Abstract
Objectives
The lung cancer is most frequently diagnosed cancer incidence worldwide. A large number of populations are heavily affected to this every year with poor prognosis.
Methods
Our study investigated the anticancer effect of alternariol, a secondary metabolite, on A549 lung cancer cell line and benzo-α-pyrene induced lung carcinoma model on balb/c mice. The cytotoxicity assay, DAPI staining and the flow cytometry was performed to assess the anticancer efficacy of alternariol in A549 lung cancer cell. For in vivo study the toxicity study was performed. The lung cancer was developed in the animals via intraperitoneal administration of benzo-α-pyrene and subsequently 2 weeks later alternariol treatment was carried out for 24 weeks. The chemotherapeutic effect of alternariol was assessed through histopathological analysis, followed by immunohistochemistry and in vivo antioxidant study.
Results
The in vitro data suggested a significant percentage of early and late apoptotic events in A549 cells with G0/G1 phase arrest which ultimately caused apoptosis followed by alternariol therapy. The in vivo study showed that alternariol therapy decreased the pulmonary fibrosis and formation of granuloma in lung tissue and restored the normal histopathological characteristics of lung. Furthermore, alternariol treatment downregulated the expression of PI3K, Akt and inflammatory mediators such as IL-6, TNF-α and increased the expression of apoptotic markers, p53.
Conclusion
In conclusion, the treatment with alternariol effectively decreased the progression of lung cancer through the inhibition of carcinogenic markers by reprogramming the p53/PI3K/Akt pathway and IL-6/TNF-α mediated cytokine signaling in mice.
Introduction
Cancer has been identified as a tumorous growth with high metastasizing potential and a high ability to invade surrounding tissues. Lung cancer is the second most commonly diagnosed cancer and a substantial cause of cancer related death worldwide with an estimated 2.2 million new cancer cases and 1.8 million deaths (18%), according to GLOBOCAN 2020. 1 In order to eradicate the cancer, a number of strategies were implemented, but the occurrence of lung carcinoma increases gradually each year with limited efficiency. This further instigates the researchers to find out a new chemotherapeutic strategy to treat lung cancer effectively.
Recently, the utilization of mycotoxins, biologically active secondary fungal metabolites, has gain interest among researchers due to its excellent cytotoxicity towards the mammalian cells. 2 The mycotoxin alternariol (AOH) is generally produced by Alternaria alternate, which can be found as natural food contaminant in small grains, wheat, fruits, vegetables and as well as in the processed fruit products like juices and wines. 3 It causes frameshift mutations through Ames Salmonella strain TA97. 4 It also induces the mutations in hypoxanthine-guanine phosphoribosyltransferase (HPRT) and thymidine kinase (TK) gene as shown in mammalian cultured cells. It is reported to cause both single and double strand DNA breaks. 5 The derivatives of AOH possess a wide range of biological activities especially anticancer activity. 6 There are mainly two mechanisms of actions which have been identified for AOH, including apoptosis, that leads to shrinkage and fragmentation of nuclear DNA, and necrosis, characterized by inflammation mediated rupture of plasma membrane. 7 AOH also has been reported to reduce the cellular proliferation, leads to cell cycle arrest, and corresponds to its DNA damaging activity. 8 Furthermore, the induction of autophagy and senescence has also been reported for AOH. 9 AOH is also associated with the stimulation of AhR/ARNT pathway, which results in the regulation of CYP1A1 and apoptosis. 10
There are several etiological factors responsible for the initiation and progression of lung cancer which includes multiple genetic and epigenetic abnormalities. These factors certainly lead to the activation of oncogenes and inactivation of tumor-suppressor genes. The chronic inflammation possesses a key triggering factor for the initiation of cancer which is influenced by genetic abnormalities. 11 It has been suggested that inflammation promotes lung cancer through several pathway such as inflammatory cell–derived reactive oxygen species that bind to DNA and lead to genomic alterations. 12 The high expression of interleukin-6 (IL-6), pro-inflammatory cytokine, has been also reported to be involved in poor survival of lung cancer patients). 13 Additionally, Tumor necrosis factor-alpha (TNF-α), an inflammatory cytokine, associated with the regulation of inflammatory responses with cancer initiation and progression in lung cancer. 14 As a result, IL-6 and TNF-α has become a popular target for lung cancer treatment. PI3K/Akt/mTOR pathway is an important signaling cascade for the cell proliferation, growth, survival. Any alteration of this pathway thus certainly leads to cancer due to mutations and amplifications of the components regulating the PI3K/Akt pathway. 15 The PI3K pathway is commonly activated in lung cancer which causes oncogenesis through cancer cell survival, growth, proliferation and migration. 16 Furthermore, p53 plays an important role in intrinsic apoptotic pathway, thus any alterations of p53 regulation due to epigenetic modification hampers the cell growth and survival machinery and leads to cancer. Taken together, targeting the cell survival machinery with the induction of apoptotic and modulation of inflammatory cytokine singling will provide a concrete chemotherapeutic strategy to eradicate lung carcinogenesis.
Till now there is lack of documented evidences demonstrating the cytotoxic effect of AOH on the lung cancer model. The model of benzo-α-pyrene induced lung carcinoma was utilized to explore the chemotherapeutic efficacy of AOH through examining the p53/PI3K/Akt cascade and cytokine signaling pathway. The chemotherapeutic effect of AOH in A549 lung cancer cell line has also been assessed, which further correlates the in vivo and in vitro approach to establish the chemotherapeutic efficacy of alternariol.
Materials and methods
Chemicals and reagents
Alternariol, benzo-α-pyrene, biotinylated goat anti-rabbit IgG, streptavidin peroxidase, 3,3’-diaminobenzidine (DAB) and proteinase K, were procured from Sigma Aldrich Chemical Co. (St. Louis, MO). Rabbit anti-rat p53, PI3K, Akt, IL-6 and TNF-α were purchased from GeneTex International Corporation (Global). All the other reagents were purchased from the local firms at its purest grade.
In vitro study
Cell culture
The lung cancer cell line A549 (CCL-185) was obtained from ATCC (American Type Culture Collection), United States. The cells were supplemented with the DMEM growth media accompanied by 10% FBS (foetal bovine serum) and penicillin/streptomycin antibiotics. The cells were incubated at 5% CO2, 95% relative humidity at 37°C temperature.
Cell viability assay
The MTT assay was conducted through the metabolism of MTT (3-(4,5 dimethylthiozol-2-yl)-2,5-diphenyl tetrazolium bromide) by succinate dehydrogenase to form formazan crystals followed by the reduction of MTT reagent. The A549 cells were seeded for 24 h in incubator at 5% CO2 and 95% relative humidity. The cells were then treated with different concentrations of AOH and incubated for 24 h. After 24 h, the cells were supplemented with MTT solution and incubated for 3 h. The absorbance of solubilized formazan crystals was determined at 560 nm. % cell viability was calculated as: % viability = 100- % of cytotoxicity
DAPI staining protocol
The DAPI staining was performed to observed nuclear blebbing and chromatin condensation in the A549 cells.
Flow cytometric detection of p53 expression
The cultured A549 cells were treated with different concentrations of AOH for 24 h in 6 well plate and further supplemented with anti-rabbit p53 polyclonal antibody for 30 min incubation period at room temperature in dark condition. The cells were then analyzed by flow cytometry.
Flow cytometric analysis
The detection of apoptotic cells in A549 cells was carried out by labeling the DNA with Annexin V-FITC and propidium iodide (PI). The FACS (fluorescence-activated cell sorting) analysis was performed to observe the nuclear arrangement in the cell cycle events. After conducting 10,000 events the flow-cytometric data was processed by Modfit tools.
In vivo study
Animal
All the animal experimentations were performed according to the Institutional Animal Ethical Committee of Zhengzhou University (Approval no. 2021ZU420) and all procedures followed ethical criteria in accordance with applicable legislation.
The animals were obtained from Zhejiang University Animal Center. For toxicity study, Swiss albino mice (18–20 g), 6–8 weeks old, of both sexes were used and for the chemotherapeutic study male balb/c mice (20–25 g) of 6 weeks old were utilized. The animals were kept in polypropylene cages at a temperature of 20–25°C with 22% relative humidity in light/dark cycle of 12 h. In 10 days of acclimatization period the food pellets and drinking water were supplied adequately for all the animals.
Toxicity study
Acute oral toxicity study
The toxicological evaluation of alternariol was assessed by acute oral toxicity study in order to determine the LD50 value, as per the guidelines of Organization for Economic Cooperation and Development (OECD) for testing of chemicals, TG 420 (adopted December 2001) with slight alterations. The animals were divided into five experimental groups, each of which contains 6 animals, three of each sex. The groups were followed as one normal control group receives only vehicle and the other four groups were selected as per the different doses selected for alternariol which includes 2000, 1000, 800 and 600 mg/kg body weight. After the drug introduction, the food and water were immediately supplied to the animals and kept under observation for 3 days for any mortality.
Sub-acute toxicity study
Following the acute toxicity study the sub-acute toxicity study was carried out in order to select the doses for chemotherapeutic study. The 28-days repeated oral toxicity study was performed according to the guidelines of Organization for Economic Cooperation and Development (OECD) for testing of chemicals, TG 420 (adopted December 2001) with slight alterations. The animals were divided into five experimental groups and each group contains 6 animals, three of each sex. The groups were followed as one normal control group receives only vehicle and the other four groups were selected as per the different doses selected for alternariol which includes 400, 200, 100 and 50 mg/kg body weight. During the experimental period the animals were observed for general clinical appearance and mortality. Along with that the food and water consumption and the body weights of the animals were also measured weekly. After 28 days of repeated oral toxicity study, all the animals were euthanized and the vital organs (liver, kidney, stomach and testis) were collected for histopathological evaluation. Furthermore, the hematological and serum biochemical parameters were also assessed.
Hematological and serum biochemical analysis
The hematological parameters were determined by using Medonic CA-620 cell analyzer systems (Boule Medical, Stock-Holm, Sweden). The blood samples were obtained from the retro-orbital plexus of the animals and it was centrifuged for 10 min at 3000 rpm in order to separate the serum form the blood. It was then assessed through automated biochemistry analyzer BIOBASE UA-240 cell analyzer systems (Biobase Biotech (Jinan) Co., Ltd.) for the evaluation of biochemical parameters of the collected serum samples.
Histopathological evaluation
After completion of sub-acute toxicity study the animals were euthanized and the vital organs were excised from them which include liver, stomach, kidney and testis. The organs were then kept into 10% formalin solution overnight for fixation. The organs were then treated with graded alcohol for dehydration and mounted on low melting paraffin wax upon the glass slides of 5 micron. It was then deparaffinized and rehydrated using xylene and alcohol respectively. The slides were then treated with heamatoxylin and eosin stain (H&E) and evaluated under light microscope.
Experimental protocol
The animals were distributed randomly into five experimental groups in which each group was consisting of 6 animals. The mice of 6 weeks old (except group I) were administered with a single i. p. Injection of benzo-α-pyrene (100 mg/kg body weight) in corn oil and the lung cancer was induced. After 2 weeks of carcinogen induction the mice were treated with alternariol which continued for 24 weeks. The experimental groups were followed as:
Group I- vehicle control animals, Group II- treated with benzo-α-pyrene and allotted as carcinogen control, Group III- treated with benzo-α-pyrene + received 50 mg/kg alternariol, Group IV- treated with benzo-α-pyrene + received 100 mg/kg alternariol, Group V- treated with benzo-α-pyrene + received 200 mg/kg alternariol.
After completion of chemotherapeutic study the animals were euthanized, lung tissue was collected and fixed in 10% formalin. The lung tissues were assessed for appearance of any tumor structure. Lung tissues were evaluated through histopathological analysis and immunohistochemistry.
Histopathological evaluation of lung tissue
The lungs were collected from the euthanized animals and fixed into 10% formalin. The tissues were embedded in paraffin wax and 5 µm thick tissues were placed on slides. The staining was done using H & E and observed under light microscope.
Antioxidant activity of lung tissue
The lung tissues were homogenized (10% w/v) in 0.1 M of phosphate buffer (pH 7.0) for the assessment of antioxidant activity. The mixture was centrifuged and the supernatant was used for enzymatic analysis. 17
Catalase activity
As per the method of Sinha et al. the catalse activity of homogenized lung tissue was carried out. The absorbance was noted at 620 nm and the catalase activity was expressed in terms of µmol of H2O2 consumed/min/mg protein. 18
Superoxide dismutase activity
The SOD activity was determined as per the method described by Awasthi et al. The SOD activity was assessed as units/min/mg protein. 19
Glutathione peroxidase activity
The activity of GPx was determined according to the method of Rotruck et al. (1973). The GPx activity was denoted as µmol of GSH consumed/min/mg protein. 20
Immunohistochemical analysis
The excised lung tissue were fixed in 10% formalin solution, embedded in paraffin, cut into 5 µm thickness and mounted on glass slides. The tissues were then deparaffinized and immersed into the solution of H2O2. The sections were then treated with goat serum for 1 h and then incubated with anti‐mouse p53, PI3K, Akt, IL-6 and TNF-α antibodies overnight at a temperature of 4oC. It was then followed by PBS washing and treated with HRP conjugated biotinylated secondary antibody for 30 min. The development of the sections was done using DAB and hematoxylin staining was carried out for counterstaining followed by evaluation under light microscope.
Statistical analysis
The statistical data were expressed as mean ± SEM. Statistical analysis was performed by variance analysis (ANOVA), accompanied with Tukey’s multiple comparison test via Graph Pad Prism (Version 5). The differences were considered as statistically significant at p < .05.
Results
In vitro study
Alternariol inhibits viability of A549 cells
The MTT assay demonstrated that treatment with AOH caused a dose dependent inhibition of cell viability of A549 cells (Figure 1(a)). AOH treatment caused a decrease in the viability of the A549 cells tends to decrease to 68.27%, 52.64%, and 40.23% at concentrations of 60, 85, and 110 μM respectively. (A) Effects of AOH on cell viability of A549 cells at 24 h where * represented p < .05 as compared to 35 μM concentration. Similarly # represented p < .05 as compared to 60 μM concentration, $ p < .05 as compared to 85 μM concentration and δ p < .05 as compared to 110 μM concentration (B) DAPI stained A549 cells after 24 h of treatment with AOH, white arrows represented nuclear fragmentations and white arrowhead represented membrane blebbing (C) detection of apoptosis in A549 cells by Flow cytometry after treatment with AOH (D) percentage of apoptotic cells versus concentration in A549 cells where * represented p < .05 as compared to control. Similarly # represented p < .05 as compared to 42.5 μM concentration, $ p < .05 as compared to 85 μM concentration (E) percentage of apoptotic cells in early and late apoptosis stage in A549 cells. * represented p < .05 as compared to control. Similarly # represented p < .05 as compared to 42.5 μM concentration, $ p < .05 as compared to 85 μM concentration (F) analysis of cell cycle phase distribution of A549 cells after the treatment with AOH (G) quantitative of distribution of A549 cells in different phases of cell cycle. Data represent means ± SEM from three different experiments in triplicate. The results were compared using ANOVA, followed by a Tukey’s multiple comparison post-hoc analysis.
Alternariol causes chromatin condensation
The induction of nuclear condensation by alternariol in A549 lung cancer cell was depicted in (Figure 1(b)). The treatment with 127.5 μM of alternariol in A549 cells demonstrated the highest condensation of chromatin, at 24 h of incubation, which indicates the apoptosis.
Effect of alternariol on the expression of p53
The A549 cells showed significant increase in the expression of p53 due to alternariol therapy in a dose dependent manner (Figure 1). The maximum number of p53 labeled cells in the M2 quadrant were detected in the alternariol treated group where the maximum number of p53 labeled cells in M1 quadrant were noted for the untreated group.
Effect alternariol in cell cycle and apoptosis
The initiation of apoptosis in A549 cells, due to AOH treatment at different concentrations for 24 h has been depicted in (Figure 1(c)). The percentages of apoptotic cell are 16.25%, 37.68% and 49.21% following treatment with 42.5, 85, and 127.5 μM of alternariol in A549 cells (Figure 1(d)). Moreover, an increase of cell population in early and late apoptotic stage was determined in alternariol treated cells at the end of 24 h (Figure 1(e)).
According to the cell cycle analysis the apoptotic cells were represented as sub diploid cells in (Figure 1(f)). Treatment of the A549 cells with 42.5, 85, and 127.5 μM of alternariol denoted 63.68%, 61.26% and 52.34% cells in the G0/G1 phase. Along with that, following the treatment with AOH an upregulation of cells was observed in the S-phase of A549 cells (Figure 1(g)).
Toxicity study
Acute and sub-acute toxicity
After performing the acute toxicity study the LD50 dose of alternariol has been found to be 600 mg/kg body weight. According to the LD50 value of AOH the doses for the sub-acute toxicity study was selected which was followed as 400, 200, 100, 50 mg/kg body weight. During the repeated oral toxicity study treatment related mortality was observed for the dose level of 400 mg/kg. Additionally, the food and water consumption was decreased for the animals treated with 400 mg/kg of AOH. The body weight of the animals was also reduced for the same treatment group.
Hematological and serum biochemical analysis
Haematological finding in male Swiss albino mice treated with alternariol for 28 days repeated-dose oral sub-acute toxicity study.
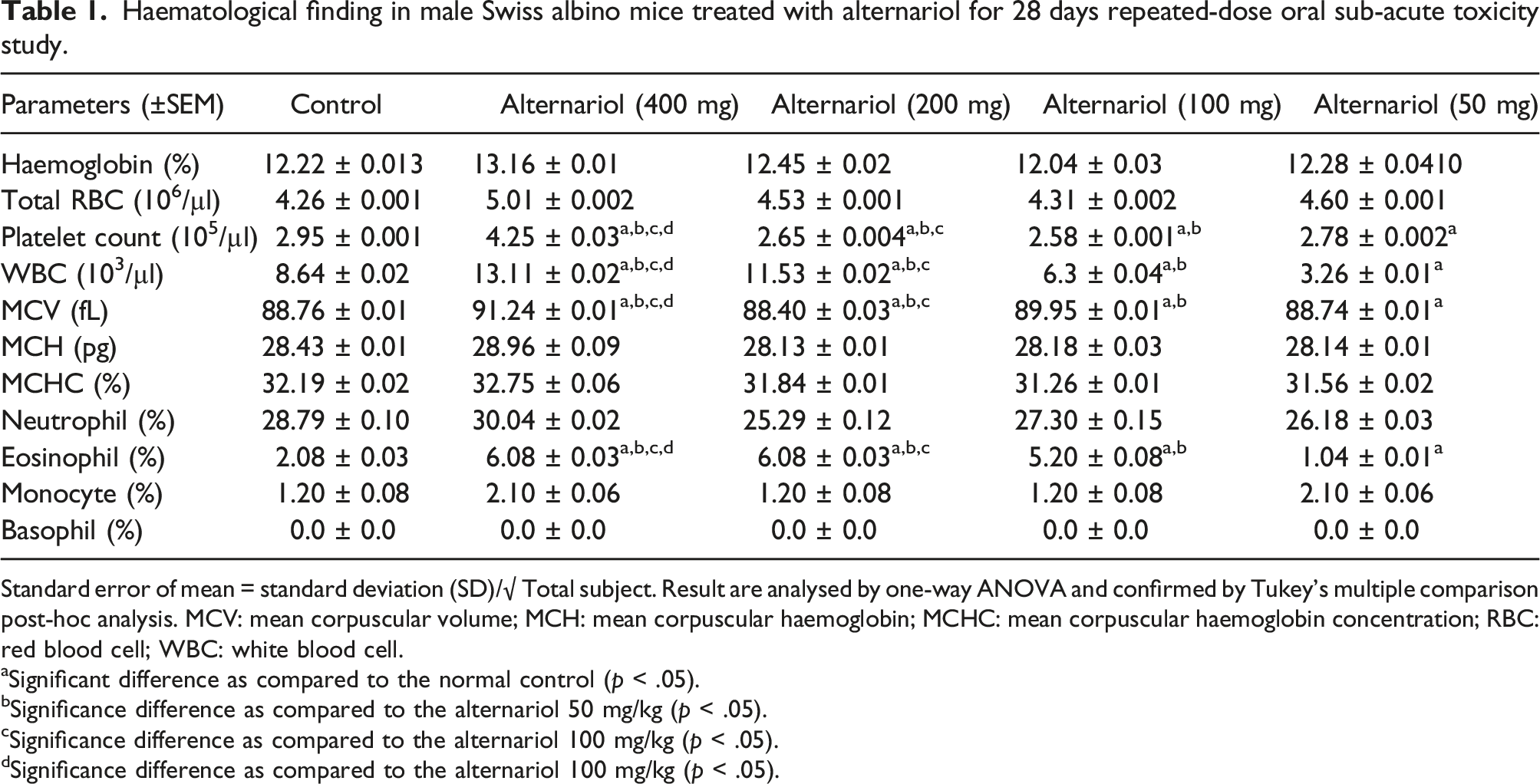
Standard error of mean = standard deviation (SD)/√ Total subject. Result are analysed by one-way ANOVA and confirmed by Tukey’s multiple comparison post-hoc analysis. MCV: mean corpuscular volume; MCH: mean corpuscular haemoglobin; MCHC: mean corpuscular haemoglobin concentration; RBC: red blood cell; WBC: white blood cell.
aSignificant difference as compared to the normal control (p < .05).
bSignificance difference as compared to the alternariol 50 mg/kg (p < .05).
cSignificance difference as compared to the alternariol 100 mg/kg (p < .05).
dSignificance difference as compared to the alternariol 100 mg/kg (p < .05).
Haematological finding in female Swiss albino mice treated with alternariol for 28 days repeated-dose oral sub-acute toxicity study

Standard error of mean = standard deviation (SD)/√ Total subject. Result are analysed by one-way ANOVA and confirmed by Tukey’s multiple comparison post-hoc analysis. MCV: mean corpuscular volume; MCH: mean corpuscular haemoglobin; MCHC: mean corpuscular haemoglobin concentration; RBC: red blood cell; WBC: white blood cell.
aSignificant difference as compared to the normal control (p < .05).
bSignificance difference as compared to the alternariol 50 mg/kg (p < .05).
cSignificance difference as compared to the alternariol 100 mg/kg (p < .05).
dSignificance difference as compared to the alternariol 100 mg/kg (p < .05).
Serum Biochemical finding in male Swiss albino mice treated with alternariol for 28 days repeated-dose oral sub-acute toxicity study
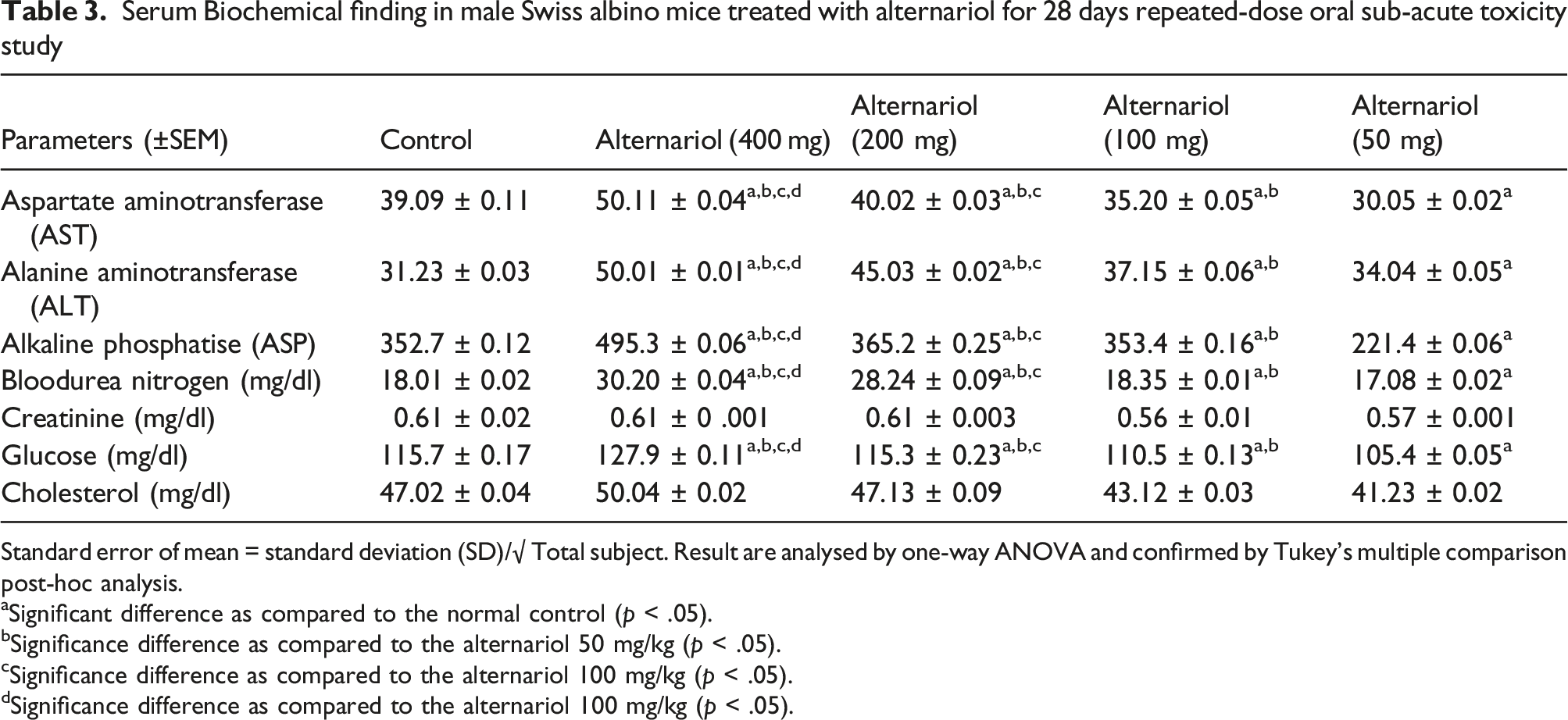
Standard error of mean = standard deviation (SD)/√ Total subject. Result are analysed by one-way ANOVA and confirmed by Tukey’s multiple comparison post-hoc analysis.
aSignificant difference as compared to the normal control (p < .05).
bSignificance difference as compared to the alternariol 50 mg/kg (p < .05).
cSignificance difference as compared to the alternariol 100 mg/kg (p < .05).
dSignificance difference as compared to the alternariol 100 mg/kg (p < .05).
Serum Biochemical finding in female Swiss albino mice treated with alternariol for 28 days repeated-dose oral sub-acute toxicity study

Standard error of mean = standard deviation (SD)/√ Total subject. Result are analysed by one-way ANOVA and confirmed by Tukey’s multiple comparison post-hoc analysis.
aSignificant difference as compared to the normal control (p < .05).
bSignificance difference as compared to the alternariol 50 mg/kg (p < .05).
cSignificance difference as compared to the alternariol 100 mg/kg (p < .05).
dSignificance difference as compared to the alternariol 100 mg/kg (p < .05).
The WBC and RBC levels were significantly high (p < .05) in the 400 mg/kg group as compared to the animals of the control group. Along with that ALT, AST, ALP levels were also upregulated in the 400 mg/kg group in comparison to the animals of the control group (p < .05). As compared to the control group the glucose and BUN levels were increased for the animals of 400 mg/kg group (p < .05).
Histopathological analysis of organs
After completion of subacute oral toxicity study the vital organs such as kidney, liver, stomach and testis were collected from the euthanized animals and analyzed for histopathology. The morphological features of kidney in the control group depicted unaltered structure of glomerulus, bowman’s capsule, proximal tubule, and distal tubule (Figure 2[i] A). Thickening of capsular membrane, ruptures, desquamated nuclei, vacuolization were detected in the kidney tissue of 400 mg/kg AOH treated animals in comparison to other treated animals (Figure 2[i] E). The kidney tissues of the animals treated with 50, 100, 200 mg/kg of AOH did not show any significant alterations (Figure 2[i] B, C, D). The liver tissue of normal control group exhibited portal vein, bile duct, hepatic artery, hepatocytes, sinusoids and the kupffer cells (Figure 2[ii] A). The liver tissue of animals treated with 400 mg/kg AOH denoted periportal mononuclear infiltrates, degeneration of hepatocytes, focal inflammation of hepatocytes (Figure 2[ii] E). No alterations were perceived in the liver tissues of the 50, 100, and 200 mg/kg of AOH treated group (Figure 2[ii] B, C, D). The histopathology of the stomach of normal control group and the AOH treated group (50, 100, and 200 mg/kg) (Figure 2[iii] A, B, C, D) showed sub-mucosa, muscularis mucosa, and mucosal layers with distinct gastric pits, chief cells, parietal cells, mucosal cells, and surface epithelial cells. The stomach tissue of animals treated with 400 mg/kg of AOH indicated hemorrhages between villus, hyperplasia, leukocyte infiltration (Figure 2[iii] E). The normal architecture of testis was observed in the animals of normal control group and in the AOH treated group (50, 100, and 200 mg/kg) (Figure 2[iv] A, B, C, D). The animals treated with 400 mg/kg AOH indicated edema in interstitial tissue, degeneration of seminiferous tubule and hyperplasia (Figure 2[iv] E). (i) Histopathological representation of Kidney of Balb/c mice. (A) Normal tissue showing (g) glomerulus, Bowman’s capsule (bc), Macula densa (md), Proximal convoluted tubule (pct), Distal convoluted tubule (dct) (B) kidney tissue exposed to 50 mg/kg alternariol (C) kidney tissue exposed to 100 mg/kg alternariol (D) kidney tissue exposed to 200 mg/kg alternariol (E) exposed to 400 mg/kg alternariol showing thickening of capsular membrane (tbm), ruptures (r), desquamated nuclei (dn), vacuolization (ii) Histopathological representation of Liver of Balb/c mice (A) Normal control showing the Central vein (cv), Bile duct (bd), Sinusoidal dilation (sd), Kupffer cell (kc), Lymph vessel (lv) (B), (C) and (D) liver tissue exposed to 50 mg/kg, 100 mg/kg and 200 mg/kg of alternariol. (E) Liver tissue exposed 400 mg/kg alternariol showing periportal mononuclear infiltrates (pmi), degeneration of hepatocytes (dh), focal inflammation (fi). (iii) Histopathological representation of stomach of Balb/c mice (A) Normal control showing muscularis externa (me), Submucosa (sm), Muscularis mucosa (mm), Lamina propia (lp), Gastric pit (gp), epithelial lining (epl) (B), (C) and (D) Stomach tissue exposed to 50 mg/kg, 100 mg/kg and 200 mg/kg of alternariol. (E) Stomach tissue exposed 400 mg/kg alternariol showing Hemorrhages (h) between villus, Hyperplasia (hyp), leukocyte infiltration (Li). (iv) Histopathological representation of testis of Balb/c mice (A) Normal control showing Sertoli cell (sc), Spermatogonia (Sp), Seminiferous tubule (St), Interstitial tissues (It) is seen within the tubular lumen (B), (C) and (D) testis exposed to 50 mg/kg, 100 mg/kg and 200 mg/kg of alternariol. (E) Testis exposed to 400 mg/kg alternariol showing edema in interstitial tissue (E), Degeneration of seminiferous tubule (D) and hyperplasia (Hyp). (H&E) 10X magnification (inset 40X).
Antioxidant status
A significant reduction of SOD, CAT and reduced glutathione levels was observed in the homogenized lung tissue of carcinogen control group (p < .05). AOH treatment significantly increases the levels of SOD, CAT and GST in the lung tissue. The 400 mg/kg group showed significant elevation of SOD, CAT and GST in the homogenized lung tissue in comparison to the carcinogen control group (p < .05) (Figure 3). Effect of alternariol on in vivo antioxidant enzymes SOD (superoxide dismutase) and CAT (catalase), (glutathione) GST. * Significant difference as compared to carcinogen control group (p < .05). # Significant difference as compared to 50 mg/kg group (p < .05). $ Significant difference as compared to 100 mg/kg group (p < .05).
Histological analysis of lung tissue
The histological evaluation of lung tissue has been depicted in the (Figure 4). The normal architecture of lung tissue was represented by the normal group (group I) which showcased the bronchiole (b), pulmonary alveoli (pa), alveolar sac (as), blood vessels (bv), pulmonary capillary (pc) of lung tissue (Figure 4(a)). The carcinogen control group (group II) showed thickened alveolar walls (t), congested alveolar cavity (cac), lymphocyte infiltration (li) in alveolar space, severe fibrosis (f) and formation of granuloma (g) (Figure 4(b)). Severe fibrosis (f), lymphocyte infiltration (li) in alveolar space, thickened alveolar walls (t) and congested alveolar cavity (cac) was identified in the AOH treated group at a dose level of 50 mg/kg body weight (Figure 4(c)). For 100 mg/kg dose of AOH showed slight lymphocyte infiltration (li) and minor thickening of alveolar walls (t) (Figure 4(d)). However, no sign of hyperplasia or cellular proliferation in lung tissue was identified in the 200 mg/kg group of AOH treated animals (Figure 4(e)), representing normal architectural profile of the lung tissue. (A) Histological appearance of lung tissue of normal control showing bronchiole (b), pulmonary alveoli (pa), alveolar sac (as), blood vessels (bv), pulmonary capillary (pc) (B) Carcinogen control shows thickened alveolar walls (t), congested alveolar cavity (cac), lymphocyte infiltration (li) in alveolar space, severe fibrosis (f) and formation of granuloma (g) (C) Lung tissue of benzo-α-pyrene induced group treated with 50 mg/kg alternariol showing Severe fibrosis (f), lymphocyte infiltration (li) in alveolar space, thickened alveolar walls (t) and congested alveolar cavity (cac) (D) Lung tissue of benzo-α-pyrene induced group treated with 100 mg/kg alternariol showing slight lymphocyte infiltration (li) and minor thickening of alveolar walls (t) (E) Lung tissue of benzo-α-pyrene induced group treated with 200 mg/kg alternariol having almost normal architecture.
Immunohistochemistry
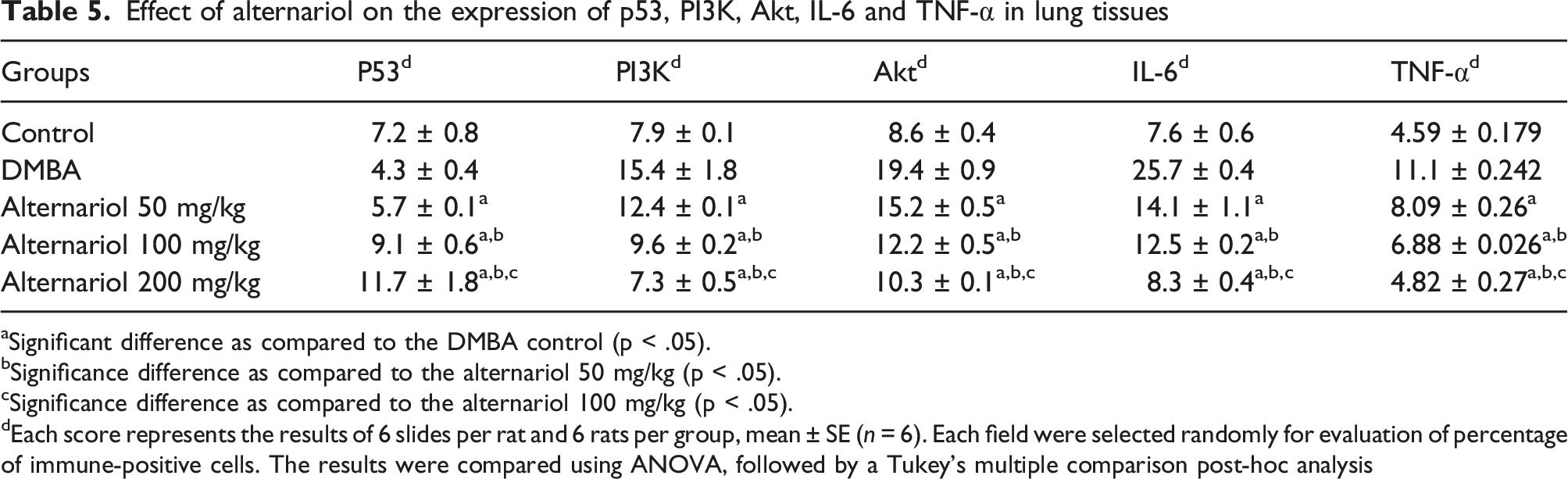
The immunohistochemical analysis of the lung tissue was performed for assessment of the protein expression which includes tumor‐suppressor protein p53, the growth regulatory proteins PI3K, Akt and inflammatory mediators IL-6, TNF-α (Figure 5). In the normal control group (group I) a moderate expression of tumor‐suppressor protein p53 was observed in the bronchiolar and alveolar wall. Along with that bronchiolar cytoplasmic localization was also observed (Figure 5(i) (a)). Whereas, in the carcinogen control group (group II) there was reduced expression of p53 (Figure 5(i) (b)). In the AOH treated group at the dose levels of 50, 100 and 200 mg/kg body weight a significantly (p < .05) high expression of p53 was detected in alveolar walls and bronchiolar epithelial membrane (Figure 5(i) C, D, E) as compared to the carcinogen control group (Table 5). There was a moderate expression of PI3K in the lung tissue of normal control group (Figure 5(ii) A) in alveolar walls. The expression of PI3K was significantly high for the carcinogen control group (Figure 5(ii) B) in comparison to the normal control group. On the other hand, a significant (p < .05) reduction of PI3K expression was noted in the AOH treated group (Figure 5(ii) C, D, E) (50, 100, 200 mg/kg) as compared to the carcinogen control group (Table 5). The expression of Akt was detected in alveolar wall and capillary lumen for the normal control group (Figure 5(iii) A). The expression of Akt was significantly increased in the carcinogen control group (Figure 5(iii) B). Whereas, there was a significantly (p < .05) low expression of Akt in the alveolar duct region for the AOH treated groups (Figure 5(iii) C, D, E) (50, 100, 200 mg/kg) in comparison to the carcinogen control group (Table 5). There was a low expression of IL-6 in the lung tissue of normal control group (Figure 5(iv) A). The expression of IL-6 was significantly high for the carcinogen control group (Figure 5(iv) B) as compared to the normal control group. On the other hand, a significant (p < .05) reduction of IL-6 expression was noted in the AOH treated group (Figure 5(iv) C, D, E) (50, 100, 200 mg/kg) in comparison to the carcinogen control group (Table 5). The slight expression of TNF-α was detected in alveolar wall for the normal control group (Figure 5(v) A). For the carcinogen control group the expression of TNF-α was significantly increased (Figure 5(v) B). Whereas, there was a significantly (p < .05) low expression of TNF-α in alveolar wall for the AOH treated groups (Figure 5(v) C, D, E) (50, 100, 200 mg/kg) in comparison to the carcinogen control group (Table 5). The immunohistochemical analysis of the (i) p53, (ii) PI3K, (iii) Akt and (iv) IL-6 (v) TNF-α expressions in the lung tissues of different groups of mice (A) the normal control (B) carcinogen control (C) 50 mg/kg of alternariol treated (D) and (E) 100 and 200 mg/kg alternariol treated. All images at 40X. Arrows represents expression of specific antibodies. Effect of alternariol on the expression of p53, PI3K, Akt, IL-6 and TNF-α in lung tissues aSignificant difference as compared to the DMBA control (p < .05). bSignificance difference as compared to the alternariol 50 mg/kg (p < .05). cSignificance difference as compared to the alternariol 100 mg/kg (p < .05). dEach score represents the results of 6 slides per rat and 6 rats per group, mean ± SE (n = 6). Each field were selected randomly for evaluation of percentage of immune-positive cells. The results were compared using ANOVA, followed by a Tukey’s multiple comparison post-hoc analysis
Discussion
In recent years the traditional chemotherapeutic medications have become obsolete as a result of their severe side effects and the emergence of drug resistance, mandating the development of novel compounds to diminish cancer incidence while avoiding the associated adverse effects. In this instance, the natural products would be the most viable option because of their potential anticancer activity and minimal side effects. 21 Thus, the current research designed to establish the chemotherapeutic effect of AOH in A549 lung cancer cell line and a benzo-α-pyrene induced lung carcinogenesis model in balb/c mice and the possible molecular mechanistic approach of AOH as an anticancer drug was further postulated. In spite of that, some limitations has been came forward that need to be reconsidered such as calculation and justification of sample size, evaluations of power analysis, expression profile of several inflammatory markers which increases the acceptance of this molecule in clinical settings.
As per the cell viability study of A549 cells, treatment with AOH caused a significant reduction of cell viability through the inhibition of cell proliferation and induction of apoptotic processes. The flow cytometric analysis demonstrated that AOH therapy caused the initiation of apoptotic events which indicated a significant percentage of early and late apoptotic events in A549 cells. Thus, leads to cell cycle arrest at G0/G1 phase that subsequently caused cellular death. The apoptotic events initiated by alternariol was further confirmed by the p53 expression on A549 cells. The dose dependent increment in the expression of p53 labeled cells in M2 quadrant suggested the alternariol mediated induction of apoptosis through p53 driven intrinsic apoptotic pathway.
In order to establish the safe dosage of AOH for the carcinogenic study the acute and sub-acute toxicity study has been performed to evaluate the LD50 dose of this novel agent. The LD50 dose of AOH was found to be 600 mg/kg body weight for Swiss albino mice. In sub-acute toxicity study significant changes were observed in the architecture of vital organs which include kidney, liver, stomach and testis at the dose level of 400 mg/kg in the AOH treated group. On the other hand, the 50, 100, 200 mg/kg AOH treated group was not associated with any histopathological alternations of their vital organs which indicates the non-toxic and safe doses of AOH that could be further utilized in the carcinogenic study.
The induction of cancer through chemical carcinogens is multiphase process which is associated with triggering of normal cells to malignant one followed by invasion into nearby tissue. 22 In this study the lung cancer was induced in balb/c mice through the administration of benzo-α-pyrene 100 mg/kg i. p. In corn oil. The histopathological evaluation of the lung tissue demonstrated formation of granuloma and severe pulmonary fibrosis in carcinogen control group. Furthermore, congested alveolar cavity, thickened alveolar walls, lymphocytic infiltration in the alveolar space was also observed in the carcinogen treated animals. The alternariol treatment causes significant reduction in the pulmonary fibrosis and restoration of normal morphological feature with substantial decrease in granuloma formation. These outcomes clearly established the anticancer activity of AOH against benzo-α-pyrene induced lung cancer in mice.
The immunohistochemical analysis of lung tissue was performed to investigate the expression of tumor suppressor protein p53, cellular growth and survival proteins PI3K, Akt and inflammatory markers such as IL-6, TNF-α to establish the signaling pathway through which AOH shows its chemotherapeutic activity against lung carcinoma. Several studies reported that, the mutation of p53, a tumor suppressor protein, is responsible for the cancerous lesions due to alteration of tumor suppressive pathways. 23 Increased expression of p53 decreased the number of cells through the induction of intrinsic apoptotic pathway which further initiates the caspase mediated downstream events, eventually causing cell death. 24 The findings of this research instigated the upregulation of p53 expression which confirms the induction of apoptosis in the lung cancer cells. Furthermore, PI3K signaling pathway stimulates the cellular growth and prevents the cancer cell from apoptosis through several cell survival pathway mediated by PI3K. In this study, the carcinogen control group showed upregulation of PI3K and Akt expression while the AOH treated group marked a significant downregulation of these proteins in the lung tissue. As a result of the AOH treatment, cancer cells experience a considerable reduction in cellular proliferation and become more sensitive to apoptosis. Moreover, there is a vivid correlation between the chronic inflammation and the induction of cancer. The chronic inflammation is mainly mediated through the proinflammatory cytokines such as TNF-α and IL-6, which also promote the epigenetic alterations in the promoter regions of the tumor suppressor proteins and cell cycle regulatory genes. Thus causes inactivation of p53. 25 In carcinogenic progression, TNF-α (tumor necrosis factor), an inflammatory cytokine, plays a critical role that is commonly found in the tumor microenvironment. 26 In inflammation-associated carcinogenesis the involvement of TNF-α has also been identified which supports tumor growth, cancer cell survival, metastasis and destabilize the immune responses. 27 The in vivo study suggested the upregulation of TNF-α and IL-6 in the carcinogen control group whereas, alternariol treatment significantly downregulates the expressions of TNF-α and IL-6 which clearly depicting the beneficial effect of AOH therapy in lung cancer.
The reactive oxygen species (ROS) plays a vital role in cellular oxygen metabolism which accounts for numerous cellular activities through the activations of various signaling pathways responsible for cellular growth and proliferation at physiological concentrations. However, increased production of ROS leads to disruption of redox homeostasis and causes the ROS mediated destruction of several important macromolecules, which includes DNA, proteins, and lipids 28 and eventually promotes the carcinogenesis. 29 The healthy cells are able to protect themselves from the harmful effects of oxidants by the action of a complex system of antioxidant enzymes such as SOD, CAT and GST. 30 From the experimental findings, it was confirmed that the carcinogen control group showed a decreased level of SOD, CAT, and GST but the treatment with AOH causes an upregulation in the levels of SOD, CAT, and GST in the homogenized lung tissue which may contribute to the prevention of ROS generation as well as inhibits the cellular growth and proliferation.
Conclusion
In conclusion, the treatment with alternariol significantly increased the p53 expression in both the A549 lung cancer cells and in benzo-α-pyrene induced lung cancer model of mice. The upregulation of p53 level is directly associated with the p53 mediated apoptotic events which caused the cellular death of the cancer cells in both in vitro and in vivo. Furthermore, the chemotherapeutic activity of AOH established through the inhibition of cellular growth and proliferation through the inhibition of PI3K/Akt mediated cell survival mechanisms with the downregulation of inflammatory cytokines TNF-α and IL-6 (Figure 6). The molecular mechanistic pathway of alternariol in lung cancer.
Moreover, this study strongly provides an insight of potential chemotherapy at a considerably low dose of drug therapy regimen through the induction of potential biomarkers responsible for apoptosis in lung cancer cells by stimulating intrinsic apoptotic pathway. Furthermore, the in vitro and in vivo studies demonstrated undeniable efficiency of the alternariol therapy that can inhibit, repeal, or interrupt the development of lung cancer. Thus, in recent future it may provide a potential strategy for lung cancer chemotherapy in clinical settings.
Footnotes
Acknowledgements
The authors are highly obliged to the Department of basic nursing, College of nursing and health, Zhengzhou University for their continuous support and encouragement throughout the study period.
Author contributions
QL, YY designed and planned the experiments. XW, XY, YZ performed the experiments. QW, YZ analyzed the data and wrote the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
All the animal experimentations were performed according to the Institutional Animal Ethical Committee of Zhengzhou University (Approval no. 2021ZU420) and all the procedures performed throughout the work on the project have complied with ethical guidelines corresponding to relevant laws.
