Abstract
Familial dysalbuminemic hyperthyroxinemia (FDH) is a familial autosomal dominant disease caused by mutation in the albumin gene that produces a condition of euthyroid hyperthyroxinemia. In patients with FDH, serum-free thyroxine (FT4) and free triiodothyronine (FT3) concentrations as measured by several commercial methods are often falsely increased with normal thyrotropin (TSH). Therefore, several diagnostic steps are needed to differentiate TSH-secreting tumor or generalized resistance to thyroid hormone from FDH. We herein report a case of a Japanese man born in Aomori prefecture, with FDH caused by a mutant albumin gene (R218P). We found that a large number of FDH patients reported in Japan to date might have been born in Aomori prefecture and have shown the R218P mutation. In conclusion, FDH needs to be considered among the differential diagnoses in Japanese patients born in Aomori prefecture and showing normal TSH levels and elevated FT4 levels.
Keywords
Introduction
Familial dysalbuminemic hyperthyroxinemia (FDH) is a familial autosomal dominant disease characterized by increased binding of thyroxine (T4) to albumin. Serum albumin concentration is quantitatively normal, and the increased binding is due to the presence of an abnormal albumin molecule that shows increased affinity for T4. 1 Serum free thyroxine (FT4) and free triiodothyronine (FT3) concentrations are often falsely elevated and serum thyrotropin (TSH) concentration is normal, laboratory findings in keeping with syndrome of inappropriate secretion of TSH (SITSH), representing a confounding pathological condition. FDH is the most common hereditary disease causing euthyroid hyperthyroxinemia in Caucasians. 2 It was first described by Hennemann et al in 1979, 3 and the R218H point mutation in the albumin gene was first identified as a cause of this disease by Petersen et al in 1994. 4 Subsequently, other albumin gene mutations (R218P,5,6 R218S, 7 and R222I 8 ) were described as the causes of FDH. FDH is rarely encountered in Japan. Only one patient had FDH among 479 patients with SITSH in Ito Hospital, and resistance to thyroid hormone (RTH) is more widely known than FDH. 9 In Japan to date, only five families with FDH have been reported.5,6,10–12 However, the birthplaces of these patients with FDH have not been described. We herein report the case of a Japanese patient born in Aomori prefecture with FDH caused by the mutation R218P in the albumin gene. The patient has given consent for publication of this report. We also report for the first time that a large number of patients with FDH in Japan might have been born in Aomori prefecture.
Case Report
A man in his 40s born in Aomori prefecture, Japan, had been feeling mild malaise. He was first found to have thyroid dysfunction at another hospital 17 months before admission and was referred to our hospital for investigation of the cause of thyroid dysfunction. On admission, his body temperature was 36.8°C, blood pressure was 136/78 mmHg, and heart rate was 82 bpm. Physical examination revealed no exophthalmos, finger tremors, skin wetness, or goiter. After the diagnosis of FDH, the patient's mother was also found to have FDH.
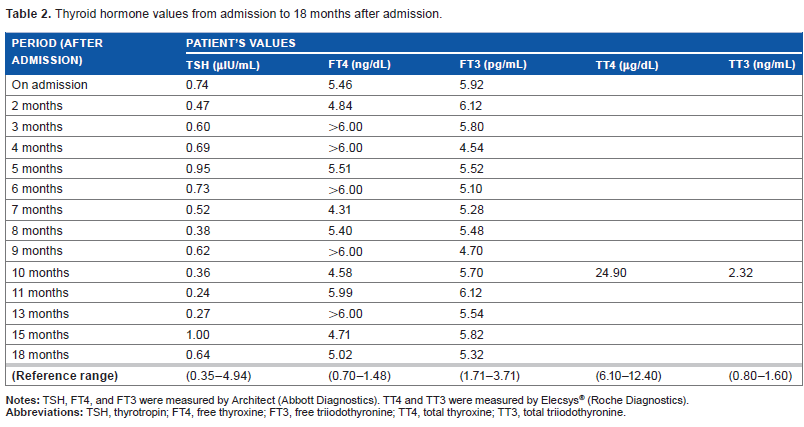
Serum TSH, FT4, FT3, total thyroxine (TT4), and total triiodothyronine (TT3) levels were measured using automated immunoassay systems (Architect from Abbott Diagnostics, Elecsys® from Roche Diagnostics, or Lumipulse® from Fujirebio). Laboratory data on admission revealed SITSH (Table 1). Negative results were obtained for TSH receptor antibody, thyroid-stimulating antibody, antithyroglobulin antibody, and antithyroid peroxidase antibody. The three-hour thyroid I-123 uptake value was 6.2%, which was within the normal range. Ultrasonography of the thyroid gland revealed no tumor, and blood flow and thyroid size were both normal. Magnetic resonance imaging of the pituitary gland revealed no tumor (Fig. 1). Based on these investigations, we considered that SITSH might be due to RTH, but ruled out TSH-secreting pituitary tumor. Then, a genomic DNA sample was obtained for clinical diagnosis, after receiving informed consent. However, genomic DNA sequencing of exons 4–10 of the thyroid hormone receptor β (TRβ) gene showed no mutation. Therefore, we investigated the possibility of albumin gene mutations. Direct genomic sequencing of exon 7 of the albumin gene revealed a substitution of cytosine (C) for guanine (G) in the second nucleotide of codon 218 in one of the two alleles, leading to the replacement of arginine by proline (Fig. 2). Therefore, we diagnosed FDH caused by R218P heterozygous point mutation.
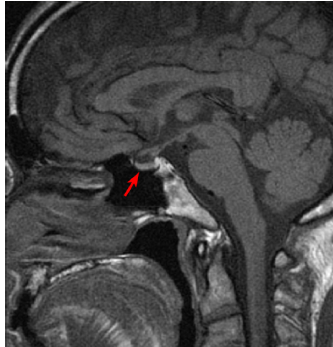
MRI of the pituitary shows no pituitary gland tumor. Red arrow indicates pituitary gland.
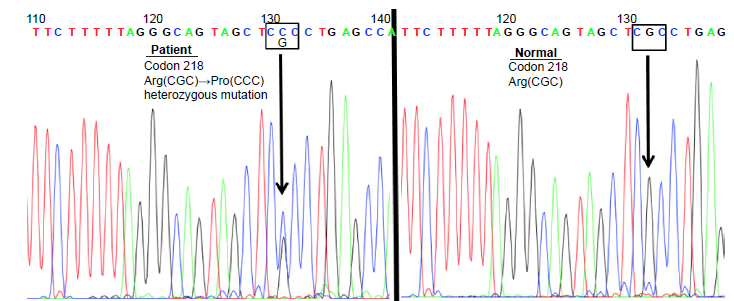
Genomic DNA sequence of exon 7 of the albumin gene in the patient (left) and a normal subject (right). The left panel shows substitution of cytosine for guanine in the second nucleotide of codon 218 for the albumin gene of the patient, resulting in the replacement of arginine (Arg) by proline (Pro).
Laboratory results related to the thyroid (on admission).
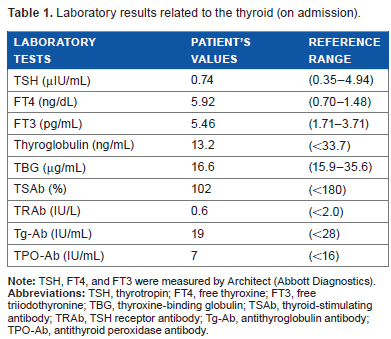
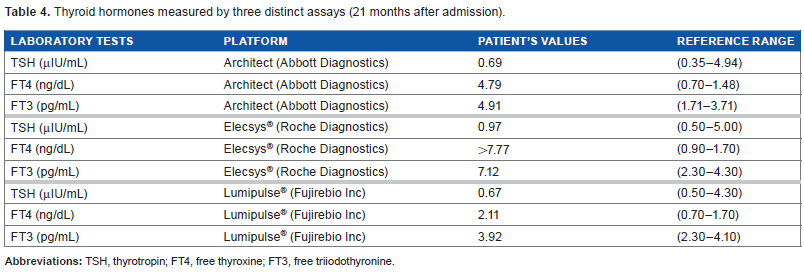
Since the diagnosis of FDH, the patient has been followed up without pharmacotherapy. The symptom of malaise disappeared spontaneously over time, but serum thyroid function tests remained unchanged. The symptom of malaise was therefore considered unrelated to thyroid hormone levels. Thyroid hormone data from admission to 18 months after admission are shown in Table 2. Serum TT4 concentration was markedly increased, but serum TT3 concentration was only slightly elevated. We evaluated the effect of a polyethylene glycol (PEG) precipitation test in the serum of FDH patients as previously reported. 13 The results of PEG precipitation testing in FDH patients and healthy controls were almost the same (Table 3). Therefore, the possible effect of anti-triiodothyronine antibody or anti-thyroxine antibody was denied, 13 but the PEG precipitation test was not able to reveal the exact value of FT4 in the serum of FDH patients. Thyroid hormone levels measured by Architect, Elecsys®, and Lumipulse® systems 21 months after admission are shown in Table 4. Thyroid hormone levels measured by Architect and Elecsys® showed similar results and suggested SITSH-like laboratory findings, but those measured by Lumipulse® showed almost normal ranges of FT4 and FT3 levels.
Thyroid hormone values from admission to 18 months after admission.
PEG precipitation test.
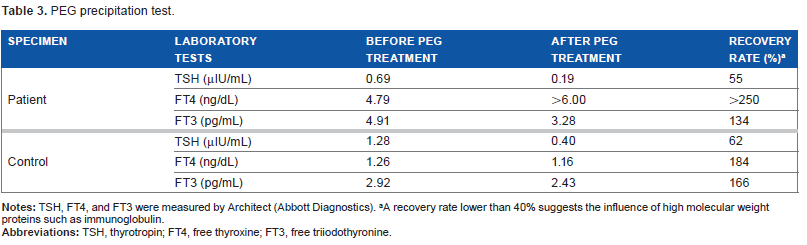
A recovery rate lower than 40% suggests the influence of high molecular weight proteins such as immunoglobulin.
Thyroid hormones measured by three distinct assays (21 months after admission).
Discussion
Thyroid hormone assays are prone to interference from a range of substances in serum, including heterophile antibodies and antithyroid hormone antibodies. Several commercial methods for measuring serum FT4 also yield high values caused by assay artifacts in patients with FDH. Serum TT4 concentration in this patient was markedly elevated and TT3 concentration was slightly elevated, characteristic of patients with FDH.5–7 In this case, thyroid hormone levels measured by Lumipulse® only showed almost normal ranges of FT4 and FT3 levels. This suggests that the Lumipulse® system may be able to measure thyroid hormone levels in patients with FDH accurately; further studies in more patients with FDH are needed. As described in the “Introduction” section, FDH is rarely encountered in Japan, and RTH is more widely known than FDH. Therefore, in patients with SITSH-like laboratory findings, the possibility of FDH must be included among the differential diagnoses. In this case report, the patient, with FDH caused by the R218P mutation in the albumin gene, was born in Aomori prefecture. Ozaki et al previously reported a case of a Japanese man in his 60s with FDH caused by the R218P albumin gene heterozygous mutation. 12 In that case, the patient had no symptoms and no abnormal physical findings, and his thyroid disorder was discovered coincidentally in association with the laboratory tests for the investigation of adrenal incidentaloma. His thyroid hormone levels measured by Elecsys® were as follows: FT3 = 9.29 pg/mL, FT4 > 7.77 ng/dL, and TSH = 0.7 µIU/mL. His R218P albumin gene heterozygous mutation was diagnosed by genomic sequencing and was the same as that of the case presented here. It should be noted that he had also been born in Aomori prefecture. 12 In addition, Yoshida et al previously reported a case of a Japanese patient with FDH caused by the R218P albumin gene heterozygous mutation, 6 and this patient had also been born in Aomori prefecture [Yoshida K, personal communications, July 2015]. Furthermore, Nakagawa et al recently discovered a case of a Japanese woman in her 30s with FDH caused by the R218P albumin gene heterozygous mutation [Nakagawa Y, personal communications, November 2015]. In that case, the patient also had no symptoms and no abnormal physical findings, and her thyroid disorder was discovered coincidentally in association with the laboratory tests for the investigation of the cause of infertility. Her thyroid hormone levels measured by Elecsys® were as follows: FT3 = 10.7 pg/mL, FT4 > 7.77 ng/dL, and TSH = 0.84 µIU/mL. Her R218P albumin gene heterozygous mutation was diagnosed by genomic sequencing and was also the same as that in our presented case. Surprisingly, she had also been born in Aomori prefecture. Therefore, at least four patients out of seven Japanese cases of FDH had been born in Aomori prefecture. The birthplaces of the other three cases could not be identified.
As described above, we have found that a large number of FDH patients reported in Japan to date might have been born in Aomori prefecture, and they have shown the R218P albumin gene mutation. In conclusion, this case suggests that FDH needs to be considered as a differential diagnosis for Japanese patients born in Aomori prefecture, showing normal TSH levels and elevated FT4 levels, and suspected of having RTH.
Author Contributions
Conceived and designed the experiments: YO and HF. Analyzed the data: YO, YH, YN, KY, HO, and HF. Wrote the first draft of the manuscript: YO and HF. Contributed to the writing of the manuscript: YH, YN, KY, and HO. Agreed with manuscript results and conclusions: YO, YH, YN, KY, HO, and HF. Jointly developed the structure and arguments for the paper: YO, YH, YN, KY, HO, and HF. Made critical revisions and approved the final version: YO, KY, and HF. All the authors reviewed and approved the final manuscript.
