Abstract
Porcine reproductive and respiratory syndrome virus (PRRSV) is an economically significant agent for which there currently are no effective treatments. Development of antiviral agents for PRRSV as well as many other viruses has been limited by toxicity of known antiviral compounds. In contrast, antibiotics for non-virus microbial infections have been widely useful, in part because of their acceptable toxicity in animals. We report here the discovery that the quinolone-containing compound Plasmocin™, as well as the quinolones nalidixic acid and ciprofloxacin, have potent anti-PRRSV activity in vitro. PRRSV replication was inhibited by these antibiotics in both cultured MARC-145 cells and cultured primary alveolar porcine macrophages (PAMs). Furthermore, sub-optimal concentrations of nalidixic acid synergized with antiviral cytokines (AK-2 or IFN-γ) to quantitatively and qualitatively inhibit PRRSV replication in MARC-145 cells or PAMs. The antiviral activity of Plasmocin and nalidixic acid correlated with reduced actin expression in MARC-145 cells. Replication of the related lactate dehydrogenase-elevating virus (LDV) was also inhibited in primary mouse macrophages by Plasmocin. These results are significant to the development of antiviral strategies with potentially reduced toxicity, and provide a model system to better understand regulation of arterivirus replication.
Introduction
Antimicrobial agents used to treat bacterial or fungal infections generally interfere with specific processes that are essential for microbe growth and/or division, for example cell wall synthesis, cytoplasmic membrane function, nucleic acid synthesis, or ribosome function (Neu and Gootz, 1996). On the other hand, antiviral chemotherapy may target the cells supporting virus replication as well as events specific to the virus, with potentially toxic side effects (De Clerq, 1996). Toxicity to the host is also well known for some antibiotics, including hematologic suppression, kidney and liver injury due to metabolism in those organs, as well as neurological side effects (Snavely and Hodges, 1984; Neu and Gootz, 1996; Chow et al. 2004), demonstrating the potential for direct host effects of antibiotics. The mechanisms of antibiotic toxicity in mammals are not well understood, but may include enzyme or membrane alterations as well as induction of apoptosis (Gerbaux et al. 1996; Van Bambeke et al. 1998; El Mouedden et al. 2000; Sliva and Schnierle, 2007).
Previous studies have demonstrated that in some cases, antibiotics may have antiviral effects (Subak-Sharpe et al. 1969; Macintyre et al. 1991), most likely due to inhibition of host cell functions required for viral replication. For the present study, several antibiotics were screened for effects on porcine reproductive and respiratory syndrome virus (PRRSV) in vitro. PRRSV is the etiologic agent of an economically-important porcine disease (PRRS; Albina et al. 1997; Prieto et al. 2005) and is also a model for chronic virus infection. The mechanism of chronic PRRSV infection is unclear. The virus induces several cytokines including interferon-gamma (IFN-γ), which inhibits PRRSV replication in vitro (Aasted et al. 2002; Cafruny et al. 2006; Johnsen et al. 2002; Rowland et al. 2001). However, the IFN-γ response in pigs may be delayed or inhibited by unknown factors, and a poorly-neutralizing Th2 response may also contribute to virus persistence (Meier et al. 2003; Meier et al. 2004). PRRSV, and the related lactate dehydrogenase-elevating virus (LDV) of mice, are resistant to vaccination strategies, and antivirals for PRRSV are still in an early stage of development. We show that several quinolone-containing compounds have potent anti-PRRSV effects, which correlated with inhibition of actin expression in MARC-145 cells. Furthermore, quinolone treatment was found to potentiate the cytokine-mediated inhibition of PRRSV replication in MARC-145 cells and in primary pig macrophages. These novel drug effects may provide insights into mechanisms of viral replication as well as the development of future therapeutic agents for virus infection.
Materials and Methods
Target Cells for Virus Replication
PRRSV was inoculated on monolayer cultures of a mycoplasma-free MARC-145 cell line (Kim et al. 1993; Meulenberg, 2000) or on primary alveolar pig macrophages (Oleksiewicz, M.B., Nielsen, J. 1999). The MARC-145 cells were routinely passaged in DMEM containing 10% fetal bovine serum, and for virus infections the medium was switched to MEM containing 2% horse serum. MARC-145 cells for virus infections were grown for 1–2 days in 8-well glass slide chambers (~30–40,000 confluent cells/culture; Lab-Tek II; Nalge Nunc International, as described (Cafruny et al. 2006). Pig cells were collected from 4–8 week old pigs by lung lavage with PBS (Mengeling et al. 1995; Duan et al. 1997; Oleksiewicz et al. 1999) and stored at −70 °C. Vials of these cells were rapidly thawed, centrifuged for 5 min at 1000 × G to obtain a cell pellet, which was resuspended in DMEM containing 10% fetal bovine serum. The cells were then distributed into wells of 8-well slide chambers, and after about 18 h of incubation at 37 °C, non-adherent cells were removed, and the adherent cells (primary alveolar porcine macrophages or PAMs) were incubated for an additional 4–6 h prior to PRRSV inoculation. Typically there were about 10,000–20,000 PAMs per culture well, with variation noted between experiments but not within a given experiment, suggesting that the variation was due to differences among the collected aliquots of alveolar cells. For LDV infection, primary mouse (outbred CF1) peritoneal macrophages were established in 8-well glass slide chambers, and inoculated with LDV within 24 h as described previously (Plagemann and Moennig, 1992; Cafruny et al. 2006).
Viruses and Virus Infections
MARC-145 cells were infected with the PRRSV isolate SD-23983, preparing high-titer (~10 6 TCID50 per ml) virus stocks from the culture supernatants of 48–96 h p.i. MARC-145 cells. For the experiments described, MARC-145 cells were inoculated with an MOI of about 0.1 TCID50 and PAMs with an MOI of about 0.5 TCID50 which are approximately optimal for these systems (Rowland et al. 2001; Kim et al. 2002; Cafruny et al. 2006). LDV-P stocks were prepared as previously described and used to inoculate mouse macrophage cultures with an MOI of about 1000 (Plagemann and Moennig, 1992).
Drugs and Drug Treatments
Plasmocin™ (product of Cayla, www.cayla.com; distributed by Invivogen; ant-mpt), nalidixic acid (Sigma; N5035), ciprofloxacin (Biochemika; 17850), erythromycin (Amresco; 0219-10G), penicillin (Sigma; P3032) and streptomycin (Sigma; S9137) were used as sterile preparations at various concentrations (2.5–250 ug/ml) as indicated in the Results. The recombinant human antiviral cytokines AK-2 (50 ug/ml) and interferon-γ (IFN-γ; 100 ug/ml) were generated by Actokine Therapeutics and used as previously described (Cafruny et al. 2006). AK-2 is a cytokine-based experimental antiviral being developed by Actokine Therapeutics, which consists of recombinant normal human proteins comprising part of the mammalian cell response to virus infection (Wong et al. unpublished; Cafruny et al. 2006).
Fluorescence (FA) Detection of Virus Replication and Actin Expression
PRRSV replication was detected using FITC-labeled IgG anti-PRRSV nucleocapsid antibodies (SDOW17; Nelson et al. 1993). MARC-145 cells or primary alveolar porcine macrophages (PAMs) were fixed in 80% acetone, and incubated for 1 h at 37 °C with a 1:100 dilution of FITC-conjugated SDOW17 antibody made in PBS containing 5% fetal bovine serum. The fixed PAMS were blocked with 5% heat-inactivated normal mouse plasma for 30 min prior to incubation with SDOW17, and in some experiments incubation of PAMs with SDOW17 was for 3 h at room temperature. Actin was stained using 592-phalloidin as described previously (Cafruny et al. 2006). After incubation with the fluorescence antibody reagents, the cells were washed three times with cold PBS prior to examination by confocal fluorescence microscopy using an Olympus BX61 microscope and Fluoview software. Quantitative analyses were performed by directly counting fluorescence-positive cells in experimental and control wells. Multiple experiments were analyzed as shown in the results; the representative images shown in the figures were obtained at equivalent PMT settings within each experiment, and display small scale bars as identified in the figure captions. LDV replication was determined in primary mouse macrophages as described previously (Cafruny et al. 1986), counting the total LDV antigen-positive cells per culture under a fluorescence microscope.
Cytotoxicity Testing
The cells used for these experiments exhibited high (>99%; Table 4) viability. Potential cytotoxicity of the antiviral drugs was assessed by trypan blue dye-exclusion (Freshney, 1987). After a period of drug exposure (Table 4), cell cultures were washed once with PBS and then incubated with 0.4% trypan blue for 5 min at room temperature. Dead cells were counted under a light microscope and comparison made between drug-treated and untreated control cultures. The cells were then fixed and stained with crystal violet, and quantitatively assessed under the microscope to provide an estimate of total-cell counts under the different culture conditions. Based on this method, the drug responses reported here did not correlate with detectable cytoxicity (see below and Table 4).
Results
Inhibition of PRRSV Replication in MARC-145 Cells by Quinolone-Containing Antibiotics
The first indication that some antibiotics were capable of inhibiting PRRSV replication emerged from studies with Plasmocin, a proprietary agent consisting of quinolone and macrolide antibiotics which has potent anti-mycoplasma activity. Treament of MARC-145 cells with Plasmocin was found to inhibit PRRSV replication in a dose- and time-dependent fashion, as shown in Table 1 A,C,D. Figure 1A illustrates the antiviral effect of Plasmocin added at the time of virus inoculation, which almost completely abrogated PRRSV-permissiveness in MARC-145 cells, with a total of only 20 weakly-PRRSV-positive cells in two treated cultures, versus a mean of 458 +/- 40 PRRSV-positive cells in control cultures. In contrast, addition of Plasmocin at 3 or 6 h p.i. resulted in progressively weaker antiviral effects (Fig. 1A and Table 1).
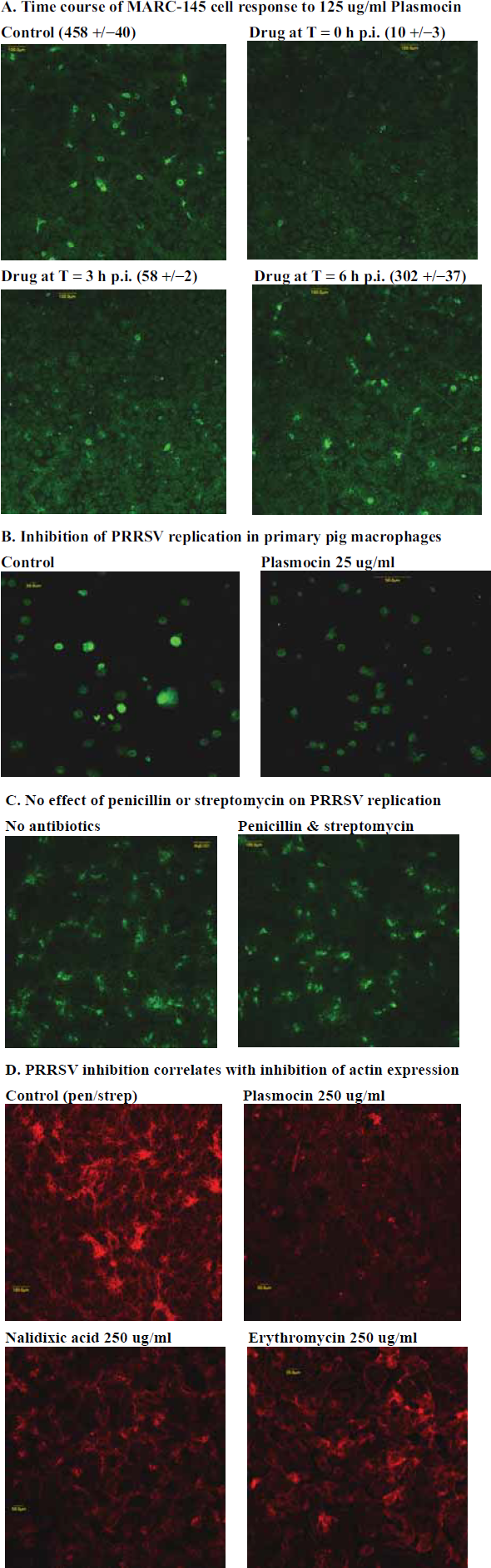
Inhibition of PRRSV replication in cultured MARC-145 cells and in primary alveolar porcine macrophages (PAMs) by Plasmocin, and correlation of the quinolone antiviral effect with inhibition of actin expression in MARC-145 cells.
Antibiotic-mediated inhibition of PRRSV replication in MARC-145 cells.

Drug dose and time of addition are indicated; for C, drugs were added at the time of virus inoculation (T = 0 h p.i.) or at 5 h p.i. (T = 5 h p.i.). Percent results are derived from means of duplicate wells, with <20% variation between well counts. For A-D, dose- and time-dependent antiviral effects of the antibiotics were apparent, and only the 12.5 ug/ml dose of nalidixic acid failed to show at least some antiviral activity.
PRRSV replication in MARC-145 cells was also inhibited by the quinolones nalidixic acid and ciprofloxacin (Table 1B and C). The dose- and time-dependency of the nalidixic acid response are shown in Table 1B and C. In contrast, neither erythromycin (a representative macrolide) nor the combination of penicillin (150 ug/ml) and streptomycin (200 ug/ml) had any significant effect on PRRSV replication in MARC-145 cells (Table 1E and F, and Fig. 1C). Combined, these data show that an early stage of PRRSV replication is inhibited by quinolone antibiotics or quinolone-containing compounds.
Cytotoxicity testing was carried out by trypan blue dye exclusion and total cell count analyses. No cytotoxicity of the antibiotics studied was observed in MARC-145 cells under our experimental conditions (Table 4). Furthermore, continuous treatment of MARC-145 cells for 2 weeks with 25 ug/ml Plasmocin had no detectable effect on cell propagation or viability (Duman et al. unpublished). Thus, the viral responses appear unrelated to general cytotoxicity.
Inhibition of PRRSV Replication in Primary Alveolar Porcine Macrophages (PAMs) by Plasmocin and Nalidixic Acid
Virus responses to Plasmocin and nalidixic acid were similar in primary alveolar porcine macrophages (PAMs) from several different pigs to those seen in MARC-145 cells (Table 2). While there were some variations in the magnitudes of the antiviral responses, both Plasmocin and nalidixic acid were potent antivirals in cultured PAMs, whereas an antiviral effect of erythromycin was not noted. The source of the variation we observed is not known, but could be due to differences between pigs or among vials of cells obtained from the same pig. Cytotoxicity studies demonstrated occasional reduction in total cell numbers and/or enhanced uptake of trypan blue at the highest Plasmocin concentration (250 ug/ml), but neither nalidixic acid nor Plasmocin at lower but effective concentrations appeared to be cytotoxic to PAMs by these methods (Table 4). Figure 1B illustrates the antiviral effect of 25 ug/ml Plasmocin in PAMs. Additional PAM responses to antibiotics are described below.
Drug responses of primary alveolar porcine macrophages.
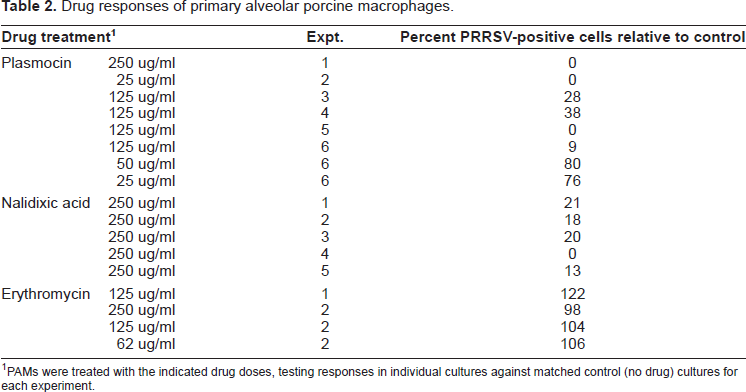
PAMs were treated with the indicated drug doses, testing responses in individual cultures against matched control (no drug) cultures for each experiment.
Correlation of Quinolone-Mediated Inhibition of PRRSV Replication with Reduced Actin Expression
Previous studies showed that PRRSV replication is dependent on an intact cytoskeletion and is inhibited by the actin-disrupting agent cytochalasin D (Cafruny et al. 2006). As illustrated in Figure 1D, actin expression was reduced in cultured MARC-145 cells by both Plasmocin and nalidixic acid, but little effect of erythromycin on actin expression was observed. Inhibition of actin expression by Plasmocin and nalidixic acid was dose-dependent (data not shown).
Potentiation of Cytokine-Mediated Inhibition of PRRSV Replication by Nalidixic Acid
AK-2 and IFN-γ are potent inhibitors of LDV and PRRSV replication, establishing an antiviral state in MARC-145 cells, as well as primary pig and mouse macrophages (Cafruny et al. 2006). For the present study, sub-optimal concentrations (below the concentrations with maximum antiviral activity (Cafruny et al. 2006, and Tables 1 and 2) of these cytokines as well as nalidixic acid (25 ug/ml) were used to treat cultured MARC-145 cells or primary alveolar pig macrophages (PAMs) just prior to or at the time of PRRSV inoculation. In most experiments, the drugs were used for 5 h pretreatment only, and after drug-removal, the cells were inoculated with PRRSV, fixed at about 14 h p.i, and stained for PRRSV antigen expression. Suboptimal concentrations of nalidixic acid, in some cases below the threshold of detectable antiviral effect by pretreatment, were found to synergize with low levels of AK-2 or IFN-γ, to inhibit PRRSV replication, as shown in Table 3. In experiment #1, the combined drug effect appeared to be additive, but in most of the experiments actual potentiation was observed since the nalidixic acid concentration was below an effective threshold. The drug effects were not due to detectable cytotoxicity (Table 4) or loss of total cells due to drug exposure. Figure 2A illustrates these viral responses to sub-optimal quinolone and cytokine concentrations, and Figure 2B illustrates partial (reduced intensity and scope of PRRSV antigen) antiviral responses which were observed in some cells due to combination drug treatment at sub-optimal concentrations.
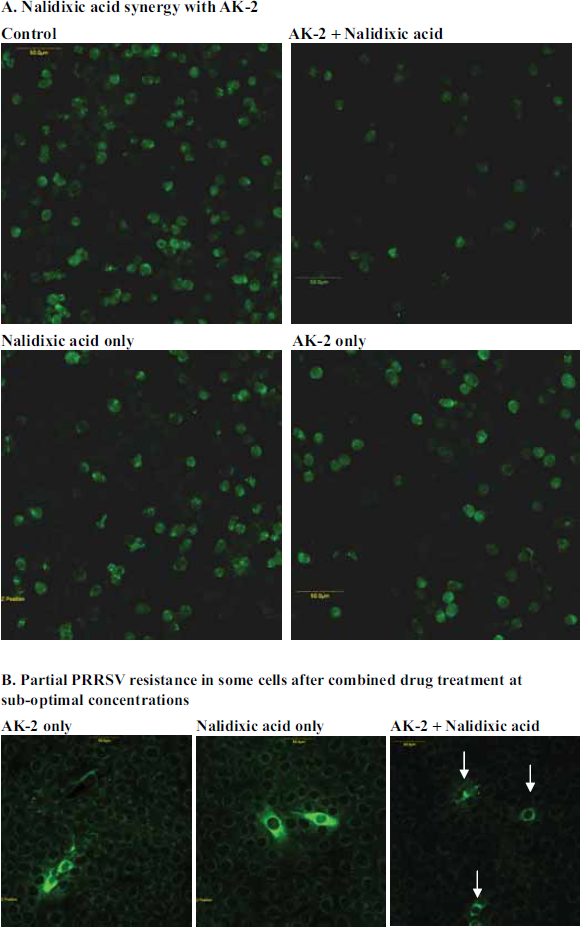
Potentiation of the antiviral cytokine response by nalidixic acid.
Potentiation of cytokine-mediated inhibition of PRRSV replication by nalidixic acid: drug synergy with sub-optimal drug concentrations.
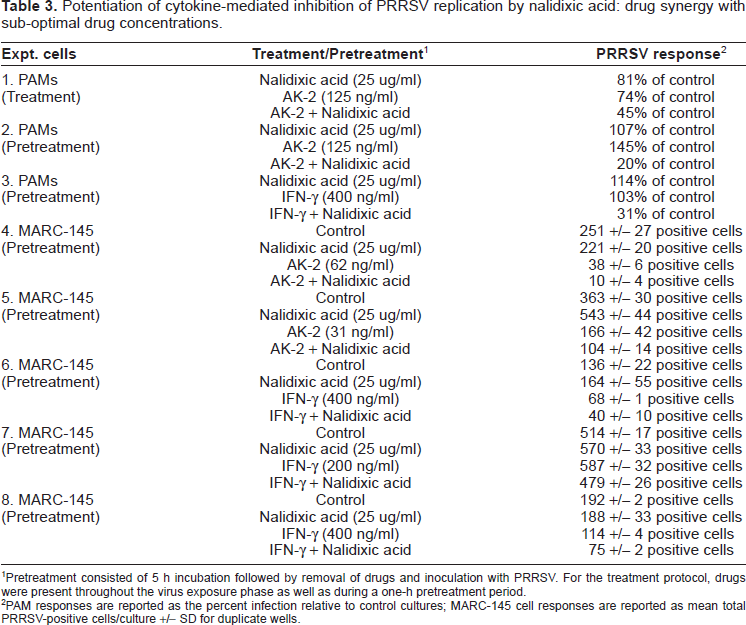
Pretreatment consisted of 5 h incubation followed by removal of drugs and inoculation with PRRSV. For the treatment protocol, drugs were present throughout the virus exposure phase as well as during a one-h pretreatment period.
PAM responses are reported as the percent infection relative to control cultures; MARC-145 cell responses are reported as mean total PRRSV-positive cells/culture +/- SD for duplicate wells.
Cytotoxicity testing by trypan blue dye exclusion.
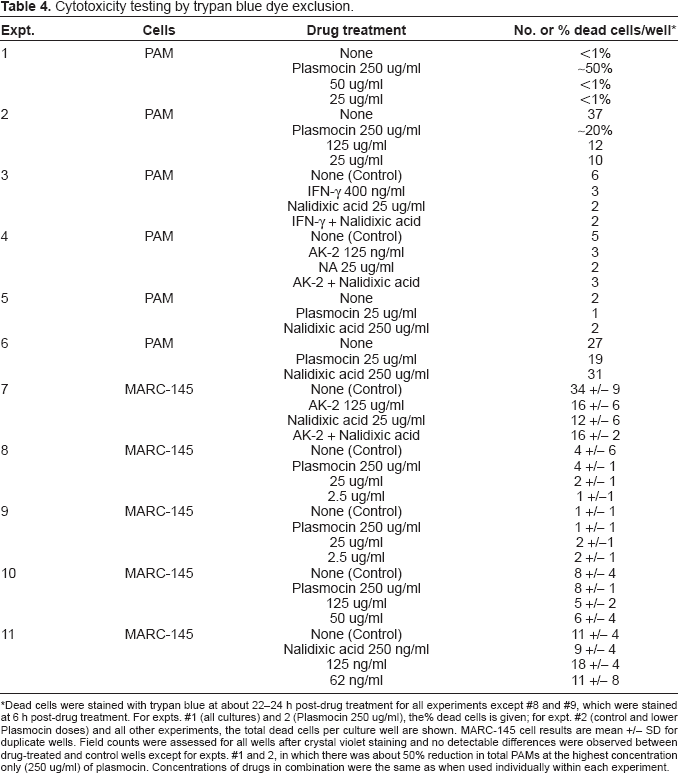
Dead cells were stained with trypan blue at about 22–24 h post-drug treatment for all experiments except #8 and #9, which were stained at 6 h post-drug treatment. For expts. #1 (all cultures) and 2 (Plasmocin 250 ug/ml), the% dead cells is given; for expt. #2 (control and lower Plasmocin doses) and all other experiments, the total dead cells per culture well are shown. MARC-145 cell results are mean +/- SD for duplicate wells. Field counts were assessed for all wells after crystal violet staining and no detectable differences were observed between drug-treated and control wells except for expts. #1 and 2, in which there was about 50% reduction in total PAMs at the highest concentration only (250 ug/ml) of plasmocin. Concentrations of drugs in combination were the same as when used individually within each experiment.
Inhibition of Lactate Dehydrogenase-Elevation Virus (LDV) Replication by Plasmocin
Primary peritoneal macrophages were LDV infected with or without Plasmocin in the culture medium. At 8 h p.i. the cells were fixed and stained for LDV antigen. Plasmocin at 250 ug/ml completely inhibited LDV replication (no positive cells per well), and at 25 ug partially inhibited LDV replication (179 +/- 22 LDV-positive cells per well, versus 272 +/- 16 positive cells per well in the untreated control, mean +/- SD given for duplicate wells.
Discussion
These results demonstrate the ability of several quinolone-containing antibiotics to inhibit arterivirus replication. Antibiotic-mediated suppression of both PRRSV and LDV was observed, the former in both primary pig cells and the MARC-145 cell line, and the latter in primary mouse macrophages. Thus, the antiviral effects of certain quinolone antibiotics are expressed in at least three different cell types. The PRRSV responses were shown to be dose- and time-dependent, and selectivity of the quinolone effects was also observed.
Nalidixic acid is the prototype quinolone (Crump et al. 2003) and was investigated further, in combination with the antiviral cytokines AK-2 and IFN-γ, employing pretreatment with sub-optimal drug doses to search for antiviral synergy. In both MARC-145 cells and primary pig macrophages, nalidixic acid was found to potentiate the antiviral effects of AK-2 and IFN-γ, even when single-drug exposures under these conditions failed to inhibit viral permissiveness. The mechanism is not clear, but potentiation under conditions in which the nalidixic acid concentration used failed to inhibit virus suggest the possibility of drug synergy, in which the combined activity is greater than the sum of individual activities (Wingard et al. 1991). In addition, exposure to the drug combinations induced partial resistance to virus replication in some cells.
Quinolones inhibit bacterial growth by interfering with the replication fork (acting on DNA gyrases and topoisomerases). The mechanism by which quinolones mediate antiviral effects in mammalian cells is not known from the present work, but does not appear to be related to direct cellular cytotoxicity, since dye-exclusion and cell count studies failed to support a detectable cytotoxic effect at the drug concentrations used. Furthermore, post-treatment by 3–6 h relative to virus inoculation was less effective than simultaneous treatment, indicating that an early stage in virus infection was the target rather than a general cytotoxicity in the cultured cells.
AK-2 and IFN-γ exert their antiviral effects by activating an antiviral gene program, consistent with the antiviral efficacy of these drugs during pretreatment followed by drug wash-out in our studies. Synergy (defined by combined activity greater than the sum of independent activities; Wingard et al. 1991) of nalidixic acid in combination with AK-2 or IFN-γ during our pretreatment strategies, suggests that this quinolone may also target viral permissiveness. Additional studies would be of interest to discriminate between the possibilities of a metabolic toxicity with antiviral side-effects and a more conventional activation of viral resistance by combination drug treatment.
The arteriviruses PRRSV and LDV are both dependent on an intact cytoskeleton for replication, and actin expression appears to be required for PRRSV replication, since inhibition of actin by cytochalasin D impairs the ability of PRRSV to infect cultured target cells (Cafruny et al. 2006). The present study demonstrated a correlation between actin and virus inhibition by the quinolone-containing antibiotics, although exposure to penicillin, streptomycin, or erythromycin failed to inhibit virus or actin expression. The cytoskeleton is a potentially important target in the identification of new antiviral molecules (Dohner and Sodeik, 2005); while it is intriguing to speculate that actin may be involved in the antiviral mechanisms of these quinolones, further studies are needed to address this possibility.
Previous studies have shown that the aminogly-coside hygromycin B inhibits mouse hepatitis virus (Macintyre et al. 1991), the macrolide bafilomycin inhibits African swine fever virus in porcine macrophages due to inhibition of the endosomal/lysosomal pump (Natale and McCullough, 1998), and rifampin inhibits poxviruses via effects on the viral envelope and protein assembly (Subak-Sharpe et al. 1969; Zhang and Moss, 1992; Sodeik et al. 1994). Coumermycin, like the quinolones an inhibitor of bacterial DNA gyrase, also inhibits retrovirus replication (Varnier et al. 1984). Thus, the present results add to the evidence that antibiotics may have antiviral activity, and furthermore demonstrate the ability of an antibiotic to potentiate antiviral cytokine effects, providing a new avenue for the study of antiviral mechanisms. These results also have implications for antiviral treatment or prevention strategies, utilizing combination therapy with the benefit of lowered toxicity, and particularly in vivo where the internal cytokine environment may help trigger antiviral responses to sub-optimal quinolone levels. While additional studies would be needed to determine clinical relevance, lipid-soluble antibiotics become concentrated in alveolar macrophages (Hand et al. 1984; Hand and King-Thompson, 1989), and ciprofloxacin levels of about 35 ug/ml have been reported in alveolar macrophages after oral dosing of humans (Gotfried et al. 2001), suggesting the potential for in vivo activity of quinolone derivatives against alveolar macrophage-tropic viruses such as PRRSV.
Footnotes
Acknowledgements
This work was supported by grants from the National Pork Board and the Center for Infectious Disease Research and Vaccinology. We thank Dr. Fran Day for her expertise in confocal microscopy.
