Abstract
BACKGROUND:
Tribbles pseudokinase 3 (TRIB3) is a member of the tribbles-related family, which has been determined in various cancers, including renal cell carcinoma, acute promyelocytic leukemia, colorectal cancer, endometrial cancer, and glioma. However, its role in retinoblastoma (RB) has not yet been explored.
METHODS:
The expression level of TRIB3 was detected in RB tissues and cell lines using qRT-PCR. The effects of TRIB3 on cell proliferation and invasion capacities were analyzed with MTT, crystal violet, and transwell assays. Western blot and rescue assays were conducted to explore the underlying mechanism.
RESULTS:
This study found that TRIB3 was upregulated in human RB tissues compared to adjacent normal tissues both at the mRNA and protein levels. Overexpression of TRIB3 significantly promoted cell proliferation and invasion of RB cells, while TRIB3 knockdown inhibited these processes. Moreover, the mechanism deciphering experiments showed that TRIB3 overexpression can increase AKT and mTOR phosphorylation. Conversely, TRIB3 knockdown decreased the phosphorylation of AKT and mTOR. Additionally, MK2206, a potent AKT inhibitor, blocked the promotive effects of TRIB3 in RB cells.
CONCLUSION:
This study demonstrated that TRIB3 acts as an oncogene and plays a crucial role in the proliferation and invasion of RB cells via regulating the AKT/mTOR signaling pathway. Therefore, TRIB3 may serve as a potential target in the diagnosis and/or treatment of RB.
Introduction
As one of the most common tumors worldwide, retinoblastoma (RB) is developed from the cells in the nuclear layer of the retina and accounts for 3 % of all pediatric tumors [1]. RB often occurs in developing countries and most cases develop to an advanced stage [2]. Progress has been made in recent years regarding RB treatment, including the development of novel anti-cancer therapies such as external beam radiation and chemotherapy [3]. However, some major problems emerge during the treatment process, including serious complications, a high recurrence rate, and generally poor prognosis [4]. Recent studies have demonstrated that the occurrence and development of RB is a very complex process. Many new RB biomarkers have been determined to be useful in diagnosis, both as prognostic indicators and in contributing to the understanding of RB pathogenesis, which aids in the determination of treatment strategies [5]. Therefore, it is imperative to search for new therapeutic strategies to improve the clinical efficacy of treatments for patients with RB.
As a member of the tribbles-related family, tribbles pseudokinase 3 (TRIB3), was first reported in Drosophila as an inhibitor of mitosis that regulates string/CDC25 during morphogenesis [6]. TRIB3 contains a serine/threonine kinase catalytic domain but lacks the conserved catalytic motif essential for kinase activity [7]. Many studies have shown that TRIB3 is an important stress-related gene that regulates phosphatidylinositol 3-kinase (PI3K) signaling pathways, transforming growth factor-beta and mitogen-activated protein kinase, as well as exerting fundamental roles in regulating blood sugar, lipid metabolism, adipocyte differentiation, and cell apoptosis [8]. Recently, mounting evidence has suggested that TRIB3 was highly involved as an oncogene or tumor suppressor gene in multiple cancers, including renal cell carcinoma [9], acute promyelocytic leukemia [10], colorectal cancer [11], endometrial cancer [12], and glioma [13]. Nevertheless, the roles of TRIB3 in RB remain unclear.
In our study, we focused on the importance of TRIB3 in the progression of RB. Our results demonstrated that TRIB3 is upregulated in human RB tissues compared to para-cancerous tissues. Assessing the biological functions of the RB cells, we demonstrated the significance of TRIB3 in regulating the proliferation and invasion of RB cell lines. Moreover, we found that the AKT signaling pathway was highly activated in TRIB3-mediated progression of RB cells.
Materials and methods
RB tissue samples
A total of 42 RB tissues and paired adjacent normal tissues (within 2 mm around the tumor site) were obtained from each patient using biopsy procedures at Huainan Chenguang Eye Hospital (Anhui, China). Following this, 30 paired samples were subjected to RNA extraction for reverse transcription-quantitative (RT-q) PCR and 12 paired samples for analyzed using western blot assay. Tissue pathology and histological diagnosis were confirmed by at least two independent pathologists. No initial therapies were applied to all the patients who had no previous malignancies. Enrolled patients signed informed consent prior to study. The Ethics Committee of the Huainan Chenguang Eye Hospital approved the study. The Table 1 demonstrates patients’ basic clinical characteristics.
The clinicopathologic features in 42 retinoblastoma patients
The clinicopathologic features in 42 retinoblastoma patients
TRIzol reagent (Invitrogen) was used to extract total RNAs from the biopsy tissues of the 30 patients. According to the manufacturer’s instructions, 2
Cell culture conditions
RB cell lines Y79, WERI, HXO-RB44, and NCI-H187 were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). All cell lines were cultured in DMEM medium supplemented with 10% fetal bovine serum (FBS) (Invitrogen), 100 U/ml penicillin and 100 smg/ml streptomycin at 37
Cell transfection
The full-length cDNA encoding human TRIB3 was extracted from human whole blood samples using RT-PCR. Following this, TRIB3 cDNA was cloned into a p23-3xflag-GFP vector according to the manufacturer’s instructions. Lentiviral supernatants were produced using the Lenti-X HTX packaging system (Clontech Laboratories, Inc.). RB cell lines were transduced with supernatants from empty vector cells to generate negative controls. The fluorescence and infection efficiency were determined using an inverted fluorescence microscope using GFP sorting. TRIB3-Flag tagged over-expression was confirmed using anti-Flag antibodies (cat. no. 8164; 1:1,000; Cell Signaling Technology, Inc.).
Short hair RNA (shRNA) plasmids for TRIB3, which were designed against the TRIB3 gene and assembled into Phblv-u6-puro vectors, were purchased from Shanghai GenePharma Co., Ltd. Non-target scrambled oligonucleotides (Shanghai GenePharma Co., Ltd) served as negative controls. The plasmid transfection was verified by sequencing. Cells were cultured and screened in medium containing 4
Western blot
Samples were lysed in protease inhibitor cocktail containing RIPA solution, and total protein was extracted. Protein concentration was determined using the Bradford reagent (Sigma). Then, proteins (10
Crystal violet assay
Briefly, the control and experimental cells were seeded into 6-well plates at a density of 1000 cells/well. The cells were cultured in complete medium supplemented with 10% FBS. The medium was changed every three days. After two weeks, the medium was removed, the cells were fixed with 20% methanol at room temperature for 10 minutes, and then stained with 0.5% crystal violet (Sigma-Aldrich; MerckKGaA). After 10 min, the fixed cells were washed with phosphate-buffered saline (PBS) and photographed. The absorbance values at 600 nm were measured using a microplate reader.
MTT cell viability assay
Cells (three replicates for every condition/treatment) at a density of 1000 cells/well were seeded into 96-well plates. Following this, 20
Transwell migration assay
Transwell 24-well chambers (8.0
Statistical analysis
All statistical evaluations were performed using GraphPad Prism 5 (GraphPad Software Inc., CA, USA). The experimental data is presented as mean
Results
TRIB3 is upregulated in RB tissues
To assess TRIB3 expression in RB, the expression levels of TRIB3 were detected in 30 pairs of RB tissues and the adjacent normal tissues using qRT-PCR. The results of qRT-PCR showed that TRIB3 was significantly upregulated in 23 out of 30 (76.7%) RB tissues compared to adjacent normal tissues (Fig. 1A). The expression levels of TRIB3 proteins were also determined using western blot, the results of which showed that the expression levels of TRIB3 were higher in RB tissues compared to those in the adjacent normal tissues (Fig. 1B). This was consistent with the qRT-PCR results. Taken together, the above data indicated that TRIB3 is upregulated in RB tissues at both mRNA and protein levels.
TRIB3 was upregulated in RB tissues. (A) TRIB3 mRNA levels in 30 pairs of tumor samples (T) relative to adjacent normal tissues (N) determined by qRT-PCR. (B) TRIB3 protein levels in 12 pairs of tumor samples (T) and adjacent normal tissues (N) determined by western blot. RB, retinoblastoma.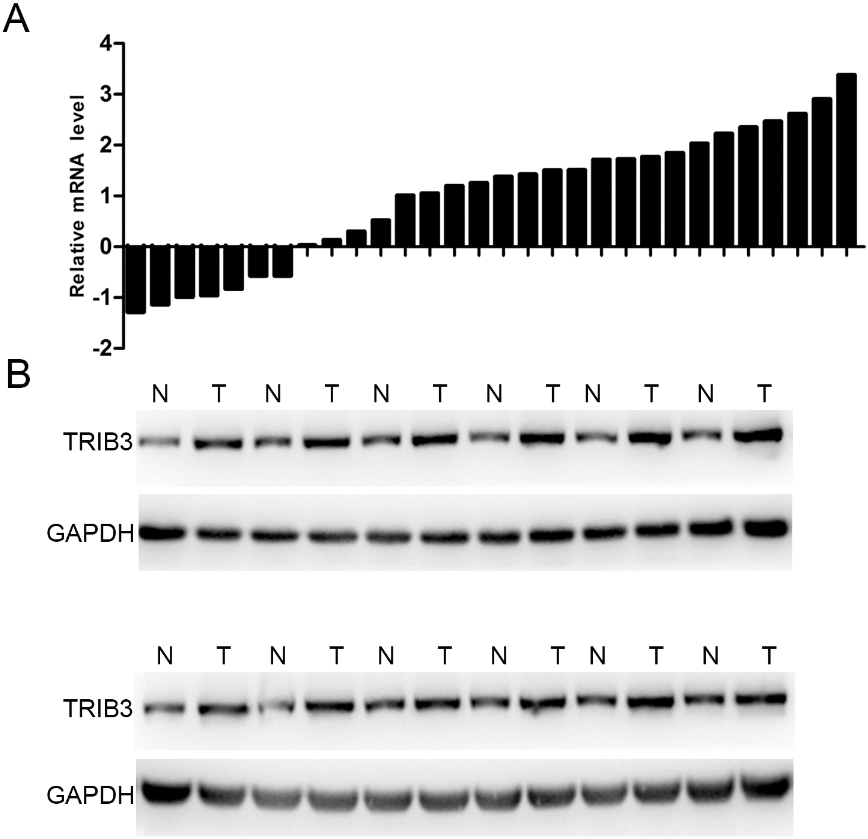
TRIB3 overexpressed and knockdown RB cell lines were constructed to further investigate the role of TRIB3 in RB cells. Firstly, the endogenous TRIB3 expression levels were detected in several RB cell lines (Y79, WERI, HXO-RB44, and NCI-H187). The results showed that the expression of TRIB3 was higher in Y79 and HXO-RB44 cells than in WERI and NCI-H187 cells (Fig. 2A). To identify the function of TRIB3 in RB cells, we established TRIB3 overexpression in WERI and NCI-H187, due to the relative lower expression of TRIB3 in these cell lines (Fig. 2B). Additionally, we used specific shRNA to knockdown TRIB3 expression in Y79 and HXO-RB44 cells, which had relatively higher expression of TRIB3 (Fig. 2C).
Overexpression and knockdown of TRIB3 in RB cell lines. (A) Western blots showing endogenous TRIB3 expression in several RB cell lines. Left panel: Result of western blot. Right panel: Grey analysis results of the western blot bands. (B) Western blots showing the overexpression of TRIB3 in WERI and NCI-H187 cells. Left panel: Result of western blot. Right panel: Grey analysis results of the western blot bands. (C) Western blots showing the knockdown of TRIB3 in Y79 and HXO-RB44 cells. Left panel: Result of western blot. Right panel: Grey analysis results of the Westernblot bands. GAPDH was used as a loading control.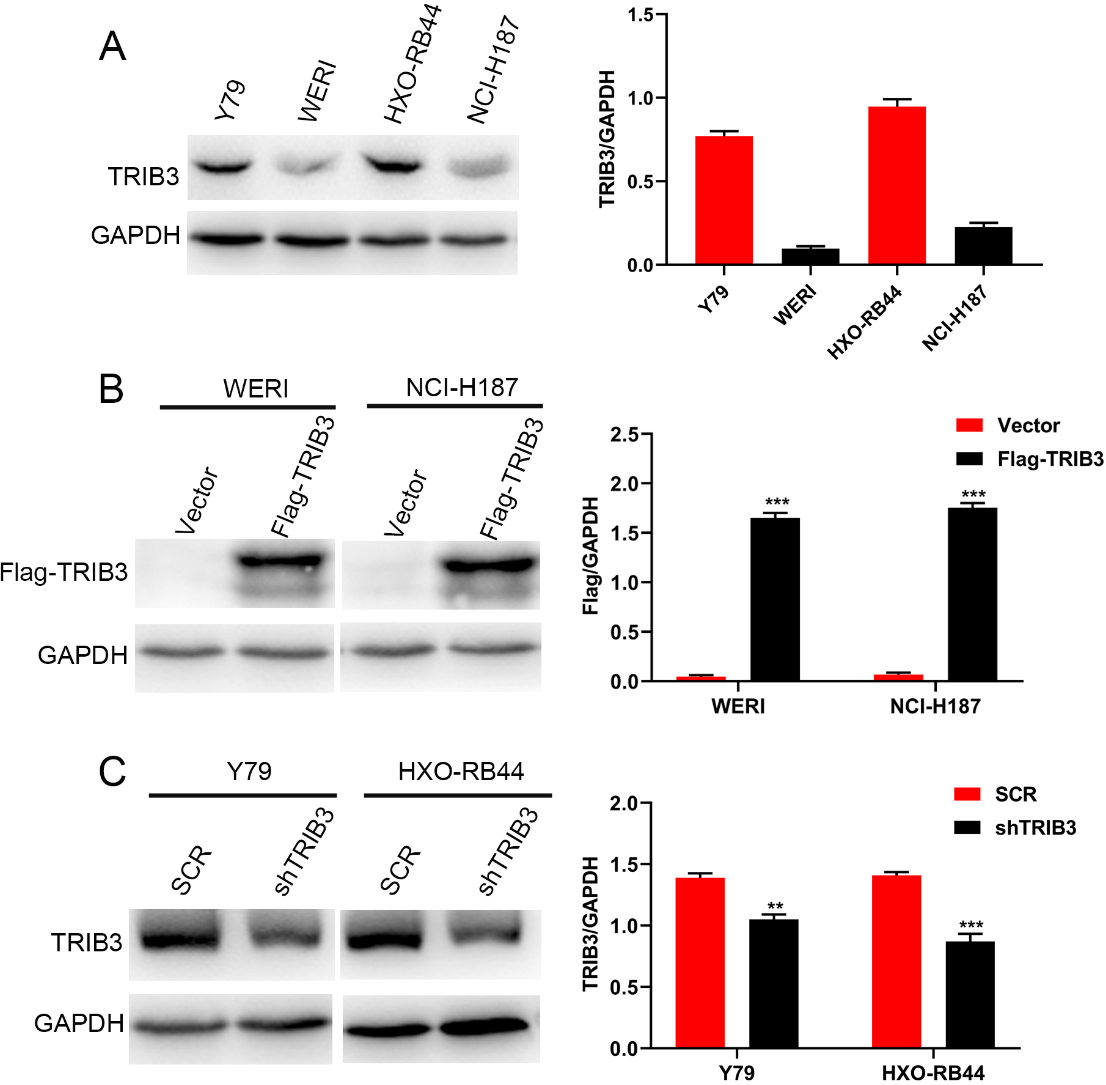
The influence of TRIB3 on proliferation abilities of RB cell lines. (A) The effect of TRIB3 overexpression on the viability of WERI and NCI-H187 cells was assessed using MTT assay. (B) The effect of TRIB3 knockdown on the viability of Y79 and HXO-RB44 cells was assessed using MTT assay. (C) The effect of TRIB3 overexpression on the viability of WERI and NCI-H187 cells was assessed using crystal violet assay. Right panel represents the OD value of crystal violet assays. (D) The effect of TRIB3 knockdown on the viability of Y79 and HXO-RB44 cells was assessed using crystal violet assay. Right panel represents the OD value of crystal violet assays. **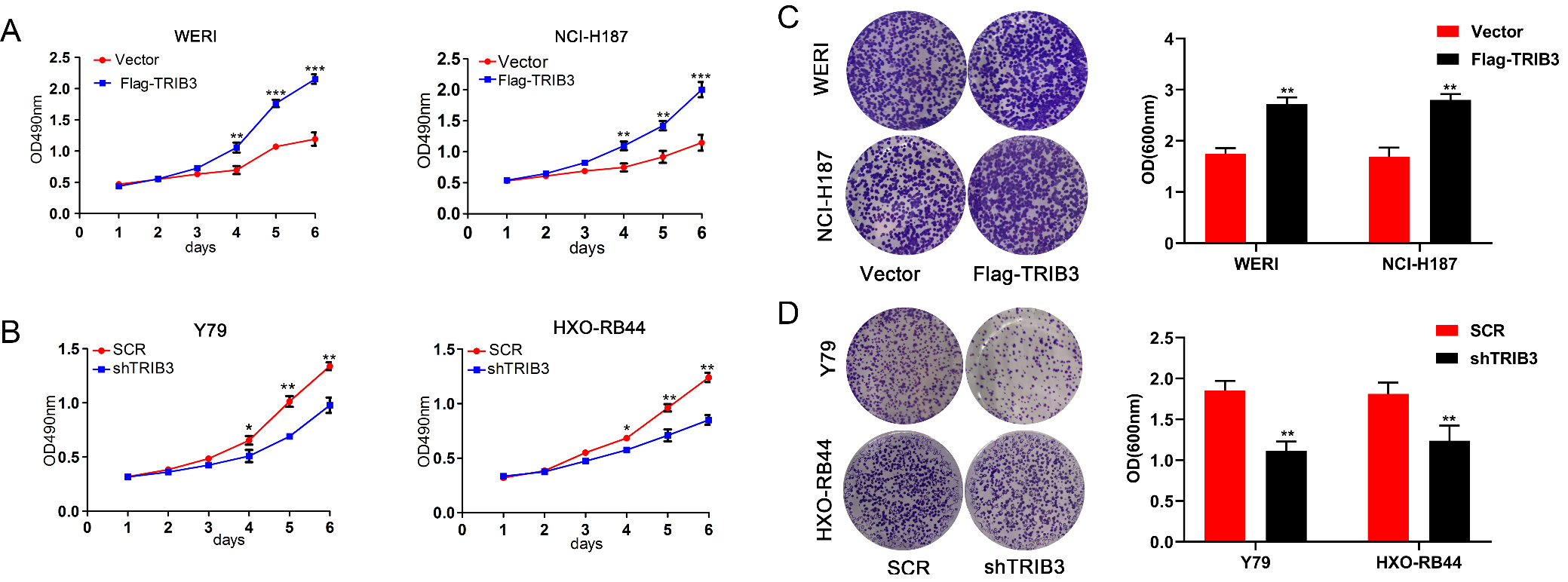
The influence of TRIB3 on invasion abilities of RB cell lines. (A–B) The effects of TRIB3 overexpression on the invasion of WERI and NCI-H187 cells were assessed using transwell assays (Magnification: 200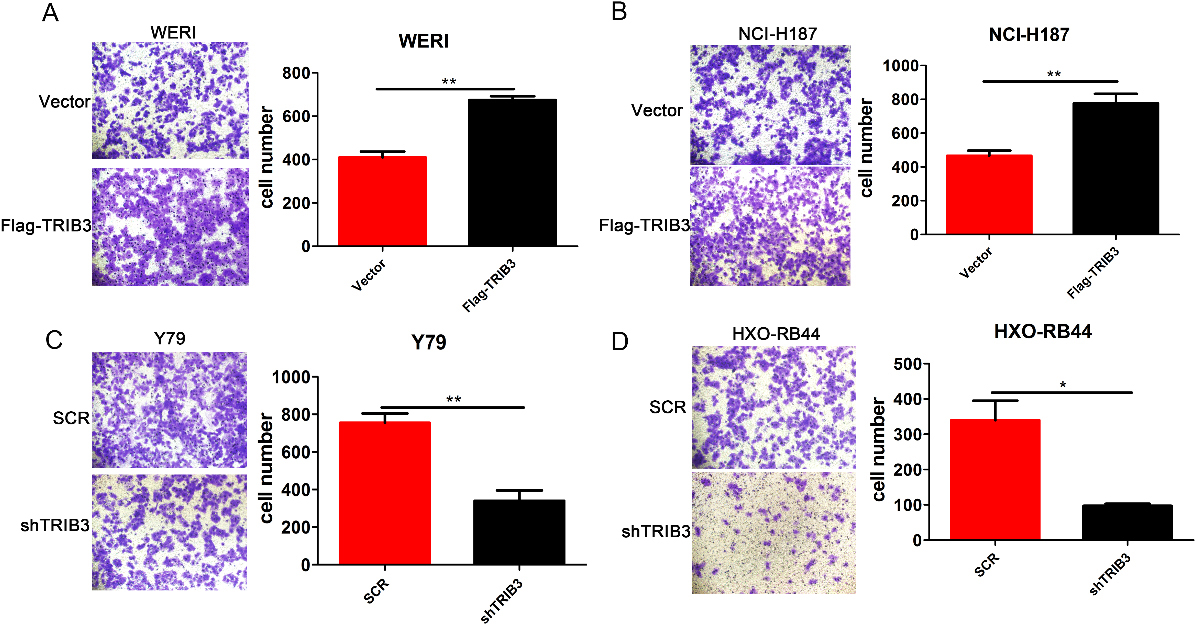
TRIB3 activated the AKT signaling pathway. (A) The overexpression of TRIB3 activated the AKT signaling pathway in both WERI and NCI-H187cells, as seen in western blot results. (B) Grey analysis results of the western blot bands of TRIB3 overexpression. (C) The knockdown of TRIB3 inhibited the AKT signaling pathway in both Y79 and HXO-RB44 cells, as seen in western blot results. (D) Grey analysis results of the western blot bands of TRIB3 knockdown. GAPDH was used as a loading control in all the cases.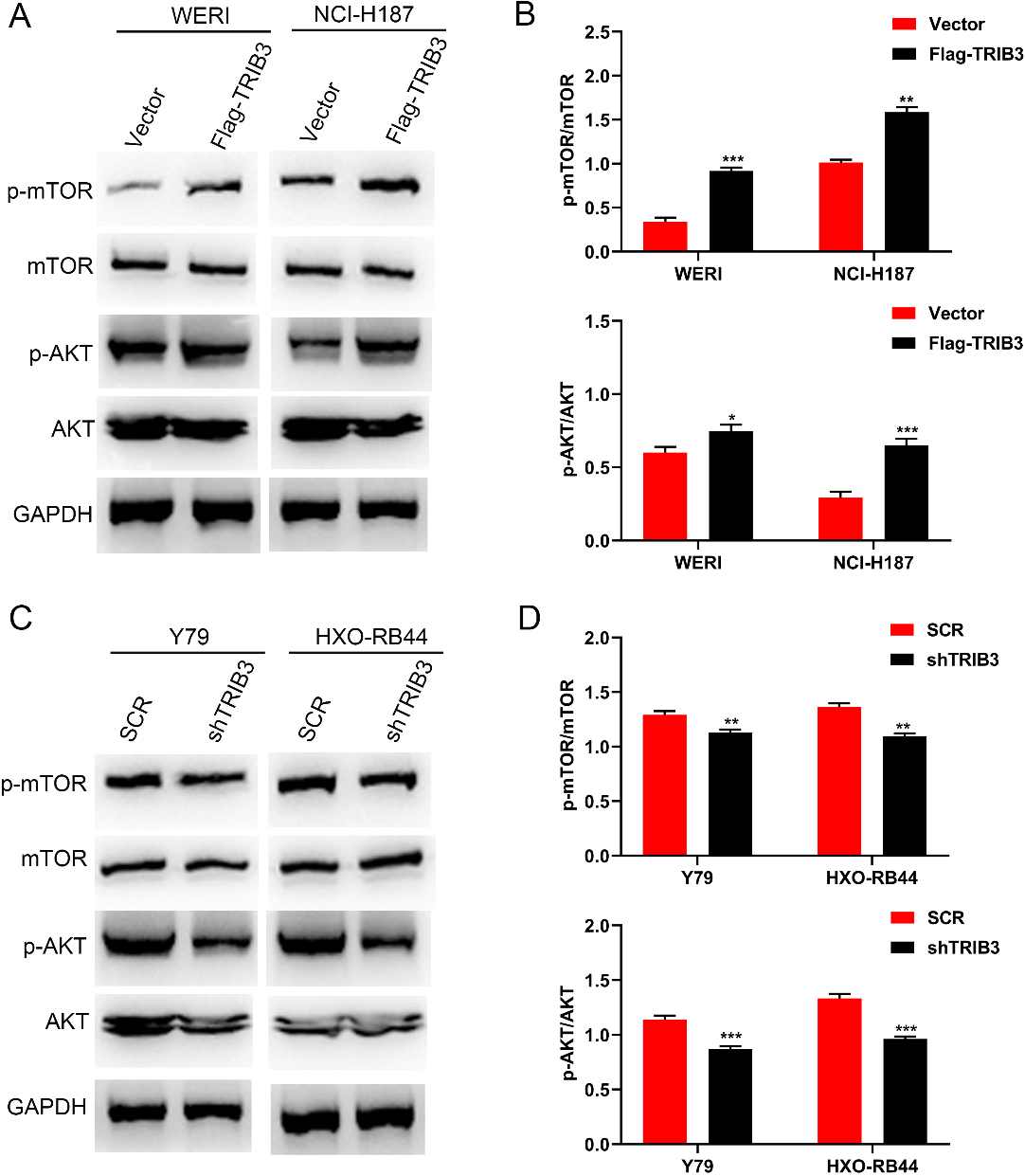
MTT and crystal violet assays were used to detect the proliferation rate of RB cells. The MTT assay showed that the absorbance values of the WERI and NCI-H187 RB cells at 4, 5, and 6 days after transfection with TRIB3 overexpression vectors were significantly higher than those of the vector-transfected control cells (
MK2206, a potent AKT inhibitor, inhibited AKT phosphorylation and reversed the RB phenotype of TRIB3 overexpression. (A) MK2206 inhibited AKT phosphorylation in WERI and NCI-H187 cells after TRIB3 overexpression according to western blot. (B) Grey analysis results of the western blot bands. (C, D) MK2206 rescued the growth promotion induced by TRIB3 in WERI (C) and NCI-H187 (D) cells, which was assessed by MTT assay. Left photograph: crystal violet assay. (E) MK2206 rescued the invasion promotion induced by TRIB3, which was assessed by transwell assay. Left photograph: transwell assay. Right photograph: calculation of number of cells that invaded through the filter following eosin staining. ns 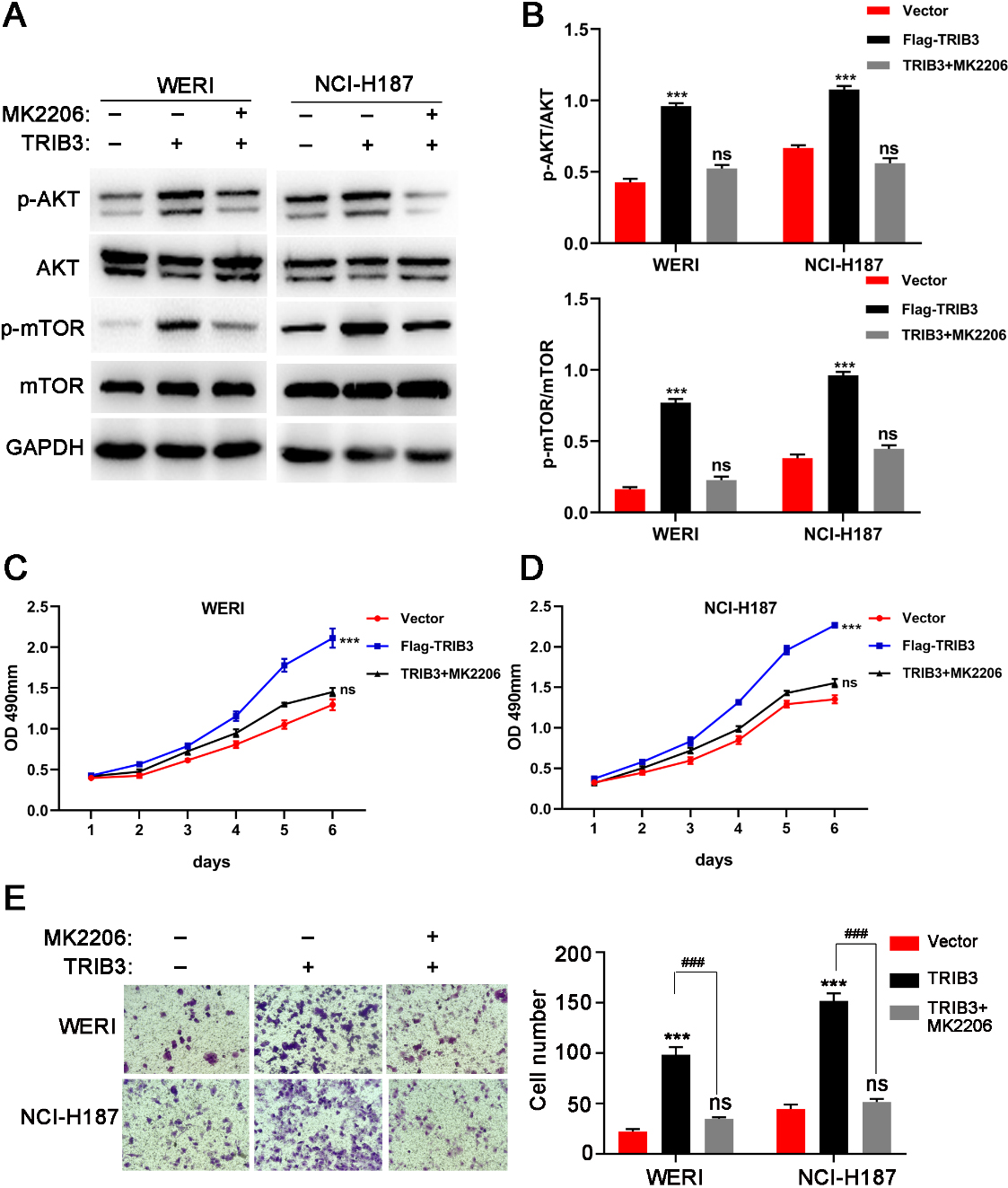
Transwell migration assays were used to estimate the invasion capacity of RB cells. As shown in Fig. 4A and B, overexpression of TRIB3 notably promoted the invasion of WERI and NCI-H187 cells (all
TRIB3 overexpression activates the AKT/mTOR signaling pathway in RB cells
To clarify the specific mechanism of TRIB3-induced development of RB, western blot analysis was performed to investigate the expression levels of phosphorylated and total AKT proteins in TRIB3-overexpressing and -knockdown cell lines. The results showed that TRIB3 overexpression in WERI and NCI-H187 cells resulted in a marked increases in AKT and mTOR phosphorylation levels with no changes in the total AKT and mTOR levels (Fig. 5A and B). Alternatively, downregulation of TRIB3 in SCC9 and SCC15 cells significantly reduced the phosphorylation levels of AKT and mTOR (Fig. 5C and D). Conclusively, these results demonstrate that AKT signaling pathway is actively involved in the TRIB3-mediated promoting effect in RB cells.
MK2206, a potent AKT inhibitor, inhibited AKT phosphorylation and reversed the RB phenotype of TRIB3 overexpression
The present study examined whether the effects of TRIB3 on the progression of RB were AKT-dependent. MK2206, a potent AKT inhibitor, was incubated with TRIB3 overexpressing WERI and NCI-H187 cells to confirm whether AKT pathway activation could be blocked to rescue the phenotype. Western blot analysis confirmed that MK2206 inhibited phosphorylation of AKT and mTOR in the TRIB3 overexpressing WERI and NCI-H187 cells (Fig. 6A and B). As shown in Fig. 6C–E, TRIB3-induced promotive effects on cell proliferation and invasion were rescued by MK2206. These observations indicated that the promotive effects of TRIB3 were AKT-dependent.
Discussion
In the last few years, numerous researchers have concentrated on exploring the underlying mechanism of pathogenesis in RB [14]. Ample evidence has demonstrated that a wide variety of molecules such as miRNAs and genes are likely associated with RB [15]. There is currently high demand for new therapeutic strategies to improve the clinical efficacy of treatments for patients with RB.
In recent years, multiple studies have shown the fundamental role of TRIB3 in cancer initiation and progression. In patients with colon cancer and breast cancer, the expression of TRIB3 was closely associated with long-term prognosis [11]. Hong et al. demonstrated that TRIB3 expression was upregulated in renal cell carcinoma tissues and that a high expression of TRIB3 was correlated with both advanced tumor stage a negative prognosis [9]. Furthermore, Hong et al. reported that TRIB3 plays an anti-apoptotic role in doxorubicin-treated gastric cancer cell lines, which may reflect the efficiency of TRIB3 for cancer therapy [16]. With respect to the opposite effect of TRIB3 in different cancers, Sack at al. reported that cell-type specificity plays an important role in cancer genetics, which reveals that some genes may paly different biological roles in different tissues or cells [17].
In this study, we found that TRIB3 was upregulated in human RB tissues, both at the mRNA and protein levels, compared to adjacent normal tissues. Regarding the biological functions of TRIB3 in the progression of RB cells, this study determined that overexpression of TRIB3 promoted the proliferation, migration, and invasion of RB cells. Accordingly, downregulation of TRIB3 inhibited the proliferation, migration, and invasion of RB cells. We also investigated the underlying mechanism of the oncogenic role of TRIB3 in RB progression. The western blot analysis showed that phosphorylation levels of AKT and mTOR were significantly increased following overexpression of TRIB3. Conversely, knockdown of TRIB3 led to a decrease in AKT and mTOR phosphorylation levels. MK2206, an allosteric inhibitor of AKT, was used to rescue the phenotype of RB cells with TRIB3 overexpression. MK2206 inhibited RB proliferation and migration by inhibiting the level of AKT phosphorylation activated by TRIB3 overexpression in RB cells. This study indicated that TRIB3 may promote proliferation, migration, and invasion of RB cells via regulation of the AKT signaling pathway. The AKT pathway is a central regulator for cancer proliferation, tumorigenesis, and metastasis in multiple cancers [18, 19, 20]. PI3K is a lipid kinase family that phosphorylates the phosphate-3-hydroxyl. Both AKT and mTOR are downstream targets of PI3K that can regulate protein synthesis, cell growth, and proliferation. mTOR is an important component of the network, as well as a PI3K-associated serine-threonine kinase that can regulate proliferation and metastasis mechanisms by phosphorylating AKT [21, 22]. Several previous studies support our findings, which demonstrated the fundamental role of AKT signaling pathway in TRIB3-mediated biological processes. Qu et al. reported that TRIB3 inhibited proliferation and migration and promoted apoptosis of endometrial cancer cells, possibly via regulation of the AKT signaling pathway [12]. Restelli et al. found that TRIB3 could inhibit several cancer-related processes such as cell growth and invasion by binding to Ser473 of the AKT protein kinase [23]. Zhang et al. claimed that TRIB3 overexpression promoted the apoptosis of tongue squamous cell carcinoma by blocking AKT activation [24]. However, it was still uncovered as regard to the more specific mechanism that how TRIB3 induce Akt phosphorylation and mTOR phosphorylation. Accumulating evidence described in the literature have shown that the PI3K/PTEN/AKT/TSC/mTOR signaling pathway may provide a novel insight into the deep molecular mechanism during the cancer progression [25, 26, 27]. TRIB3 may induce Akt phosphorylation via suppressing the expression of PTEN. TRIB3 may also stimulates progression of RB cells by activating the mTOR pathway through the inhibition of the heterotrimeric complex consisting of tuberous sclerosis 1 or hamartin (TSC1) and tuberous sclerosis 2 or tuberin (TSC2). Hence, our future study needs to focus on the potential role of PI3K/PTEN/AKT/TSC/mTOR axis as regard to the oncogenic TRIB3 in RB cells.
To our knowledge, this is the first study to uncover the biological role of TRIB3 in the progression of RB. However, the study has its limitations. For instance, animal experiments should be conducted to further explore the regulatory mechanism of AKT signaling pathway in vivo.
This study demonstrated that TRIB3 acts as an oncogene and plays a crucial role in the proliferation, migration, and invasion of RB cells via regulation of the AKT/mTOR signaling pathways.
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
