Abstract
Major species differences in tryptophan (Trp) metabolism and disposition exist with important physiological, functional and toxicity implications. Unlike mammalian and other species in which plasma Trp exists largely bound to albumin, teleosts and other aquatic species possess little or no albumin, such that Trp entry into their tissues is not hampered, neither is that of environmental chemicals and toxins, hence the need for strict measures to safeguard their aquatic environments. In species sensitive to toxicity of excess Trp, hepatic Trp 2,3-dioxygenase (TDO) lacks the free apoenzyme and its glucocorticoid induction mechanism. These species, which are largely herbivorous, however, dispose of Trp more rapidly and their TDO is activated by smaller doses of Trp than Trp-tolerant species. In general, sensitive species may possess a higher indoleamine 2,3-dioxygenase (IDO) activity which equips them to resist immune insults up to a point. Of the enzymes of the kynurenine pathway beyond TDO and IDO, 2-amino-3-carboxymuconic acid-6-semialdehyde decarboxylase (ACMSD) determines the extent of progress of the pathway towards NAD+ synthesis and its activity varies across species, with the domestic cat (Felis catus) being the leading species possessing the highest activity, hence its inability to utilise Trp for NAD+ synthesis. The paucity of current knowledge of Trp metabolism and disposition in wild carnivores, invertebrates and many other animal species described here underscores the need for further studies of the physiology of these species and its interaction with Trp metabolism.
Keywords
Introduction
Many species differences in metabolism and disposition of the essential amino acid L-tryptophan (Trp) exist. Remarkable differences are seen when comparing species such as fish and humans. For example: (1) the rainbow trout (Oncorhynchus mykiss) utilises 60% of dietary Trp for protein synthesis, 1 whereas humans hardly use 1%, if at all, as, in a person in nitrogen balance, the Trp released from protein breakdown is reutilised for protein synthesis 2 ; (2) the plasma Trp-binding protein, albumin, is totally absent from the Antarctic toothfish, the New Zealand eel3,4 and many other aquatic species (see below), whereas, in humans, albumin is the major plasma protein with a normal range of 35 to 50 g/L. Interest in species differences in Trp metabolism probably began with the demonstration during 1949 to 1954 of differences in the conversion of Trp via the kynurenine (Kyn) pathway (KP) to oxidised nicotinamide-adenine dinucleotide (NAD+), with the domestic cat (Felis catus) being unable to utilise Trp instead of nicotinic acid and to possess a high activity of picolinic acid carboxylase (2-amino-3-carboxymuconic acid-6-semialdehyde decarboxylase: ACMSD), a key enzyme whose increased activity can undermine the production and subsequent conversion of quinolinic acid (QA) into NAD+. 5 Studies that followed involved assessment of species differences in activity of the major Trp-degrading enzyme, hepatic Trp 2,3-dioxygenase (TDO: formerly Trp pyrrolase; EC.1.13.11.11), probably prompted in part by the observations by Knox 6 that adrenalectomized rats cannot handle exogenous Trp efficiently and do not survive repeated injections of the amino acid due to the absence of the glucocorticoid TDO induction mechanism, but can survive after administration of cortisol. A number of other animal species were subsequently shown to suffer from excess Trp and to lack the glucocorticoid induction mechanism, and the concept that these species, which are mainly herbivores, lack the free apoenzyme form of TDO that is inducible by glucocorticoids was proposed. 7 A wide array of comparative studies of various aspects of Trp biochemistry and physiology in animal, including aquatic, species have since been reported and the purpose of the present review is to provide an updated wider account of this fascinating area of research that has a wide range of biological, clinical and toxicological implications. The following text is not intended to be a comprehensive one, but of sufficient content to illustrate the general features of, and key advances in research on, Trp metabolism across a range of animal species and to stimulate further Trp research across many species, particularly wild and aquatic ones, to fill in the many gaps in our knowledge of the physiology of these species and its interaction with Trp metabolism. Wherever applicable, data from humans will be the comparative reference.
Overview of Tryptophan Metabolism
Detailed accounts of the Trp degradative pathways have been published2,8,9 and the following is a brief overview that will be followed later on in this text by detailed species comparisons. Apart from being essential for protein synthesis, Trp is metabolised in mammals by 4 pathways (Figure 1), 3 of which (the serotonin and melatonin, the tryptamine, and the indolepyruvate) are of quantitatively minor importance, though of a great functional one. The fourth, the kynurenine (Kyn) pathway (KP), is quantitatively the most important, accounting for 95% of dietary Trp degradation. The 3 minor pathways exist in both the periphery and brain. The KP exists mainly in the liver, where 90% of dietary Trp is degraded, with the remaining 5% occurring elsewhere. The host microbiota also degrade Trp, mainly to indoles, but some can also synthesise Trp. Microbiota may therefore play an important role in human, and particularly in ruminant, health.

The tryptophan degradative pathways.
The serotonin or hydroxylation pathway is controlled by the first enzyme, Trp hydroxylase (TPH), although the second enzyme (aromatic L amino acid decarboxylase: ALAAD) which is pyridoxal 5′-phosphate (PLP)-dependent, could also be limiting under conditions of vitamin B6 deficiency, particularly in humans, wherein its activity in brain is very low, 10 or with certain drugs inactivating the PLP cofactor. Brain [Trp] is the major determinant of cerebral serotonin synthesis, as its cerebral levels (5-25 µM) are well below the Km (50 µM) of TPH2. 11 In mammals, TPH2 is the brain enzyme isoform, as distinct from the TPH1 isoform in the periphery. TPH2 is therefore at most only 50% saturated with its Trp substrate. 12 It is notable that both TPH isoforms are expressed in the rainbow trout brain. 13 Melatonin is synthesised from 5-HT in both the pineal and periphery and its synthesis is also subject to availability of the serotonin precursor Trp. 14 Alkylamine N-acetyl transferase, is the rate-limiting enzyme of melatonin synthesis.
The indolepyruvate (IPA) or transamination pathway and the tryptamine or decarboxylation pathway have in common the end product indol-3-ylacetic acid (IAA), a plant auxin. Not shown in Figure 1 is the conversion of IPA into kynurenic acid (KA), a KP metabolite, via an unstable kynuric intermediate that is produced by the action of reactive oxygen species (ROS) on the enol form of IPA and then cyclisation into KA.
The main products and intermediates of the above 3 pathways exert a range of important functions in both the central nervous system and periphery that impact many physiological processes of particular importance in immune, neurological and psychological disorders.
The (oxidative) KP is controlled mainly by the first enzyme: TDO mainly in liver and indoleamine 2,3-dioxygenase (IDO) elsewhere. The hepatic KP in most mammals contains all the enzymes necessary for the conversion of Trp into NAD+, whereas extrahepatically, not all enzymes are expressed. Accordingly, metabolite formation outside the liver will depend on the enzymes present. Kyn can however be metabolised outside the liver, especially in kidney. NAD+ is synthesised in the main de novo pathway from quinolinic acid (QA), the ‘Preiss-Handler’ pathway from nicotinic acid or the ‘salvage’ pathway from nicotinamide (see Badawy 8 for enzymatic details). As shown in Figure 1, the KP produces a range of metabolites. These Kyn metabolites possess various biological activities both affecting, and being influenced by, many physiological processes, including the endocrine, haematopoietic, intermediary metabolism, immune, neuronal and vital body systems, with major clinical implications for a wide range of disorders.8,9,15
Tryptophan Disposition
Dietary tryptophan disposition
Plasma (or serum) is the major source of dietary Trp for utilisation by tissues. In humans and many other animal species, Trp exists largely bound to albumin, with a small fraction (5%-10%) being free. The proportion of free Trp varies according to the analytical procedure, with ultrafiltration of plasma samples being more accurate than equilibrium dialysis. 16 Heparinised plasma contains higher free [Trp], because of the lipolytic action of heparin causing a non-esterified fatty acid (NEFA)-mediated Trp displacement from albumin-binding sites. Differences in levels of free Trp occur across many species and are determined by variations in concentrations of the 2 major physiological determinants of Trp binding: the binder albumin and the displacers NEFA. Drugs and other chemicals with a high albumin binding affinity can also cause direct displacement of bound Trp (eg, salicylate) and this is an important issue to consider in Trp metabolic and clinical studies in humans and in exposure of various species to industrial chemicals. Albumin levels can be decreased, leading to release of bound Trp, in liver cirrhosis, late pregnancy and infections, whereas those of NEFA are subject to modulation by nutritional, pharmacological and physiological factors, for example, by fasting, intake of the 3 major dietary classes, the antilipolytic insulin and nicotinic acid and the lipolytic catecholamines and phosphodiesterase inhibitors. 16 Thus, as free Trp is a labile parameter subject to modulation by many factors, albumin levels represent the more stable determinant of Trp binding. Trp binding is generally expressed as the percentage free Trp (100 × [free Trp]/[total Trp]). The following are accounts of species differences in albumin and NEFA which can impact Trp binding and hence its availability.
Species differences in plasma albumin
Table 1 illustrates the wide range of plasma albumin concentrations (0-59 g/L) in various animal species. As not all species listed have been examined for Trp binding to albumin, the albumin values given can provide an indication of the likely level of Trp binding in future studies of these species. As stated above, the normal range of plasma albumin in humans is 35 to 50 g/L. Albumin levels of many species fall within this range, including some Trp-tolerant and Trp-sensitive species, all the herbivores and ruminants listed and carnivores and Perissodactyls. By contrast, most Trp-sensitive species have albumin levels of ⩽35 g/L and fish have even-lower levels varying between 0 and 20 g/L. In such species, Trp availability to tissues is likely to be greater than that in those with higher albumin levels and, as will be discussed below, the flux of plasma free Trp through the hepatic KP and other Trp-degradative pathways could be an important factor in their Trp metabolism. That changes in albumin can alter Trp binding has been demonstrated under various conditions, including increased binding by addition of albumin in vitro 72 and decreased binding in hepatic cirrhosis 73 and in late pregnancy in rats. 19 In this latter study, a decrease in serum [albumin] of 19% on day 16 of pregnancy was associated with a significant 33% increase in the % free Trp (from 4.54% to 6.03%). On day 20, the decrease in albumin was little altered (21%), whereas the % free Trp rose by more than expected (by 121%) due to a simultaneous 67% elevation of [NEFA]. A minimum decrease in albumin of ~20% can therefore be expected to increase significantly plasma free Trp within any particular species or experimental setting. Simultaneous elevation of NEFA and depletion of albumin can therefore provide the maximum impact on plasma Trp binding.
Plasma or serum albumin concentration in various animal species.

Numbers in parentheses next to species are those of animals tested. Where no numbers are given indicates too many or information not given. Albumin concentration is expressed in g/L.
Species differences in plasma tryptophan binding to albumin
Table 2 lists the total plasma Trp concentration in a wide range of species, with a smaller number of species (36) in which also free Trp was measured and the % free Trp calculated. As shown, Trp-sensitive species (cat, channel catfish, cow, frog, guinea pig, hamster, rabbit, sheep and steer) possess less total Trp in plasma than Trp-tolerant species. Other species with even-lower plasma total [Trp] include many teleosts, lamprey and crustacea. Binding expressed as the % free Trp varies among most species between 6.29% and 26.39%, but is minimal in aquatic species, with % free Trp values of 81% to 100%.
Plasma tryptophan concentrations and binding in various animal species.
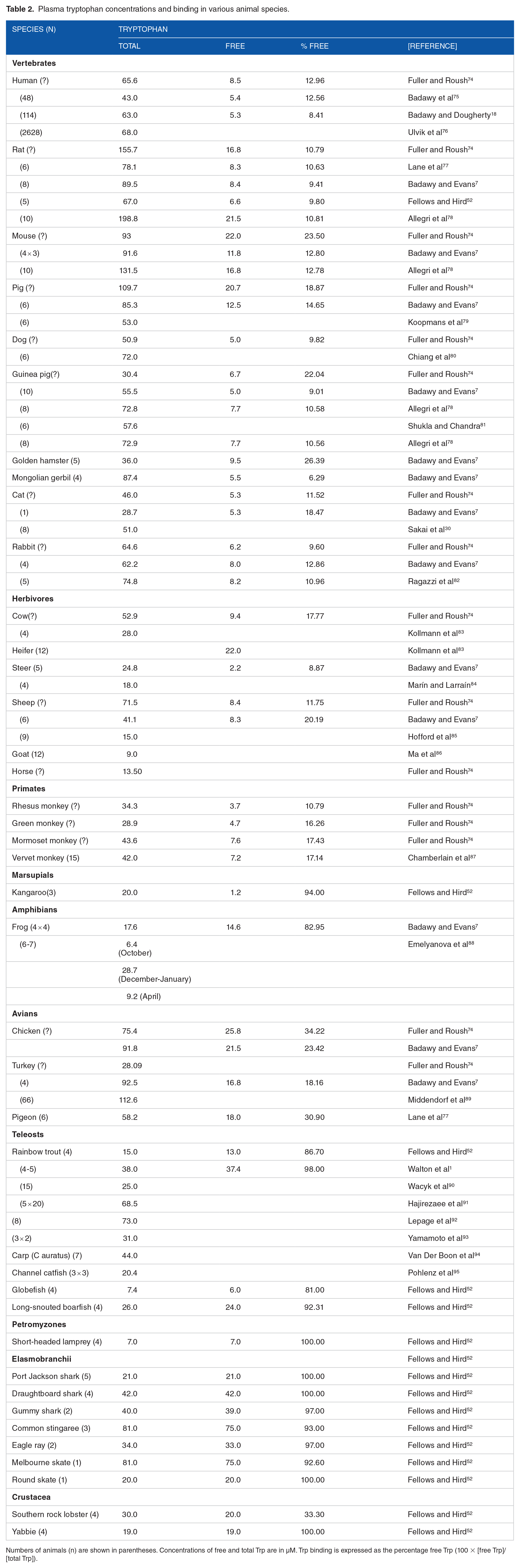
Numbers of animals (n) are shown in parentheses. Concentrations of free and total Trp are in µM. Trp binding is expressed as the percentage free Trp (100 × [free Trp]/[total Trp]).
Relationship between plasma tryptophan binding and albumin concentration
As stated above, a ~ 20% decrease in plasma [albumin] can cause a significant increase in the % free Trp. Figure 2 shows the relationship between these 2 parameters in normal human subjects (Figure 2A) and across various species (Figure 2B–E). In our previous study, 18 in fasting human subjects (n = 114), average plasma albumin and % free Trp were 49.6 ± 0.3 g/l and 8.40% ± 0.21%, respectively (means ± SEM). However, there was no significant (Pearson Product Moment) correlation between these 2 parameters (r = −0.0318, P = .737) (Figure 2A) nor between albumin and free Trp, total Trp or bound Trp (r = 0.114-0.143; P > .1) (data not shown), thus reflecting individual variations in a single species for albumin values within the normal range. By contrast, comparison of 35 different species with wider variations in plasma albumin revealed highly significant negative correlations between albumin and either the % free Trp (Figure 2B) or [free Trp] (Figure 2C). A borderline positive correlation was, however, observed between [albumin] and [total Trp] (r = 0.329; P = .0536) (Figure 2D), whereas a highly significant positive correlation was observed between [albumin] and bound Trp (Figure 2E).

Correlations between plasma albumin and tryptophan concentrations and tryptophan binding in humans and other species.
Physiological role of albumin across species
A brief account of this role can point to potential species differences in Trp binding as a function of albumin levels and rate of degradation. In vertebrates, albumin is the major plasma protein and is essential for normal body physiology. Its main functions include maintenance of intravascular oncotic pressure, being responsible for ~80% of colloidal osmotic pressure, binding and transport of substances, with a high capacity for binding water, and free radical scavenging through its SH content.96,97 Albumin is synthesised only in liver, at a rate in humans of ~ 0.2 g/kg body weight per day. Its daily metabolism depends on plasma levels and occurs at a fixed ~ 10% rate per day. 98 Its degradation is inversely related to its plasma levels, with human albumin having a half-life of about 19 to 21 days.99,100 The albumin T1/2 of horse (19-20 days), cow (14-19 days) and sheep (14-28 days) 101 are closer to that of human and are parallelled by the plasma albumin values in Table 1 here. Some species with lower plasma [albumin] than the above species, such as the cat, mouse, rabbit, pig and dog (25.8-28.2 g/l; see Table 1 here) have shorter albumin half-lives of 1.5 to 8.2 days.102,103A plot of averaged plasma [albumin] versus the albumin T1/2 of these 9 species (Figure 3) confirms the above inverse relationship between albumin concentration and its rate of degradation. It is remarkable that a highly significant positive correlation between [albumin] and its T1/2 is observed with a relatively small number (9) of species with moderate differences in their plasma [albumin]. Adding 13 species from Table 1 with zero albumin, such as sharks, skates, rays and eels to the calculations only raised the correlation coefficient slightly to 0.923, but much more significantly to a P value of 9.76 × 10−14. Species differences in albumin degradation can be due to a range of determinants, for example, rate of synthesis, disposition across compartments and expression of the neonatal Fc receptor (FcRn) that binds to albumin and protects it against lysosomal proteolysis.103,104Engineering this receptor prolongs the albumin T1/2 105 and infusion of albumin into an analbuminaemic human neonate prolongs the T1/2 of the infused albumin to 115 days, as compared with the ~ 20-day duration in normal subjects. 106 These latter authors also observed that plasma [Trp] was extremely low in this infant, and suggested that this Trp deficiency might have impaired albumin synthesis. FcRn binding to albumin of various animal species requires systematic investigation, as currently available data are extremely limited.
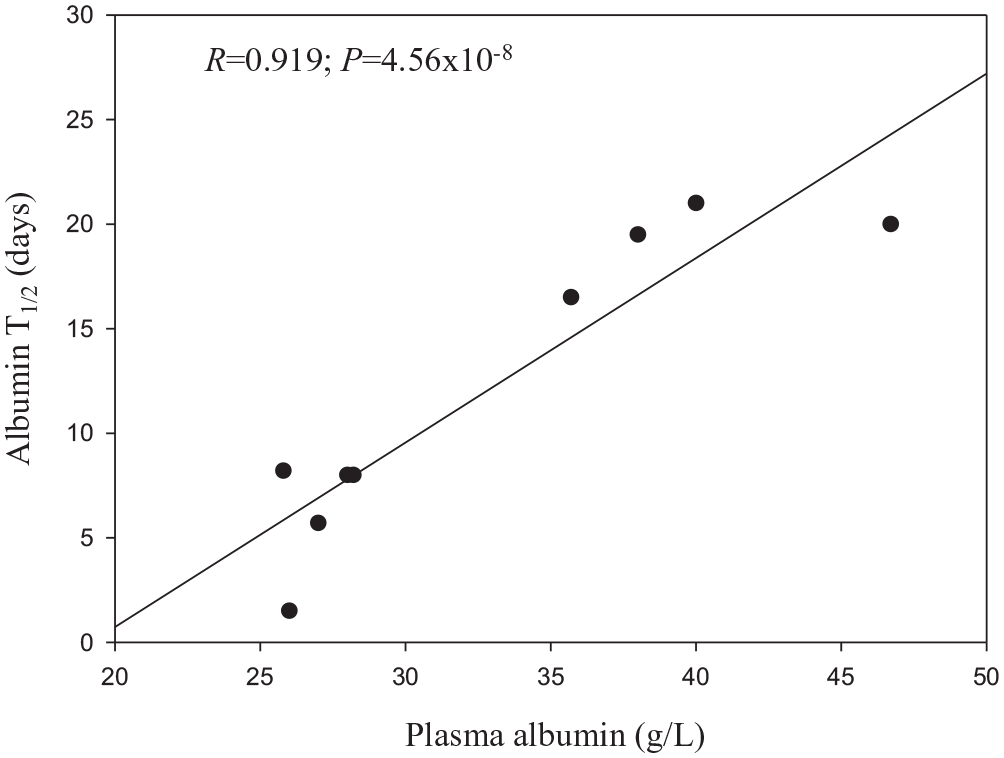
Correlation between plasma albumin concentration and turnover rate.
Prolonging the albumin half-life has been used to enhance the transport function of albumin for therapeutic purposes, for example albumin-based therapeutics with tailored FcRn binding properties to study and improve drug pharmacokinetics.22,100Binding of chemicals by albumin can also be used as a biomarker of recent marine life exposure, for example, albumin-benz(α)pyrene diol epoxide adducts. 107
In contrast with vertebrates and others, aquatic species show marked differences in plasma albumin. Of the 25 aquatic species listed in Table 1, albumin values (in g/L) varied between zero (n = 14), 1.9 to 4.4 (n = 2), 9 to 20.8 (n = 8) and 39.5 (n = 1: Nile tilapia). It is almost certain that the lower albumin in aquatic life is related to osmotic activity, fluid and electrolyte balance and needs within the aquatic macro-environment, as well as the species microenvironment. A discussion of these aspects is outside the scope of the present article, but it is of great interest to note that urea plays a major role as a more suitable substitute for albumin under these conditions. For example, urea levels in shark blood are very high (0.19-6 M), thus enabling them to have an osmotic pressure higher than that in sea water, and teleosts also have raised urea levels, but ~ 100-fold lower than in sharks. 108 Whereas the impact of differences in albumin turnover on Trp binding may be difficult to assess, the potential effects of low plasma albumin on Trp binding and its consequences are certainly by-products of the physiological adaptations of aquatic species to their environments.
Species differences in plasma non-esterified fatty acids (NEFA)
As the physiological displacers of albumin-bound Trp, NEFA play an important role in determining plasma free [Trp] and hence its flux and availability. Table 3 shows the wide species variations in plasma [NEFA]. The report by Fellows et al, 130 is the most comprehensive. Comparing differences among 7 species, these authors reported plasma [NEFA] levels of 0.04-0.53 mEq/L and in the following decreasing order: pigeon > rat > domestic fowl > human = cane toad > Eastern grey kangaroo > axolot. They also compared [NEFA] in aquatic species. Levels in 5 teleosts varied between 0.09 and 0.64 mEq/L, thus resembling those in the above species. By contrast, apart for a [NEFA] of 0.13 mEq/L in white-spotted stingaree, 0.1 in Short headed lamprey, 0.04 in Yabbe and 0.02 in Southern rock lobster, NEFA were not detectable in the common stingaree, eagle ray, southern fiddler, Port Jackson shark, draughtboard shark or Melbourne skate. As described in Table 2 here, binding of Trp to albumin was studied by the same group 52 in many of these species.
Species differences in plasma nonesterified fatty acid (NEFA) concentration.

[NEFA] was determined but not reported in this study. **Blood was taken from cut liver.
Despite the available information on albumin and NEFA levels, it is difficult to dissociate the impact of one or the other on Trp binding, except in species in which albumin is either absent or very low. Here, differences in [NEFA] are unlikely to have a significant impact. It is of interest, however, that where albumin is absent, NEFA are also absent (compare species in Tables 1 and 3). The potential impact of species differences in [NEFA] is, however, difficult to assess, except for changes within a single species in response to stress and dietary manipulations, for which significant evidence exists. As the data in Table 3 show, there is no dichotomy in plasma [NEFA] between Trp-tolerant or – sensitive species or between vertebrates and teleosts. Nutritional, metabolic and other physiological factors, and differences are potential explanations.
Fate of Plasma Tryptophan in Species
Physiological aspects
Plasma Trp levels are determined by dietary protein intake and metabolism in tissues. Trp is essential for protein synthesis, and also for NAD+ synthesis in the absence of adequate intake of niacin (nicotinic acid or nicotinamide: the 2 forms of vitamin B3). With protein synthesis, wide variations in dietary Trp requirements exist among species. Thus, as stated in the Introduction, whereas humans hardly use 1%, if at all, the rainbow trout uses 60%. 1 However, many studies of dietary Trp requirements by teleosts are controversial and suggest that requirements are subject to many determinants and confounders other than growth and wellbeing. 131 In most mammals, NAD+ synthesis from Trp is quantitatively more important than that from niacin, with only 1 mg of niacin arising from intake of 60 mg of Trp in humans.8,132 Species are known to differ in their nutritional Trp requirements and their Trp:niacin ratios reflect their ability to convert Trp into niacin with any defects being attributable to altered activities of KP enzymes, as will be described further below. Examples of Trp:niacin ratios in species other than humans are: rats: 33:1 133 or 40-53:1,134,135 turkeys: 103-119:1, chick 45:1, broiler chicken 47-54:1,136,137Pekin and mule ducks 172-181:1 137 and both cat5,66,138 and rainbow trout 139 lack an efficient conversion of Trp into niacin. The mouse has an even-more efficient conversion rate than the rat. 140
The primary determinant of plasma Trp disposition is activity of the major Trp-degrading enzyme, liver TDO. This is best illustrated by the observed 9.3 to 12.7-fold increase in plasma [Trp]141,142 and the 10.6-fold elevation of brain [Trp] 143 following deletion of the mouse TDO2 gene. By contrast, no changes in brain [Trp] occur after deletion of the genes encoding the other Trp-degrading enzyme indoleamine 23-dioxygenase (IDO) isoforms 1 and 2. 143 Plasma Trp availability is also controlled at the secondary, but more immediate, level by its albumin binding and, additionally for uptake by the brain, by extent of competition from a number of neutral (competing) amino acids (CAA), mainly Val, Leu, Ile, Phe and Tyr, with the ratio of [free Trp]/[CAA] or [total Trp]/[CAA] being a predictor of likely changes in brain [Trp] and hence serotonin synthesis. Many studies involving measurement of this ratio have been performed in humans to assess the role of serotonin in behavioural and other disorders using the acute Trp depletion (ATD) and loading (ATL) tests, by administering amino acid mixtures respectively deficient or supplemented with Trp (see review by 144 ). Originally proposed for use in rats,145,146 the test was adopted for humans 147 and the formulations reported by Young et al 148 became standard. Other species underwent the ATD and/or ATL test, including vervet monkey to study aggression, 87 rat to study catecholamines 149 and pigeon 150 to study injurious behaviour. 151 These latter authors suggested that ATD may be a useful test to explore in avian species the role of serotonin in abnormal behaviours such as feather picking in laying hens or compulsive feather picking in parrots.
Irrespective of TDO activity, the flux of plasma free Trp down the Kyn and other pathways of Trp degradation can play an important role in production of Trp metabolites. Flux is determined primarily by free Trp levels through changes in albumin and NEFA.72,152 The low levels in, or absence of albumin from, aquatic species is likely to facilitate Trp entry into tissues and thus enhance its utilisation and degradation. Similar changes can also be expected under stressful conditions leading to elevation of NEFA, for example, after exercise in camels. 153
Toxicity aspects
It is important at this point to establish the safety or otherwise of Trp, especially at high levels, among various species before discussing in detail Trp metabolism, as the latter plays a key role in Trp disposition and hence its potential toxicity. As stated earlier, the first report of a harmful effect of a large dose of Trp was that of death of adrenalectomized rats, which could be prevented by cortisol administration, suggesting that TDO synthesis is important for ‘detoxifying’ Trp. 6 There then followed reports of harmful effects of Trp in other species: death from large or repeated doses of Trp administered to steer,154,155the channel (USA) catfish Ictalurus punctatus, 156 the Mongolian gerbil (Meriones unguiculatus) 157 and the intact guinea pig. 158
Little information is available from toxicological studies on safety of Trp in other species, though a certain degree of speculation is possible. For example, the high Trp intake by carnivorous wild animals is unlikely to be harmful given their Trp-rich protein consumption, though they may suffer potential cardiovascular abnormalities mediated by homocysteine produced from protein-derived methionine. Cardiomyopathy associated with coronary arteriosclerosis has been reported in the Eurasian lynx. 159 While death of wild animals has many causes, it would be of interest to assess the potential involvement of homocysteine and frequency of cardiovascular abnormalities in these species. Plasma homocysteine in horse is within the normal range for humans (<15 µM) 160 and Trp is not toxic to horse, at least at doses of up to 120 mg/kg body weight (producing blood Trp levels of 350 µM). 161 Trp exerts no effect on reaction time to startle or clinical pathology parameters in horse. The above authors observed no calmative effect of Trp (ie, for the horse to travel at a lower speed). While Trp may exert a calmative effect in excitable horses, evidence for this is controversial. 162 Several studies have established the role of dietary Trp in stimulating growth and immunity and decreasing anxiety in fish species.163-166 Trp administration is well tolerated by Rhesus monkey (Macaca mulatta) and inhibits self-injurious behaviour in males with a history of such behaviour. 167 Clearly the safety of Trp in many other species requires assessment.
From available evidence so far, Trp toxicity is associated with the absence of the TDO apoenzyme and its glucocorticoid induction mechanism. Table 4 lists species possessing and lacking apo-TDO and/or its glucocorticoid induction mechanism in relation to Trp toxicity. A clear distinction between the above 2 parameters and toxicity is obvious, except for the rainbow trout, in which Trp is safe (see above) despite the absence of the TDO apoenzyme. 168 It is also unknown if the trout TDO is inducible by cortisol. Indirect evidence for induction in another teleost, Meagre (Argyrosomus regius) is suggested by the finding 169 that stress-induced elevation of plasma cortisol is associated with increased production of Kyn and quinolinic acid (QA) in liver in dietary Trp-supplemented fish. As will be discussed below, stress in meagre receiving the basal diet increases the flux of Trp through TDO, increasing both liver Trp and Kyn simultaneously. Whereas turkey liver TDO is inducible by cortisol, 170 indirect evidence suggests that chicken and pig liver TDO may also be inducible by glucocorticoids. Thus, Bordetella avium infection of poults increases serum corticosterone concentration and enhances TDO activity, 171 and treatment of weaned piglets with Diquat enhances TDO mRNA expression, 172 most likely by elevating circulating cortisol. 173 Cortisol does not induce TDO activity in Ictalurus punctatus. 156 These latter authors also reported the failure of Trp to activate the enzyme at ⩾4 hours, though this could be explained by a potential activation by the dose given having taken place at earlier time intervals, as species lacking apo-TDO appear to respond rapidly to activation by Trp. 7 Much work is clearly required to assess glucocorticoid induction of TDO in other species in relation to Trp toxicity. Potential mechanisms of Trp toxicity in Trp-sensitive species are discussed below under IDO.
Summary of species differences in plasma tryptophan, liver apo-TDO and its induction by glucocorticoids, and sensitivity to tryptophan.
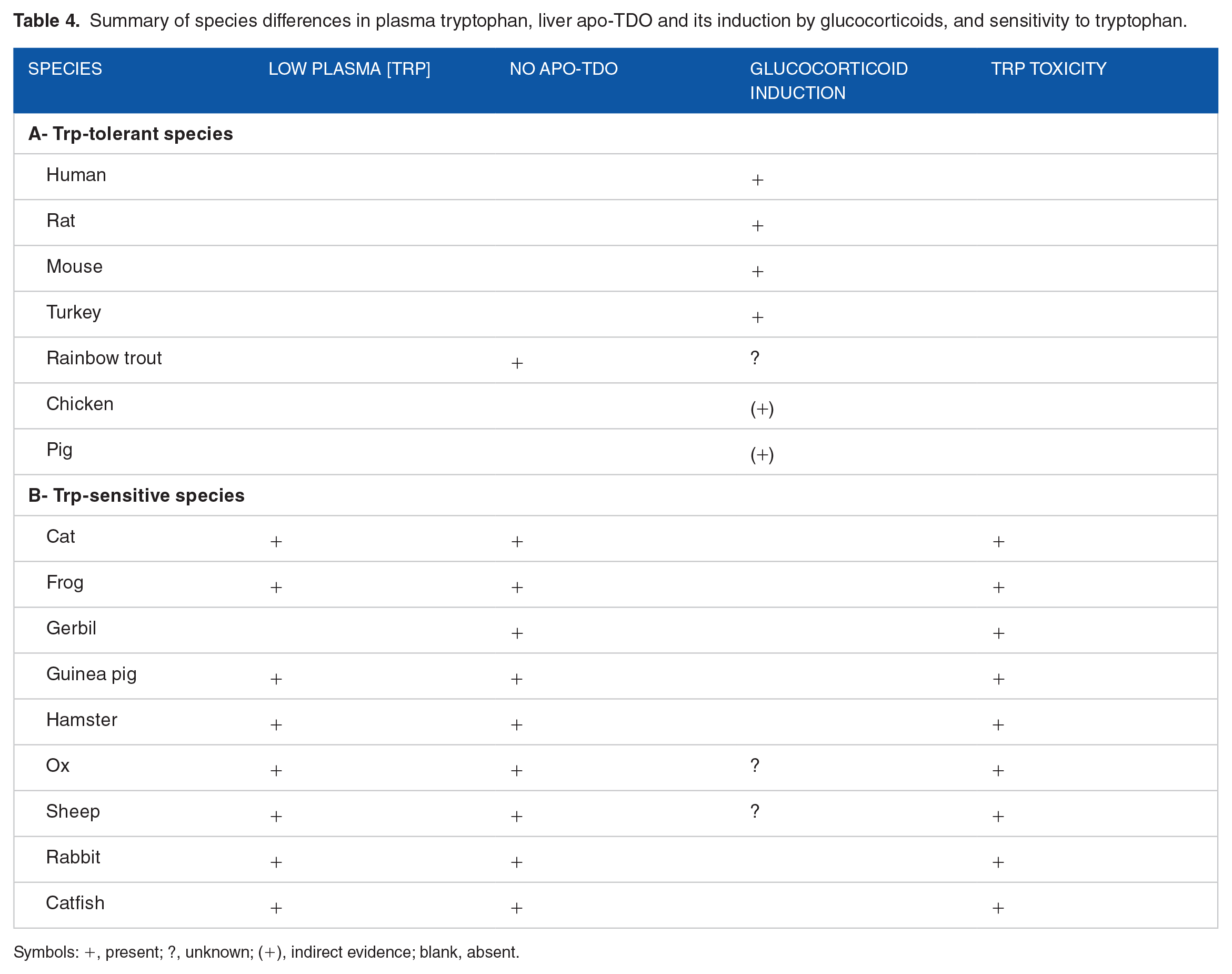
Symbols: +, present; ?, unknown; (+), indirect evidence; blank, absent.
Although fish species lack or possess very little albumin to bind Trp and generally have low Trp levels, these limitations could benefit fish in at least 2 ways. (1) Because of their low TDO activity, the low plasma Trp does not impose an immediate (strong) burden on TDO. (2) The absence of albumin enables Trp to have a much easier access to liver and other tissues, with the Trp flux playing a particularly important role in production of kynurenine metabolites. For brain, Trp availability can enhance serotonin synthesis, whereas for liver, synthesis of KP metabolites, niacin and NAD+ will depend on the KP enzyme complement. Lack of albumin in most aquatic species, however, also facilitates tissue uptake of industrial chemicals and other pollutants and consequent organ damage and death, thus further emphasising the need to protect these vulnerable species by strengthening measures to control the disposal of pollutants.
Metabolic aspects
Given that TDO activity and its induction by glucocorticoids are likely to be involved in Trp toxicity, it is important to assess the role of TDO and other enzymes of Trp metabolism along the KP in some detail, especially as this is the most studied Trp-metabolic pathway in animal species. This will be followed by a discussion of the role of Kyn and other pathway metabolites in immune modulation and other physiological processes.
Species Differences in Enzymes of the KP
Most studies of species differences in enzymes of the KP have been limited to relatively small numbers of species and have focused on the 2 rate-limiting enzymes of the pathway, TDO and IDO, with very few comparative studies of subsequent pathway enzymes. The most detailed of these studies are those involving TDO enzymatic determination, 7 development of a TDO assay procedure in liver cytosol from multiple species 174 and a comparison of TDO and IDO in metazoans. 175 However, as will be discussed below, studies of urinary Trp metabolite excretion can also provide indirect indicators of enzyme activities. The following are accounts of data available for individual enzymes of the KP
Tryptophan 2,3-dioxygenase (TDO)
Species comparisons
In addition to assays of TDO activity in various species discussed above, other measures of enzyme activity have been published and involved determination of TDO activity in vivo by measuring the evolution of 14CO2 from administered 2-14C Trp and formation of niacin or NAD+ from exogenous Trp. For example, Green et al 182 reported that TDO activity in vivo in gerbils is much lower than that in rats and is not inducible by cortisol. These authors also reported that, whereas TDO induction by cortisol in rats decreases brain Trp and serotonin synthesis and turnover, no changes in brain indoles are observed in gerbils. By contrast, α-methyl tryptophan, a potent activator of TDO, is effective in activating liver TDO and decreasing serotonin in both species. As the comparisons in Table 5 show, the studies listed reported variations in the relative TDO activities among species, possibly because of experimental and other study differences. For example, whereas in one study 174 only the holoenzyme was assayed (ie, in the absence of added cofactor) in pooled liver cytosols obtained from commercial suppliers, the total (holoenzyme + apoenzyme) activity was measured in the presence of added haematin in the other 4 studies. The rat TDO activity in these latter 4 studies was the highest or second highest among species, whereas that in the other study 174 was the second lowest. The rat TDO holoenzyme activity is by no means the highest among the various species studied. 7 This latter study showed a clear distinction in TDO activity between species that tolerate, and those that are sensitive to, excess Trp, with the latter lacking the inactive haem-free apoenzyme and the former possessing both forms.
Comparison of enzymes of the kynurenine pathway of tryptophan metabolism across animal species.

Comparisons of TDO among strains
Strain differences in TDO activity have been studied largely in mice, but also in rat strains developed for alcohol preference. In mice, TDO activity differs among strains as follows: C57BL > AKR > C3H = DBA. 183 TDO of the C57BL is double that of the CBA strain.184,185 TDO activity in albino NCL mice is 52% higher than in Swiss albino mice. 186 The C57BL strain is notable for its preference over other strains for alcohol intake in a free choice drinking situation. 185 In rats, TDO activity is comparable among the Wistar, heterozygous Gunn and Sprague-Dawley, and higher than in the Long-Evans strain. 187 However, TDO activity of Sprague-Dawley rats was reported to be moderately (31%) higher than that of the Wistar strain. 188 Rat strains have also been developed to prefer alcohol and have been shown to possess a higher TDO activity over their corresponding controls, for example, the Indiana preferring (P) versus non-preferring (nP) and the Sardinian preferring (SP) versus non-preferring (SnP) strains. 189 The higher TDO activity may be due to the higher [corticosterone] in both preferring strains and, additionally enhanced gene expression of the enzyme, as demonstrated with the SP strain and also with the C57BL versus CBA. 189 TDO also exists in flies. The enzyme in Drosophila melanogaster has been assayed without added haematin and its activity appears to be equally distributed between head, thorax and abdomen. 190 Expressed per weight, TDO is more active in flies than in rats. Activity of the enzyme in flies is also strain-dependent in the following descending order: BW > Sevelon > Oregon-R, with minimal activities in several other strains. 190
TDO in other species
In addition to mammals, TDO exists in many non-mammalian species, including teleosts [rainbow trout, 168 Senegal soles (Solea senegalensis), 166 Atlantic salmon (Salmo salar), Japanese rice fish (Oryzias latipes) and zebra fish (Danio rerio). 131 It also exists in poults, 171 honey bees, 191 mosquitoes 192 and across metazoans. 175 The TDO of the mosquito Culex pipiens appears to be fully saturated with haem, 193 thus possibly existing in the holoenzyme form, and, its kynurenine pathway is limited to production of xanthurenic acid. Wild carnivores are almost certain to express TDO, otherwise they would be unable to handle their high Trp intake.
Species TDO response to glucocorticoids and tryptophan
As stated above, species sensitive to Trp toxicity lack the TDO apoenzyme and are also unresponsive to its induction by glucocorticoids. As shown by Knox 6 glucocorticoid induction of apo-TDO synthesis protects adrenalectomized rats against Trp toxicity. Glucocorticoid induction of TDO can therefore be regarded as a Trp detoxification mechanism. Species lacking the haem-free apoenzyme, however, process Trp somewhat differently from those possessing the apoenzyme in an apparent attempt to dispose of it rapidly. For example, their TDO is activated earlier and by smaller doses of Trp than the rat TDO. Thus, at 30 minutes after Trp administration, the TDO of golden hamster > guinea pig > frog is activated by a 50 mg/kg dose and maximally by a 150 mg/kg dose, whereas the rat TDO is unresponsive to any Trp dosage up to 500 mg/kg at this time-interval. 7 Whereas haem (haematin) and its precursor 5-aminolaevulinate and agents that displace Trp from albumin-binding sites, such as ethanol and salicylate, activate the rat TDO by cofactor or substrate- mediated mechanisms, they fail to do so in guinea pigs or golden hamsters.7,194 Different responses are also observed in mouse strains. Thus, TDO of the C57BL strain exhibits a greater response to induction by cortisol or dexamethasone than other strains.183,189 By contrast to TDO, Tyr aminotransferase induction by dexamethasone or cortisol is comparable between C57BL and CBA. 189 In rabbits, where the glucocorticoid induction mechanism is absent, repeated triamcinolone acetonide administration, which induces TDO in rats, 195 does not decrease plasma [Trp], but decreases plasma [Tyr], 196 suggesting induction of Tyr aminotransferase, but not TDO. Response of TDO to Trp, however, is less variable among mouse strains, with C57BL = DBA = AKR > C3H. 183
Trp, however, has been reported not to activate TDO at 4 to 16 hours after administration to the channel catfish (Ictalurus punctatus). 156 These latter authors suggested seasonal variations as an explanation, though activation at an earlier time-interval is also possible. If this fish TDO does not respond to Trp, it could be especially vulnerable to its toxicity, as suggested by the rapid death of half the treated fish within 12 to 16 hours, 156 relative to steers dying from Trp over 1 to 7 days. 154
Regulation of TDO
TDO regulation has been extensively studied following the pioneering work of W. Eugene Knox, Olga Greengard, Phillip Feigelson and others in the ~ 1950s with the discovery of its induction by glucocorticoids and activation by Trp. TDO has since been at the centre of the concept of enzyme regulation. Many reviews of TDO regulation have been published. Briefly, the enzyme is regulated by at least 4 major mechanisms: hormonal induction by glucocorticoids, substrate activation and stabilisation by Trp, cofactor activation by haem and feedback or end-product inhibition by NAD(P)H. While this is a general outline of these mechanisms, more complex aspects are involved, such as the roles of insulin, glucagon and other hormones and of the haem cofactor in the glucocorticoid mechanism, and the ability of some kynurenine metabolites (KA and 3-HAA) to activate TDO. 8
Functions of TDO
TDO performs a range of important functions.8,15 These include detoxification of excess Trp, control of Trp availability to other tissues, including the brain for serotonin synthesis, regulation of hepatic haem biosynthesis by its preferential utilisation of the small regulatory-haem pool in the hepatic cytosol, control of production of kynurenine metabolites and of NAD+ synthesis. These functions and the resulting physiological changes extend across many body systems with important implications for a range of health issues.
Indoleamine 2,3-dioxygenase (IDO)
Species comparisons and expressions of IDO
As well as the comparative studies of IDO activity in the mammalian species outlined in Table 5, and that comparing IDO activity in mice, opossum (Monodelphis domestica) and the semiaquatic mammal the platypus (Ornithorhynchus anatinus), 178 most other studies of IDO were performed in aquatic species. In the Yuasa et al 178 study, IDO1 activity in descending order was: platypus > opossum > mouse, and IDO2 was platypus > mouse > opossum. IDO is expressed in many fish species, including the rainbow trout (Oncorhynchus mykiss), 197 the grass carp (Ctenopharyngodon idella) 198 and many other teleosts. 199 The IDO of the grass carp can be activated by the haem precursor 5-aminolaevulinate, thus suggesting that it is not fully haem-saturated. As well as in the mammalian species studied above, IDO is also expressed in invertebrates in at least 15 species within 6 phyla. 175
IDO isoforms and variants and their functional differences
All vertebrae express both IDO1 and IDO2. IDO1 is expressed in human, gorilla, macaque, goat, chiru, yak, cattle, bat, domestic cat, rat, giant panda, dog, hamster, Tasmanian devil and all other mammalian species. 199 IDO2 is also expressed in the above mammals and in amphibia, avians, reptiles (lizards and alligators) and certain teleosts. 199 Seven teleost and 2 turtle species express both isoforms, whereas many other teleosts express only IDO2. 199 As well as IDO1 and IDO2, an IDO3 variant is expressed in a mollusc (M. yessoensis) and variants across many mammalian and other species with altered catalytic activity have also been identified. 175
Kinetic parameters differ among IDO isoforms. With IDO1, substrate (L-Trp) affinity (expressed as the Km in µM) is generally relatively high (14-22 µM for human < mouse < platypus), compared with Japanese Medaka (351 µM). 199 In rat and mouse tissues, predominantly placenta, lung and intestine, the IDO1 Km varies between 20 and 50 µM. 8 IDO1 thus exhibits a greater affinity for its Trp substrate, compared with TDO2, whose Km is 190 µM. The greater capacity of TDO2 towards Trp enables it to handle large increases in dietary [Trp], whereas, under these conditions, IDO1 is at a disadvantage, as its activity is inhibited by Trp 200 with a Ki of 50 µM for the human enzyme. 201 IDO1 inhibition by Trp involves reversed sequence of binding of O2 and Trp: with O2 binding preceding that of Trp at low [Trp] and the opposite sequence occurring at high [Trp]. 202 IDO2 has a much reduced affinity for its Trp substrate, with Km values in the high mM range, except that (81 µM) of the lizard (Anolis carolinensis). 199 While IDO2 of many species exhibits a very low affinity for its Trp substrate, occasionally wild-type species IDO2 shows comparable affinity to that of IDO1, for example, the sponge Amphimedon queenslandica, with Km values of 4.7 and 7.7 µM respectively. 175 Variants with amino acid residue modifications can alter the affinity of IDOs considerably in both directions. 175
IDO and immune activity
IDO’s major role is that of interaction with the immune system. 203 Immune activation induces IDO1 and results in accelerated Trp degradation along the KP, leading to depletion of Trp and simultaneous production of its immuno-reactive and other metabolites. In particular, 3-HK, 3-HAA and QA cause immunosuppression of allogeneic T-cell proliferation additively 204 and by an apoptotic mechanism. 205 At 10 µM, induction of apoptosis is significant with 3-HAA > QA > 3-HK, with Kyn or anthranilic acid (AA) being inactive. 205 The Kyn transamination product kynurenic acid (KA) possesses both pro- and anti-inflammatory properties206,207 and is the KP metabolite with the greatest affinity for the aryl hydrocarbon receptor (AhR), followed by xanthurenic acid (XA), with Kyn itself being the weakest. 208 While Kyn has generally been assumed to be an active KP ligand of the AhR, its role in vivo is likely to be that of the KA precursor, in view of the importance of substrate availability for kynurenine aminotransferase, given the high Km of the enzyme for Kyn (0.96-4.7 mM). 8 The importance of KA in AhR activation is further suggested by the discovery of a new metabolic immune check-point: IL-4-induced gene 1 (IL4I1 or L-phenylalanine oxidase), which activates the AhR via KA and also the Trp transamination metabolite IPA. 209
The AhR, a ligand activated transcription factor, can elicit both destructive and protective effects through binding respectively to exogenous or endogenous ligands. Exogenous ligands are activated to toxic metabolites through induction of cytochrome P-450 enzymes and this presents danger to aquatic and other wild life when exposed to industrial chemicals. Exogenous AhR ligands include the chemical 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) and other halogenated aromatic hydrocarbons, for example, dibenzofurans and biphenyls, polycyclic aromatic hydrocarbons, such as 3-methylcholanthrene, benzo-[a]-pyrene, benzanthracenes and benzoflavones, indirubin and 6-formylindolo(3,2-b) carbazole (FICZ). Their harmful effects depend on the rate of their metabolic clearance, with effects of the high affinity AhR ligand TCDD lasting for days, because of its poor metabolism due to its 4 Cl atoms impeding access by metabolising enzymes. 210
Endogenous ligands derived from Trp metabolism via the KP are KA, XA and Kyn, whereas many other ligands are derived from other Trp-metabolic pathways and include indole, indol-3-ylacetic acid, indol-3-yllactic acid, indol-3-ylpyruvic acid, indol-3-ylaldehyde and indoxyl sulphate.211-215 AhR activation by endogenous ligands is generally associated with positive immune changes, but excessive activation can induce negative effects. The AhR controls IDO1 expression through an autocrine loop of AhR-IL-6-STAT3 signalling216,217 and whereas KA induces IL-6, IL-6 generation by inflammation can induce IDO1 to produce enough KA to activate the AhR. 208
Another important function of the AhR is control of expression of poly (ADP-ribose) polymerases (PARPs).209,218 PARPs are NAD+ consumers and their excessive activation can deplete this vital cellular effector and also ATP, resulting in cell death.
Because of the wider exposure of aquatic species to environmental pollutants and other chemicals, the role of the AhR in their susceptibility to chemical toxicity has received a great deal of research interest, a detailed discussion of which is outside the scope of this review. However, a number of aspects of AhR biology in aquatic species are noteworthy, for example, the lower affinity of the AhRs of the frog, salamander and other extant amphibian orders to TCDD, compared with vertebrates and teleosts, 219 the greater sensitivity to AhR induction by β-naphthoflavone of brown trout > rainbow trout > brook trout > European eel > white sturgeon > Atlantic salmon over several other fish species, 220 the wide variations in AhR activation potencies of a broad range of poly aromatic hydrocarbon and heterocyclic compounds in fish, avian and mammalian systems, 221 the wide variations in susceptibility of fish species to early life mortality induced by TCDD, with trout showing the least susceptibility and zebra fish the greatest 222 and the tissue specificity of AhR expression in rainbow trout as a function of the CYP isoforms. 223
Whereas the role of the major cytokine and IDO1 inducer interferon-γ (IFN-γ) in vertebrate physiopathology is well known, less information is available on its role and that of its variants in teleosts 224 and potentially other aquatic species. Despite the large literature on the AhR and the likely IDO1 induction by IFN-γ in aquatic species, Trp metabolism along the KP has received little attention in these species. Persistent exposure of the common minke whale (Balaenoptera acutorostrata) to organochlorines downregulates liver kynureninase, 225 suggesting a potential elevation of kynurenine and KA levels and a consequent AhR activation. As will be discussed below, inhibition of N′-formylkynurenine formamidase by the organophosphate insecticide diazinon paradoxically raises levels of Kyn and KA in mice and a potentially similar effect in aquatic species could occur. Clearly more studies of the KP in aquatic species are desirable.
Regulation of IDO
Unlike TDO, IDO is not inducible by glucocorticoids and its activation by Trp is limited, partly because of its high affinity for Trp (Km = 20-50 µM) and its inhibition by [Trp] ⩾50 µM (see above). Interferons (IFNs) are the major IDO effectors, with IFN-γ being the strongest inducer. 226 Glucocorticoids, however, exert differential effects on IDO induction by IFNs, namely inhibition and potentiation. Thus, IDO induction by IFN-α is inhibited by dexamethasone, whereas that by IFN-γ is potentiated 226 and this may explain in part the efficacy of dexamethasone in therapy of COVID-19 infection, wherein IFN-α appears to play a major role. 227 Other proinflammatory cytokines such as interleukin IL-1β, IL-6 and tumour necrosis factor-α (TNF-α) also induce IDO or potentiate its induction by IFN-γ, whereas anti-inflammatory cytokines such as IL-4, IL-10 and TGF-β inhibit IDO induction by IFN-γ. 228 Thus, the balance between proinflammatory and anti-inflammatory cytokines determines the IDO status. NO is also a negative IDO effector.
Although IDO is a haemoprotein, many enzymatic studies using tissue preparations do not include added cofactor, thus suggesting that the enzyme exists fully saturated with haem. An exception to this notion is that of haem activation of the grass carp IDO described above. However, in instances where newly synthesised apo-IDO is studied, addition of haem activates the enzyme, whereas haem deprivation inhibits it. 203 Regulation of IDO is a complex and multi-faceted process extending beyond these aspects, as detailed in the excellent review by Yeung et al. 203
As TDO and IDO are both hemoproteins, the question of whether their activities could be influenced by changes in body iron levels and whether species differences in these levels could be involved has not been studied in detail. Species differences in body Fe have been compared in a limited number of studies. Thus, whole blood [Fe] is the highest in dog > horse > rat > human > rabbit > hamster > mouse. 229 Wide variations in the total body Fe content in 3 Clarias (walking catfish) species have been reported. 230 Of 8 fish species in India, C. fasciata shows the largest Fe content. 231 As far as we could ascertain, no comparative studies of any potential effect of differences in iron content on the above 2 hemoproteins have been reported. However, differences in Fe content could, at least theoretically, influence the 2 enzymes, especially if Fe levels are extremely large or small. The wide variation in body Fe in above 3 Clarias could render these species a suitable model for such a study. In experimental Fe deficiency in rats, liver TDO activity is doubled and the response of the enzyme to induction by glucocorticoids or activation by Trp is not impaired. 232 The increased TDO activity in this study 232 however appears to be glucocorticoid-mediated, rather than Fe-related, as Fe-deficiency induced weight loss, akin to that caused by starvation, which is associated with a corticosterone-mediated TDO induction. 233 In this model of iron deficiency anaemia, which induces significant decreases in levels of blood haemoglobin, serum Fe and liver non-heme iron, levels the major hepatic hemoprotein, cytochrome P-450, which utilises ~ 66% of hepatic heme, 234 are not impaired. 235 As far as we could ascertain, the potential effects of iron overload on TDO activity are unknown. By contrast, the effects of Fe on IDO1 activity appear to be cell- and/or tissue-dependent. Thus, Fe activates IDO1 in brain, and cultured microglia and the purified human recombinant enzyme, 236 but inhibits the IFN-γ induction of the enzyme in Hep-2 cells. 237 Whereas iron deficiency anaemia can exert profound effects on the immune system including cytokine abnormalities,238,239 sequestration of Fe blocks its IDO activation. 236 Clearly more systematic studies in this area are warranted.
Does IDO play a role in tryptophan toxicity?
The mechanism of the toxic effects of Trp in sensitive species is not fully delineated. Knox 6 suggested that the absence of glucocorticoid induction of TDO by adrenalectomy in rats diverts Trp metabolism to other pathways, the products of which may be toxic. 5-Hydroxytryptamine was implicated in death of rats treated with large doses of Trp, based on increased death by co-administration of the monoamine oxidase inhibitor pargyline and protection by the TPH inhibitor p-chlorophenylalanine. 240 In cattle which are prone to emphysema, ruminant bacteria may enhance the conversion of excess Trp to indole metabolites. 155 In fact, Trp metabolism in steer occurs mainly (78%) in indole-forming pathways, with the kynurenine pathway contributing only ~10%. Many indoles are AhR agonists, including indole itself, 5-hydroxyindole, 3-methylindole, indol-3-ylacetic acid and tryptamine. 3-Methylindole causes pulmonary oedema and emphysema in cattle and goats. 241 Moffett and Namboodiri 242 suggested that Trp toxicity in Trp-sensitive species may involve immune activation with consequent IDO induction and production of immunosuppressive kynurenine metabolites. However, this implies an initial development of an inflammatory state to elicit toxicity. In this regard, it would be of interest to know if Trp-sensitive species are more prone to infections and if the greater induction of IDO by LPS reported in gerbils 243 (see above) could be demonstrated in other Trp-sensitive species.
Serotonin is unlikely to be involved in Trp toxicity, as excess Trp exerts a substrate inhibition of TPH, 244 hence the failure of brain 5-HT to rise further beyond levels observed at brain [Trp] of ⩾ 79 µM by doses of Trp of ⩾ 25 mg/kg body weight in rats. 245 Although p-chlorophenylalanine irreversibly inhibits TPH thereby decreasing 5-HT synthesis, it also inhibits the production of IFN-γ in human blood cells at a relatively low concentration (5 µM), 246 an effect likely to weaken or block IDO1 induction by this cytokine. Whereas microbiota in ruminants play a significant role in Trp metabolism, the indole metabolites produced can collectively induce a significant activation of the AhR and a consequent induction of IDO. However, in non-ruminant Trp-sensitive species, the role of gut microbiota and/or IDO in Trp toxicity is less clear and thus requires investigation. A rank score of IDO1 activity in the limited comparative studies summarised in Table 5 suggests that the 3 Trp-sensitive species (gerbil = rabbit > guinea pig) possess higher IDO activity than the 2 Trp-tolerant species (rat > mouse). Heyes et al 243 showed that basal IDO activity in gerbil lung is 13.2-fold greater than that in rats and that IDO induction by lipopolysaccharide (LPS: endotoxin) is 153-fold greater in gerbil. From this limited information, it can be suggested that IDO may play a role in Trp toxicity in Trp-sensitive species, but more comparative studies in both sensitive and tolerant species are required before this possibility can be established with certainty.
N′-Formylkynurenine formamidase
Formamidase exists mainly in liver, kidney and brain, with a Km of 50 to 180 µM, 8 and is widely distributed across species, including flies, 190 mosquitos 192 cattle and rainbow trout. 247 In mice, formamidase activity is high in liver, almost double that in kidney and deletion of its gene almost completely abolishes its activity in liver and reduces it by 87% in kidney. 248 Deletion, however, does not inactivate some formamidase variants or enzymes with formamidase activity, as suggested by their presence in Drosophila melanogaster, 249 pig liver 250 and mouse brain. 251 Deletion of the formamidase gene does not influence plasma [Trp]. 248 Inhibition studied of the liver enzyme from rainbow trout and cattle 247 show that Trp is a poor inhibitor, whereas Kyn and 3-HK cause significant inhibition at 50-150 µM. By contrast, certain metal cations are powerful inhibitors at 1 mM. In rainbow trout, HgCl2 and ZnCl2 cause 84% and 95% inhibition, whereas in cattle, ZnCl2, BaCl2 and CaCl2 cause 50%, 81% and 86% inhibition respectively. Studies with purified formamidase from adult chicken liver show that Ag+ is the strongest inhibitor, causing 89% inhibition at 10 µM. 252 It is currently unknown if formamidase inhibition by metal cations can paradoxically lead to elevation of Kyn and KA levels, as is the case with the organophosphate insecticide diazinon, which causes 5-fold elevation of plasma [Kyn] and greatly increased urinary [KA] and [XA] and to a lesser extent urinary [QA], 253 suggesting increased transamination of Kyn to KA and of 3-HK to XA and that, in view of the relatively greater affinity of KMO for Kyn and of kynureninase for 3-HK (14-25 and 77 µM, respectively), greater conversion of kynurenine to the immunosuppressive metabolites 3-HK, 3-HAA and QA can be expected with formamidase inhibition.
Other enzymes of the Kynurenine pathway
The 2 studies in Table 5 comparing liver KMO activity across 5 species suggest the following decreasing order: gerbil > rat = guinea pig > mouse > rabbit. In liver, KMO activity is also higher in rats > cats. Other tissues contain lower enzyme activity and, in lung, the enzyme is highest in mouse > rat = gerbil > rabbit. Thus, there is no distinctive pattern of differences in KMO between Trp-sensitive and –tolerant species. KMO is the gateway for production of 3-HK, 3-HAA and QA and also, by inference, NAD+. Its activity is therefore vital for immune, neuronal and other cellular activities. Also, as shown in Table 5, little information is available to establish a distinction between Trp-sensitive or – tolerant species in activities of kynurenine aminotransferase, kynureninase or 3-hydroxyanthranilic acid 3,4-dioxygenase, perhaps because of the different experimental conditions between studies. The only exception is the lower activities of TDO, KAT and KMO in cats, relative to those in rats. 179 Only activity of hepatic 2-amino-3-carboxymuconate-6-semialdehyde decarboxylase (ACMSD) (picolinate carboxylase) has been compared across 14 species in a single study by Ikeda et al 5 Assigning a score of 1 for the human ACMSD activity in Figure 4, of the species studied by Ikeda et al 5 the rat enzyme exhibited only ~50%, whereas that of toad and hamster was comparable to that, of the human enzyme. All other 10 species listed exhibited greater enzyme activities than humans. In particular, frog < lizard < cat showed the highest activities. ACMSD is an important KP enzyme, as its activity controls the progress of the pathway towards QA and hence NAD+ synthesis or towards picolinic acid (PA) and eventual formation of acetyl CoA. Inhibition of ACMSD in humans by phthalates increases QA production and its urinary excretion. 254 The particularly higher ACMSD activity in the above 3 species renders them unable to effectively utilise Trp as a source of NAD+. In the cat, this is further compromised by the lower activity of 3-HAAO compared to the rat, though the 2 species do not differ in quinolinate phosphoribosyltransferase (QPRT) or AMS dehydrogenase. 5

Ranking of ACMSD activity among species.
Urinary metabolite excretion patterns can provide indirect pointers to enzyme activities. In rat strains, urinary excretion of Trp metabolites of the KP is similar among the Wistar, heterozygous Gunn and Sprague-Dawley strains and higher than in Long-Evans, consistent with the lower TDO activity in the latter strain. 187 Data on urinary excretion of kynurenine and other KP metabolites were compared in 4 species. 255 Urinary kynurenine levels were highest in dog > man > cat > rat (23.6, 14.3, 1.4, and 0.5 µmol/day respectively: n = 5, 9, 2 and 5, respectively). After Trp loading, kynurenine metabolite excretion as a % of the administered dose was highest in dog = rat > man > cat, thus further confirming the low TDO activity in cats.
Species differences in efficiency of tryptophan conversion to niacin and NAD+
In mammals, Trp is a more efficient precursor of NAD+ than the 2 forms of niacin, nicotinamide and nicotinic acid.256,257 The elegant work of the group of D. A. Bender on kinetic and other activities of enzymes involved in NAD+ synthesis from its various precursors has established the mechanisms behind this concept (see Badawy 8 for discussion and references). As discussed above, ACMSD activity is a major determinant of the progress of the KP towards QA and NAD+ synthesis in the de novo pathway or diversion towards PA and eventual formation of acetyl CoA. As the cat possesses the greatest ACMSD activity among species, its ability to form NAD+ from Trp is severely impaired, with its production of nicotinic acid ribonucleotide from 3-HK being only 11% of that of the rat, 5 hence its reliance of exogenous niacin in the Preiss-Handler pathway from nicotinic acid and/or the salvage pathway from nicotinamide. Trp is also a poor precursor of NAD+ synthesis in the channel catfish Ictalurus punctatus, 258 the rainbow and brook trout and many other salmonid species, due to their high ACMSD activity139,259,260 Other than frog, (see above), very little is known about ACMSD activity or efficiency of Trp conversion to NAD+ in other Trp-sensitive species.
Species Differences in the Serotonin Pathway
As far as we could ascertain, little work has been published on species differences in Trp metabolism by pathways other than the KP. Only the serotonin pathway has received some attention and available comparative data are discussed below. No attempt will therefore be made here to consider the tryptamine or indolepyruvate pathways, nor microbiota Trp metabolism.
While serotonin is synthesised from Trp in a whole range of species and performs important functions in many physiological processes, including mood in humans, 148 aggression in vervet monkey, 87 injurious behaviour in pigeon, hens and parrots 150 or catecholamines metabolism in rats, 149 surprisingly little comparative studies of serotonin pathway activity across species or strains thereof have been reported and these are mainly concerned with response of the pathway to manipulations, such as stress. 261 alterations in dietary Trp, 262 monoamine transporters in humans, rats and mice, 263 behavioural variability in Medaka fish strains 264 and mouse strain differences in serotonin-specific reuptake inhibitor sensitivity. 265
Table 6 summarises data published on species content of brain Trp and its 5-hydroxyindole metabolites 5-HT and 5-HIAA and a few species or strain comparisons. It is clear that much information is required to fill in the gaps in our knowledge of serotonin metabolism in various species. The brain Trp concentrations in rats and gerbils reported by Green et al 182 appear excessive, at least in rats, compared to other data in the Table. Whereas no clear distinction exists between Trp-sensitive and – tolerant species in 5-HT metabolism, the data in Table 6 suggest provisionally that teleosts form less 5-HT than vertebrates and this appears consistent with their lower plasma [Trp]. In the absence of more detailed information, no other conclusions can be drawn from the data in Table 6, particularly given that brain 5-HT levels are controlled by several mechanisms, including brain [Trp] and activities of TPH2, ALAAD and MAO. TPH2 is widely distributed across species, even if its activity varies with species, for example, the rat brain TPH2 is more active than that of human or chimpanzee. 272 TPH2 activity is, however, limited mainly by brain [Trp] because of the partial saturation of the enzyme with its substrate. ALAAD activity also varies across species. Thus, in a detailed study by Robins et al 10 the brain enzyme activity in decreasing order was as follows: guinea pig > mouse > hamster > rat > rabbit > chicken > squirrel monkey > cat > turtle > carp > Macaca monkey > Cynomolgus monkey, with the human brain enzyme activity being lower than the last of these species. By contrast with the brain, the liver ALAAD is most active in rat > mouse > guinea pig > hamster > rabbit, 10 with the human enzyme activity averaging just below that of turtle. With MAOA, the subtype that prefers 5-HT as substrate, few studies of species differences have been published, for example, gene functional variability in macaque species in relation to aggressive behaviour 273 and oxidative capacity of the enzyme in human, rabbit, rat, mouse, bovine and subhuman primate liver. 274
Concentrations of tryptophan and its 5-hydroxyindole metabolites in brains of various species.
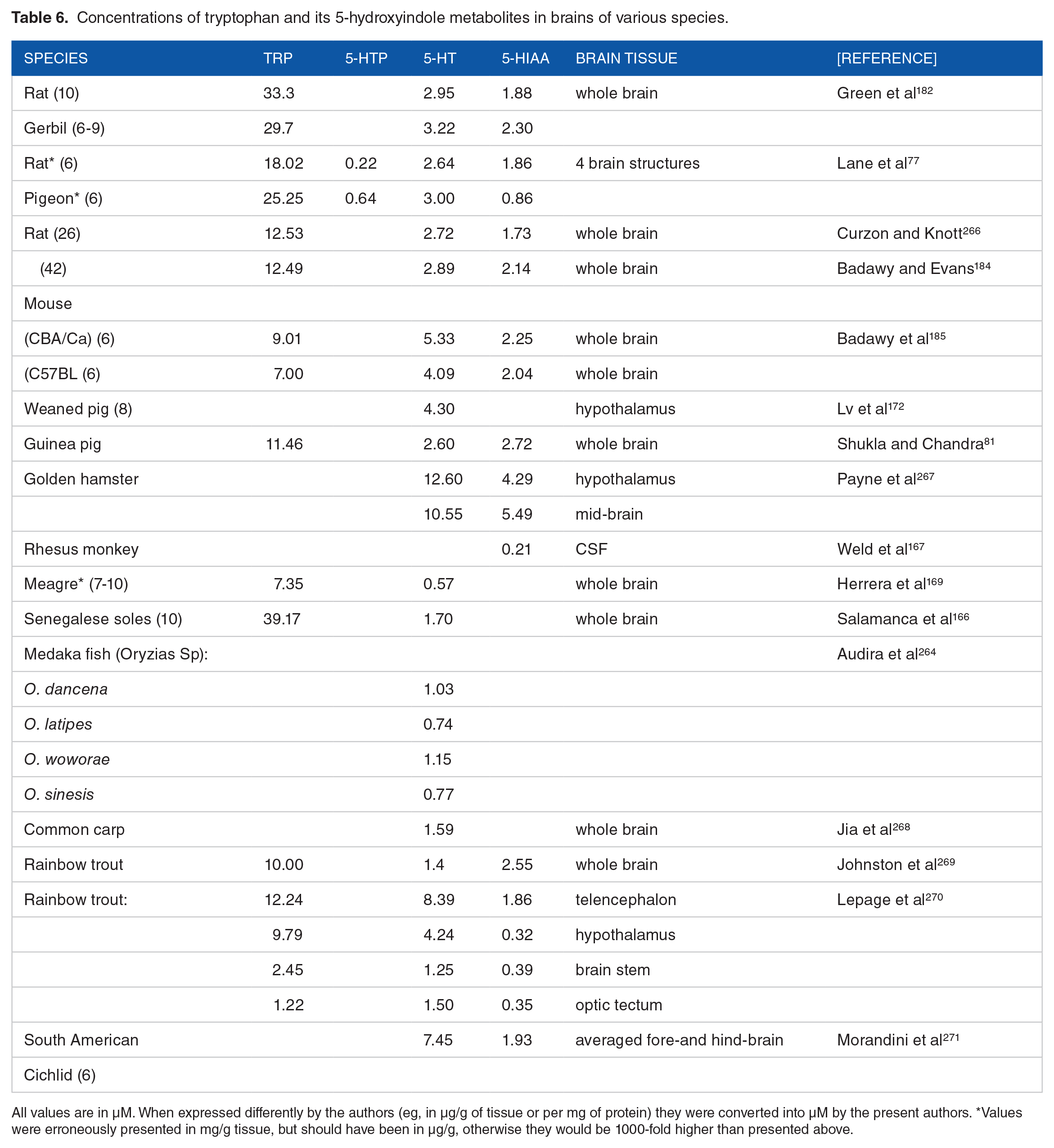
All values are in µM. When expressed differently by the authors (eg, in µg/g of tissue or per mg of protein) they were converted into µM by the present authors. *Values were erroneously presented in mg/g tissue, but should have been in µg/g, otherwise they would be 1000-fold higher than presented above.
Conclusions and Comments
Whereas the present review has provided an updated comparison of tryptophan metabolism and disposition across a range of animal species, it has exposed the many gaps to be addressed in our knowledge in this area in many species. It is hoped that this account will provide a platform and a stimulus for further studies of species differences in Trp metabolism. A number of aspects are noteworthy in this regard. The wide species variations in plasma albumin can predict likely levels of Trp binding and availability to tissues of many species. In wild carnivores, much of plasma Trp can be expected to be albumin-bound. In aquatic species, the low levels or absence of albumin suggests a largely unimpeded entry into tissues of environmental pollutants: a vulnerability that reinforces the need for stringent safeguards against industrial pollution. A limited number of species have been shown to be sensitive to toxicity of excess Trp due to their lack the free apoenzyme form of Trp 2,3-dioxygenase and its glucocorticoid induction mechanism. While wild carnivores are almost certain to tolerate excess Trp by virtue of their Trp-rich prey diet, many aquatic species could be vulnerable to Trp. Because aquatic species are particularly vulnerable to the harmful effects of chemical pollutants, the AhR has received much attention in immune studies in these species. However, by contrast, little is known about changes in IDO1 elicited by the immune insults induced by AhR activation by chemicals. The potential role of IDO in Trp toxicity requires assessment in comparative studies. The importance of the kynurenine pathway in species physiology further extends to synthesis of the vital cellular effector NAD+ with ACMSD activity playing a decisive role in this process. Nutritional studies in species should explore the role of this enzyme. 85
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AA-BB received no financial support for the research, authorship, and/or publication of this article. GJG is supported by the National Health and Medical Research Council (NHMRC) and Macquarie University.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AA-BB wrote the first draft. AA-BB and GJG revised it and approved the final version.
AA, anthranilic acid; ACMSD, 2-amino-3-carboxymuconic acid-6-semialdehyde decarboxylase; ALAAD, aromatic L-amino acid decarboxylase; 3-HAA, 3-hydroxyanthranilic acid; 3-HK, 3-hydroxykynurenine; 5-HIAA, 5-hydroxyindoleacetic acid; 5-HT, 5-hydroxytryptamine or serotonin; IDO, indoleamine 2,3-dioxygenase; IAA, indol-3-ylacetic acid; IPA, indol-3-ylpyruvic acid; KA, kynurenic acid; Kyn, kynurenine; NAD+, oxidised nicotinamide-adenine dinucleotide; PA, picolinic acid; PLP, pyridoxal 5′-phosphate; QA, quinolinic acid; ROS, reactive oxygen species; Trp, L-tryptophan; TDO, Trp 2,3-dioxygenase; TPH, Trp hydroxylase; XA, xanthurenic acid.
