparison of the luminal subtypes revealed significantly more luminal B tumors presenting high Ki-67 expression than luminal A tumors. Although lymph node status was not associated with breast cancer subtypes, significant differences were observed regarding the histological and nuclear grades of tumors, whilst comparison of the mitotic counts of breast cancer subtypes approached significance. Histologically, the prevalence of high-grade tumors was higher in luminal B than luminal A subtypes. No significant associations among breast cancer subtypes were detected for histological type, lymphovascular invasion, lymph node involvement or tumor stage. Even though not reaching significance, HER-2 and triple-negative subtypes were more often at a higher stage (stage III or IV).
Clinicopathological Characteristics Of Breast Cancer Cases According To Ihc Subtypes
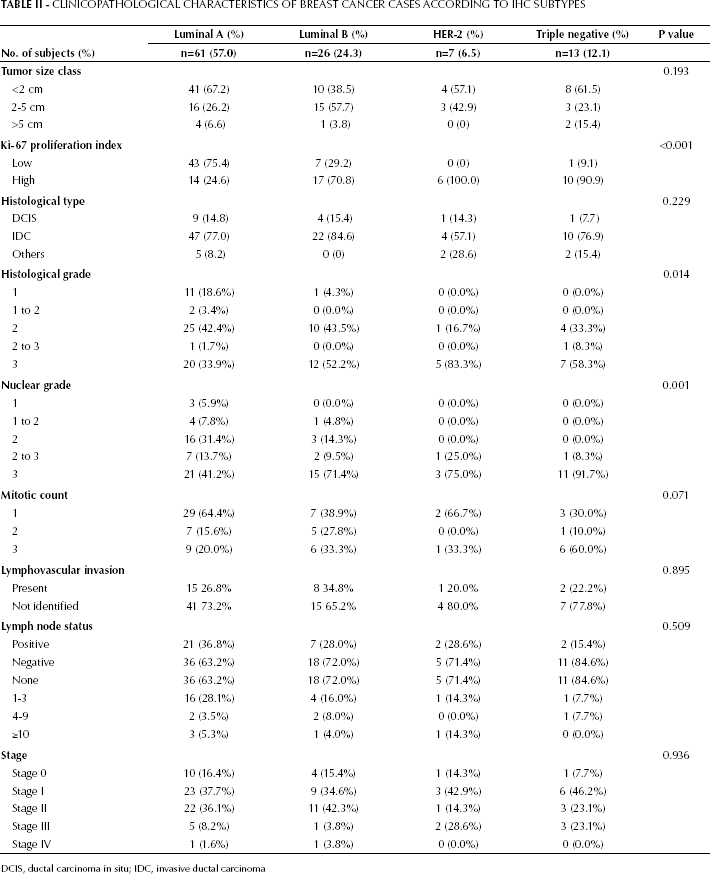
DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma
Predictive value of IHC subtypes regarding treatment options
The majority of patients with invasive carcinoma (57/92, 62.0%) had undergone mastectomy as definitive treatment, of whom 15.4% received breast reconstruction. Where treatment data were available, there was a substantial difference in adjuvant therapy among breast cancer subtypes. Although the IHC subtypes did not differ significantly with regard to chemotherapy, the highest proportion of patients receiving chemotherapy (53.8%) were triple-negative cases. Women who had negative lymph node status were more likely to have received adjuvant chemotherapy if they had the triple-negative subtype (46.2%) rather than others (luminal A, 22.8%; luminal B, 32%; HER-2, 14.3%). There was a significant difference in patients receiving hormonal therapy (p<0.001) and targeted therapy (p<0.001) among breast cancer subtypes. Drug choice for hormonal therapy was significantly different (p=0.046) for patients receiving tamoxifen, aromatase inhibitors or a combination of both between luminal subtypes, but there was no significant relationship in patients receiving radiation therapy (p=0.344). For women who had breast-conserving surgery, the likelihood of receiving radiation therapy was statistically significant (p<0.001).
General characteristics of tumor staging
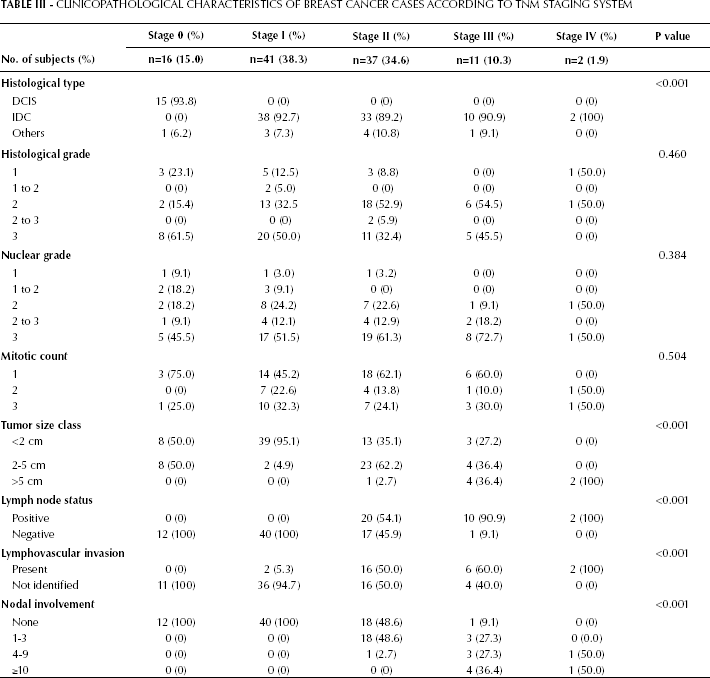
In contrast to molecular subtyping, histological type, tumor size class, lymph node status, lymphovascular invasion, and nodal involvement were significant (p<0.001) with respect to the traditional histopathological classification (Tab. III). No relationship was detected for histological grade, nuclear grade and mitotic count if tumors were classified by the TNM staging system. Unlike mo lecular subtypes, there was a significant difference in patients receiving chemotherapy (p<0.001), but no significant association was found in patients having hormonal therapy or targeted therapy when compared by tumor stages.
Clinicopathological Characteristics of Breast Cancer Cases According to TNM Staging System
Survival and breast cancer subtypes
Assessment of survival time after diagnosis was performed for noninvasive and invasive carcinomas. During the follow-up period 4 patients died, giving an OS probability of 96.3%. The DFS rate was 86.9%. Of the 10 patients suffering relapse, 6 cases were local and 8 cases regional. The molecular subtypes differed significantly in OS (log-rank test=14.2, p=0.003), with HER-2 and triple-negative subtypes showing reduced OS (71.4% and 92.3%, respectively) compared to luminal A and luminal B cases (100% and 96.2%, respectively) (Fig. 2A). Although not reaching significance (log-rank test=5.6, p=0.135), the HER-2 and triple-negative subtype DFS rates (71.4% and 76.9%) were lower than those of luminal cases (93.4% and 80.8% for luminal A and B), as shown in Figure 2B.
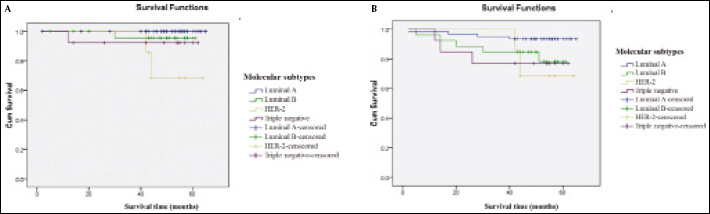
Kaplan-Meier (A) overall and (B) disease-free survival by molecular breast cancer subtypes.
In terms of molecular biomarkers, patients with ER-positive tumors had a significantly higher DFS rate (91.0%) than those with ER-negative tumors (75.9%), (log-rank test=4.4, p=0.036) (Fig. 3A). Patients with PR-positive tumors also had a higher DFS rate (90.2%) than patients with PR-negative tumors (76.0%), although this difference did not reach significance (log-rank test=4.4, p=0.066) (Fig. 3B). A similar nonsignificant difference in DFS rate (log-rank test=2.6, p=0.105) was observed in patients having tumors with HER-2 overexpression (78.8%) (Fig. 3C). Finally, a significant difference in DFS (log-rank test=5.4, p=0.02) was observed for patients having tumors with a low Ki-67 proliferation index, who were more likely to have a favorable prognosis (94.1%) compared with those having a high proliferation index (78.7%) (Fig. 3D).
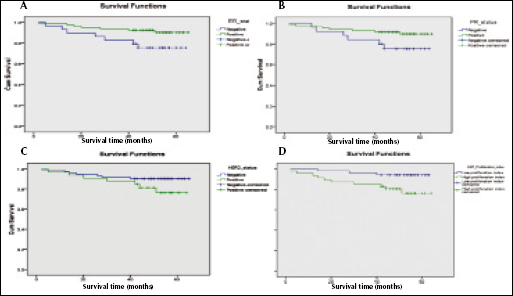
Kaplan-Meier disease-free survival by (A) ER, (B) PR, (C) HER-2, and (D) Ki-67 status.
Discussion
Our case series showed that the distribution of the molecular subtypes luminal A, luminal B, HER-2 and triple negative (57%, 24.3%, 6.5% and 12.1%, respectively) was broadly similar to the distribution reported by the Shanghai Breast Cancer Survival Study (13) (48.6%, 16.7%, 13.7% and 12.9%). This finding also indicates that the incidence of the triple-negative subtype among Chinese women is similar to that of Western populations (10-16%) (14-16). However, cases of the HER-2 subtype were somewhat higher in the Shanghai study, the 6.5% HER-2 incidence in the Hong Kong sample being closer to that reported in European or African-American populations (4-8%) (17, 18).
The associations with risk factors did not differ significantly between breast cancer subtypes. This suggests that additional confounding variables may be involved. Although definitive conclusions could not be drawn regarding exposure, hormonal risk factors appeared to have a greater impact on luminal-type than HER-2 and triple-negative tumors. Based on limited data, some reproductive factors including early menarche, late menopause, nulliparity, and first childbirth at late age were more frequently observed in luminal A tumors, the most common breast cancer subtype. Postmenopausal hormone use was also more common in luminal A patients, although heterogeneity across breast cancer subtypes was not statistically significant. In both the Nurses’ Health Study (19) and a Polish breast cancer study (18), risk factors such as age at menarche, age at first birth, parity and postmenopausal hormone use were associated with luminal A subtype. Our data supported the hypothesis of etiological heterogeneity across breast cancer subtypes.
Our results suggested that early onset of breast cancer was more likely in patients with luminal and triple-negative tumors even though the difference between IHC subtypes did not reach significance. No significant association of menopausal status with molecular subtypes was observed. This may be due to the data being derived from a case series including all ages with a higher percentage of premenopausal women. Nevertheless, HER-2 cases appeared to be more frequent in postmenopausal women in our study. This agreed with the findings of an earlier population-based study in southern Switzerland in which HER-2 cases were more frequent in the age group 50-69 (20).
As reported elsewhere, the luminal A subtype was predominant and differed in clinical and pathological characteristics from other subtypes (21), comprising a higher percentage of noninvasive cases, the lowest frequency of poorly differentiated carcinomas, and the highest proportion of tumors with a diameter <2 cm at the time of diagnosis. Specifically, histological grade was significantly associated with molecular subtypes, of which HER-2 and triple-negative cases had a greater frequency of high-grade tumors and poor prognosis. These clinicopathological characteristics of breast cancer subtypes have been consistently observed in both Western (15, 16) and Chinese populations (22, 23). Such findings are closely correlated with previous analyses showing triple-negative and HER-2 subtypes to be associated with larger tumors, higher histological grade, more advanced TNM stage and high proliferative activity. Despite being high grade, triple-negative tumors are reported to have less nodal metastasis (24). In this study, 84.6% of triple-negative cases did not present with positive nodal involvement. Recent data suggest that lymph node status is not a major prognostic factor for the triple-negative subtype (25). Besides, both triple-negative and HER-2 cases presented with a high Ki-67 proliferation index. However, there was no significant difference for traditional prognostic parameters when the mitotic count was used to indicate the tumor's proliferative activity. Clearly, Ki-67 expression, as a marker of proliferation, displays a strong prognostic effect relative to the mitotic count, but no standard methodology and cutoff point have so far been defined for routine use.
Interpretation of the study results was somewhat limited because the statistical power of all analyses was reduced by the inclusion of only 7 HER-2 overexpressing and 13 triple-negative cases. As this was a retrospective study, the reliability of patients’ demographic data necessary for evaluation of risk exposure between breast cancer subtypes may suffer from a recall bias of self-reported data. It is possible that there was some misclassification of molecular subtypes resulting from the use of ER, PR and HER-2 data provided by different laboratories, as assay methods and criteria for interpretation of results vary between laboratories. Similarly, assessment of histological tumor grading as well as traditional prognostic parameters was performed by different pathologists without central review; this aspect may also be vulnerable to observer bias. There may also be residual confounders from therapeutic options that were not considered during analysis, especially related to patients treated with lumpectomy and mastectomy as well as any changes in treatment over time due to drug resistance.
Another important aspect was observed when clinicopathological characteristics were stratified by traditional classification. Histological features, including histological grade, nuclear grade and mitotic count, did not differ significantly across tumor stages. Our findings are consistent with other studies suggesting that histological grading is an independent prognostic factor to assess the current state of disease and the extent of breast cancer (26, 27). As expected, tumor size and lymph node status were associated with tumor stage using the TNM classification. No significant relationship was observed for molecular biomarkers between different pathological stages. With respect to additional treatment, chemotherapy differed significantly by tumor stages in our case series. As the existing TNM classification does not incorporate histological grade, which may be clinically relevant in influencing treatment options, a new molecular classification based on IHC markers appears to provide better prognosis and guidance regarding the need for additional adjuvant therapy and optimal management.
With respect to the prognosis of breast cancer in terms of survival, molecular classification of breast cancers yielded results comparable with those of earlier studies (20, 28). A case-control study revealed that patients with triple-negative and HER-2-positive tumors exhibited the highest mortality (29). After a follow-up of 5 years, the log-rank test showed that cases with HER-2 and triple-negative subtypes had a significantly poorer OS compared to women with luminal subtypes. Triple-negative tumors showed a reduced survival time as early as 2 years after diagnosis. It has been suggested that women with triple-negative subtypes have higher initial mortality compared with other subtypes, but mortality decreases compared to other types if they survive the first 5 years (30-32). In terms of DFS, breast cancer patients with luminal subtypes had more favorable prognoses than women with HER-2 and triple-negative tumors. However, our results also revealed a difference in DFS among luminal subtypes, with luminal B having a significantly poorer prognosis than luminal A. Molecular subtypes also differed significantly in DFS by ER status, patients with ER-positive tumors having prolonged DFS following primary treatment. With respect to recurrence, early relapse has been more commonly observed in hormone receptor-negative and triple-negative breast cancer patients (33). Furthermore, Ki-67 expression has also been significantly correlated with DFS (34). Patients whose tumors exhibited low Ki-67 expression survived longer than those with high expression (35).
It is important to note that luminal A is associated with relatively good prognosis and better survival (36). The luminal and HER-2 subtypes are well characterized and correlate strongly with prognosis and response to adjuvant therapy. By contrast, the triple-negative subtype is associated with tumors of high histological grade and nuclear grade (37). This subtype is more common in premenopausal women with an aggressive clinical course and poor disease outcome as chemotherapy seems to be less effective.
It should be acknowledged that this study was performed on a population of women attending private clinics in Hong Kong and the findings may not apply to the general population of patients with breast cancer. Additional research is needed to conduct a population-based study in order to assess the value of molecular classification.
Future directions and perspective
Microarray analysis has identified 5 distinct molecular subtypes with gene expression profiles (38), including luminal A and B tumors, which are ER-positive, and ER-negative tumors that comprise the HER-2 type, triple-negative and normal breast-like/unclassified subtype. However, the existence of the fifth group as an individual entity remains unclear. Currently, gene expression profiling can only be employed using fresh or frozen tissue and not tissue from paraffin blocks. To overcome this limitation, methods are being developed to extract RNA from formalin-fixed or paraffin-embedded tissue for genomic studies (39). Recently, emerging molecular biomarkers such as epidermal growth factor receptor (EGFR), cytokeratin, p53 and bcl-2 have been utilized in the characterization of breast tumors, defining relationships between histological parameters and providing reliable prognosis for breast cancer patients (40). With the understanding of the molecular evolution of breast cancer, it is anticipated that recent interest in personalized therapy may lead to the extended use of molecular predictors for management of breast cancer.
Although molecular classification correlates well with prognosis, it is not yet possible to replace the conventional morphological classification because some subtypes have not been fully characterized and some gene signatures are not fully validated. However, it is increasingly recognized that the molecular characteristics of breast cancer affect the treatment choice. The present study indicated that clinical outcomes were significantly different between breast cancer subtypes.
Molecular classification provides a framework for the study of breast cancer but further evaluation is needed. The application of molecular predictors can assist in selecting those patients who will benefit most from adjuvant therapy and identifying those who are at high risk of recurrence. It is of great importance to avoid overtreatment in patients who receive little benefit whilst suffering from toxic side effects of systemic therapy. Conversely, undertreatment or suboptimal treatment should also be avoided.
CONCLUSION
Molecular classification aims to reveal the heterogeneity of breast cancer and provide important prognostic information to guide clinical decision-making. The application of IHC-based molecular classification allows categorization of tumors at reasonable cost in the absence of fresh or frozen tissue specimens. However, the use of IHC expression assay remains controversial and quantitative methods using computer-aided image analysis are being developed to improve its accuracy. Apart from conventional diagnosis, molecular predictors have been extensively used to help selecting appropriate treatment for patients. Currently, prognosis and treatment decisions are guided by histological features, tumor stage, hormone receptor status and HER-2 expression. Molecular subtypes are used only in research settings, not forming a part of standard medical practice.
With the movement towards personalized treatment, molecular classification is of particular importance for the clinical management of breast cancer patients. It has been shown that prognostic classification based on molecular predictors is superior to that based on pathological parameters alone, allowing patients to receive the most appropriate treatment and prediction of disease outcome. Both hormone receptor and HER-2 expression had direct impact on the effects of adjuvant therapy. With continued efforts in characterizing molecular characteristics, the emerging biomarkers can improve optimization of tailored therapies and individualized breast cancer patient care.
