Abstract
A number of tyrosine plus phenylalanine double auxotrophic mutants were isolated by N-methyl-N-nitro-N-nitrosoguanidine (MNNG) treatment of a locally isolated strain of Aureobacterium flavescens of which 11A39 and 11A17 were selected on the basis of their tryptophan production in a mineral salt medium over other isolated mutant strains. The mutational block in the aromatic amino acid biosynthetic pathway of the selected double auxotrophs were determined. By controlling pH of the production medium to near neutrality, the active growth period could be extended up to 72 h and more tryptophan was accumulated compared to pH unregulated culture where the active growth ceased after 48 h. Further improvement of the tryptophan production has been achieved by stepwise isolation of a mutant strain resistant to the tryptophan analogues p-fluorotryptophan (FT) and 5-methyl tryptophan (MT) from the 11A39. Demand for L-tryptophan as food additive and therapeutic agent is increasing day by day throughout the World, particularly in the underdeveloped and developing countries like India. Still to date India depends on other countries for L-tryptophan. The aim of this work is to develop a potent high yielding, feed back insensitive mutant strain and optimization of its medium pH for maximum production of tryptophan.
Keywords
Introduction
L-tryptophan is an aromatic amino acid that was first proved to be essential for animal nutrition. It is mainly used therapeutically especially as a component of solution for transfusion and to treat depression, schizophrenia, alcoholism and amnesia.1–4 Apart from these two antimicrobial compounds viz indolocidin and violacein are produced from tryptophan. 5 As most of the cereal grains are lacking in tryptophan, use of L-tryptophan as food additive is increasing steadily in countries like India where the staple food is either rice or wheat. 6 Tryptophan is also important for its key role in many biosynthetic pathways like Indole-3-acetic acid, Phenyl acetic acid etc.7–11 So, demand for L-tryptophan is ever increasing, particularly in those countries which are largely dependent on others for the supply of this essential amino acid.
Though microbial production is the only source for obtaining tryptophan for commercial use, reports of microbial over production of tryptophan is rare because of its complicated branched biosynthetic pathway controlled by effective regulatory mechanisms. L-tryptophan has traditionally been produced by an enzymatic method12,13 but the direct fermentation method using strains of Corynebacterium and E. coli has several advantages. It is well known that bacteria have stringent regulatory mechanisms controlling the over-production of these metabolites. The over production of amino acids occur only in strains with inherited anomalies in their metabolism or in organisms with relaxed regulatory mechanisms through induction of mutagenesis. The production of tryptophan has been reported using auxotrophic and analogue resistant mutants of Bacillus subtilis, 14 Corynebacterium sp., 15 Brevibacterium lactofermentum, 16 Corynebacterium glutamicum, 17 Citrobacter freundii, 18 E. coli, 19 Pseudomonas hydrogenothermophila, 20 Arthrobacter parafineus. 21
The present paper describes a fermentation processs for the L-tryptophan production by double auxotrophs of A. flavescens by controlling the pH in a shake flask culture. It also includes improvement of yield of L-tryptophan by isolation of FT plus MT resistant mutant from the selected double auxotroph (11A39). The aim of this work is to develop a potent high yielding, feed back insensitive mutant strain and optimization of its medium pH for maximum production of tryptophan.
Most of the amino acid producing soil isolates are generally auxotroph of biotin, which is a very costly item and main barrier on the way of amino acid production by microbial fermentation in commercially feasible cost. A. flavescens is a potent bacterial strain excreting L-tryptophan in growth medium without biotin. We selected A. flavescens for the following reasons:- a) ability of the organism to grow rapidly in mineral salt medium without any requirement of biotin, b) non-pathogenic nature and absence of toxic products, c) inability to degrade amino acid L-tryptophan, d) availability of methods for isolation of stable mutants. All data incorporated in this work are mean value of three replicates.
Material and Methods
Microorganisms, maintenance of culture and cultivation
The Aureobacterium flavescens, which required pyridoxine HCL for normal growth was isolated from soil sample of Burdwan and maintained on Alfoldi's agar slant. 22 Tyrosine plus phenylalanine double auxotrophic mutant were derived from the parent isolate by a two step mutagenic treatment with MNNG. 23 These mutants were grown on a rotary shaker at 30 °C in the same liquid medium supplemented with the required amount of tyrosine and phenylalanine. Bacterial cell growth was determined turbidimetrically in an EEL (UK) colorimeter.
Determination of minimum inhibitory concentration (MIC) of the analogue
The sensitivity of the auxotrophic mutants to the analogue was tested by plating cells of double auxotrophic mutant strain on an agar medium 23 with an optimum level of nutrient supplementation and with various concentration of the analogues. The plates were incubated at 30 °C for 96 h. Bacterial growth was detected visually to find out the MIC level of the analogue.
Isolation of the analogue resistant mutant
Freshly grown cells of the mutant strains were suspended in 0.05 M Tris Maleate™ buffer (pH 6.0) at the cell concentration of 10 6 –10 7 cells ml–1 and treated with MNNG solution at 500 μg ml–1 final concentration for 60 minutes at 37 °C. The cells were washed twice with sterile TM buffer and then were spread on the surface of the agar medium containing the varying concentration of analogue higher than the MIC. The plates were incubated at 30 °C for 96–120 h, and the colonies that appeared were harvested and checked again for the analogue resistance.
Detection of L-tryptophan
Quantitative estimation of L-tryptophan was carried out spectrophotometrically following Hassan 24 as well as by microbiological assay using the L-tryptophan auxotroph. Residual sugar in the culture filtrate was estimated by the dinitrosalicylic acid method. 25
Results
Detection of the site of the mutational block in the tyrosine and phenylalanine biosynthesis pathway of the double auxotrophs of A. flavescens
For detection of actual point of mutational block in the tyrosine and phenylalanine biosynthetic pathway, both the liquid and agar medium of Alfoldi's were used. Eleven different combinations (Table 1) of aromatic amino acids and their precursors were supplemented to the medium. All the supplemented media were inoculated separately with the suspension of freshly grown culture of double auxotrophs (11A39 11A17) and incubated at 30 °C for 72 h. The result (Table 1) showed that two double auxotrophs could grow in medium with tyrosine plus phenylalanine and tyrosine plus phenylpyruvate. But the double auxotroph 11A39, could grow in the medium supplemented with prephenic acid plus phenyl pyruvate, phenyl pyruvate plus hydroxyphenyl pyruvate and phenylalanine plus hydroxyphenylpyruvate. From the result it was clear that mutational metabolic blockage took place in case of 11A39 in between prephenic acid to phenyl pyruvate and chorismic acid to prephenic acid. In case of double auxotroph 11A17 mutational block in all probability lies in between hydroxyphenylpyruvate to tyrosine and prephenic acid to phenylpyruvate (Fig. 1).
Detection of the site of mutational block on the tyrosine and phenylalanine biosynthetic pathway.


Site of mutational metabolic block (//) of 11A39 (a) and 11A17 (b).
Effect of controlling the pH on the growth and production of tryptophan
The tyrosine plus phenylalanine auxotrophs were grown in an Alfoldi's medium supplemented with pyridoxine HCl and required amino acids. The culture medium turned acidic with growth that might be one of the causes of the early cessation of the active growth. It was expected that there might be more cell growth and more tryptophan accumulation if the pH of the growth medium was maintained in the range near neutrality because preliminary experiments revealed that maximum tryptophan production takes place at pH of the cell medium to near neutrality. Two parallel sets of shake cultures were used. In one set of flasks the pH was kept unregulated. In other set the medium contained 0.1% CaCO3 to neutralize the anions set free by the bacteria during growth. For visualizing the shift of pH, 0.2 ml of 0.5% ethanolic solution of bromothymol blue was added to the medium as an indicator and 1 (N) NaOH was added at interval when required (Fig. 2). The result indicated that the regulation of the pH of the medium near neutrality lead to a longer growth period (72 h) resulting in a higher population density and accumulation of more tryptophan in the medium whereas in unregulated pH growth period lasted for about 48 h. As a result of this extended growth period of pH regulated medium, all the glucose was nearly consumed and tryptophan production was also significantly higher than flasks with unregulated pH (Fig. 2)
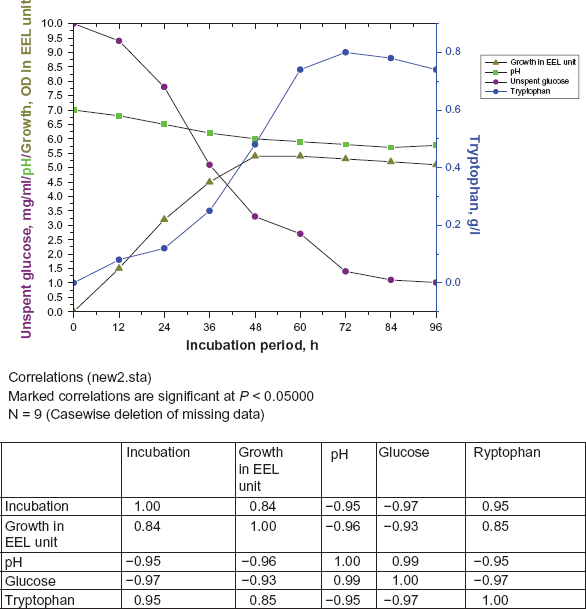
Effect of maintaining the pH of the medium near neutrality on growth and tryptophan production of the selected strain (pH unregulated).
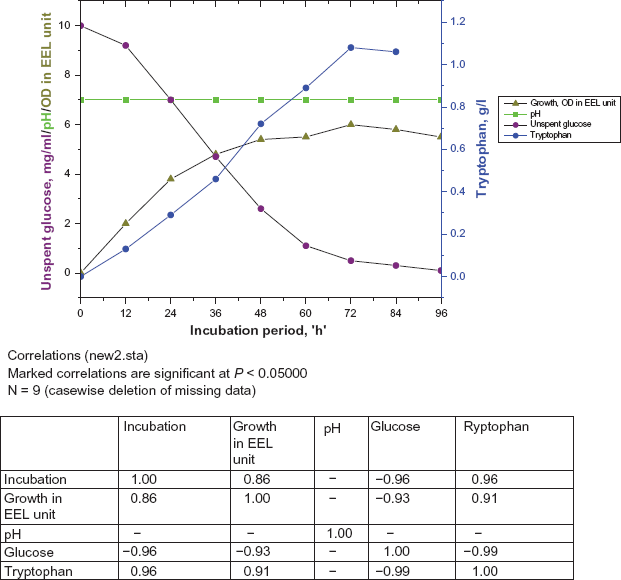
Effect of maintaining the pH of the medium near neutrality on growth and tryptophan production of the selected strain (pH regulated).
Isolation of p-fluorotryptophan (FT) resistant mutant
Preliminary experiment (data not shown) revealed that molasses (N2 content 0.02 mg/g) and NH4Cl were the best carbon and nitrogen sources with optimum at 7% and 60 mM respectively. After optimization of the culture conditions of the double auxotroph the yield of tryptophan was 2.6 g/l. Further improvement of yield of the L-tryptophan has been done by the isolation of mutant resistant to FT and MT, the structural analogues of the tryptophan. For obtaining analogue resistant mutant, prior to mutagenesis the sensitivity of the double auxotroph to these analogues were tested (Table 2). It was observed that the double auxotroph did not grow on the plate containing FT at a concentration of 4 mM or higher. So MIC of FT appear to be 4 mM for these auxotrophs. FT resistant mutants were isolated from the best tryptophan producing double auxotrophic mutant 11A39. It was treated with MNNG and plated on an agar medium containing 20 mM of FT. Twelve FT resistant mutants were isolated and tested for tryptophan production. Amongst these some mutants showed increased tryptophan yield over the parent double auxotroph (Table 3). The resistant mutant 11A39FTrM2 showed highest yield of L-tryptophan.
Determination of the minimum inhibitory concentration (MIC) of FT and MT on the growth of the mutant strains.

L-tryptophan accumulation by FT resistant mutant strains.

Isolation of 5-methyl-tryptophan (MT) resistant mutant
Before mutagenic treatment the MIC of 5-MT of the best FT resistant mutant (11A39FTrM2) was determined as described earlier. The MIC of 5-MT for the 11A39FTrM2 was found to be 10 mM (Table 2). In order to isolate MT resistant mutants from the 11A39FTrM2, a cell suspension was treated with MNNG as described earlier and plated on an agar medium containing 30 mM of 5-MT plus 20 Mm of FT. Six mutants were isolated and the tryptophan production was tested. Growth and tryptophan production of the different auxotrophs are presented in Table 4. All the double auxotrophic analogue resistant mutants showed an increasing tryptophan yield in comparison to 11A39FTrM2. Among these six strain 1139FTrMTrD1 was the best and yielded 6.7 g/l of tryptophan.
L-tryptophan accumulation by double auxotrophic FT plus MT resistant mutant strains.

Discussion
Production of tryptophan in substantial amount by micro-organisms is restricted due to the complicated and branched pathway with stringent regulatory mechanism. For overcoming this stringent regulation it is necessary to obtain double auxotrophic analogue resistant mutants with relaxed regulatory control where a metabolite in question accumulated in large quantities preventing an excess drainage of metabolite to other branch of the same pathway. With this idea in view, tyrosine plus phenylalanine double auxotrophs were isolated from the parent A. flavescens by the MNNG treatment. It was observed that in the mutant 11A39 blockage lies in between chorismic acid and prephenic acid which permits maximum drainage of metabolite towards tryptophan biosynthesis. Similar production of L-tryptophan by auxotrophic mutants has been reported in C. glutamicum, 17 Brevibacterium lactofermentum, 16 E. coli 26 and Candida utilis. 21
It was observed that during the progress of fermentation the pH of the medium gradually become acidic and after 72 h it became as low as 5.6. By regulating the pH of the medium by periodic neutralization, it was observed that the active growth period could be extended up to 68–72 h, where as in unregulated culture this lasted up to 48 h (Fig. 2). This seems to indicate clearly that the shift of pH of the medium towards acidity may be one of the main causes for early cessation of growth. Due to prolonged growth period tryptophan accumulation also proportionally increased. At the favourable pH with an increased cell growth tryptophan accumulation was increased by 35% over control, where pH was unregulated. Similar increase in tryptophan yield by maintaining pH of the fermentation medium near neutrality have been reported with Hansenula anomala, 27 Corynebacterium glutamicum KY9456, 16 Brevibacterium Sp., 28 Bacillus subtilis 29 and Corynebacterium glutamicum K 81. 28
Analogues function as a competitive inhibitor for the allosteric site like a true end product. Analogues, particularly of amino acids, effectively function as true feed back inhibitors without participating in other useful functions in the cells. Mutants resistant to analogue have altered and deregulated enzyme and such mutants in absence of the analogues synthesize the metabolite in excess which is eventually excreted in the medium. With a view to improve the yield of tryptophan by the selected double auxotrophic mutant, attempts were made to isolate mutant resistant to the analogue of tryptophan viz. p-fluorotryptophan (FT) and 5-methyl-trytophan (MT). Of the two tryptophan analogues tested, FT was more potent as compared to MT (Table 3). By mutagenesis of the best double auxotroph (11A39) with MNNG and screening on agar plates containing tryptophan analogue (FT, MT) a number of analogue resistant mutants were isolated and among them FTrM2 produces highest amount (6.5 g/l) of tryptophan (Table 3). Further mutagenesis of the best mutant FTM2 with MNNG following screening on agar plates containing 5-MT (25 mM), a number of mutants resistant to both FT plus MT were isolated. Among these double analogue resistant mutants, FTMTrD1 was the best for tryptophan production and produced 6.7 g/l tryptophan under optimal cultural condition in shake flask culture (Table 4). Thus by selecting double analogue resistant strain about 56% increase in the yield of tryptophan over the double auxotrophic strain (11A39) has been achieved. Similar improvements of yield of tryptophan by analogue resistant mutant have been reported in E. coli, 30 B. subtilis 29 C. glutamicum,15,16,18 P. hydrogenothermophila, 20 H. henrichii, 27 C. utilis 23 and P. uncinatum. 31
Considering the above mentioned fact we can say that the isolated double auxotrophic analogue resistant mutant strains of Aureobacterium flavescens has potency to be used for commercial production of L-tryptophan. However, scale up of flask culture conditions in various laboratory fermenters are required before considering the strain finally for commercial venture.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgement
The authors are thankful to the UGC for a fellowship to A. Roy.
