Abstract
Tumor hypoxia results in poor treatment response and is an indicator of poor outcome in cancer patients. TRIB3 is a hypoxia-upregulated protein involved in the ability of breast cancer cells to survive in hypoxic conditions. It is also involved in the prognosis of cancer patients, possibly by affecting several kinase-signaling pathways. We set out to establish which kinase-signaling pathways are regulated by hypoxia and whether these kinases are relevant for breast cancer prognosis. Using a phosphokinase antibody array comparing cells cultured under hypoxic conditions with those cultured during normoxia, we found that the phosphorylation status of ERK1/2, AKT, p70 S6 kinase, Lck and STAT3 was altered in both MCF7 and MDA-MB-231 breast cancer cells. Using Western blotting, we found that phosphorylated AKT (pAKT) increased in hypoxic conditions. Knockdown of TRIB3 attenuated this effect of hypoxia on AKT activation. Both pAKT and TRIB3 were expressed in pimonidazole-positive, hypoxic areas of human breast cancer tumors. In breast cancer patients significantly lower 5-year disease-free survival was observed for the pAKT-positive compared to the pAKT-negative group (64.6% vs 86.1%, p=0.03).
In conclusion, the phosphorylation status of AKT is increased in hypoxic conditions and TRIB3 knockdown attenuates this response. Furthermore, pAKT expression denotes a worse prognosis in breast cancer patients. The hypoxia-related activation of AKT could explain the resistance to various treatments including chemotherapy and radiotherapy.
Introduction
Tumor hypoxia is an important microenvironmental factor determining the prognosis of cancer patients and therapy sensitivity in solid tumors (1-3). Often, tumor cells are capable of surviving in a hypoxic microenvironment (4). Kinases play an important role in signal transduction from the environment into the cell and thus in the adaptation to stressful microenvironmental conditions such as hypoxia.
Tribbles homolog 3 (TRIB3) has been described to regulate multiple kinase-signaling routes such as the NFκB, MAPK and PI3K/AKT pathways (5-7). TRIB3 is a pseudokinase containing 12 subdomains that belongs to the protein kinase superfamily but has no kinase activity itself (8). It functions as a scaffold protein, for example by binding to the phosphorylation domains of AKT, thereby preventing the activation of this protein (5). In breast cancer cells, TRIB3 mRNA is upregulated by hypoxia via the transcription factors ATF4 and CHOP (9), which are both part of the unfolded protein response (UPR) (10). The UPR is a pathway that has a cytoprotective effect during stress conditions by protecting cells from apoptosis and induction of autophagy (11, 12).
Recently we described how TRIB3 is involved in the ability of breast cancer cells to survive in hypoxic conditions and in the prognosis of cancer patients (13). We hypothesized that TRIB3 affects specific kinase-signaling pathways induced by hypoxia that are involved in providing tumor cells the chance to survive under stressful microenvironmental conditions. Here, we determined which pathways are hypoxia-regulated in breast cancer cells in vitro, what the effect of TRIB3 knockdown is on this hypoxia-induced kinase profile, and whether these phosphoproteins have a relation to prognosis in breast cancer patients.
Materials and Methods
Breast cancer cell lines
The human breast cancer cells MDA-MB-231 (ER-/PR-/HER2) and MCF-7 (ER+/PR+/HER-) (ATCC, LGC Promochem, London, UK) were cultured for a limited number of passages in standard culture medium (DMEM with 10% dialyzed FCS, 2 mM L-glutamine, 20 mM HEPES, penicillin/streptomycin, and nonessential amino acids [all PAA Laboratories, Pasching, Austria]) at 37°C with 5% CO2, unless stated otherwise.
Treatment of cells
For 1% O2 exposure cells were transferred to a hypoxic culture chamber (H35 hypoxystation, Don Whitley Scientific Limited, West Yorkshire, UK). Knockdown of TRIB3 was performed using siRNA transfection reagent SAINT-RED (Synvolux Therapeutics B.V., Groningen, the Netherlands). The siRNAs MISSION̄ siRNA Universal Negative Control #1 (SIC001) and TRIB3 (SASI_Hs01_00197511) were acquired from Sigma-Aldrich (Sigma-Aldrich Chemie B.V., Zwijndrecht, the Netherlands). Knockdown efficiencies were determined using RT-qPCR and less than 40% mRNA expression compared to negative control was seen as effective knockdown.
Phosphokinase antibody array
The phosphokinase antibody array was performed using a human phosphokinase array kit (Proteome Profiler, R & D Systems Inc., Minneapolis, USA) and the supplemented protocol. In this assay, the capture antibodies are spotted in duplicate on nitrocellulose membranes, which are incubated with the cellular extracts. After washing, the membranes are incubated with a cocktail of biotinylated detection antibodies and streptavidin-HRP; subsequently, chemiluminescent detection reagents are applied to produce a signal. Capture antibodies used on the membranes are directed against AKT (S473), AKT (T308), AMPKα1 (T174), AMPKα2 (T172), Chk-2 (T68), c-Jun (S63), CREB (S133), eNOS (S1177), ERK1/2 (T202/Y204, T185/Y187), FAK (Y397), Fgr (Y412), Fyn (Y420), GSK-3α/β (S1/S9), Hck (Y411), HSP27 (S78/S82), JNK pan (T183/Y185, T221/Y223), Lck (Y394), Lyn (Y397), MEK1/2 (S218/S222, S222/226), MSK1/2 (S376/S360), p27 (T157), p27 (T198), p38α (T180/Y182), p53 (S15), p53 (S46), p53 (S392), p70 S6 kinase (T229), p70 S6 kinase (T389), p70 S6 kinase (T421/S424), paxillin (Y118), PLCγ-1 (Y783), Pyk2 (Y402), RSK1/2 (S221), RSK1/2/3 (S380), Src (Y419), STAT1 (Y701), STAT2 (Y689), STAT3 (Y705), STAT4 (Y693), STAT5a (Y699), STAT5a/b (Y699), STAT5b (Y699), STAT6 (Y641), TOR (S2448), Yes (Y426) and β-catenin.
The integrated optical density of each spot was measured using Image J (14). The average background signal from the negative controls was subtracted from the average optical densities for the 2 spots of each protein. Subsequently, these values were divided by the average optical density of the positive controls, resulting in a value between 0 and 1 for each protein.
Western blot analysis
Cells were harvested in RIPA buffer (PBS [Klinipath, Duiven, the Netherlands], MQ [sterile H2O, Versol̄, Lyon, France], NP-40 [Sigma-Aldrich Chemie B.V.], sodium desoxycholate [VWR International B.V., Amsterdam, the Netherlands] and SDS) with phosphatase and protease inhibitors (Roche, Indianapolis, USA). Samples were sonicated and centrifuged and the supernatant was stored at −80°C. Protein quantification was performed using a Piercē BCA Protein Assay Kit (Thermo Scientific, Etten-Leur, the Netherlands). Thirty micrograms of protein was fractionated on 4%-20% Criterion XT Bis-Tris gels (Bio-Rad Laboratories B.V., Veenendaal, the Netherlands). After electrophoresis, samples were transferred to PVDF membranes (Millipore Immobilon, Millipore B.V., Amsterdam, the Netherlands).
Membranes were blocked with 5% NFDM (Blotting Grade Blocker Non-Fat Dry Milk, Bio-Rad Laboratories B.V.) and incubated with the appropriate antibodies (mouse anti-α-tubulin [Calbiochem, Millipore B.V.], rabbit anti-phospho p44/p42 MAPK ERK 1-2 [Cell Signaling Technology, BIOKE, Leiden, the Netherlands], rabbit anti-phospho AKT [ser 473, Cell Signaling Technology], goat anti-phospho p70 S6 kinase [R & D Systems Inc.], rabbit anti-phospho STAT3 [Cell Signaling Technology], rabbit anti-phospho eNOS [Cell Signaling Technology] and goat anti-AKT1 [Santa Cruz Biotechnology Inc., Heidelberg, Germany]). Proteins were detected using chemiluminescent peroxidase s ubstrate (Sigma-Aldrich Chemie B.V.) and visualized with a ChemiDoc XRS + imaging system (Bio-Rad Laboratories B.V.). Images and protein band intensities were acquired using Quantity Onē 1-D Analysis Software (Bio-Rad Laboratories B.V.). Signal intensities were quantified using gel analyze tools from Image J (14) and corrected for α-tubulin intensities detected on the same blot.
Immunohistological analysis of breast cancer samples with an exogenous hypoxia marker
To obtain patient material containing the exogenous hypoxia marker pimonidazole, 38 patients with newly diagnosed breast cancer were enrolled in a tumor hypoxia study in accordance with a research protocol approved by the Institutional Review Board at the University of North Carolina Hospitals in the period 1997-1999. The patients signed informed consent prior to their participation in the study. Prior to tumor biopsy, patients received an intravenous infusion of pimonidazole hydrochloride (0.5 g/m2, Hypoxyprobe-1™, NPI Inc, Belmont, USA) diluted in 100 mL NaCl 0.9% over 20 minutes. Between 16 and 24 hours later, biopsies were obtained from primary tumors. After biopsy, fresh tumor samples were placed in cold 10% neutral buffered formalin, kept at 4°C for 12-24 hours, and processed into paraffin blocks.
Four-micron-thick sections were mounted on glass slides for immunohistochemical staining. One slide per block was stained with hematoxylin and eosin for pathological review to confirm the presence of tumor. Three adjacent slides were stained for pAKT, pimonidazole and TRIB3. Slides were incubated with the primary antibodies rabbit anti-pAKT (S473) (Cell Signaling Technology) and rabbit anti-pimonidazole or chicken anti-TRIB3 (13), and donkey anti-rabbit/chicken biotin-labeled secondary antibodies were used (Jackson ImmunoResearch Europe Ltd., Suffolk, UK). This was followed by incubation with ABC reagent and visualization with DAB reagent (Vector Laboratories Ltd., Peterborough, UK). Sections were counterstained with Mayer's hematoxylin solution. Negative control included substitution of the primary antibody with primary antibody diluent (PAD, AbD Serotec, Oxford, UK).
All microscopic images were acquired using the IP-Lab for Macintosh software (Scanalytics Inc., Fairfax, USA) in combination with a monochrome CCD camera (Retiga SRV, 1392×1040 pixels) and an RGB filter (Slider Module; QImaging, Burnaby, Canada) attached to a motorized microscope (Leica DM 6000, Wetzlar, Germany). For assessment of colocalization of pAKT, TRIB3 and pimonidazole, whole tumor sections were scanned with a 10× objective at 100× magnification (15). The individual colors (DAB [brown] and hematoxylin [blue] signals) were extracted and unmixed from the bright field images (16).
Tissue microarray of human breast cancer tissues
Breast cancer tissue sections were available from 95 patients who had undergone resection of their primary tumors, as described before (13). Breast cancer patients were selected from a cohort treated between January 1991 and December 1996 that did not undergo systemic treatment and had at least 5 years’ follow-up or a recurrence before that. The selection criteria resulted in a patient cohort including 25% ER-positive, 13% PR-positive, 30% HER2-positive, and 44% triple-negative patients. As approved by the institutional review board and according to national law, coded tumor tissues were used. This study was performed according to the REMARK guidelines (17).
Hematoxylin and eosin stained 4-μm sections of formalin-fixed paraffin-embedded tumor sections were used to identify tumor areas by an experienced pathologist. The tumor areas were outlined on the glass slides and used to select the appropriate area in the tissue blocks to punch out tissue (2 mm ø) for the tissue microarrays (TMA) using a Tissue-Tek Quick Ray puncher (Sakura Finetek Europe B.V., Leiden, the Netherlands).
Four-micron-thick sections of the TMA were incubated with chicken anti-TRIB3 (13) and donkey anti-chicken biotin-labeled secondary antibody (Jackson ImmunoResearch Europe Ltd.), followed by incubation with ABC reagent and visualization with DAB reagent (Vector Laboratories Ltd.) or rabbit anti-pAKT (Ser 473) (Cell Signaling Technology) and PowerVision (ImmunoLogic, Duiven, the Netherlands) and visualized with PowerVision DAB (3,3′-diaminobenzidine) (ImmunoLogic). Sections were counterstained with Mayer's hematoxylin solution. Negative control included substitution of the primary antibody with PAD (AbD Serotec).
Scoring was performed blinded to clinical patient data. For TRIB3 scoring was done as described previously (13). pAKT staining was scored both for nuclear and cytoplasmic staining, and was considered either negative or positive.
Statistical analyses
Statistical analyses were carried out using the SPSS 16.0.5 software (SPSS Benelux B.V., Gorinchem, the Netherlands). Normality of distribution of variables was tested using Kolmogorov-Smirnov testing. Non-parametric correlations were assessed using Spearman's rank correlation testing. A χ 2 test was performed to test differences in the distribution of pAKT-negative and pAKT-positive patients among groups with the clinical parameters age (below vs above 50), menopausal status, nodal category, tumor type, tumor size, histological grade, ER expression, PR expression, HER2 expression, surgery, radiotherapy, and adjuvant systemic therapy. Disease-free survival (DFS) time (defined as the time from surgery until diagnosis of recurrent or metastatic disease) was used as follow-up endpoint. Survival curves were generated using the method of Kaplan and Meier. Cases with more than 60 months of follow-up were censored at 60 months because of the rapidly declining number of patients thereafter. Equality of survival distribution was tested using Cox regression univariate and multivariable survival analyses. Two-sided p values below 0.05 were considered statistically significant.
Results
Hypoxia-induced phosphokinase profile in breast cancer cells
Using the phosphokinase antibody array, we tested the expression levels of 45 phosphorylated kinases in 2 breast cancer cell lines exposed to either normoxia or hypoxia for 6 hours (Tab. I). The detection of each kinase was performed for 2 independent spots and the average difference between duplicate spots was 5.6%. We observed that in MDA-MB-231 cells AKT (S473), eNOS (S1177), Fyn (Y420), JNK pan (T183/Y185, T221/Y223), Lck (Y394), p38α (T180/Y182), p70 S6 kinase (T389), STAT3 (Y705), STAT5b (Y699) and STAT6 (Y641) were upregulated more than 1.5-fold in hypoxic conditions. CREB (S133) and ERK1/2 (T202/Y204, T185/Y187) were downregulated more than 2-fold (0.5 induction or lower) in this cell line. In MCF-7 cells, only eNOS was upregulated more than 1.5-fold, whereas AKT (S473), ERK1/2 (T202/Y204, T185/Y187), Fgr (Y412), Lck (Y394), p27 (T157), p53 (S392), p70 S6 kinase (T389 and T229) and STAT3 (Y705) were downregulated more than 2-fold.
Fold Induction Of Protein Spot Intensities On Phosphokinase Antibody Array
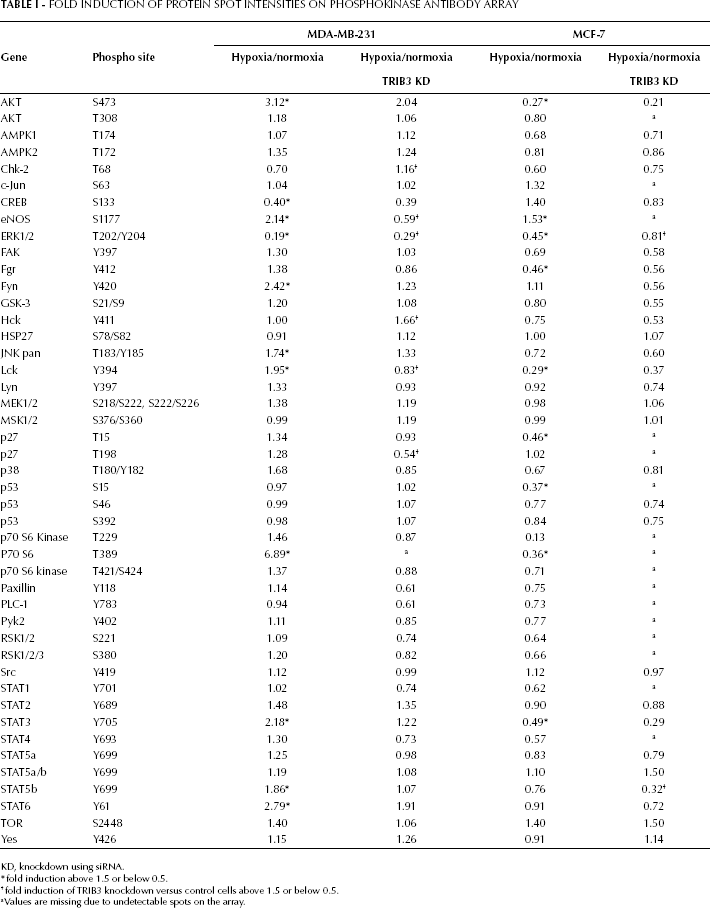
KD, knockdown using siRNA.
fold induction above 1.5 or below 0.5.
fold induction of TRIB3 knockdown versus control cells above 1.5 or below 0.5.
a Values are missing due to undetectable spots on the array.
The role of TRIB3 in these hypoxia-induced changes in kinase expression was determined by siRNA-mediated knockdown of TRIB3 in normoxic and hypoxic incubated MDA-MB-231 and MCF7 breast cancer cells. TRIB3 knockdown attenuated the hypoxia response of ERK1/2 in both cell lines. In MDA-MB-231 cells the hypoxia effect on eNOS and Lck phosphorylation was the opposite for the TRIB3 knockdown cells compared with the control cells. For Chk-2 (T68), Hck (Y411) and p27 (T198) there was a significant effect of TRIB3 knockdown on their response to hypoxia. However, these proteins were not affected by hypoxia in the control cells. A similar observation was made in the MCF-7 cells for STAT5b (Y699).
Western blot reveals pAKT to be hypoxia-regulated and TRIB3 sensitive
Thus, although sometimes contradictory, the phosphorylated forms of AKT, ERK1/2, Lck, p70 S6 kinase and STAT3 were affected by hypoxia in both cell lines. Due to the effect of TRIB3 knockdown on its hypoxia response, eNOS was also considered of interest. Next, the effect of hypoxia and/or TRIB3 knockdown on the expression of these phosphorylated proteins was determined by Western blot analysis (Fig. 1). Unfortunately, no antibody for Western blot analysis was available for Lck (Y394) and therefore validation of the results for this protein could not be performed. Further, p70 S6 kinase (T389 and T229) was below the detection limit in MDA-MB-231 cells. In MCF-7 cells, we observed no effect of hypoxia on the expression of p70 S6 kinase (T389 and T229) by Western blotting. In both MDA-MB-231 and MCF-7 cells, we could not observe an increase in p-eNOS during hypoxia, neither were differences in expression levels detectable after TRIB3 knockdown. Although ERK1/2 (T202/Y204, T185/Y187) was clearly downregulated on the phosphokinase array, we were not able to validate this result using Western blot analysis.
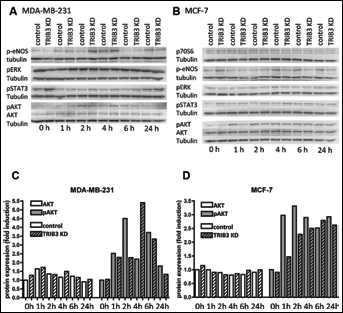
Western blot analysis of selected phosphokinases.
For STAT3 phosphorylation in MDA-MB-231 cells we observed a decrease in expression during hypoxia that was most pronounced at 1-6 hours of hypoxia (0.5-0.8-fold reduction, Fig. 2). TRIB3 knockdown had no effect on p-STAT3 in MDA-MB-231 cells. In contrast, in MCF-7 cells a small increase in the expression of p-STAT3 was found over time after hypoxia (up to 1.7-fold induction at 24 hours). Furthermore, TRIB3 knockdown slightly increased the expression of p-STAT3, most pronounced during normoxia with a fold induction of 1.8 compared to control cells.
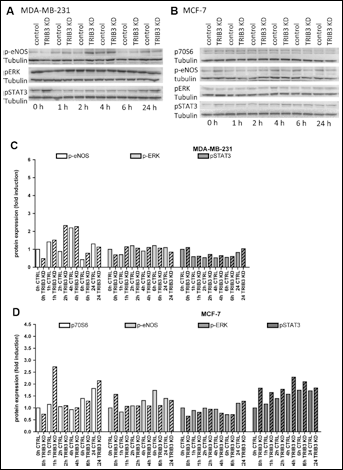
Western blot analysis of selected phosphokinases. Western blots stained for p70S6, p-eNOS, pERK and pSTAT3 of breast cancer cells treated with control siRNA or TRIB3 siRNA (TRIB3 KD) exposed to 1% O2 for 1, 2, 6, and 24 hours. A) MDA-MB-231 cells, B) MCF-7 cells.
Hypoxia induced an increase in AKT (S473) levels in both cell lines that was maximal at 2 hours’ incubation; MDA-MB-231 cells showed a 5.4-fold induction and MCF-7 cells a 4.2-fold induction. The hypoxia response of pAKT seemed to be delayed after TRIB3 knockdown in these cell lines. Both cell lines exhibited a lower induction of AKT (S473) levels after knockdown after 2 hours of hypoxia exposure (2.5-fold and 2.8-fold). At later time points the TRIB3 knockdown effect was less pronounced and for the MDA-MB-231 cells even somewhat reversed.
Effect of hypoxia and TRIB3 knockdown on pAKT is not due to alterations of total AKT expression
Based on the results of the validation of the phosphokinase antibody array on Western blot, we further investigated pAKT. It was upregulated in both cell lines during hypoxia and this upregulation was attenuated by TRIB3 knockdown (Fig. 1). To establish whether hypoxia induces the activation of AKT or the expression of total AKT, we determined total AKT expression after normoxia or hypoxia using Western blots. In both cell lines there was no increase in AKT expression during hypoxia. Furthermore, there was no effect of TRIB3 knockdown on total AKT levels. Thus, the effect of hypoxia and TRIB3 knockdown are specific for the phosphorylation of AKT.
pAKT and TRIB3 localization in hypoxic areas of human breast tumor tissue
To determine whether pAKT and TRIB3 are localized in hypoxic tumor areas, we used tumor tissue from 38 breast cancer patients who were injected with the exogenous hypoxia marker pimonidazole prior to surgery. In these specimens we observed abundant pAKT staining located in the hypoxic tumor areas, as was indicated by pimonidazole positivity (Fig. 3). This colocalization was not limited to tumors with large hypoxic areas but was also observed in tumors that had only focal hypoxic areas. Furthermore, TRIB3 staining was also observed in areas positive for pAKT and pimonidazole (Fig. 3B). Thus, in human breast cancer tissues hypoxic regions are present that express pAKT and TRIB3.
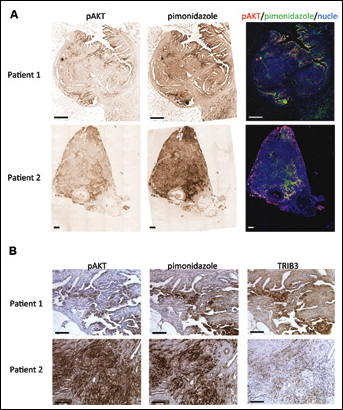
pAKT, pimonidazole and TRIB3 staining in human breast cancer tissues.
pAKT is prognostic in breast cancer patients
To determine the prognostic value of pAKT expression, we stained a tumor microarray with tissue from breast tumors of 95 patients for pAKT, and correlated these results with previously established TRIB3 scoring (13) and outcome. The pAKT staining was scored for both nuclear and cytoplasmic staining, and was considered either negative or positive. Nuclear staining was scored positive in 7 of these 95 patients. Cytoplasmic staining was positive for 51 patients. For further analysis we only used the cytoplasmic staining score because of the low number of positive scoring for nuclear staining. A weak but significant positive correlation between TRIB3 expression and pAKT expression was observed (rs=0.243, p=0.018). pAKT expression showed no difference between groups in the clinical parameters tested besides a significant difference of pAKT expression between HER2-negative versus HER2-positive patients (p=0.04) and patients who received a mastectomy versus patients who received a lumpectomy (p=0.04) (Tab. II). Furthermore, a significantly higher 5-year DFS rate was observed for the pAKT-negative group compared with the positive group (86.1% [80.8-91.4] vs 64.6% [57.9-71.3]) (Fig. 4). Cox regression analyses (Tab. III) revealed a hazard ratio (HR) of 2.87 (95% CI, 1.14-7.23, p=0.03) for DFS for patients with pAKT-expressing tumors compared to patients with pAKT-negative tumors. This remained significant (p=0.007, HR 3.77 (95% CI, 1.43-9.94) after correction for ER, PR and HER2 status, histological grade, tumor type, tumor size and type of treatment (surgery and/or radiotherapy).
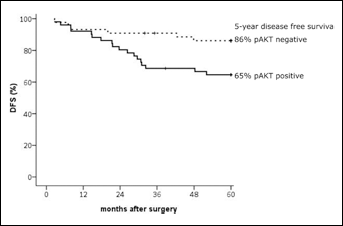
Disease-free survival in patients stratified based on pAKT expression. Kaplan Meier curve of disease-free survival for patients without pAKT expression (dotted line) and with pAKT expression (continuous line).
Patient Characteristics Among Pakt Expression Groups
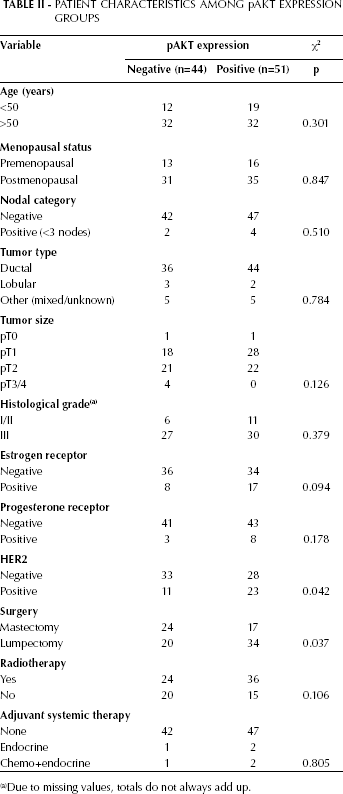
Due to missing values, totals do not always add up.
Univariate And Multivariate Cox Regression Analyses Of Disease Free Survival
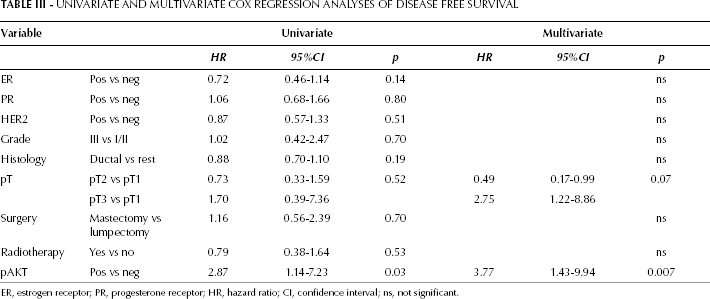
ER, estrogen receptor; PR, progesterone receptor; HR, hazard ratio; CI, confidence interval; ns, not significant.
Discussion
In this study, we profiled the phosphokinase signaling pathways regulated by hypoxia in 2 different breast cancer cell lines. AKT activation by phosphorylation at serine 473 was clearly induced during hypoxia, but total AKT levels remained constant. Knockdown of the scaffold protein TRIB3 attenuated the early hypoxia-induced upregulation of pAKT. Furthermore, pAKT was predominantly localized in hypoxic areas of human breast cancer samples, and in a separate cohort of breast cancer patients pAKT expression was associated with a poor prognosis.
This is the first study to profile the phosphoprotein response to hypoxia in breast cancer cells. We found 6 phosphorylated proteins that were altered during hypoxia in both breast cancer cell lines tested. Several of these proteins have been suggested to have a relationship with hypoxia (18-23). However, their direct regulation by oxygen depletion was not specifically studied before. For pAKT and pERK1/2 the upregulation by hypoxia has been described in multiple cell lines including the breast cancer cell line MCF-7 (19, 22). We could not confirm this result for pERK1/2, but observed similar results for pAKT.
In addition, using Western blot analysis we observed that TRIB3 knockdown attenuated the early hypoxia-induced upregulation of pAKT. This effect was most pronounced at the earlier time point (2 hours). As the phosphokinase array was performed after a 6-hour hypoxic incubation period, this effect was smaller and therefore just outside the limits set for further validation. The result that TRIB3 knockdown attenuates the activation of AKT is in conflict with earlier results describing that TRIB3 blocks the phosphorylation of AKT (5, 24). However, some of these results are under debate (25). The here described effect of TRIB3 knockdown on pAKT levels was only observed at 1 and 2 hours of hypoxia exposure and not during normoxia or longer hypoxia exposure. This indicates that the putative regulatory effect of TRIB3 on AKT activation specifically holds true in the early activation of AKT during hypoxia. Apparently, TRIB3 knockdown delays the activation of AKT, which would be in line with the scaffolding function of TRIB3 in fine-tuning multiple survival pathways during hypoxia. However, to fully understand the mechanism behind this delay and the effect of the delay on cell survival, the function of TRIB3 during hypoxia needs to be examined to a greater extent including other major survival pathways like the UPR.
In addition to the increased activation during hypoxia, the observation that pAKT is located in hypoxic areas of breast cancer tissues suggests a role for pAKT in hypoxic cell survival in breast cancer. Hypoxic cells are resistant to a variety of treatment regimens and, in fact, in vitro studies also show increased pAKT expression to be associated with treatment resistance (26, 27). Thus, targeting pAKT could provide a new method to overcome treatment resistance and improve patient outcome. In vitro, promising results have been obtained in reducing resistance to chemo- or radiotherapy and tamoxifen treatment (28, 29). Multiple specific AKT inhibitors have already been tested in phase I/II clinical trials and they are generally well tolerated (30).
It should be noted that hypoxic cells are associated not only with treatment resistance but also with an increase in genomic instability and metastatic propensity. This fits well with our observation that tumor pAKT expression denoted a poorer prognosis in breast cancer patients, in the absence of adjuvant systemic treatment. This result is in line with earlier results showing that pAKT is related to a higher risk of relapse (reviewed in 31). In earlier studies, the effect of pAKT expression on tumor progression was mainly found in a subset of breast cancer patients, namely patients treated with endocrine therapy or radiotherapy, or patients overexpressing HER2 (32-34).
In our cohort the effect of pAKT expression on prognosis was seen in patients who did not receive adjuvant systemic treatment. Thus, pAKT has specific prognostic value even in the absence of treatment and is not only indicative of sensitivity to systemic therapy. Inhibiting AKT activation might lead to more hypoxic cell death and thereby less hypoxic tumors, an increase in the response to treatment, as well as less aggressive tumor growth. In conclusion, our findings that AKT is activated in hypoxic conditions could provide additional targets for cancer treatment that also targets hypoxic cells (which are usually less sensitive to treatment) and thereby improves patient outcome.
Footnotes
Acknowledgments
We are grateful to M. Pluk, M. Smeekens, M. Roeffen and W. Peeters for their technical assistance.
As approved by the Radboud University Nijmegen Medical Centre Institutional Review Board and according to the “Code Proper Secondary Use of Human Tissue” developed by the Federation of Medical Societies (FMWV) in the Netherlands and Dutch national law, coded tumor tissues from anonymized patients were used. Additional patient material was acquired using a research protocol approved by the Institutional Review Board at the University of North Carolina Hospitals. These patients signed informed consent prior to their participation in the study.
