Abstract
In humans, hippocampal damage typically produces temporally graded retrograde amnesia, with relative sparing of remote memories compared to recent memories. This observation led to the idea that as memories age, they are reorganized in a time-dependent manner. Here, we evaluate evidence for time-dependent memory reorganization in animal models. We conclude that, although hippocampal lesions may not always produce temporal gradients under all conditions, studies using alternate experimental approaches consistently support the idea that memories reorganize over time—becoming less dependent on the hippocampus and more dependent on a cortical network. We further speculate on the processes that drive memory reorganization such as sleep, memory reactivation, synaptic plasticity, and neurogenesis.
Since at least the late 19th century, it has been recognized that “the dissolution of memory is inversely related to the recency of the event,” 1 or, in other words, that new memories are more vulnerable to brain insults than old memories. In the mid-20th century, the treatment of intractable seizures or psychosis via surgical resection of the brain unexpectedly revealed the neuroanatomical locus of this effect. In the most famous case, 2 Henry Molaison (patient H.M.) underwent resection of a large, bilateral portion of the medial temporal lobe including the hippocampus. After surgery, he exhibited partial retrograde amnesia, with memories formed shortly before the surgery forgotten but memories formed earlier in life remembered. 3 Over the last several decades, numerous individuals who sustained damage to the hippocampus exhibited similar patterns of retrograde amnesia, with recent memories lost but remote memories left relatively intact. 4 7
These cases of temporally graded retrograde amnesia led to the idea that the hippocampus plays a time-limited role in the storage and retrieval of memory.
8
11
In particular, two major theories of systems-level memory consolidation have emerged (Fig. 1A, B). The first theory—known as standard consolidation theory—proposes that the hippocampus is initially required for memory expression but that, over time, memory expression becomes dependent on a cortical network.12,13 The second theory— known as multiple trace theory—proposes that the brain regions that support memory expression differ depending on the type of memory; semantic or context-free memories depend less on the hippocampus and more on extrahippocampal structures as they age, whereas episodic or contextually-rich memories always depend on the hippocampus.14,15 Although there are some key differences between these two theories, both agree that memory can undergo time-dependent reorganization across brain regions.
Two theories of time-dependent memory reorganization. 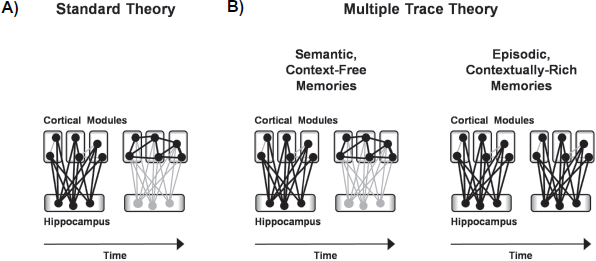
While clinical cases of retrograde amnesia in humans form the basis of the idea that memory reor-ganizes in a time-dependent manner, they involve relatively few patients and there is no control over the extent and location of brain damage as well as the age and nature of memories that may be affected. Experimental studies using animals, therefore, have made a significant contribution to our understanding of how memory is reorganized over time. Here, we review evidence for time-dependent memory reorganization in experimental animals and speculate on the processes that might drive this reorganization.
Temporal Gradients after Hippocampal Damage: Sometimes There, Sometimes Not
In one of the first studies to model temporally graded retrograde amnesia in animals, Winocur and colleagues 16 made electrolytic lesions of the dorsal hippocampus in rats at varying time points after training in a socially-transmitted food preference task. They found that lesions made 1–2 days after training abolished memory for the learned food preference, but memory was spared when lesions were made 10 days after training. This temporal gradient of retrograde amnesia supports the idea that memories become independent of the hippocampus as they age. Subsequent studies employing hippocampal lesions or disruption of hippocampal function replicated and extended this basic effect to encompass not only socially-transmitted food preference, 17 19 but also contextual fear conditioning, 20 27 trace fear conditioning, 27 spatial discrimination, 28 32 visual discrimination, 33 trace eyeblink conditioning, 34 36 object discrimination, 37 inhibitory avoidance,38,39 and some variations of the water maze.25,40–43
Not all studies, however, report evidence for a temporal gradient of retrograde amnesia. Instead, many found that hippocampal lesions or disruption of hippocampal function abolished both recent and remote memory. 44 64 Although the conditions that lead to the presence—or absence—of a temporal gradient of retrograde amnesia following hippocampal damage are not yet well defined, a number of potential factors have been proposed.
Extent of training
An emerging theme from recent studies is that the extent of training may influence whether remote memories are spared following hippocampal damage. That is, extensive training protocols may produce memories that survive hippocampal damage, whereas minimal training protocols may result in memories that are vulnerable to hippocampal damage.
In one study, Winocur and colleagues 65 reared rats in a complex environment, or “village”, for 3 months. After hippocampal lesions, rats retained accurate memory for the spatial layout of the environment. The authors speculated that the extensive pre-operative experience in the spatial environment allowed the formation of a schematic representation of the environment that did not require the hippocampus. Because the timing of the surgery relative to training was not manipulated in this study, whether memory for the spatial layout was initially dependent on the hippocampus and later dependent on extrahippocampal regions could not be determined. Rather, it may be the case that extensive experience or intense training enabled extrahippocampal regions to encode memories separately from the hippocampus. 66
Further insight into these issues comes from a study by Morris and colleagues, 67 who trained rats over several weeks to associate particular flavors of food to specific locations in a complex spatial environment. After this lengthy training period, rats were able to acquire novel flavor-place associations in just one trial, suggesting that extensive experience with the task led to the formation of an associative schema that allowed for the rapid encoding of new information. After hippocampal lesions, rats retained memory for the well-learned flavor-place associations, indicating that the schema was stored in extrahippocampal areas. As for the flavor-place associations acquired over a single trial, whether they survived hippocampal damage depended on their age. Associations that were 3 hours old were abolished by hippocampal lesions, but associations that were 48 hours old were left intact. Because this temporal gradient of retrograde amnesia was significantly steeper than that typically observed after hippocampal damage (e.g. several days to a few months), this suggests that the creation of an extrahippocampal schema increases the rate at which memory becomes independent of the hippocampus. Furthermore, when hippocampal lesions were made before training, rats failed to learn the flavor-place associations, indicating that extrahippocampal structures were not capable of independently encoding the spatial memories, even after extensive training.
Context-dependent versus context-free memory
In contrast to standard consolidation theory,12,13 which predicts that all remote memories should survive hippocampal damage, multiple trace theory14,15 predicts that whether remote memories survive hippocampal damage depends on whether they are context-dependent or context-free. Remote memories that remain entangled with the context in which they were encoded should remain permanently dependent on the hippocampus and, thus, would not survive hippocampal damage. In contrast, remote memories that have become unraveled from their original context should become independent of the hippocampus and, thus, would be spared after hippocampal damage.
Consistent with multiple trace theory, studies show that fear 68 70 and food preference 68 memories, which are initially dependent on the particular context in which they were encoded, lose contextual specificity as they age. As these memories lose contextual specificity, they may become independent of the hippocampus. This might explain why remote fear 20 26 food preference16,17,19,71 memories typically survive damage to hippocampus. In contrast, remote memories that remain tied to a particular context, such as water maze memory,44–46,50,52–57,60 typically do not survive hippocampal damage.
Several studies, however, do not support the predictions of multiple trace theory. For instance, remote memories that are relatively context-free do not always survive hippocampal damage. Ungraded retrograde amnesia has been found for contextual fear memory,48,51,59 which is expected to lose contextual specificity over time, 68 70 and object47,49,60 and picture 61 memories, which need not be associated with a particular context at any time point. Furthermore, attempts to reduce the spatial complexity or the need for spatial navigation in the water maze, which should conceivably allow memory to become independent of the hippocampus, failed to reveal a temporal gradient of retrograde amnesia after hippocampal lesions.55,56
Another problem for multiple trace theory is that remote memories that remain tied to the context in which they are encoded are sometimes preserved after hippocampal damage. In a study by Wang et al
72
mice were trained in a 3-day context fear discrimination protocol in which one context was always paired with shock and a second context was never paired with shock. Different from standard fear conditioning protocols, this context fear discrimination protocol produced precise, context-specific fear memory lasting several weeks (Fig. 2A–C). Although extensive hippocampal lesions made 1 day after training abolished discrimination between contexts, discrimination was spared when lesions were made 42 days after training (Fig. 2D). Thus, the hippocampus may not be permanently necessary for the expression of precise, context-specific memory.
Precise, context-specific remote memories do not require the hippocampus. 
Extent of lesion
Another factor that may influence whether remote memories are lost after hippocampal damage is the size of the lesion. Unlike standard consolidation theory,12,13 which predicts that all remote memories will survive hippocampal damage regardless of lesion size, multiple trace theory14,15 predicts that context-dependent memories may or may not survive hippocampal damage depending on the size of the lesion. Specifically, multiple trace theory predicts that as context-dependent memories age, they become supported by a greater number and wider distribution of hippocampal-cortical traces. Because the hippocampal-cortical traces supporting recent memory are fewer in number and sparser in distribution compared to remote memory, partial hippocampal lesions are more likely to disrupt recent compared to remote memory and thus result in a temporal gradient of retrograde amnesia. Extensive hippocampal lesions, on the other hand, are likely to disrupt both recent and remote memory and thus result in ungraded retrograde amnesia. To some degree, experimental findings support the prediction of multiple trace theory, as many studies reporting temporal gradients employed partial hippocampal lesions16,20–24,30,31,34,35,38,39 and many studies reporting ungraded amnesia employed extensive hippocampal lesions.44,47,49,54,58,60,61 However, it is sometimes found that remote context-dependent memory is spared after extensive lesions,17–19,72 which is problematic for multiple trace theory. Conversely, it is sometimes found that remote context-dependent memory is disrupted by partial lesions,45,46,48,51,53,55,57,62 which is problematic for multiple trace theory as well as standard consolidation theory.
Memory involving a taste or odor component
A fourth factor that may determine the fate of remote memory following hippocampal damage is whether it involves a significant taste or odor component. One type of remote memory that is consistently found to survive hippocampal damage is socially-transmitted food preference memory.16,17,71,73 Furthermore, it has been argued 51 that when remote contextual fear memory is found to survive hippocampal damage, it is often in studies that incorporated a prominent odor (e.g. acetic acid, ethanol) into the training context.20–22,24 Thus, it may be possible that memory involving taste or odor becomes completely independent of the hippocampus over time, whereas other types of memory tend to engage the hippocampus for as long as they exist.
Alternate Approaches: Consistent Evident for Time-Dependent Memory Reorganization
Although hippocampal damage or disruption of hippocampal function does not always result in a temporal gradient of retrograde amnesia, studies utilizing alternate experimental approaches offer nearly unanimous support for the idea that memories reorganize over time.
One such approach is to map region-wide changes in brain activation corresponding to the expression of recent and remote memories. In the first study of this kind, performed by Bontempi and colleagues,
74
mice were trained to find food rewards in an eight-arm radial maze and given a retention test either 5 or 25 days later. Retrieval of memory for the food locations 5 days after training corresponded with increased metabolic activity in the hippocampus. In contrast, retrieval of memory 25 days after training corresponded with increased metabolic activity in the anterior cingulate, medial prefrontal, and temporal cortices. This suggests that, over time, initially-hippocampal dependent memory becomes dependent on a widespread cortical network. Studies examining changes in immediate early gene (e.g.
A strength of these brain-mapping studies is that they allow for the observation of whether the hippocampus is engaged during retrieval of remote memory, which is not possible in hippocampal lesion studies. From the studies performed so far, it appears that whether the hippocampus is activated by recall of remote memory depends on the type of memory being recalled. Specifically, retrieval of remote socially-transmitted food preference memory did not activate the hippocampus.19,76 In contrast, the hippocampus was activated by retrieval of remote water maze memory.57,77 Notably, however, the pattern of activation across hippocampal subregions was more sparse and of lesser magnitude during retrieval of remote compared to recent water maze memory, 77 suggesting that time-dependent reorganization of this type of memory can occur within the hippocampus itself. For spatial discrimination30,74 and contextual fear 75 memory, levels of hippocampal activation were lower than control conditions, suggesting that for these types of memories, the hippocampus is not just passively disengaged during recall of remote memory but is actively inhibited by other brain regions.
Despite its strengths, there are some potential disadvantages of the brain-mapping approach. One is that the choice of an appropriate control condition is difficult. For some types of tasks, such as contextual fear conditioning, there is relative consensus on an appropriate control group (e.g. no-shock or immediate-shock). The situation, however, is not as clear for tasks such as the water maze, for which highly conservative control groups (e.g. swim or visible platform) could lead to a high false-negative rate, but less conservative control groups (e.g. home cage) could produce a high false-positive rate. A second issue is that immediate early gene expression is only an indirect marker of brain activation. Although the expression of these genes is tightly correlated with neuronal activity, 78 differences in endogenous activity patterns across brain regions may limit their utility in brain-wide studies. A third issue is that reorganization may not necessarily result in an overall increase in activity in a given region. For example, there was no overall increase in activation of the parietal cortex after recall of remote, compared to recent, spatial discrimination memory, but a closer examination showed that activation shifted from deep to more superficial cortical layers over time. 30 This example highlights the complexity of defining reorganization and raises the question of what level of anatomical resolution is appropriate, or even feasible, for this type of analysis.
Another alternate approach to studying time-dependent reorganization of memory is to target specific extrahippocampal regions that are implicated in the recall of remote memory. If certain extrahippocampal regions gradually assume a critical role in memory, then an increase in task-relevant neural firing or synaptic transmission should be observable within those regions as memories age. Takehara-Nishiuchi and McNaughton 79 trained rats in a trace eyeblink conditioning protocol and recorded neural firing in the medial prefrontal cortex. Across several weeks of tone-shock pairings, neural firing became selective for the learned associations, evidenced by increased firing during the delay between the tone and the eyelid shock. This suggests that as trace eyeblink memories age, they gradually engage the medial prefrontal cortex. In another study, Hugues and Garcia 80 trained rats in a contextual fear extinction protocol and recorded field potentials at two different inputs to the medial prefrontal cortex. As the extinction memory aged, there was a depotentiation of field potentials at ventral hippocampal-medial prefrontal synapses and a potentiation of field potentials at mediodorsal thalamic-medial prefrontal synapses. This suggests that, over time, processing of the extinction memory was relinquished by hippocampal inputs and overtaken by other inputs to the medial prefrontal cortex.
Likewise, if certain extrahippocampal regions play an increasingly critical role in memory over time, then damage to those regions should disrupt remote memory to a greater extent than recent memory. Indeed, inactivation of the anterior cingulate cortex blocked expression of remote, but not recent, memory for contextual fear, 75 spatial discrimination, 30 water maze, 57 and conditioned taste aversion. 81 With one exception, 82 inactivation or lesions of the medial prefrontal cortex resulted in greater disruption of remote memory than recent memory for spatial discrimination, 30 trace eyeblink conditioning, 34 and contextual and trace fear conditioning. 27 Finally, inactivation of the parietal cortex blocked the expression of remote, but not recent, inhibitory avoidance memory.38,39 These studies provide direct evidence that certain cortical areas are necessary for the recall of remote, but not recent, memory, which provides support for the idea that memories gradually come to rely on cortical regions for storage and retrieval.
Although the selective involvement of cortical areas in the expression of remote memory may reflect time-dependent memory reorganization, an alternative interpretation is that cortical areas are situationally recruited for the effortful retrieval of memories that are weak due to their age. 83 As memories tend to become weaker with time, distinguishing between these two interpretations requires memory strength to be uncoupled from memory age. In a study that addressed this issue, Ding et al. 81 used a taste aversion protocol in which mice were treated with a high or low dose of lithium chloride after drinking saccharin-flavored water to create strong or weak taste aversion memory, respectively. They found that inactivation of the anterior cingulate cortex disrupted retrieval of remote taste aversion memories regardless of their strength. This is contrary to the prediction of the effortful retrieval hypothesis—that cortical damage should disrupt retrieval of weak memories regardless of their age.
Mechanisms of Time-Dependent Memory Reorganization
It is believed that the time-dependent reorganization of memory is not passive, but rather actively driven by memory reactivation.9,11 Successive reactivations of memory could lead to the reinstatement of memory-relevant patterns of hippocampal and cortical activity and the stabilization of cortical traces, thereby propelling memories to become independent of the hippocampus.
One situation in which memory reactivation may promote memory reorganization is during sleep. Evidence that memory is reactivated during sleep comes from studies showing that the sequence in which neurons fired when rats explored a spatial environment was replayed during subsequent slow-wave sleep.84,85 Because replay of waking experience was coordinated between the hippocampus and the cortex,
86
88
this suggests a possible dialogue between the two brain areas during slow-wave sleep that contributes to memory reorganization. While these studies indicate that slow-wave sleep is important for memory reorganization, other studies point toward an important role for rapid eye movement (REM) sleep. In a series of studies, Ribeiro et al. examined the expression of
Another situation in which memory reactivation may promote memory reorganization is during the explicit retrieval of memories. For instance, observations that extensive training can produce contextually-rich memories that survive hippocampal lesions65,67,72 might reflect reorganization driven by the repeated retrieval of task-relevant information inherent in extensive training protocols. Furthermore, even after memories have become independent of the hippocampus, retrieval of memory may initiate additional rounds of reorganization. Debiec and colleagues 23 found that remote contextual fear memory, which was independent of the hippocampus, briefly returned to a hippocampal-dependent state after rats were reminded of the training context. This re-engagement of the hippocampus during memory retrieval may serve to further refine or to incorporate new information into existing cortical memory traces.
Regardless of the exact situations during which time-dependent memory reorganization occurs, it most certainly involves plasticity both within and between hippocampal and cortical regions. Studies utilizing transgenic lines of mice have implicated several plasticity-related molecules in the establishment of remote memory, including α-calcium/calmodulin kinase II,75,95,96 p21-activated kinase, 97 type 1 adenylyl cyclase, 98 L-type voltage-gated calcium channels, 99 integrin β2, 100 and steryl-O-acyl transferase 1. 100 For example, mice that were heterozygous for a null mutation of α-calcium/calmodulin kinase II (αCamKII) showed normal acquisition and retention of contextual fear and water maze memory over short delays (1–3 days) but pronounced forgetting at longer retention delays (10–50 days). 75 Because this mutation causes a deficit in cortical LTP, it may be the case that αCamKII-mediated synaptic plasticity in the cortex is essential for the establishment of remote memory. Although some studies indicate that the maintenance of remote memory requires ongoing synaptic plasticity for up to several months after training,101,102 others suggest a time-limited role for synaptic plasticity.25,26,103 In particular, survival of remote memories required activation of N-methyl-D-aspartate (NMDA) receptors in the hippocampus 25 and medial prefrontal cortex 103 only during the first 2 weeks after training but not thereafter. Likewise, survival of remote memories required normal αCamKII activity in the forebrain—including the hippocampus and cortex—only during the first week after training. 26 These studies suggest that hippocampal and cortical synaptic plasticity is critical for the transition of memories from a temporary, hippocampal-dependent state to a stable, cortex-dependent state.
Summary and Future Directions
A relatively conservative reading of the studies employing inactivation or damage of the hippocampus indicates that temporal gradients of retrograde amnesia exist. Certain conditions, however, may prolong the period during which memories depend on the hippocampus, resulting in ungraded amnesia at the time points at which memory is probed. The inconsistency with which temporal gradients are found suggests the interplay of a number of influential factors (e.g. training intensity, extent of lesion, qualitative aspects of the target memory) that, at the present moment, are poorly understood. Thus, more systematic examination of how these factors determine the temporal pattern of retrograde amnesia after hippocampal damage is needed.
In contrast to studies employing hippocampal damage, studies using alternate approaches have provided nearly unanimous support for the idea that as memories age, they become less dependent on the hippocampus and more dependent on a broad network of cortical regions. Our current picture of how remote memories are organized, however, is still incomplete. For example, the analysis of six cortical regions in the Frankland et al. brain-mapping study 75 represents only 2%–3% of total forebrain volume. Brain-wide examinations are necessary to comprehensively map the networks supporting remote memories and to understand how different types of remote memories may be differentially organized.
Although evidence is accumulating that memory reactivation drives memory reorganization either in online (e.g. memory retrieval) or offline (e.g. sleep) situations, this evidence remains correlative. To provide a causal link between memory reactivation and reorganization, future studies would need to determine, for example, whether blocking memory replay during sleep prevents reorganization or whether repeated reminders of the learning event accelerates reorganization.
Finally, some possibilities—such as the role of neurogenesis in time-dependent memory reorganization—are just beginning to be explored. Tsien and colleagues 104 found that transgenic mice with deficient hippocampal neurogenesis exhibited longer retention of contextual fear memory. This led them to hypothesize that the addition of new neurons into the hippocampal network via neurogenesis may lead to instability and eventual erasure of the hippocampal memory trace, thereby making room for the acquisition and temporary storage of new memories. In line with this hypothesis, Inokuchi and colleagues 105 found that irradiation-induced reduction of hippocampal neurogenesis prolonged the hippocampal-dependency of contextual fear memories, suggesting that hippocampal neurogenesis may expedite and enhance the efficiency of time-dependent memory reorganization.
Disclosure
The authors report no conflicts of interest.
