Abstract
Electrophysiological studies have shown that gonadotropin-releasing hormone (GnRH) modifies chemosensory neurons responses to odors. We have previously demonstrated that male Plethodon shermani pheromone stimulates vomeronasal neurons in the female conspecific. In the present study we used agmatine uptake as a relative measure of the effects of GnRH on this pheromone-induced neural activation of vomeronasal neurons. Whole male pheromone extract containing 3 millimolar agmatine with or without 10 micromolar GnRH was applied to the nasolabial groove of female salamanders for 45 minutes. Immunocytochemical procedures were conducted to visualize and quantify relative agmatine uptake as measured by labeling density of activated vomeronasal neurons. The relative number of labeled neurons did not differ between the two groups: pheromone alone or pheromone-GnRH. However, vomeronasal neurons exposed to pheromone-GnRH collectively demonstrated higher labeling intensity, as a percentage above background (75%) as compared with neurons exposed to pheromone alone (63%, P < 0.018). Since the labeling intensity of agmatine within neurons signifies the relative activity levels of the neurons, these results suggest that GnRH increases the response of female vomeronasal neurons to male pheromone.
Introduction
Gonadotropin releasing hormone (GnRH) is an important reproductive hormone and neuromodulator that is synthesized and released by three sets of neurons in the brains and nasal cavities of vertebrate animals. The GnRH decapeptide varies somewhat in its amino acid sequence between these neuronal groups within a given species. The neurons that produce GnRH reside in preoptic/hypothalamic region, mescencephalic area and terminal nerve/ventral forebrain, and are designated as neural groups GnRH I, II and III, respectively. In addition, these three designations also refer to the form of the neuropeptide in the vertebrate brain that is released from the specific group of neurons. Each GnRH-producing set of neurons is derived from a separate embryological precursor cell set within the neural plate. 1 5 GnRH I secreted from cells in the hypothalamus controls the release of LH and FSH from the pituitary gland, whereas GnRH II and III potentially act as neuromodulators at multiple brain sites.
Within the nasal cavity, GnRH is released from neurons of the terminal nerve and is thought to function as a neuromodulator of chemosensory neurons in the periphery. We and others have shown that GnRH fibers generally reach only as far as the lamina propria of the chemosensory mucosa fibers normally do not project into the sensory epithelium. However, we have evidence that GnRH reaches the surface of the epithelium via its incorporation into the mucus secreted by nasal glands. Radioimmunoassays of nasal mucus from the tiger salamander have substantiated the presence of GnRH in the mucus (unpublished observations).
Previous research in our laboratory and in that of others utilizing patch clamp recording technology has demonstrated that GnRH alters the responses of olfactory neurons to odors. In the nasal cavity of salamanders, GnRH has been shown to influence both voltage-activated and odor-activated responses of olfactory neurons. 6 8 It should be noted that in all of these studies, GnRH does not have an effect by itself; ie, GnRH does not evoke any change in membrane potential indicating that GnRH by itself does not open ion channels. GnRH is not the only neuroactive substance that modulates olfaction. Another neuroactive substance synthesized in the terminal nerve that modulates olfaction is a molecule with immunological similarity to both neuropeptide Y 9 and molluscan cardio-excitatory peptide or FMRFamide. 10 In rodents, dopamine from sympathetic fibers has been shown to alter olfactory- and electrically-induced currents in olfactory receptor neurons.11,12 Thus, there appears to be multiple avenues of altering the responses of chemosensory neurons in the nasal cavity to chemical stimuli.
We are specifically interested in how behaviors and physiology are coordinated by the interplay of neurohormones and modulators with sensory and motor systems. In this context, we are asking the question: does GnRH, a sex hormone and potential neuromodulator, modify the neural responses of a species specific pheromone in the plethodontid salamander Plethodon shermani, our model system of pheromonal control of reproduction? In behavioral studies, P. shermani female salamanders are more receptive to males when male pheromone is delivered to their vomeronasal organs.13,14 In the present study, we specifically tested the hypothesis that GnRH increases the responses of at least some vomeronasal neurons to pheromone stimuli.
Materials and Methods
We conducted two separate experiments: (1) to verify that P. shermani had GnRH in the terminal nerve and that these neurons could be traced to the peripheral nasal chemosensory system, and (2) to determine the effects of GnRH on the pheromone-induced uptake of agmatine in vomeronasal neurons.
Animals
Plethodon shermani
Eight female salamanders (P. shermani) were used in these experiments. Two animals were used to ascertain the presence of GnRH in the brain and nasal cavity. Six animals were used as olfactory subjects to test the effects of GnRH on pheromone-induced agmatine uptake. Animals were collected from Wayah Bald (Macon County, NC) during August, the beginning of the plethodontid mating season. Animals were maintained individually, each in a plastic box (31 × 17 × 9 cm) lined with moist paper towels and containing crumpled moist towels. The salamanders were exposed to a 14:10 light/dark illumination schedule and were fed wax worm larvae or fruit flies.
Immunocytochemical Analysis of GnRH in Brain and nasal cavity
Salamanders were anesthetized by immersion in 1% tricane methane sulfonate (MS-222; Sigma Chemical Company, St. Louis, MO) for 3 min and were perfused with 0.5 mL of 0.9% NaCl (saline) and 10 mL of Zamboni's fixative (2% paraformaldehyde-15% picric acid in 0.1 M phosphate buffer, pH 7.3) at a rate of 0.5 mL/min. The heads were immersed in the fixative overnight, then in DeCal (Decal Chemical Corporation, Congers, NY) for 3 days to decalcify the cranial bone, and in 30% sucrose in phosphate buffer as a cryoprotectant for 3 days. Heads were sectioned at 30 μm in the horizontal plane using a cryostat microtome. Sections were picked up directly onto slides and air-dried. Immunocytochemical procedures were used to visualize GnRH immunoreactivity (-ir) in the terminal nerve within the brain and nasal cavity. The tissue was rinsed in phosphate-buffered saline (PBS) pH 7.4, followed by a pre-incubation in 0.5% Triton-X-100 with 1% normal goat serum in PBS for 20 minutes and then an incubation in GnRH antisera (1:4000; Diasorin, Stillwater MN) for 3 days. Controls consisted of incubation of tissue in solutions without the primary antisera and incubation in GnRH antisera preabsorbed with 2 μM GnRH (Peninsula, San Carlos, CA). Again, the tissue was rinsed in PBS and labeled with Alexa 488-conjugated goat anti-rabbit IgG (Molecular Probes, Eugene, OR) for 30 minutes. Some sections were counter-labeled with DAPI to label cell nuclei. The tissue was examined with an Olympus microscope to identify fluorescently labeled neural cell bodies and fiber projections. Digital photographs were collected of the labeled terminal nerve cells and fibers.
Delivery of pheromone/Hormone stimuli and Tissue Fixation of plethodontid salamanders
We used relative amount of agmatine uptake and immunocytochemical labeling intensity as an indication of the magnitude of neural activation. Methods of mental gland removal and preparation of the male pheromone solution followed established protocols. 13 Female salamanders received a 3 mM agmatine/whole male pheromone solution with (n = 3) or without (n = 3) 10 μM GnRH. One microliter of solution was delivered to the nasolabial groove of salamanders every 2 minutes for 45 minutes. Following several saline rinses, animals were decapitated; heads were fixed overnight in 4% paraformaldehyde/2% glutaraldehyde, decalcified in DeCal and cryoprotected in 30% sucrose/PBS. Sequential frozen coronal sections (20 microns thick) were collected for processing with immunocytochemical procedures. Tissue from each experimental group was placed on the same set of slides to ensure identical immunocytochemical processing.
Immunocytochemical procedures to Visualize Agmatine
Immunocytochemical procedures for labeling AGB were conducted as previously reported. 15 Tissues were rinsed in PBS to remove the fixative, incubated for 30 min in a preincubation buffer and subsequently incubated in rabbit anti-AGB antisera (1:4000; Chemicon International, Inc., Temecula CA; Lot # 18112624) for three days. Following additional rinses in PBS, the sections were incubated in secondary antibody, avidin-HRP (Vector Laboratories, CA) and labeled with diaminobenzidine (DAB).
Histological Analysis of plethodontid Vomeronasal Organ
Coronal sections of female salamander head were examined for identifying chemosensory cells containing DAB. Digital images of chemosensory mucosae were obtained using an Olympus microscope with a SPOT camera (Diagnostic Instruments, Sterling Heights, MI). Digital images of vomeronasal neurons were obtained from sections every 100 microns through the vomeronasal organ.
DAB-labeled vomeronasal neurons were counted in each of these sections of salamander head. The number of labeled neurons per section from each animal was used to compare the number of labeled vomeronasal neurons between pheromone and pheromone-GnRH groups.
In addition, all neurons demonstrating an entire cell body with a nucleus and a clearly labeled dendrite were tagged to be included in the densitometric analysis. Densitometric data were obtained from live images of vomeronasal neurons at one time using Scion Image. Maximum and minimum density values were 256 and 0 respectively corresponding to black and white intensity levels. An oval marquee was placed over the cell body as shown Figure 1, and a densitometric value was obtained. The same marquee was then moved adjacent to the cell body to obtain a background measurement. Sixty-nine densitometric values were obtained from the three animals in the pheromone-only group (n = 31, n = 22, n = 16), and seventy-nine values were obtained from the three animals in the pheromone-GnRH group (n = 36, n = 10, n = 33). Densitometric data were standardized by expressing the intensity of label as a percentage of background levels for each data point. Data are graphically plotted as the mean of the average cell densities from each of the 3 animals per treatment group.
Micrograph of AgB-labeled neurons illustrating the strategy for collecting densitometry data. (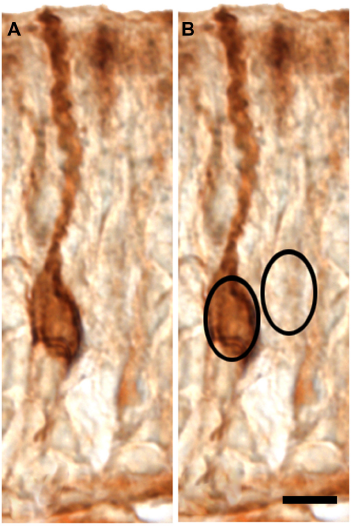
Statistical Analysis of Plethodontid Vomeronasal Organ
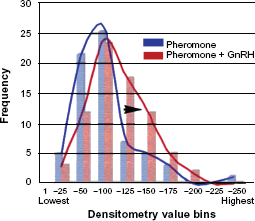
Generalized estimating equation analyses were used to determine whether number of cells showing labeling (Poisson/log-linear link model) or density values (Normal/linear link model) from pheromone vs. pheromone-GnRH groups were significantly different while accounting for the repeated measures made from each animal.16,17 A Kolmogorov-Smirnov test of the residuals from the latter model showed the Normal/ linear link to be appropriate for the density values. Results are provided as mean ± SEM. In addition, standardized densitometric values were grouped into bins of 25 value point increments. The number of value points in each bin was graphed on a frequency distribution curve for pheromone and pheromone-GnRH groups to visualize the distribution in intensity values demonstrating any overlap of the two groups and any shift in values between the two groups. Ethical approval was obtained for the use of animals in this research from the University of Oklahoma IACUC
Results
GnRH immunocytochemistry of the terminal nerve
The distribution of the terminal nerve plexus in P. shermani was very similar to that of other salamander species, eg, Ambystoma tigrinum
18
(Fig. 2). Female P. shermani salamanders possessed GnRH-ir neural cell bodies and fibers in the ventral forebrain extending from the olfactory bulbs to the preoptic region (Fig. 3A). Labeled fibers were apparent also in olfactory and vomeronasal nerve fascicles attached to the brain and within the nasal cavity (Fig. 3B). The appearance of the GnRH-ir cell bodies and fibers were similar to those in other vertebrate species that demonstrate GnRH neuron along the terminal nerve.
Illustration of the ventral surface of P. shermani brain and nasal cavities demonstrating the distribution of the terminal nerve (red). Horizontal section of P. shermani brain showing GnRH-ir (green) neurons along the terminal nerve. Horizontal section of olfactory nerve within the nasal cavity.


GnRH effects on pheromone Responses in P. shermani
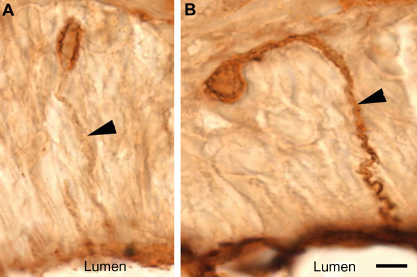
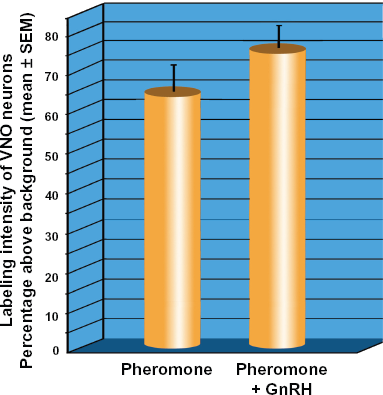
Pheromone application and pheromone-GnRH application both produced labeling of vomeronasal neurons in female P. shermani salamanders (Fig. 4A and B). The number of labeled vomeronasal neurons per section was not significantly different between pheromone (13.4 ± 0.9) and pheromone-GnRH groups (14.0 ± 0.4, P = 0.82). However, visual observation of the two sets of tissues suggested that GnRH application along with the pheromone produced more robust labeling of both the cell body and dendrites of the vomeronasal neurons (Figs. 4A, 4B and 5). Standardized densitometric values of vomeronasal labeling intensity were significantly greater from animals exposed to pheromone-GnRH (75% ± 6% above background) compared to animals that received pheromone alone (63% ± 7% above background; P = 0.018; Fig. 6). While pheromone and pheromone-GnRH groups demonstrated overlapping densitometric values, as evidenced from the frequency distribution, there was a shift to the right for the pheromone-GnRH group to greater numbers of neurons demonstrating more intense labeling (Fig. 7).
Low magnification micrographs of AGB-labeled vomeronasal neurons following the application of pheromone-only ( Micrographs of neurons labeled with AGB illustrating difference in dendrite labeling. ( Graph demonstrating percentage above background intensity of agmatine labeling of vomeronasal neurons. Frequency distribution of percentage above background labeling intensities of Pheromone (blue) and Pheromone-GnRH (red) groups.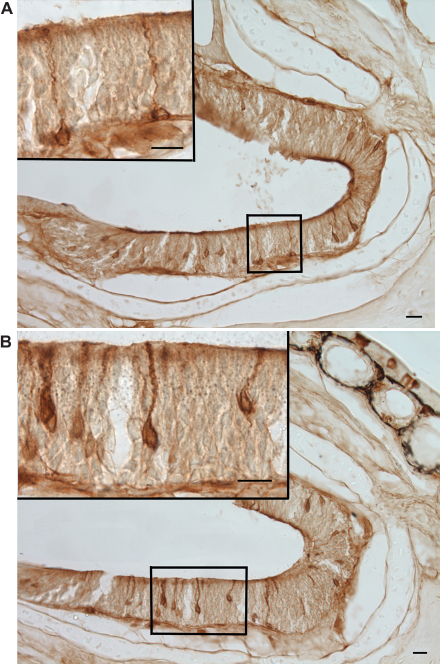
Discussion
Agmatine, decarboxylated arginine, is taken up into neurons upon their stimulation and the opening of nonspecific cation channels.19,20 Previous experiments have shown that only calcium blockers prevented agmatine from entering synaptosomes. 21 Although other channels may be involved in different cell membrane locations, it is highly likely that calcium or nonspecific cation channels are responsible for the uptake of agmatine in our experiments. In some animals agmatine is an endogenous neuroactive substance in discrete areas of the brain. Our immunocytochemical studies have demonstrated that agmatine is not endogenously present in neurons of P. shermani nasal cavity, and that application of agmatine alone to chemosensory neurons leads to very little or no uptake. Therefore, in this species, agmatine can be used to assess stimulation of chemosensory receptor neurons. We have previously used this method to study the neural activation of female P. shermani vomeronasal neurons to male pheromone and its components. 22 While the labeling intensity in our experiment does not represent a direct quantification of the agmatine uptake by chemosensory stimulation, it does allow a relative comparison of different stimulatory conditions. In addition, since all tissue was processed and labeled on the same slides, we are confident that all tissue was processed in exactly the same manner with the same concentration of reagents. Our choice of neurons with identifiable nuclei and dendrites for the densitometric analysis of vomeronasal neurons also provided a level of standardization of our data between groups and obviated the need to collect random samples.
The more intense labeling of vomeronasal neurons with stimulation by pheromone and GnRH suggests that one or more sets of ion channels were modulated by GnRH. Since stimulatory agents were applied to the apical dendritic knobs covered with microvilli, and these knobs are isolated from the remainder of the neuron by tight junctions, 23 we assume that any modulatory activity occurred in the region of the dendritic knob and microvilli.
In the nasal cavity, terminal nerve cell bodies and fibers are located within other chemosensory nerve fascicles as far as the lamina propria underlying the chemosensory epithelium. Therefore, under normal conditions, GnRH from the terminal nerve would have to gain access to the surface of the mucosa to stimulate the dendritic knobs. Our previous unpublished study demonstrating that GnRH is present in nasal mucus provides evidence of the most obvious route by which GnRH reaches the surface of the chemosensory mucosa.
In other species including reptiles and amphib-ians, vomeronasal neurons are thought to transduce pheromonal signals through V1R and V2R receptors coupled to G-proteins Gq and Go, respectively; PLC synthesis of DAG; and a TRPC2 channel, a channel that allows calcium to enter the cell; 24 28 For reviews see. 29 31 In P. shermani, the male pheromone is a mixture of proteinaceous molecules. Large nonvolatile molecules generally stimulate V2R receptors, and in line with this V2R mRNA has been detected via in situ hybridization in the female P. shermani vomeronasal epithelium. 32 Therefore, we would expect that pheromone signaling in P. shermani is accomplished via V2R, Go and TRP channels.
In electrophysiological studies, GnRH applied to olfactory neurons does not alter membrane potential by itself. This fact demonstrates that GnRH does not directly stimulate olfactory neurons as an odorant and does not have the potential for activating neural electrical activity leading to action potentials. However, GnRH does modify both odorant-induced and electrically induced changes in olfactory neuron membrane potential, thereby suggesting that GnRH is acting as a neuromodulator of stimulus-induced activation. In our present experiment, the increased agmatine labeling of vomeronasal neurons suggests that the pheromone stimulation effects are intensified by GnRH through the activation of an intracellular signaling pathway or by some other mechanism that may serve to increase the number of or prolong the opening of ion channels following stimulation by pheromone. The most likely place for this modulation of ion channel activity is in the region of the dendritic knob.
GnRH in the terminal nerve system has been suggested to act as a neuromodulator throughout the brain. 33 In dwarf gourami, Oka has shown that individual terminal nerve cell bodies located in the terminal nerve ganglia (located caudal to the olfactory bulb) send processes bilaterally to areas such as the olfactory bulb, preoptic area, dorsal thalamus, hypothalamus, midbrain tegmentum, optic tectum, medulla and even to the rostral spinal cord. Processes also project along the olfactory nerve fascicles to the olfactory mucosa. 34 Processes that enter the optic nerve presumably project to the retina as occurs in numerous other fish species. 35 It has been suggested that GnRH acts as a neuromodulator not only in the retina but also in other visual areas of the brain or areas involved with reproduction. 36 In the optic tectum, GnRH has been shown to modulate incoming signals from the optic nerve, and yet produce no neural activity by itself. 34 Recently, GnRH has been shown to enhance olfactory activation of a specific reproduction-related region of the medial amygdala. 37 Thus GnRH appears to modulate a number of both peripheral and central neural systems. The benefit of having a widely distributed neuromodulatory GnRH system is to coordinate the functioning of systems, including sensory systems, motor systems, and neuroendocrine systems, during reproductive activities. In addition, GnRH may help to reduce the intensity of nonreproductive stimuli that would be distracting. Further studies on the chemosensory system and other sensory or CNS systems are necessary to finely elucidate the molecular mechanisms by which GnRH can modulate the transmission of neural signals during reproductive activity.
Conclusions
We have shown that GnRH is a potent modulator of pheromone signaling at the level of the primary chemosensory neuron. In female P. shermani, GnRH appears to increase the pheromonal signal as evidenced by the increased uptake of agmatine. We hypothesize that GnRH performs this modulation through molecularly altering the functioning of ion channels involved in the pheromonal response. Because GnRH is undoubtedly released at multiple levels of the reproductive neural networks, it has the potential for coordinating changes to many sensory and motor systems at reproductively appropriate times.
Funding
Funding was supported in part by National Science Foundation grants IOS-0416724 (LDH), IOS-0818554 (LDH), IOS-0416834 (RCF), and IOS-0818649 (RCF).
Authors Contributions
LDH supplied reproductively-screened female salamanders, RCF and PWF isolated and purified the male pheromone, CRWW and RD conducted the animal stimulation paradigms and tissue fixation, CRWW, JC and RD conducted tissue sectioning, immunocytochemistry and digital imaging, JC conducted densitometry analysis, RD, CRWW and CEA conducted statistical procedures, CRWW wrote the manuscript and created the figures, all authors provided additions and editing of the manuscript.
Author Information
Justin Colvard was a senior high school student at the Oklahoma School of Science and Mathematics while he participated in this research study. Radhika Dighe was a Research Assistant I when she participated in this research study. Lynne Houck, Richard Feldhoff, Pamela Feldhoff, and Celeste Wirsig-Wiechmann have collaborated since the year 2000 on studies involving P. shermani pheromone stimulation of the chemosensory system. Christopher Aston is an Associate Professor of Pediatrics who provides statistical expertise in the design and analysis of research projects at the University of Oklahoma Health Sciences Center.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
Footnotes
Acknowledgements
We thank Kathleen Bowen for expert technical assistance.
