Abstract
The effects of sanitation treatments including chlorination (ca 10 ppm available chlorine) of agricultural water and ethyl alcohol (70%) spraying on packing shed equipment on microbial contamination on fruits and the environment were determined and compared with those in conventionally managed field and packing shed in persimmon and satsuma mandarin orchards. Chlorinated water reduced the microbial counts to levels below the lower limit of detection (1.4 log CFU/ml for bacteria and 2.0 log CFU/ml for fungi) in most agricultural water samples. Microbial counts of pesticide solution, which contained the agricultural water or chlorinated water for the mixture, were lower in sanitary field than in control field in both fruit orchards. The number of bacterial and mold species detected in agricultural water, chlorinated water, and pesticide solution were almost proportional to microbial counts in each sample throughout the year. The chlorination treatment of agricultural water tended to reduce the counts of mesophiles and fungi on the peel of persimmon fruit during production season. The ethyl alcohol spray treatment on packing shed equipment resulted in a substantial microbial reduction on plastic harvest basket and container in persimmon orchard and plastic harvest basket and container, gloves, scissors, and size sorter in satsuma mandarin orchard. The spray application on packing shed equipment reduced the counts of mesophiles and fungi on the peel of persimmon fruit by >1 log CFU/g. The number of satsuma mandarin packing shed equipment containing the species found on fruit peel was higher in control than in sanitary packing shed. No human pathogens such as verotoxin-producing Escherichia coli and Salmonella were detected in any of the fruit and environmental samples. These results indicate that uses of sanitizers such as chlorine for agricultural water and ethyl alcohol for packing shed equipment would be useful in a good agricultural practices program of persimmons and satsuma mandarin.
Keywords
Introduction
Fresh fruits and vegetables can become contaminated with pathogenic microorganisms and have been identified as the vehicle for Escherichia coli O157:H7 and Salmonella infection (Beuchat, 1996; NACMCF, 1999; De Wall et al. 2007), which are the organisms most likely to cause an outbreak that needs to be studied for produce safety (Aruscavage et al. 2006; Mukherjee et al. 2006). Surveys were performed in several countries to determine prevalence of the two enteric pathogens. The surveys of a total of 7,686 product samples in the United States (Riordan et al. 2001; Thunberg et al. 2002; Mukherjee et al. 2004; Johnston et al. 2005; Mukherjee et al. 2006), the European Union (McMahon and Wilson, 2001; Sagoo et al. 2001; Johannessen et al. 2002) or Japan (Konishi et al. 2001; Murase et al. 2002) detected no E. coli O157:H7 but Salmonella in 3 samples of cantaloupes in the U.S. (Johnston et al. 2005), 2 samples of lettuce and green pepper in the EU (McMahon and Wilson, 2001), and 1 sample of alfalfa sprouts in Japan (Konishi et al. 2001). Although prevalence of the pathogens on the products was very low, the cross contamination may have occurred from irrigation water, soil, manure, transport equipment, and farm worker where the pathogens present (Beuchat, 1996; Beuchat and Ryu 1997; NACMCF 1999; Knabel et al. 2003). Our previous report on persimmon orchard in Japan indicated that E. coli O157:H7 was identified from agricultural water in May and Salmonella was detected in agricultural water, pesticide solution containing the agricultural water for the mixture, and soil after application of the pesticide solution in June (Izumi et al. 2008b). We also found that pesticide solution collected from satsuma mandarin orchard in July was positive for Salmonella (Izumi et al. 2008a). Since persimmon and satsuma mandarin fruits were not fully developed when E. coli O157:H7 or Salmonella was detected in the environment, neither of these pathogenic bacteria was detected in any of the fruit samples. However, treatment of agricultural water, which was one of the most important potential preharvest contamination sources, might be required for products consumed raw to reduce the levels of microorganisms and the risks for human illness according to safety guidelines.
Several researchers have suggested that postharvest strategies to minimize microbial contamination were important as well as preharvest strategies with cantaloupe (Gagliardi et al. 2003; Duffy et al. 2005; Johnston et al. 2005), oranges (Duffy et al. 2005), and grapefruit (Parish and Higgins, 1990). We found that some packing shed equipment such as gloves, plastic harvest basket and container, and size sorter were assumed as postharvest contamination sources for persimmon (Izumi et al. 2008b) and satsuma mandarin (Izumi et al. 2008a), although some of the preharvest sources could also be postharvest sources. Thus, sanitizing treatment of equipment may be needed to reduce populations of pathogenic and other microorganisms on the equipment and fresh produce by following on-farm food safety program.
In this study, we proposed sanitation measures for reducing the risk of microbial contamination on persimmon and satsuma mandarin fruits in the field and packing shed. The measures focused on control of microbial contamination by chlorination of agricultural water in the production field and alcohol spraying of equipment in the packing shed. Chlorine is routinely used as a sanitizer for agricultural and processing waters (Beuchat and Ryu, 1997; Steele and Odumeru, 2004; Aruscavage et al. 2006). A solution of 50 to 200 ppm available chlorine has been recommended for postharvest washing treatment of fresh produce (Anonymous, 1998; Aruscavage et al. 2006). In this study, the agricultural water used for pesticide solution was chlorinated to 10 ppm available chlorine. A higher chlorine level was avoided, because when using chlorine, there is a potential danger of chemical reaction of chlorine gas in the pesticide solution, which is hazardous to health and safety of workers. In plain aqueous systems, only 0.5 to 10 ppm available chlorine has been shown to be effective for rapid inactivation of Listeria monocytogenes (El-Kest and Marth, 1998a; El-Kest and Marth, 1998b), although the effectiveness of chlorine is reduced in water with high organic matter (Boyette et al. 1993). While chlorine is the disinfecting agent in wash, spray, and flume waters used in the fresh fruit and vegetable industry, ethyl alcohol is effective disinfectant in hospitals and other Health Care Services to prevent nosocomial infection from environmental surfaces (Rutala, 1996; Vieira et al. 2005). Therefore, a 70% ethyl alcohol spray was evaluated on disinfection of packing shed equipment such as gloves, scissors, plastic harvest basket and container, and size sorter during packing facility operations to minimize postharvest contamination. Our objective is to provide a scientific framework such as sanitation program to design a good agricultural practices (GAP), such as that developed for on-farm food safety of tree fruits, because the GAP program by Ministry of Agriculture, Forestry, and Fisheries of Japan (Anonymous, 2006) is still in its infancy for implementation as compared with that by the U.S. Food and Drug Administration/U.S. Department of Agriculture/Centers for Disease Control and Prevention (Anonymous, 1998).
Materials and Methods
Sanitation treatments for on-farm food safety
The sanitation treatments were implemented in a persimmon (Diospryos kaki Thunb. cv. Fuyu) and satsuma mandarin (Citrus unshiu Marcow. cv. Miyagawa Wase) production field (sanitary field) and packing shed (sanitary packing shed) in Wakayama prefecture during 2006 season. The same producer also managed conventional field (control field) and packing shed (control packing shed) of persimmon or satsuma mandarin in each district in Wakayama prefecture. The treatments included chlorination of agricultural water and ethyl alcohol spraying on packing shed equipment as critical control points to prevent contamination. Chlorination of agricultural water was made by adding Chemi-chlon G (Nippon-soda, Tokyo, Japan) containing calcium hypochlorite as granules in formulations of 90% to agricultural water. The granules were dissolved in agricultural water at a rate of one granule (ca. 0.1 g) to 10-liter water to attain about 10 ppm available chlorine. Concentration of available chlorine was confirmed by the sodium thiosulfate titration method (Asada et al. 1981) and the average throughout a season was 7.9 ppm and 8.4 ppm in persimmon and satsuma mandarin orchards, respectively. The Chemi-chlon G is approved as a pesticide in the Japanese Pesticide Control Law. Packing shed equipment such as gloves, scissors, plastic basket, plastic container, and size sorter were sprayed once with 70% (v/v) ethyl alcohol (Nakarai Pharmaceutical, Tokyo, Japan) at the beginning of the work day and following breaks. Ethyl alcohol has been reported to have the optimum antimicrobial activity in concentrations between 60% and 90% (Rutala, 1996). The 70% ethyl alcohol ranging from 2.5 ml for gloves and scissors to 40 ml for plastic basket and container was used for each spray application.
Persimmon orchard and satsuma mandarin orchard were not irrigated during the study period from May to November 2006. The agricultural water to dilute pesticide solution was from the Kino River, mountain spring, or rainwater in persimmon orchard and from mountain spring or rainwater in satsuma mandarin orchard, depending on the field and month. The pesticides applied from May to October in persimmon orchard included fungicide (Zimman dithane, Quinone-do, M-diphar, Anvil flowable, Topsin M, and Score WP), insecticide (Orhtene, Clef-Non, Albarin water solble granule, Mr. Joker, and Agrosrin), and acaricide (Supracide). In satsuma mandarin orchard, fungicide (Ziman dithane, Strobiluin, M-Diphar, and Topsin M), insecticide (Mospilan, Mineral oil, Kotetsu flowable, Orhtene, and Omi-88 15% EC), and acaricide (Supracide and Kanemite flowable) were applied from May to October; liquid fertilizer (Powerful green) was applied along with the pesticide solution in May to July. In persimmon and satsuma mandarin orchards, the pesticides and liquid fertilizer were applied at same concentration and same time in both sanitary and control fields.
Fruit and environmental samples
Nine fruit each of persimmon and satsuma mandarin in each field were picked monthly with a gloved hand from selected 3 trees from August (ca. 7 cm diam of persimmon and ca. 4 cm diam of satsuma mandarin) to November (harvest period of both fruits). Samples were also collected after harvest (persimmon) and sorting (satsuma mandarin) at packing shed in November. Environmental samples were obtained monthly from the field agricultural water (ca. 100 ml), chlorinated water (ca. 100 ml) in only sanitary field, and pesticide solution (ca. 100 ml) from May to October. Samples were collected in a sterile glass bottle within 3 days after pesticide application. Surface contact plates (ca. 30 cm2) of packing shed equipment including plastic basket and container at harvest (persimmon) and gloves, scissors, and size sorter at fruit sorting (satsuma mandarin) were utilized and collected using the replicate organism direct agar contact (RODAC) method (Jay, 1992) in November. The site of contact plate on equipment was the surfaces where the fruit came in contact (i.e., side and bottom of plastic basket and container, palm part of glove, blade of scissors, and part of inlet, drum, and outlet of size sorter). All samples were carried on ice to the laboratory at Kinki University within 2 h after sample collection.
Microbial counts and identification
Microbial analyses of the samples were replicated three times. Each sample was assessed for counts of mesophilic aerobic bacteria, coliform group, and fungi and identification of bacteria and molds as previously described (Izumi et al. 2008b). The peel and flesh were aseptically separated from the fruit with sterile scalpels. The portions of the flesh that came into contact with the scalpels were removed to avoid transfer of microorganisms from the surface peel to the flesh. A 10-g sample of fruit peel and flesh and 10-ml sample of agricultural water, chlorinated water, and pesticide solution were used for enumeration and isolation of microorganisms. The serial dilutions from each sample were made in sterile saline solution and then plated in duplicate standard method agar (SMA; Nissui Pharmaceutical, Tokyo, Japan) for enumeration and onto solidified SMA for isolation of mesophiles, in duplicate desoxycholate agar (DA; Nissui Pharmaceutical) for enumeration of coliforms, and in triplicate potato dextrose agar (PDA; Nissui Pharmaceutical) with 100 ppm chloramphenicol for enumeration and onto solidified PDA for isolation of fungi. Incubation conditions were 48–72 h at 37 °C for SMA, 24 h at 37 °C for DA, and 5–7 days at 26 °C for PDA. Triplicate Food Stamp “Nissui” SMA (Nissui Pharmaceutical) for mesophiles, Food Stamp “Nissui” DA (Nissui Pharmaceutical) for coliforms, and Food Stamp “Nissui” PDA (Nissui Pharmaceutical) for fungi were used as RODAC plates that made direct contact with the surface of plastic harvest basket and container, gloves, scissors, and size sorter. Each RODAC plate was incubated for the enumeration and isolation of microorganisms in the same manner as each agar plate.
Since yeasts within fungi were rarely found in persimmon and satsuma mandarin fruits, only molds were identified to genus and species. Thus, sixty-nine bacterial and 66 mold isolates and fifty-five bacterial and 42 mold isolates were selected from different appearing colonies on petri plates from persimmon fruit and satsuma mandarin fruit, respectively. The isolates were from peel and flesh of both fruits in control and sanitary fields during development (September), harvest (November), and sorting (November). A total of 469 bacterial and 155 mold isolates and a total of 533 bacterial and 168 mold isolates were also selected for the identification from 26 environmental samples (agricultural water, chlorinated water, pesticide solution, and plastic harvest basket and container) in persimmon orchard and from 34 environmental samples (agricultural water, chlorinated water, pesticide solution, gloves, scissors, plastic harvest basket and container, and size sorter) in satsuma mandarin orchard, respectively. The MicroSeq Microbial Identification and the MicroSeq D2 LSU rDNA Fungal Identification (Applied Biosystems, Foster City, CA, U.S.A.) were used for identification of bacteria and molds, respectively, as previously described (Poubol and Izumi, 2005). The sequencing data were analyzed using Analysis Software (MicroSeq Analysis Software v. 1.40 and MicroSeq 16S rDNA Sequence Databases v.1.01) and the nucleotide database at GenBank using BLAST to determine bacterial and fungal identities, respectively. A cutoff of the lowest distance score from the sequence in the database was chosen for species identity.
Detection of Salmonella and verotoxin-producing E. coli (VTEC)
Detection of foodborne pathogenic bacteria, Salmonella and VTEC, on all fruit and environmental samples were determined using the loop-mediated isothermal amplification (LAMP) method as previously described (Izumi et al. 2008b). The LAMP reactions were performed using Loopamp Salmonella Screening Kit (LMP601, Eiken Chemical, Tokyo, Japan) and Loopamp Verotoxin-producing E. coli Screening Kit (LMP621, Eiken Chemical) following the manufacturer's instructions. The LAMP reaction was carried out by incubation at 65 °C for 60 min using Loopamp Realtime Turbidimeter (LA-200, TERAMECS, Tokyo, Japan) and the gene amplification was monitored by measuring turbidity of white precipitates of magnesium pyrophosphate formation.
Data analysis
Three replicated microbiological plate count data were converted to log CFU/g of fruit peel and flesh; log CFU/ml of agricultural water, chlorinated water, and pesticide solution; and log CFU/100-cm2 surface area of gloves, scissors, plastic harvest basket and container, and size sorter. Statistically significant differences (P ≤ 0.05) between paired sanitary and control samples within each month of analysis were determined for microbial population detectable data in each organism based on analysis of variance using the SAS system release 6.12 (SAS Inst. Inc, Cary, NC, U.S.A.).
Results and Discussion
Effect of chlorination of agricultural water on microbial decontamination of fruit and the environment in the field
Microbial population of persimmon fruit and the environment during production managed in control and sanitary fields.
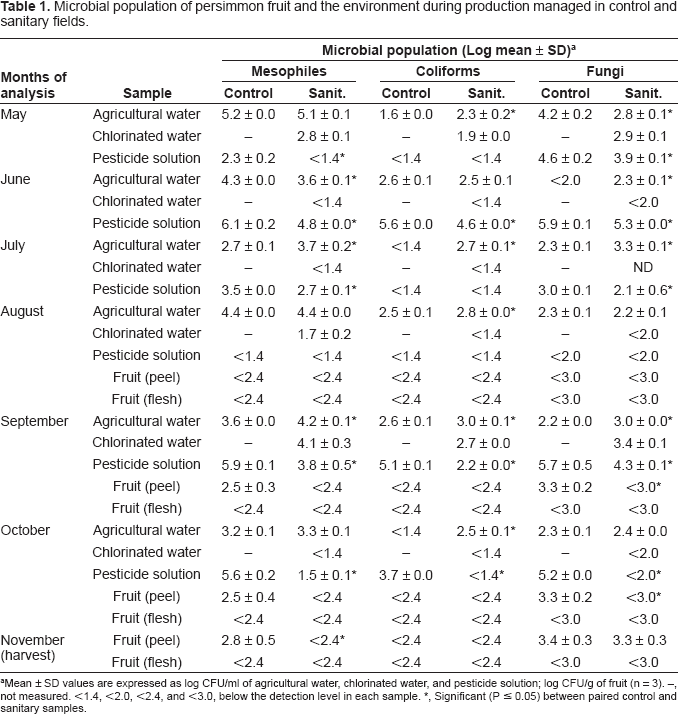
Mean ± SD values are expressed as log CFU/ml of agricultural water, chlorinated water, and pesticide solution; log CFU/g of fruit (n = 3). –, not measured. <1.4, <2.0, <2.4, and <3.0, below the detection level in each sample.
Significant (P ≤ 0.05) between paired control and sanitary samples.
Microbial counts of pesticide solution, which contained the agricultural water or chlorinated water for the mixture, were lower in sanitary field than in control field from May through October. Exception to this was August, when the microbial counts in pesticide solution were below the detectable level in both samples from control and sanitary fields. Guan et al. (2001) found that some pesticide products, when diluted with contaminated water, might have the potential to promote the growth of pathogens such as E. coli O157:H7, Salmonella, Shigella, and Listeria. Ng et al. (2005) reported that the agricultural water used for reconstitution of pesticides had initial bacterial populations from 10 3 to 10 6 CFU/ml, when the growth of bacteria in the pesticide solution was supported. Thus, chlorination of agricultural water in this study helped in reducing microbial counts in pesticide solution and also may reduce the risk of widespread contamination from agricultural water to pesticide solution and other environment.
Bacteria and molds isolated from agricultural water, chlorinated water, and pesticide solution managed in control and sanitary fields in persimmon orchard in July.
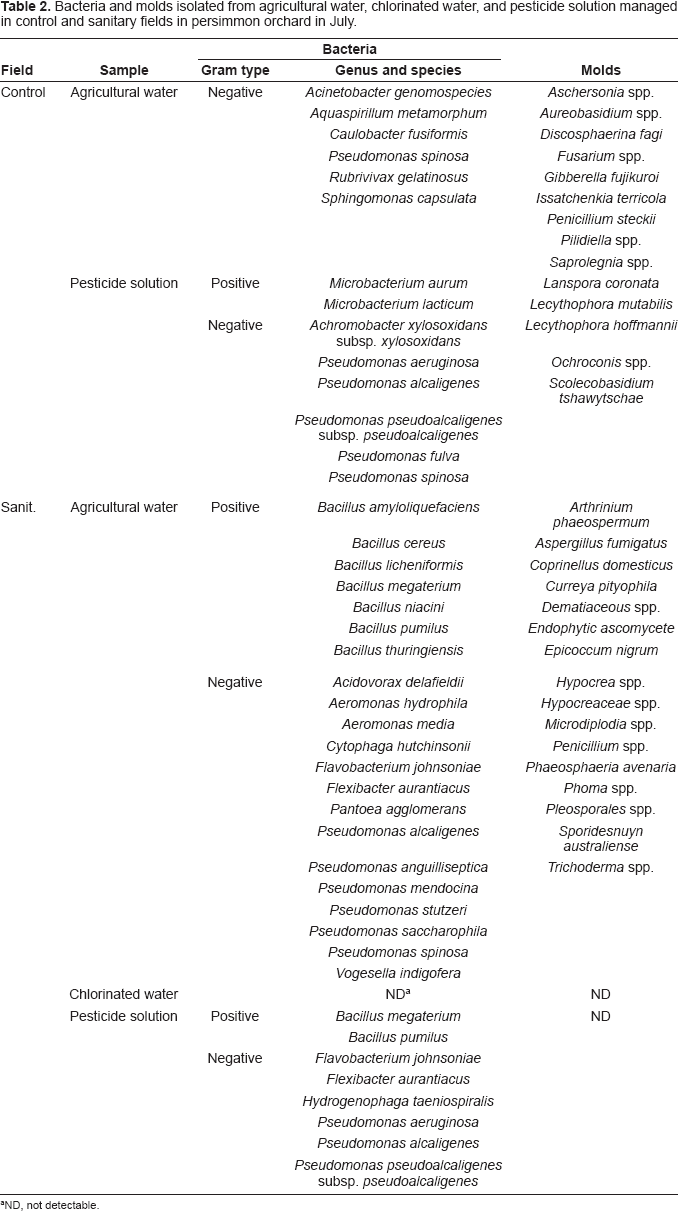
ND, not detectable.
With persimmon fruit, mesophiles and fungi were at the detectable levels in the peel of control fruit from September through November, while the counts in the peel of sanitary fruit were below 2.4 log CFU/g for bacteria and 3.0 log CFU/g for fungi, except for the fungi count in November (Table 1). Since chlorination of agricultural water reduced the microbial counts in the pesticide solution, it may have reflected the counts on the fruit peel due to the direct contact between the pesticide solution and fruit peel. Coliforms in the peel of all fruits and microbial counts in the flesh of all fruits were below the limit of detection level. The same species of bacteria such as Bacillus, Flavimonas, and Microbacterium and molds such as Cladosporium, Dematiaceous, and Penicillium were found frequently on persimmon fruit and in the agricultural water, chlorinated water, and pesticide solution (data not shown), suggesting that the water or solution is the possible source for fruit contamination, although the genetic diversity of the isolates has not been evaluated. This result was in confirmation of our previous repot on persimmons (Izumi et al. 2008b).
Microbial population of satusma mandarin fruit and the environment during production managed in control and sanitary fields.
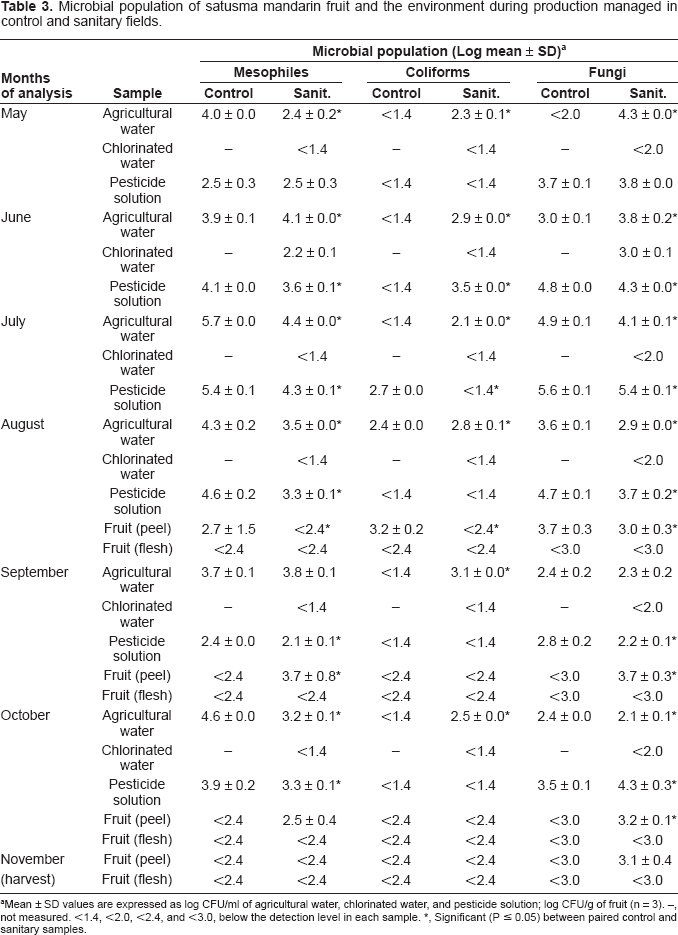
Mean ± SD values are expressed as log CFU/ml of agricultural water, chlorinated water, and pesticide solution; log CFU/g of fruit (n = 3). –, not measured. <1.4, <2.0, <2.4, and <3.0, below the detection level in each sample.
Significant (P ≤ 0.05) between paired control and sanitary samples.
Microbial population in the peel of satsuma mandarin fruit picked in control field in August was only detectable during fruit development and was higher than that in sanitary field in August (Table 3). However, the counts of mesophiles in September and fungi in September and October were higher in the fruit peel from sanitary field than that from control field. The remainder of peel samples and all flesh samples showed microbial level below the lower limit of detection and thus no differences were found between control and sanitary samples. The chlorination treatment of agricultural water was not necessarily effective in reducing the microbial counts on fruit, although Bacillus species and Cladosporium species found on fruit peel seemed to be transferred from the agricultural water and pesticide solution (data not shown).
Effect of ethyl alcohol spraying on microbial decontamination of fruit and the environment in the packing shed
Microbial population of persimmon fruit, satsuma mandarin fruit, and the environment managed in control and sanitary packing shed.
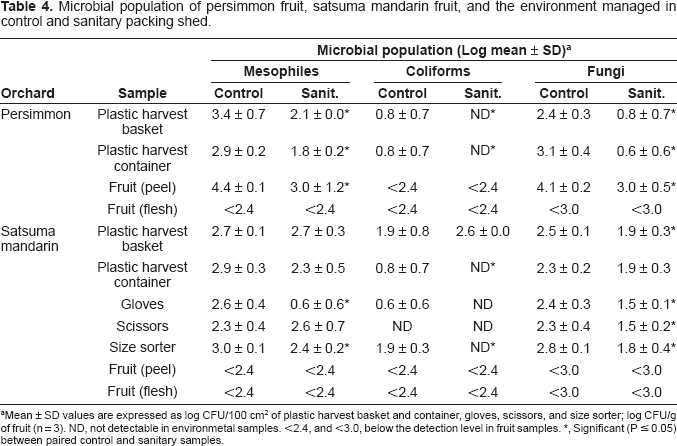
Mean ± SD values are expressed as log CFU/100 cm2 of plastic harvest basket and container, gloves, scissors, and size sorter; log CFU/g of fruit (n = 3). ND, not detectable in environmetal samples. <2.4, and <3.0, below the detection level in fruit samples.
Significant (P ≤0.05) between paired control and sanitary samples.
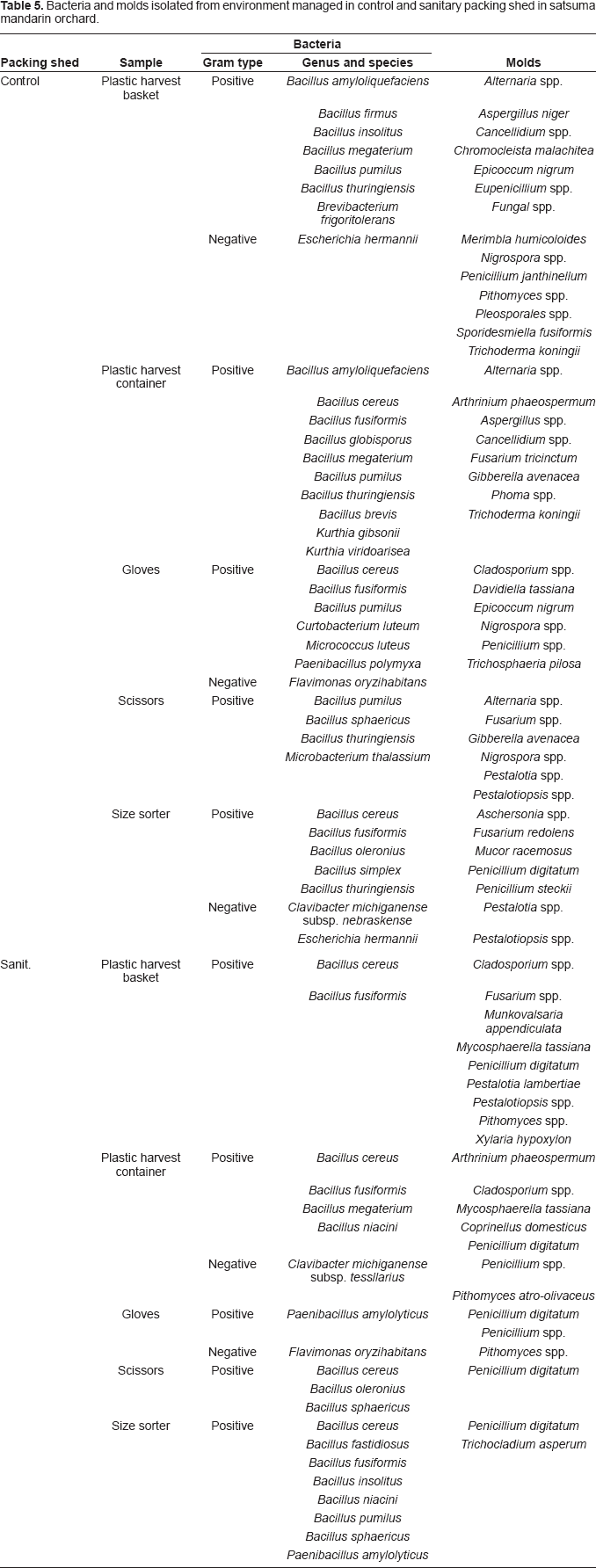
Bacteria and molds isolated from environment managed in control and sanitary packing shed in satsuma mandarin orchard.
Bacteria and molds isolated from satusma mandarin fruit after sorting managed in control and sanitary packing shed.
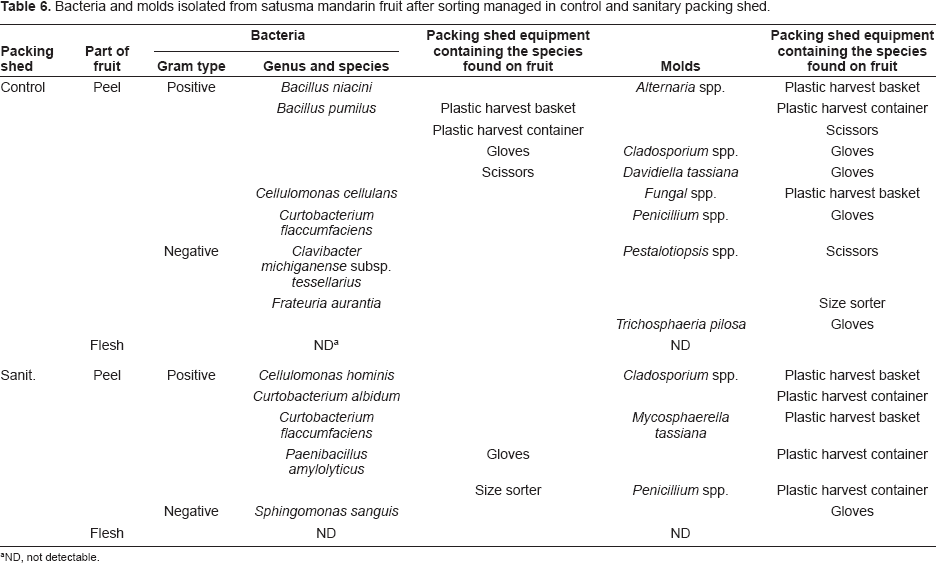
ND, not detectable.
The diversity of mold flora was less in the peel of sanitary fruit (4 isolates; 3 species belonging to 3 genera) than in those of control fruit (8 isolates; 7 species belonging to 7 genera). The mold genera such as Cladosporium and Penicillium detected in the peel of both control and sanitary fruit were frequently found in the peel of satsuma mandarin fruit as noted in our previous reports (Izumi et al. 2007; Izumi et al. 2008b). The mold species in the peel of control fruit and sanitary fruit were also detected in 10 and 6 packing shed equipment samples, respectively. This suggests that the equipment would be the source of contamination during and after harvest and the cross-contamination would occur more frequently in control packing shed than in sanitary packing shed. No bacteria and molds were detected in any fruit flesh. These results indicate that ethyl alcohol spray application in satsuma mandarin packing shed helped in reducing the microbial counts on the surface of packing shed equipment directly and also the microbial diversity on the peel of fruit after sorting indirectly.
The similar relationship in microbial diversity between control and sanitary satsuma mandarin fruit after sorting was obtained in persimmon fruit (data not shown). The diversity was less in the peel of sanitary fruit (10 bacterial isolates; 6 species belonging to 2 genera and 7 mold isolates; 7 species belonging to 7 genera) than in control fruit (16 bacterial isolates; 8 species belonging to 5 genera and 12 mold isolates; 11 species belonging to 11 genera). Considering that the number of packing shed equipment containing the species found on fruit was higher in control than sanitary packing shed, the cross-contamination would occur more frequently in control packing shed than in sanitary packing shed in persimmons as confirmed with satsuma mandarin in this study.
Microbiological safety in fruit and environment
In our previous studies in 2005, E. coli O157:H7 and Salmonella were detected in agricultural water in May and pesticide solution in June in persimmon orchard (Izumi et al. 2008b) and only pesticide solution in July was positive for Salmonella in satsuma mandarin orchard (Izumi et al. 2008a). However, in this study conducted in the same orchards, no human pathogens such as VTEC and Salmonella were detected in any of the fruit and environmental samples based on LAMP assay. Therefore, it cannot be determined from this study whether the chlorine and ethyl alcohol treatments could remove the human pathogen in the environment, although the sanitizers were effective in reducing the spoilage microorganisms in agricultural water and pesticide solution and on packing shed equipment.
B. cereus can cause two distinct forms of food poisoning syndrome (Gilbert and Kramer, 1984). Since B. cereus is ubiquitous in soil environment (Watterson, 1985), the occasional presence on fruits and vegetables can be expected. However, most cases of B. cereus food poisoning in Japan are of the emetic type due mostly to the ingestion of rice dishes and derived from rice paddy field soil (Ueda and Kuwabara, 1993). The incidence of the emetic strain was very rare in B. cereus isolated from soil other than paddy field, animal feces, and vegetable produce (Altayar and Sutherland, 2006). Thus, we assumed that B. cereus strains detected in the satsuma mandarin orchard in previous (Izumi et al. 2008a; Izumi et al. 2008b) and present studies would be the indicator bacteria for cross-contamination with soil rather than the bacteria that have human pathogenic capabilities. Since B. cereus, spore-former, was often identified from not only control equipment but also sanitary equipment (Table 5), application of physical sterilization such as gamma irradiation (Chervin and Boisseau, 1994; Prakash et al. 2000) and hot water treatment (Izumi et al. 2001) may be required to eliminate the spore-forming bacillus.
Conclusion
Based on microbiological results of this study with comparison of sanitary and control fields and packing sheds, uses of sanitizers such as chlorine for agricultural water and ethyl alcohol for packing shed equipment would be useful in GAP program of persimmons and satsuma mandarin. Whereas these sanitizers were only minimally effective at reducing surface contamination of produce by human pathogens (Beuchat and Ryu, 1997; Beuchat, 1999; Francis and O'Beirne, 2002;), they would reduce risk of cross-contamination via water and equipment. We propose that these practices for tree fruits can serve as a general model of on-farm food safety program to be developed in a stepwise manner in Japan.
Footnotes
Acknowledgements
This research was part of the Research Project for Utilizing Advanced Technologies in Agriculture, Forestry, and Fisheries sponsored by the Ministry of Agriculture, Forestry, and Fisheries of Japan. We thank Mr. Kiyonori Uchishiba and Mr. Tadashi Taniguchi at the Japan Agriculture (JA) of Wakayama prefecture for assistance in surveying the orchard and sampling. We also thank Dr. Alley E. Watada for reading the manuscript.
