Abstract
The purpose of this pilot study is to measure the exposure to SHS among Richmond bar and restaurant workers and identify the prevalence of the tobacco-specific lung carcinogen-4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNAL) among nonsmoking bar and restaurants workers. Hair and urine samples were obtained from 40 nonsmoking bar and restaurant workers from Richmond establishments that do not have smoke free policies. Workers' exposure to SHS was estimated through measuring nicotine concentration in hair (NG/MG). Self-reported tobacco use was assessed in addition to performing urine analysis and listing sources and intensity of SHS exposure. Urine specimens were analyzed for total NNAL.
Results
Hair nicotine ranged from 0.05 to 42.15 ng/mg among workers with an average of 23.3 hours of self reported exposure in workplaces where smoking was permitted, indicating that workers in smoking restaurants and bars are exposed to SHS. Also, 60% of workers had a detectable level of NNAL that ranged from 0.019 to 1.9 PMol/ML. Increased levels of NNAL were mostly associated with the number of continuous hours of a single workplace exposure.
In the city of Richmond, most bar and restaurant workers are continuously exposed to SHS in their workplace. To achieve complete protection for all workers and patrons in Richmond, Virginia, smoke free initiatives in all occupational settings are required. This information can be used to advocate for smoke free policies when discussing potential ways to strengthen the law.
Introduction
Tobacco is one of the major causes of cancer related mortality worldwide. 1 Globally, lung cancer, in addition to trachea and bronchus cancers, are the most common causes of death from cancer among men. This is also the case in five of the seven World Health Organization (WHO) regions. In recent years, 1.3 million people have died from lung cancer worldwide.2,3 WHO estimated that tobacco use accounted for 40%–45% of all cancer-related deaths and 90%–95% of lung cancer deaths in developed countries. 4 Secondhand tobacco smoke (SHS) is considered as a form of tobacco use. SHS causes about 600,000 premature deaths per year worldwide. 5 About 11% of the annual tobacco-related deaths in the US (n = 50,000) are attributable to exposure to SHS. 6 Many studies have shown that bar and restaurant workers, who work at places that permit indoor smoking, experience 2 to 6 times more exposure to SHS than other workers in smoke free bars and restaurants.6–9 Furthermore, studies have indicated that SHS exposure at the work place was related to lung cancer or deaths in many regions of the world. For example, SHS causes about 7,600 deaths per year in the European Union.7–12
SHS is the smoke produced from the burning end of a cigarette or from other non-smokeless tobacco products. It is usually in combination with the smoke exhaled by the smoker and has similar components to inhaled smoke. 13 However, studies have indicated that SHS is three to four times more toxic per gram of particulate matter than active tobacco smoke and the toxicity of SHS is higher than the sum of the toxicities of its constituents. 14 The evidence is overwhelming; the National Toxicology Program has listed environmental tobacco smoke (ETS) exposure as a workplace carcinogen in its Tenth Annual Report on Carcinogens.6,9,15 Furthermore, in a number of the U.S cities that do not have restrictive smoke free-laws, it was reported that full time bar and restaurant workers are exposed to SHS at work more than four times the average annual outdoor limits of fine particular air pollution.16,17
In 2006, the U.S. Surgeon General's office advocated for a law that would eventually ban smoking in bars and restaurants. 18 The Office's report clearly stated that only a smoke free environment would protect all workers and the public from SHS. Thus, there is a significant danger from SHS, especially in the work place. Studies have shown that cigarette smoke is two to five times higher in bars and restaurants than in a residential home where smokers reside.19–23 The enactment of comprehensive smoke-free workplace laws in some U.S. states has provided an opportunity to examine the effect of these laws on tobacco toxicant exposure among workers in theses venues. The results of these studies provide the evidence needed to support the positive health benefits that arise from comprehensive bans. The Commonwealth of Virginia is one such state that has enacted a smoking ban law.21,22 However, no published studies have examined the effect of SHS on workers of bars and restaurants to date. Assessing Virginia's restaurant and bar workers' SHS exposure level and its related health effects is vital at this stage. The assessment is anticipated to have two benefits: first, it will help strengthen the case for improving the ban, and, second, it will serve as baseline data for a follow up cohort study in order to measure any improvements in health effects from applying the smoking ban.
Virginia has approximately 10,221 restaurants and bars. Employees in these establishments are a particularly vulnerable sector of the workforce. These workers experience higher SHS exposure in the workplace relative to other workers, and a high proportion of them are still in their twenties. Therefore, policies that can promote a smoking ban within their working environment could have significant benefits far into the future. The states that adopted smoke free legislation have reduced the SHS levels among hospitality workers.22,23 Recently, 77% of Virginians (~2 million) reported that their workplace has a comprehensive ban on smoking in all indoor work areas. The remaining 647,000 adult employees are not fully protected from SHS at work. This includes nearly 475,000 non-smokers (~21% of the population). 23
As mentioned earlier, there has been no published data that has either reported nicotine exposure among unprotected exposed workers in Virginia or quantified the uptake of certain lung carcinogens. This study measures the amount of SHS exposure among nonsmoking restaurant and bar workers as measured by hair nicotine level and self reporting of exposure. The prevalence of 4-(methylnitrosamino)-1-(3-pyridyl)-1- butanone (NNK) was also reported.
Materials and Methods
The study was approved by the Virginia Commonwealth University institutional review board. Eligible participants were nonsmokers who worked in bars and restaurants in Richmond, Virginia where smoking was allowed. Participants were recruited through local newspaper advertisements and advertisements in restaurants and bars where employers reported work exposure to SHS. All participants gave informed consent prior to answering the study questionnaires.
Participants were hospitality industry workers who reported abstaining from tobacco or nicotine products and worked in a hospitality location where they had been exposed to SHS for shifts of 6 or more hours. Participants were provided with a self reporting SHS exposure questionnaire that contained questions about typical work hours and the extent of tobacco smoke exposure at work and at home. The questionnaire also included questions about their places of employment and their employers' policies on smoking. Self reported SHS exposure was assessed by asking “thinking about the last 7 days altogether, what was the intensity of exposure?” The response was assessed on a three point scale ranging from light to moderate to heavy exposure. Assessment of home, car, and other out of work sources of SHS exposure was assessed by asking about frequency of exposure on a four point scale ranging from never exposed to highly exposed. Data were collected from October 2008 through January 2009. Data collection ended prior to February 2009 when the statewide smoke free-law took effect. Participants were compensated $20 for their time and participation.
Smoking status was measured by asking “did you smoke any amount of cigarettes, even just one, in the last 30 days”. Several studies have compared the self-reported SHS exposure with nicotine levels in hair and found significant associations such as high sensitivity, steady exposure-dose ratio, and good reproducibility over time.24–26 Hair nicotine measurement was found to have a very good sensitivity and excellent specificity as a biomarker for cumulative tobacco smoke exposure. Hair samples of approximately 0–50 mg and/or approximately 10–15 strands of hair of at least 1 cm in length were cut from the back of the participants' scalps with scissors. After cutting the sample, it was kept in a paper envelope in one group and oriented in such a way that the cut end was distinguished from the loose end; after the samples had been collected, they were then shipped overnight to a lab in New Zealand for analysis. Hair nicotine levels were analyzed by dissolving the hair and then examining its contents using reversed-phase high performance 3 liquid chromatography with electrochemical detection (HPLC-ECD).26,27 The lower detection limit for HPLPC_ECD is 0.05 ng nicotine/mg of hair.
Participants were also requested to provide a urine sample to be collected after their shift ended. The urine samples were frozen and shipped overnight to the University of Minnesota in thermal coolers containing ice cubes. The urine specimens were tested for cotinine (a nicotine metabolite) to confirm the non smoking status of the participants. This test was used to validate the self report of nonsmoking status by the participants. The Nicalert® test strips; Nyamox, Maywood, New Jersy, USA were used for measuring cotinine level.
Analysis was done by the University of Minnesota Transdisciplinary Tobacco Use Research Center Bio-markers Core laboratory. Urine samples were analyzed for urinary metabolites of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) and its glucuronides (NNAL-Glucs), or total NNAL.28–32 Summary statistics included the number and percentage of participants for categorical variables, the mean, standard deviation, and range. Total NNAL levels below the detection limit of 0.0050 were given a value of 0.0025 PMol per milligram of creatinine, a value that is half the detection limit. The Pearson's correlation coefficients were calculated for the log NNAL and hair nicotine versus length of work and working hours. between selected SHS exposure variables and for the total NNAL level and hair nicotine level.
Results and Discussion
Self-report of SHS exposure outside the workplace (N = 40).

Self-report of intensity of SHS exposure at the workplace (N = 40).

Hair nicotine levels and total NNAL levels in nonsmoking hospitality workers (N = 41).

LOD < 0.015 PMol/ML.
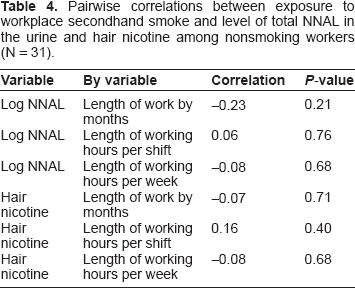
Furthermore, Table 3 shows the mean and standard deviations (SD) for both the NNAL and hair nicotine for the final 31 subjects. 60% (18 of 32) of the workers had detectable total NNAL in the urine. Total working hours per shift or week were not significantly related to NANAL detachability. The mean (SD) value of the Hair nicotine was 2.09 (5.6) ng/Mg, while the mean (SD) value for the NNAL total was 0.20 (0.42) PMol/ML.
Two sample t-tests were used to determine any difference in hair nicotine levels by duration of exposure. We found that the duration of exposure to SHS as measured in weekly total hours was not significantly associated with the total hair nicotine.
Pairwise correlations between exposure to workplace secondhand smoke and level of total NNAL in the urine and hair nicotine among nonsmoking workers (N = 31).
Discussion
The main purpose of this study was to identify the amount of exposure to SHS as measured by hair nicotine level among nonsmoking restaurant and bar workers, the self reporting of that exposure, and the prevalence of NNAL. This study determined both, long term hair nicotine accumulation and carcinogen uptake by workers exposed to SHS. The presence of NNAL in the urine of these workers can be explained only by their exposure to the tobacco-specific carcinogen NNK. This exposure was most likely through SHS in the workplace. Although uptake of NNK from surfaces, such as furniture or carpet and rugs, cannot be excluded, the NNK originated from tobacco products, which is its only known exogenous source. Our findings of increased metabolites of NNK among exposed nonsmoking bar and restaurant workers show health risks and possibly adverse outcomes among nonsmokers exposed to SHS in the workplace. It is worth mentioning that most of the previous studies have been conducted mainly by a two research groups in the states of Minnesota and California.28–36 The current study was one of the few studies conducted outside the above mentioned states and the only such study in the Commonwealth of Virginia.
The major finding of this study is that hospitality workers in bars and restaurants exposed to SHS have both high detectable levels of nicotine and NNAL. This finding is consistent with the previous SHS studies.31,33,34 However, there are some results in this study that are not consistent with previous studies. These are the assessment of SHS exposure in bar and restaurant workers and carcinogen uptake in terms of timing of urine samples collection (ie, NNAL).35,36 One of the previous studies measured the total NNAL level by collecting participants' total urine over a 24 hour time period; 34 however, in our study a onetime urine sample (1 ounce) was collected and analyzed. This sample was collected and obtained after the working shift ended. While a number of studies,29–31 found strong associations between exposure to SHS for a single shift in the workplace and total NNAL levels, our results have only showed a significant association between workplace length of exposure for the total weekly hours and NNAL levels. This result could be explained by the fact that we measured hours of exposure by the self report only. However, the current study did not compare smoking and nonsmoking restaurants and bars as previous studies had done. For example, Benowitz et al 37 reported that NNAL level in urine was higher for passive smokers compared with active smokers. Although inconsistent with some results of previous studies, the current study presents significant information as it examines the relationship between SHS exposure at the workplace, nicotine hair levels, and total NNAL levels. It is also one of the few studies that examine the relationship of length of exposure for more than one shift.
It is worth noting that 11 participants in the current study did not have detectable levels of total NNAL. This can be attributed to low level of SHS exposure on the day of data collection, which could have been due to slow business hours, bad weather, and working in the non-smoking section or outdoors.
Prior studies using hair nicotine as an outcome measure showed significant levels of association between hair nicotine and the magnitude of SHS exposure.27,38–40 The current study did not show such associations. Differences in study designs make it difficult to compare our results with previous studies. Still, one can conclude that, with regards to hair nicotine levels, our study was not consistent with the others. 41
Another explanation of the lack of significant association between NNAL and hair Nicotine in our study could be that hair nicotine is a long term biomarker, while NNAL is a short term biomarker. Hair nicotine could reflect 1 month of cumulative tobacco-smoke exposure. NNAL, on the other hand, could reflect up to 12 hours of exposure. Therefore, it could be assumed that some of the participants who had high hair nicotine did not have a full working shift or had light SHS exposure when they gave the urine sample for analysis. Such an issue could be avoided in future studies by measuring acute biomarkers of SHS exposure, such as urine cotinine, and long-term SHS exposure by hair nicotine levels.
A limitation of this study is that the study did not use a random sampling technique. Therefore, it is possible that the participants were supportive of comprehensive smoke-free workplace policies. Thus they may have altered their self response which might have influenced the study outcome. Additionally, another limitation is related to a lack of sensitivity in the NNAL analysis method; this was also reported recently by the Minnesota cancer center laboratory. 33 Also, the small number of participants must be viewed as an overall limitation to our study. Finally, there was no alternate objective measure of SHS exposure, such as personal passive air nicotine monitoring. This could have been used to provide a comparison between hair nicotine and self-reported exposure. On the other hand, if we were to repeat the study, we would collect the urine for 24 hours so that we could have an accurate representation of the total NNAL.
The results of this study will provide a baseline data for future studies to evaluate cohort groups of bar and restaurant workers in the Commonwealth of Virginia, since the implementation of the new law has been in effect since March 2009. 42 In addition, this preliminary study leads to the recommendation of future rigorous studies that control covariates, including air quality in the few smoking sections that are still allowed in 10% of Virginia's restaurants. Such additional information would prove to be a helpful advocacy tool for smoke free advocates to lobby for more comprehensive smoke free laws and enforcement in Virginia. Comprehensive smoke free laws safeguard nonsmokers from the negative consequences of SHS exposure, are associated with decreasing smoking incidence rates among adults and youth,43–45 and are associated as well with decreased levels of carcinogenic uptake by bar and restaurants workers. Weak laws, such as the new Virginia law that limits smoking to separately enclosed areas, do not protect workers from the risk of being exposed to SHS. 46 Our study findings support the need to make all Virginia public places totally smoke-free to protect employees from the harmful effects of SHS.
Footnotes
Acknowledgements
The study was funded by award # p20 NR008988 (N. McCain, PI), National Institute of Nursing Research, NIH; Pilot Study PI, 2008–2009. The author's are also grateful to the University of Minnesota's Transdisciplinary Tobacco Use Research Center for Biomarkers Core and Laboratories for Chemical Analysis of NNal. We thank Dr. Ellen Hahan, University of Kentucky, College of Nursing, for training the PI on hair sample collection protocol. We also thank Dr. Todd Starkweather for editing our final manuscript.
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
