Abstract
Wheat grain and its products are widely consumed as fodder and basic daily food stuffs in Kyrgyzstan. Mycobiota is known to produce hazardous effects to a consumer since it produces mycotoxins. Henceforth, mycobiota starting from the field stage to flour, grain and flour samples were selected for mycological analysis from eight sites of flour manufacture: grain stored in storehouses before milling, mechanically cleaned grain, washed grain, grain dried and prepared for mill, roughly-milled flour, first grade flour and high grade flour. The samples were analyzed using classical mycological and immunoassay methods in order to detect mycotoxins producing fungi species. We isolated overall 27 species belonging to 7 genera. Mycotoxins producing species like Aspergillus flavus, Aspergillus ochraceus and Penicillium cyclopium were detected in the stored grains and in mechanically-cleaned grains. The species of Penicillium, Alternaria and Fusarium genera dominated in roughly-milled flour samples, so this site of flour manufacture still has a risk and danger of contamination with mycotoxins producing fungus. Only the final product i.e. the high grade flour lacked any fungal contamination. We recommend to scrutinize flour samples at the last stages of processing, particularly in the mills like B1, C1 and C4.
Keywords
Introduction
Certain species of fungi growing in food are able to produce metabolites toxic for animals, birds and human. Such metabolites are incorporated into a group of mycotoxins. Physicochemically, mycotoxins are thermostable and in most cases aromatic and nonantigenic low-molecular metabolites. Mycotoxins exert a diverse range of toxic evicts because their chemical structures are very heterogeneous. They develop a cancerogenic, mutagenic and teratogenic effect on human beings, animals and birds.
Mycotoxicosis is a disease that results from the ingestion of food contaminated with mycotoxins. For many centuries in some developed European countries Mycotoxicosis was not considered as a dangerous disease. Since 1970 cases of Mycotoxicosis caused by mycotoxins have intensively increased in many countries of the world. 1 In particular, cases of Mycotoxicosis diagnoses have increased in developing countries lately. Thus, the analysis conducted in Kyrgyzstan in 2004 shows that 44 samples out of 1123 samples of raw food taken from Osh, Chu and Issyk-Kyl regions were infected with mycotoxins. 2
Representatives of Aspergillus, Penicillium, Fusarium and Alternaria genera are more responsible for producing mycotoxins. Microscopic fungi can produce these poisonous substances not only during the vegetation on grain crops, but also during storage time. 36 High frequency of Alternaria species in grains of wheat, barley, oats, rye and other cereals has been discovered in many countries of the world.7,8 Scientists from Russia have marked a wide spread of fusarioz (Fusarium) of cereal heads in the European part of Russia, in the Urals and West—Siberian regions. 4
Aflatoxin producing strains of A. flavus and A. parasiticus has been isolated from the fields of California. In California, on a fig plantation scientists have found isolates of A ochraceus, A. melleus and A sclerotiorum3,9 Regular pollution of grapes with Aspergillus sp. has been marked in tropical and subtropical countries. For example, A. niger var. niger, A. flavus, A. niger var. awamori, A. foetidus, and A. candidus strains have been detected.10,11 A. niger, A. flavus, A. terreus and A. ustus strains have been found in grapes from Argentina and Brazil. 12
Penicillium species are different on their needs for climatic conditions from Aspergillus. Representatives of Penicillium sp. can grow at the temperature of 4–31 °C, whereas Aspergillus species can grow at the temperature of 12–39 °C.9,13 Penicillium fungi are spread out in all regions of the globe. Penicillium chrysogenum was isolated in France from grape samples and this strain has a potential to produce the Ochratoxin A. 14
The first step in the diagnostic process of the pathogen or disease associated with the production of mycotoxins is to know and understand the biology of organisms responsible for mycotoxicosis. The biology, ecology and dissemination of mycotoxins producing fungi found in agricultural crops and food is still unknown in Kyrgyzstan.
This research describes the results of microbiological analysis of grain and its products after harvesting and during processing in order to detect contamination with mycotoxins produced by fungi and risk points at the manufacture.
Material and Methods
Grain and flour samples, the points of their collection for the analysis
Wheat grains were stored at the temperature of 27–28 °C (in summer months) and at the temperature of 15 °C (in winter months), air humidity was 16.5%–17%, so these parameters do not meet standard requirements for the storage of grains in optimum conditions. Samples were collected once in two months from the sites:
Grain stored in storehouses before processing
Grain after mechanical cleaning
Grain after washing
Grain after drying and preparation for grinding
Flour after rough mill
Flour after middle mill
First-grade flour
High-grade flour
Samples were taken by 3 kg from each point and were subjected to a triple microbiological analysis.
Nutrient mediums used for fungi cultivation
Czapek-Dox Agar (CDA) medium (by adding antibacterial antibiotics 200 mg cycloheximide/litre) was used in order to isolate microscopic fungi and their pure culture; also a peptone—dextrose agar and a potato agar were used to study their culture, morphological and physiological characteristics.
Mycobiota analysis
From each collection, 20–25 grains per plate, usually in triplicate, were superficially sterilized for 5 minutes in 70% spirit to enumerate the storage fungi, then washed thoroughly in sterile distilled water for 2 minutes and plated onto Czapek-Dox Agar (CDA). The plates were incubated at the temperature of 22–27 °C for 10 days. Colonies of fungi growing around the grains were isolated to a pure culture by a standard technique. Individual species of fungi were counted separately and their numbers were expressed as Colony Forming Units per gram (CFU/g).
The percentage of abundance, density, frequency, occurrence and contribution was calculated for each species. 15 Common fungi species were identified using the determinants.6,11,16–21,32,36,37 The Aspergillus and Penicillium genera were identified after.22,23 Nomenclature of Aspergillus was accomplished. 24
The strains were deposited in the Microbiology Laboratory of Kyrgyz-Turkish International University. The Shenon index 25 was used to estimate biodiversity of fungi species in wheat grain. The Simpson's index 26 was used to determine a domination rate.
The frequency criteria were used to determine the importance of fungi species.
Test on the antagonistic activity of micromycetes cultures to bacteria
To test the activity against bacteria species, the fungus was spotted at the center of the plate containing MRS.02 agar. After 48 hours of incubation of fungus culture at 25 °C, the bacteria culture of Bacillus thuringiensis, Staphylococcus sp., and Erwinia sp. were spotted to the fungus mycelium. Bacterial growth inhibition was estimated using measurements of the colony density towards the edge in the direction of the grown fungal colonies. Controls of bacterial growth in the selected media were performed at the absence of fungus culture.
All tests were conducted in duplicates.
Results and Discussion
Level of fungal contamination of grain at flour-manufacture
Wheat grain stored in storehouse before processing
Grains covered and not covered with mycelium were recorded to calculate the level of fungal contamination. Grains in the storehouse before processing showed quite a high level of fungal contamination -15 to 30.5% (Fig. 1). Apparently, this mycobiota contains various species of fungi like representatives of ephytic microflora in plant grains.
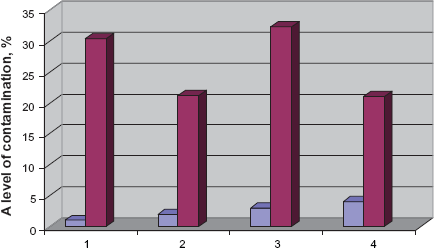
Fungal contamination of grains at the flour manufacture. 1. The grain stored in storehouse before processing 2. The grain after mechanical cleaning 3. The grain after washing 4. After drying and prepared for mill.
Wheat grain after mechanical cleaning
Grain from the granary goes to a cleaning machine where the grains are exposed to 70% of mechanical cleaning. The number of fungal spores on the cleaned grains reduced almost twice in comparison with the first point (Fig. 1). Obviously, during the mechanical cleaning fungal spores are eliminated with dirt and dust stuck on the grain coat.
Wheat grain after washing with room-temperature water
Wheat grain after mechanical cleaning goes to a washing machine to be washed up with water without disinfectants. The temperature of water during the winter time is 14°–16 °C and 20 °C in summer. Grain moisture was 14.8%–15.1% before washing in the machine, but after washing moisture rose up to 20%. The level of fungal contamination of the washed grain increased to 32.3% (Fig. 1). This might be caused by the increase of moisture contents in grains that leads to intensive growth and reproduction of fungi spores.
Wheat grain after drying and preparation for mill
At this point of flour-manufacture the grain gets dried. Grain moisture reduces to 16%–17%, and then the grain is sent to a mill. The level of fungal contamination of the dried grain decreased to 21%. It occurs because moisture contents decrease in grains before going to a mill (Fig. 1).
Mycobiota of grain and flour
Mycobiota of grain stored in storehouse before processing
Fifteen species classified under seven genera were isolated from the grains which included Penicillium, Alternaria, Helmintosporium, Cladosporium, Aspergillus and Mucor. Representatives of Penicillium genus showed maximum contribution (40,0%) and appeared to be the most predominant species in the grain stored before processing. Cladosporium species took the second position with 31.0%. Alternaria species took the third position with 13%. The percentage contribution of other fungi (Fusarium, Helmintosporium, Aspergillus and Mucor was equal (4.3%) (Fig. 2).
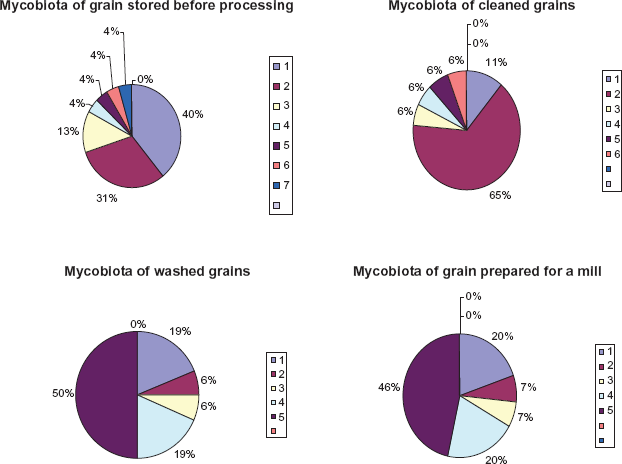
Species of fungi frequently isolated from different grain samples and their percentage contribution: 1. Penicillium 2. Cladosporium 3. Alternaria 4. Fusarium 5. Helmintosporium 6. Aspergillus 7. Mucor.
Mycobiota of grain after mechanical cleaning
Twelve species belonging to six genera were isolated from the cleaned grains. They included Cladosporium, Penicillium, Alternaria, Fusarium, Helmintosporium and Aspergillus species. Cladosporium species were abundant in the grain and constituted maximum percentage (65,0%) of the total. Although Penicillium species took the second position, its percentage contribution was much lower (11.0%) compared to that of stored grain. Alternaria, Fusarium, Helmintosporium and Aspergillus species were recorded more frequently than in stored grains (6,0%) (Fig. 2).
Mycobiota of grain after washing with water of room temperature
Ten species classified under five genera were isolated from the grains washed up with room-temperature water. They included Helmintosporium, Fusarium, Penicillium, Cladosporium and Alternaria species. Helmintosporium representatives showed maximum contribution (50.0%) and appeared to be the most predominant species. Fusarium species occupied the second position with 19.0%. It was much higher than in the stored and washed grains. Penicillium species occupied the same position with 19.0%. Percentage contribution of remaining genera (Cladosporium and Alternaria) was notable too (6.0%). Aspergillus species were not detected (Fig. 2).
Mycobiota of grain after drying and preparation for mill
At this phase grains are dried. Grain moisture reduces to 16%–17%. After drying the grains went to a mill. Ten species classified under five genera were isolated from the grains dried and prepared for mill. They included Helmintosporium, Penicillium, Fusarium, Cladosporium and Alternaria species (Fig. 3). Helmintosporium representatives showed maximum contribution (46. 6%), and appeared to be the most predominant species. Penicillium and Fusarium species showed higher and more notable contribution (20.0%) compared to that of the washed grains. Aspergillus species were not detected (Fig. 2).
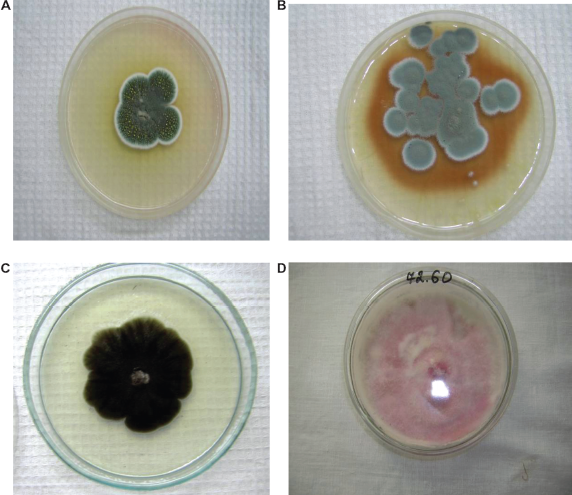
The colonies of fungi. A,B) Penicillium species C) Alternaria sp. D) Fusarium sp. found in grain before processing in mill.
Mycobiota of flour samples subjected to different degree of fineness and milling
Mills at flour-manufacture line up in the following sequence: B1, B2, B3, B4, C1, C2, C3, and C4. Each mill fulfills 40% of grinding. For example, grains which were not grinded in B1 mill do not go to the next B2 mill. Grains which were not grinded in “B” mills are not accepted in “C” mills. Flour after complete processing in mills “B” and “C” and going through a sieve is separated into supreme-grade flour, first-grade flour, second-grade flour, and then packed in bags on electronic scale and transported in tanks. We selected samples from mills B1, B4, C1 and C4 for mycological analysis.
Mycobiota of flour after processing in B1 mill
Ten species belonging to six genera were isolated from a rough fraction of flour. They included Cladosporium, Mucor, Penicillium, Alternaria, Fusarium and Helmintosporium species. Cladosporium and Mucor species dominated and constituted maximum percentage (29,0%) of the total. Helmintosporium and Alternaria species formed 14.0%; Fusarium and Penicillium formed 7.0% (Fig. 4).
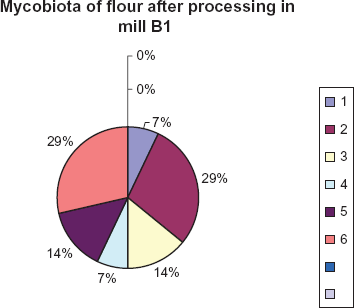
Species of fungi frequently isolated from flour samples and their percentage contribution. 1. Penicillium 2. Cladosporium 3. Alternaria 4. Fusarium 5. Helmintosporium 6. Mucor
Mycobiota of flour after processing in B4 mill
Eight species belonging to three genera were isolated from a rough fraction of flour. They included Cladosporium, Mucor and Penicillium species. Cladosporium and Mucor species were recorded frequently and made a maximum contribution (43.7% each) of the total. Penicillium species reached 12.5% (Fig. 5).
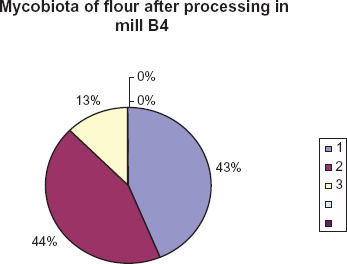
Species of fungi isolated from flour samples and their percentage contribution. 1. Mucor 2. Cladosporium 3. Penicillium.
Mycobiota of flour after processing in C1 mill
Again the representatives of Cladosporium and Mucor genera were abundant in flour fractions (43,0% and 50.0%). However, representatives of Alternaria were also detected (7.0%.) (Fig. 6).
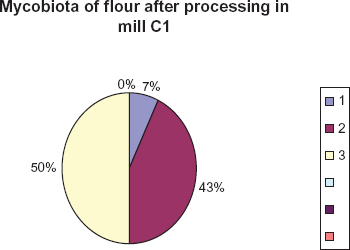
Species of fungi isolated from flour samples and their percentage contribution. 1. Alternaria 2. Cladosporium 3. Mucor.
Mycobiota of flour after processing in C4 mill
Only 2 species were isolated from a fraction of flour after processing at the last mill. They included Cladosporium and Mucor species. They demonstrated equal percentage—50.0%. (Fig. 7).
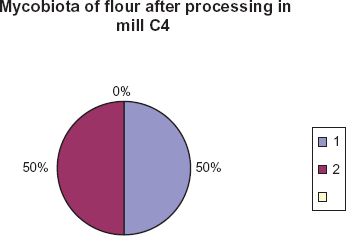
Species of fungi isolated from flour samples and their percentage contribution. 1. Mucor 2. Cladosporium.
Differentiation of Mycotoxins Producing Micromycetes from Saprophytic Species
Species belonging to 7 fungi genera (Aspergillus, Alternaria, Cladosporium, Helmintosporium, Fusarium, Penicillium, and Mucor) were isolated from grain and flour samples taken at different stages of flour-manufacture.
In further studies we paid more attention to the isolates of Aspergillus and Penicillium genera as producers of the most important mycotoxins responsible for mycotoxicosis of animals and human.
As it is known from some researches,8,10 mycotoxins producing micromycetes have specific physiological properties. They can grow at the temperature of 37 °C.
To specify fungi properties and to grow at the temperature of 37°, their pure cultures were cultivated on the Czapek-Dox Agar at the temperature of 4°, 25° and 37 °C.
We checked growth rate, the sizes of radial circles of growing colonies within 20–25 days. Only one isolate—Aspergillus sp. Asp-4 formed colonies at 37 °C on the 5th day (Fig. 8). At the temperature of 25 °C growth of this culture was more intensive, the diameter of a colony reached 4–4. 5 cm. Other Aspergillus isolates were not able to grow at 37 °C; however, at 25 °C their colony growth was normal. At 4 °C growth of Aspergillus strains was not noted within 20 days.
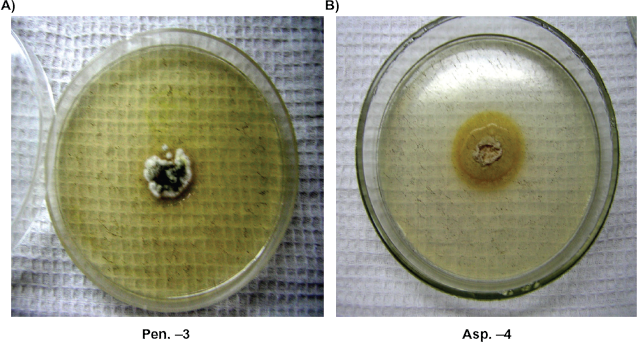
The growth of Pen-3, Penicillium sp and Asp-4, Aspergillus sp colonies at 37 °C temperature.
Penicillium cultures displayed a more distinctive need for temperature than Aspergillus isolates. Strains such as Pen-4, Penicillium sp., and Pen-3, Penicillium sp., were able to grow at 37 °C. In particular, Pen-3, Penicillium s. showed intensive growth, the diameter of a colony reached 2.8 cm in 5 days (Fig. 8). Penicillium cultures did not show intensive growth and showed a small size of colonies (1.6 cm) at 25 °C. All analyzed Penicillium isolates were able to grow at 4 °C (Fig. 9).
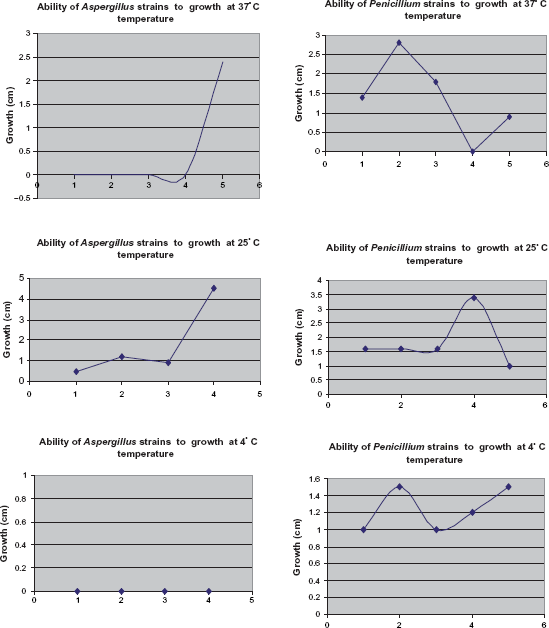
Growth ability of Aspergillus and Penicillium strains at different temperature.
Antagonistic Activity of Micromycetes Cultures to other Microorganism Groups
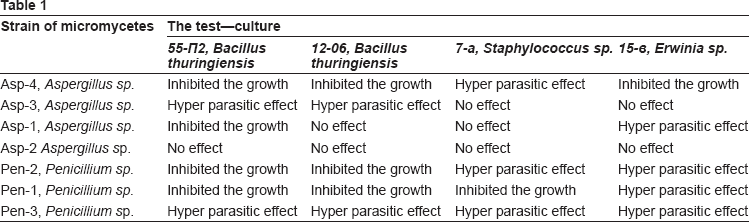
Antagonistic activity of Aspergillus and Penicillium species isolated from grain and flour was assayed against the Bacillus thuringiensis, toxins forming bacteria for insects, Staphylococcus—toxin forming bacteria in food that causes human intoxication; Erwinia sp a phytopathogen.
One strain of Aspergillus, Asp-4 showed a high inhibition activity towards all test-cultures. Totally this strain inhibited the growth of two strains of Bacillus thuringiensis (55-P2 and 12-06), which produce crystal toxins—intestinal poisons of Lepidoptera insects. This strain not only inhibited the growth of Staphylococcus aureus, but also showed a hyper parasitic effect on this strain growing in its colonies. Also it inhibited the growth of Erwinia bacteria (strain Erwinia sp.15-ó), that is a phytopathogen of plants. The isolate Asp-3 showed a hyper parasitic effect on the two strains of Bacillus thuringiensis. However, it did not affect any other test-cultures. The isolate Asp-1 had a hyper parasitic effect on Erwinia bacteria, it inhibited the growth of Bacillus thuringiensis 55-P2 strain, but it had no antagonistic effect on the other strain—Bacillus thuringiensis 12-06. The isolate Asp-2 was not active towards all test-cultures.
The isolate Penicillium, Pen-3 had the best antagonistic capacity against all test-cultures. Mycelium of this isolate fully covered the surface of the test-culture and used it as a source of supply. Other two isolates of Penicillium—Pen-2 and Pen-1 also showed the highest inhibition capacity when tested against Bacillus thuringiensis, Staphylococcus aureus and Erwinia sp. (Table 1). Three isolates of Penicillium genus— Pen-3, Pen-2 and Pen-1 can be preliminarily ranked to mycotoxins producing fungi. Two isolates of Aspergillus genus—Asp-4 and Asp-1 can be ranked to mycotoxins producing fungi. These cultures showed a high antagonistic activity towards all tested bacteria species. Among them only one Asp-4 was able to grow at the temperature of 37 °C.
Identification and Characterization of Selected Aspergillus and Penicillium Strains which were Considered to be Producers of Mycotoxins
Strain Asp—4
For 10–14 days this strain produces colonies 3–4 cm in the diameter at the temperature of 24–26° on Czapek-Dox agar. Colonies are usually flat or slightly furrow.
Air mycelium is of ochre—light yellow color. The reverse part of colonies has red—purple shades. Exudation is of amber color and produces a drop on the colony surfaces. A vesicle is spherical, colorless and 35–50 μ in diameter. Mature vesicle departs into 2–3 wide complete columns. The phialides are in two tiers and closely covers the vesicle. Chains of conidia are 1–1.5 mm in length, 10–14 μ in thickness; they have a pale—brown color and a thick (1–2 μ) rough coat. Conidia are spherical 2.5–3 μ. Sclerotia are 1 mm in diameter, purple and cylindrical. (Fig. 10). According to its cultural and morphological characteristics, this strain was determined as Aspergillus ochraceus (Wilhelm, Raper, Fennell).
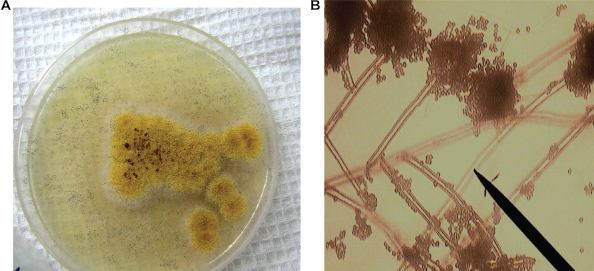
A) Aspergillus ochraceus colony on Czapek-Dox agar B) Aspergillus ochraceus conidiophores and conidia, × 900.
Aspergillus ochracues was isolated from the second point where wheat grains were cleaned mechanically. Aspergillus ochracues is known as a species producing the following mycotoxins—Ochratoxin A, Ochratoxin B, Ochratoxin C, Penicillic acid, Viomellein.
Strain Asp—1
For 10 days, at the temperature of 24–26 °C on Czapek-Dox agar this strain produces colonies 6–7 cm in diameter. Colonies are usually radially folded; air mycelium is of olive-yellowish-green color. Exudation is plentiful of yellow red color with an unpleasant smell. Immature vesicle is oblong, later more spherical, 10–65 μ in diameter. Mature vesicle is fine radial; they split into badly expressed columns. Chains of conidia are colorless, 3 mm in length. The phialides are in one tier. Conidia is spherical and thorn, 3.5–4.5 μ in diameter (Fig. 11). According to its cultural and morphological characteristics this strain was determined as Aspergillus flavus (Link; Raper, Fennel).
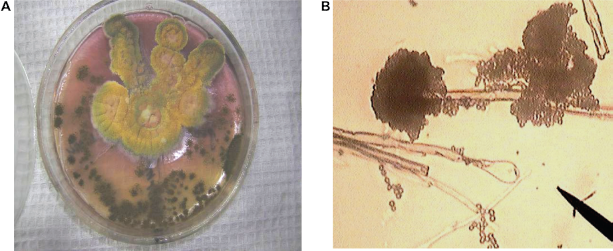
A) Aspergillus flavus Colonies on czapek-Dox agar B) Aspergillus flavus conidiophores and conidia, × 900.
Aspergillus flavus was isolated from the first point where grain was stored in storehouses before processing. Aspergillus flavus is considered to be a producer of the following toxic metabolites: Aflatoxin B1, Aflatoxin B2, Aflatoxin M1, Aflatrem (alkaloid), Aflatrem (indol alkaloid) and other.
Strain Asp—3
For 10–14 days, at the temperature of 24–26 °C on Czapek-Dox agar this strain produces colonies 3.5–4.5 cm in diameter. Colonies are of a furrow form with radial traces. Air mycelium is from white to the cream or pale yellow color forming a dense layer. The reverse part of colonies is pale yellow and has pink shades. Exudation is light and imperceptible. The vesicle is spherical and colorless, sometimes oblong. The phialides are in two tiers, of brightly yellow color. They are friable radial. Chains of conidia reach 1 mm in length with a smooth and colorless coat. Conidia are spindle—shaped and slightly rough, 2–2.5 μ in diameter. Sclerotia are of a cubical form of dark blue color (Fig. 12). According to its cultural and morphological characteristics this strain was determined as
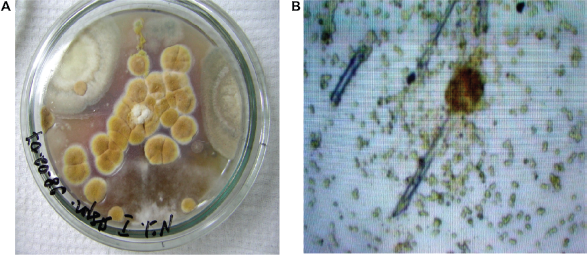
A) Aspergillus sulphures colonies on Czapek-Dox agar B) Aspergillus sulphures conidiophores and conidia, × 900.
strains Pen-2 and Pen-5
These strains produce fast-growing colonies on Czapek-Dox agar. Colonies are of a radially furrow form with a granular surface. Air mycelium of immature colonies is bluish-gray. Air mycelium of mature colonies is dim grey. The reverse part of immature colonies is orange brown, of mature ones is in purple tones. Exudation is plentiful and light and has a strong and mouldy smell. Metula depart from a substratum, 200–400 × 3–3.5 μ, slightly rough, with 2 branches. The phialides of 4–8 in a bunch, 7–9 × 2.5 μ. Conidia are from elliptic to spherical, 3.3–4 × 3–3.3 μ., chains form irregular mass (Fig. 13). According to its cultural and morphological characteristics these strains were determined as
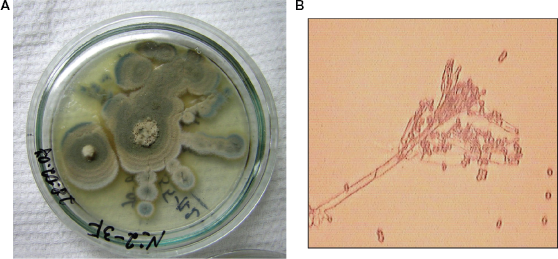
A) Penicillium martensii colonies on Czapek-Dox agar B) Penicillium martensii conidiophores and conidia, × 900.
Penicillium martensii was isolated from the first and second point of manufacture when grains were in storehouse after mechanical cleaning. There are no data about the mycotoxins produced by Penicillium martensii.
Strain Pen-1
This strain on Czapek-Dox produces fast-growing colonies. Colonies are of a less radial furrow form with abundant spore forming. Air mycelium of immature colonies is light blue, of mature ones is dark green and mealy. The reverse part of immature colonies is yellowish, of mature ones is orange—brown. Colonies have a strong mouldy smell. Metula is slightly rough and depart from a substratum, 200–400 × 3–3.5 μ, with several branches. The phialides are 7–10 × 2.2–2.8 μ, of 4–8 in a bunch, with the truncated top. Conidia are more or less spherical, 3.5–4 μ in diameter (Fig. 14). According to its cultural and morphological characteristics this strain was determined as
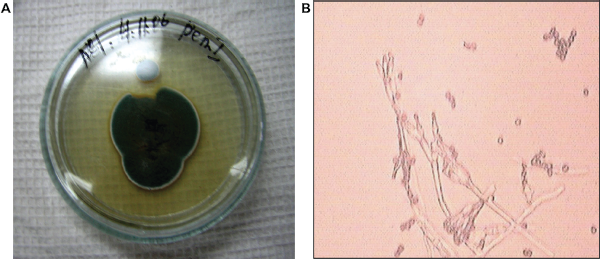
A) Penicillium cyclopium colonies on Czapek-Dox agar B) Penicillium cyclopium conidiophores and conidia, × 900
Strain Pen-3
On Czapek-Dox agar this strain produces radially folded and velvety colonies. Air mycelium is abundantly spore forming from bright—yellow green to bluish dark green shades. The reverse part is of yellow—brown color. Exudation is white like brilliant drops with a mouldy smell. Metula reach 100–200 μ in length, 3–3.5 μ in thickness. Phialides are of 8–10 in a bunch (Fig. 15). According to its cultural and morphological characteristics this strain was determined as
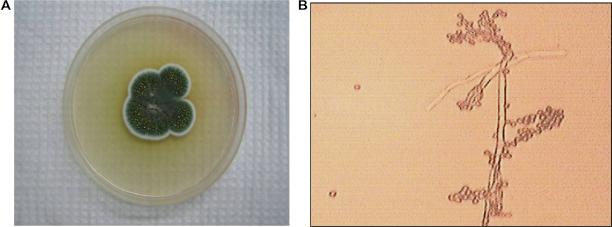
A) Penicillium frequentans colonies on Capek's agar B) Penicillium frequentans conidiophores and conidia, × 900.
Immuno-Enzyme Assay of Fungi Isolates Able to Produce Mycotoxins
The study of morphological, cultural, physiological, biochemical properties of fungi strains can give basic information on their ability to produce toxins; however, only specific researches, particularly an immuno-enzyme analysis method can detect the toxic level.27,28 The evaluation of toxin production was conducted in laboratory conditions according to a general scheme, which includes preparation of a fungi subculture, plating on a sterile grain substratum (on rice) with humidity of 30%, stationary incubation in darkness at the temperature of 25–26 °C for 14 days. Subculture inoculum was represented by homogeneous suspension of fungi cells in 0.01%-s’ water solution of Tween 80 with approximate concentration of 20 thousand spores per ml. Mycelium—grain biomass was homogenized in the mix of acetonitrol—water (6:1), kept for 14 hours at room temperature, intensively mixed and filtered through a paper filter. The received extract was analyzed by immune enzyme method to detect concentration of Ochratoxin A, aflatoxin B1 and Patulin. Special antibodies were used to each species of mycotoxins. The results showed that Aspergillus ochraceus isolated from wheat grain had ability of Ochratoxin biosynthesis in a number of 1.2–4. 8 mg/kg. Aspergillus flavus also was able to synthesize aflatoxin B1; however, this strain was poor toxigenic. The number of toxins in a mycelium—grain biomass was only 0.04–1.0 mg/kg. Toxin producing Penicillium cyclopium constituted 0.08 to 2.4 mg/kg.
Discussion
In Kyrgyzstan wheat grain, cereal products and flour are used for food preparation daily. Under favorable environmental conditions, some toxigenic molds can produce mycotoxins on agricultural commodities during plant growth and storage. Being overspread in the environment, toxigenic molds can be found in food products and forages without visible mycelium growth. Therefore, the absence of microscopic fungi mycelium does not indicate that a product does not contain mycotoxins. On the contrary, the presence of toxin producing fungi in food products is not a ground to assume that they contain mycotoxins. Generally, there are three types of food contamination with mycotoxins. Primary pollution of agricultural products occurs in the field during vegetation, after harvesting and during storage time. Secondary pollution can occur during processing in poor sanitary conditions. Finally, the last pollution can occur as mycotoxins residual effect as a result of feeding animals by fodder containing mycotoxins. 29
This study with the wheat grain and flour samples asserts that fungal infection occurs during harvest as well as storage and further continues until the last processing stages of flour though their incidence vary with the different samples. This might be caused by the differences in their moisture contents and cleaning degrees. Grain samples showed high levels of fungal contamination due to their moisture content. As for grains washed with water, contamination with fungi increased abruptly (32.3%) corresponding to the increase of moisture content (20%). Fungal contamination decreased to 21% in dried grain samples with the decreasing moisture content (to 16%). In cleaned grains due to the elimination of dirt and dust stuck on the grain coat, fungal contamination decreased almost twice (7.0%–15.0%) in comparison with grains in the storehouse before processing.
High levels of mycotoxins producing fungi were found in grains during storage time and at the mechanical cleaning point at flour-manufacture. Here Penicillium, Aspergillus, Alternaria and Fusarium species abounded in grain samples. At the fourth point, where the grains were prepared for mill after drying, there were no Aspergillus species but there was a significant number of Penicillium and Fusarium species (20.0%). Thus, grain before going to mill was free from Aspergillus species; however, Penicillium and Fusarium species were still detected. They cause a risk as they are mycotoxin-producing agents.
Three different species of Aspergillus, mycotoxin producers, were detected from grain samples. Aspergillus flavus was detected from the grains stored in storehouses before processing. Aspergillus flavus is the main source of aflatoxin, the most important mycotoxin in the world's food supplies. 30 These toxins badly affect the liver of warm-blooded animals. Aflatoxin B1 is the most dangerous in this group (LD50 = 6.5–16.5 mg/kg). However, for chickens and ducklings this rate amounts for 1 mg/kg. In a doze of 0.7 mg/kg aflatoxin B1 depresses DNA, concentration of vitamin A in the liver reduces, and concentration of fat rises.10,14,31,32
Aspergillus ochraceus was detected from the second point where wheat grains have mechanical cleaning. Aspergillus ochracues is known to be the source of the following mycotoxins—Ochratoxin A, Ochratoxin B, Ochratoxin C, Penicillic acid, Viomellein. Ochratoxin production occurs over a wide temperature range and, apparently, below the freezing point too. Ochratoxin— contaminated fodder has its major economic impact on the poultry industry.3,12,14,33 Aspergillus sulphureus was isolated from the first point where grain was stored in storehouses before processing. Aspergillus sulphureus is known as Ochratoxin A producing species.
During this study 3 different species of Penicillium were isolated from the grain samples. Penicillium martensii and Penicillium frequentans were recorded from the first and second point of research where the grains were in the storehouse after mechanical cleaning. They are not pathogen. Penicillium cyclopium was isolated from the second point where wheat grains have mechanical cleaning. Penicillium cyclopium is known as Patulin producing species.
Penicillium species produce toxins—Patulin and Citrinin. Citrinin is basically related to cereals and rice grain. It is produced by cells of P. citreonigrum, P. citrinum, P. expansum and P. verrucosum. Patulin is produced by cells of P. expansum, P. claviforme, P. cyclopium, P. equinum, P. glandicola, P. commune, P. lapidosum, P. melinii, P. novaezeelandiae, and P. griseofulvum.5,9,11,33
The researches show that Patulin occurs on bananas, pineapples, grapes, peaches, apricots, plums, and tomatoes.34,35 Inoculation experiments showed that Patulin may be produced by Penicillium species on a variety of food and especially fruit, but natural occurrence of Patulin is limited to fruit and is mainly associated with apples and apple products.
Demirici et al 36 detected Patulin in cherries, mulberry, raspberry, and strawberry. Cherries were contaminated with Patulin most frequently, 9 of 10 samples contained Patulin with concentration of 37 mg/kg.
Since the present study detects mycotoxin-producing species like Aspergillus flavus, Aspergillus ochraceus and Penicillium cyclopium in the grain samples, it is apparent that wheat bran as a waste of flour grinding manufacture contains these toxins; thus it is not suitable fodder for chicken or other animals. Even flour ready for sale contains a significant number of saprophytic fungi.
Only the final product, i.e. high-grade flour was free from Aspergillus, Penicillium and Fusarium contamination. We assert that the grinding process under several-sequence mills and through sieves eliminates the fungal spores with bran from grain covers. But rough grain fractions still contain fungal spores which are used for preparation of porridge and other food for daily need. Wheat bran, which is proven to have a high level of mycotoxins produced by their fungal component, is given to animals as fodder. Therefore, it is suggested that a severe scrutiny of the samples to be given to animals be done. Also, it is found that supreme-grade flour is safe for consumption since there is no possibility of mycotoxins production.
Publish with Libertas Academica and every scientist working in your field can read your article
“I would like to say that this is the most author-friendly editing process I have experienced in over 150 publications. Thank you most sincerely.”
“The communication between your staff and me has been terrific. Whenever progress is made with the manuscript, I receive notice. Quite honestly, I've never had such complete communication with a journal.”
“LA is different, and hopefully represents a kind of scientific publication machinery that removes the hurdles from free flow of scientific thought.”
Available to your entire community free of charge
Fairly and quickly peer reviewed
Yours! You retain copyright
Footnotes
Acknowledgements
This work was supported by the Kyrgyz Science Foundation “Food Biosafety”.
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author reports no conflicts of interest.


 2, Bacillus thuringiensis
2, Bacillus thuringiensis
 , Erwinia sp.
, Erwinia sp.