Abstract
Immune system activation and inflammation accompanies immune dysfunction in trauma and sepsis patients. Immunodeficiency may develop in such patients as one consequence of an activated chronic pro-inflammatory response. According to recent data, degradation of L-tryptophan (TRP) via the kynurenine (KYN) pathway by the cytokine-inducible enzyme indoleamine 2,3-dioxygenase (IDO) could represent an important contributor to the deficient responsiveness of immunocompetent cells. Compared to healthy controls, patients post trauma or with sepsis had increasing KYN concentrations and KYN to TRP ratios (KYN/TRP) whereas TRP concentrations decreased. Likewise, concentrations of cytokines tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) and of immune activation marker neopterin increased in patients (all p < 0.001). Furthermore in patients KYN/TRP, KYN and neopterin concentrations were further increasing (all p < 0.001), whereas the changes of TRP, TNF-α and IL-6 concentrations were not significant. Compared to the survivors, the non-survivors had a higher concentration of KYN, neopterin, TNF-α and IL-6 as well as a higher KYN/TRP ratio. KYN/TRP correlated with neopterin (p < 0.001) and also with TNF-α (p < 0.01) and IL-6 concentrations (p < 0.05) and inversely with the in vitro response of stimulated monocytes. We conclude that increased TRP degradation in patients post trauma is closely associated with immune activation. Cytokines released during the pro-inflammatory response may induce the activity of IDO and thus accelerate TRP degradation. Thus, increased IDO activity most likely represents a result of host response to pro-inflammation in patients. Data support a possible role of inflammation-induced IDO in the diminished immunoresponsiveness in patients.
Introduction
Trauma is the main cause of death for people below the age of 40 years. 1 Multiple trauma—which is synonymous to severe trauma, major trauma or polytrauma—is defined as physical insults or injuries occurring simultaneously in at least two different body regions. The sum of those injuries represents a life threatening situation. In literature the Injury Severity Score (ISS) of more than 16 points is widely used to define a multiple trauma. 2 After a huge physical impact like a trauma, destroyed tissue needs to be cleared and this leads to an activation of several immune system compartments and cytokine cascades followed by a systemic inflammatory response. 3 This reaction, often accompanied by a counter-regulation, diminishes immune function and can lead to immune paralysis (Fig. 1). 4 The extent of this immune dysfunction is crucial for the outcome of the patient.

Schematic drawing of immunoregulatory events that are critically involved in the pathogenesis of a systemic inflammatory response syndrome (SIRS) and sepsis with increased production of pro-inflammatory cytokines like interleukin (IL)−1, −6, −8 an tumor necrosis factor-α (TNF-α). SIRS and sepsis are often followed by severe immunosuppression due to a compensatory anti-inflammatory reaction syndrome (CARS) which involves overproduction of anti-inflammatory cytokines IL-10, −4, −13, −10 and reduced functional immunoresponsiveness as is indicated by reduced human leukocyte antigen (HLA)-DR expression and diminished TNF-α release upon exposure to lipopolysaccharide (LPS).
Monocytes play a key role in the innate immune system. After activation for example by lipopoly-saccharid (LPS) via toll-like receptors (TLR) or by destroyed tissue, monocytes produce pro-inflammatory cytokines such as interleukin-(IL)-1, IL-6, IL-8 and IL-12, interferon-γ(IFN-γ), transforming growth factor-β (TGF) and tumor necrosis factor-α (TNF-α) as well as prostaglandins, complement and neopterin (Fig. 3). After trauma, the TNF-α concentration in patients' is reduced.5,6 Furthermore, diminished TNF-α production in blood following ex vivo LPS stimulation is a very important predictive marker in traumatic patients and more sensitive than other markers in predicting unfavorable outcome. 7 In a similar way, IFN-γ-production in stimulated peripheral blood mononuclear cells of patients as compared to controls and this deficient IFN-γ-response is considered to be crucially involved in poor survival expectations. 4 But notably, IFN-γ deficiency is often observed only upon in vitro-stimulation of cells and is paralleled by signs of activated immunity in vivo. 8
IFN-γ, next to other effects, strongly induces the enzyme indoleamine 2,3-dioxygenase (IDO). IDO catalyzes the initial and rate-limiting step of tryptophan (TRP) to kynurenine (KYN). 9 This TRP degradation by IDO inhibits the growth of viruses, bacteria and parasites but also tumor cells10,11 because TRP is the least available and therefore most important essential amino acid for their growth. In trauma patients, a higher IDO activity not only leads to a decline in lymphocyte counts but is also associated with an unfavorable outcome.12,13
In patients with trauma and sepsis increased levels of IL-6, TNF-α and neopterin are also well documented to predict outcome.13–17 TNF-α enhances the IFN-γ-induced IDO activity and at the same time triggers neopterin production in a synergistic fashion. 18 Accordingly, in several groups of patients suffering from different diseases like infections, auto-immune syndromes, cancer and trauma increased IDO activity is paralleled by higher levels of neopterin.12,13,19–22
In a recent study we analyzed plasma samples from trauma patients and determined the amounts of TRP and KYN and calculated the KYN to TRP ratio (KYN/TRP) as an index of IDO activity. The objectives of this study were to compared those values to the plasma concentrations of IL-6, TNF-α, neopterin as well as the ex vivo production of TNF-α after LPS stimulation and finally to correlate those results with the outcome.13,23
Study
Eighteen patients were included in this study: 14 males, 4 females; aged mean ± S.D.: 45 ± 19 years, range: 20–77 years, 15 trauma patients and 3 surgical patients who developed sepsis; Apache score: 18.9 ± 6.75 range: 8–34; ISS: 39 ± 13.1, range: 18–57. Patients were either admitted to the Intensive Care Unit (ICU) of the Lorenz Boehler Trauma Center or the ICU of the Medical University of Vienna. Samples were collected every third day during the first 14 days of follow up. For statistical analyses, samples were divided into five groups: one sample collected between days 1–2 of each patient was referred to as group I, day 3–5 = group II, day 6–8 = group III, days 9–12 = group IV, and days 13–14 = group V. In total, every patient contributed 3–5 plasma samples to the total number of 84 sera analyzed. 23
Samples from 49 healthy donors (21 women and 28 men; age, 35 ± 13.5 years) served as a reference group for kynurenin, tryptophan and neopterin. 20 A different group of healthy volunteers served as a reference group for TNF-α, IL-6 and ex vivo LPS stimulated TNF-α as published recently. 7 The study was performed according to the Helsinki declaration. The protocol was approved by the local ethics committee, and written consent was granted by the next of kin. The observed values of all study parameters had no influence whatsoever on the course of the therapy.
Concentrations of TRP and KYN were determined by high performance liquid chromatography (HPLC) as described before using 3-nitro-L-tyrosine as external standard.24,25 Tryptophan was detected by its natural fluorescence at an excitation wavelength of 285 nm and an emission of 365 nm. UV absorption of both compounds, KYN and L-nitrotyrosine, was monitored at a wavelength of 360 nm. KYN/TRP was calculated and expressed as μmol KYN per mmol TRP. Neopterin was measured by ELISA (BRAHMS, Hennigsdorf, Germany) according to the manufacturer's instructions with a detection limit of 2 nM. Serum levels of TNF-α and IL-6 were measured with Immulite semi-automated chemoluminescent immunoassay analyzer (DPC, Los Angeles, CA; detection limits were 1.7 pg/mL for TNF-α and 2 pg/mL for IL-6). Ex vivo stimulation of monocytes with LPS was performed with a commercially available test kit (Ex vivo Stimulation Kit, Milenia Biotec GmbH, Bad Nauheim, Germany), 7 and measurement of TNF-α on the Immulite semi-automated chemiluminescent immunoassay analyzer (DPC, Los Angeles, CA).
Statistical analysis was performed with the statistical software package SPSS for Windows (version 17.0; SPSS Inc., Chicago, Ill) applying non-parametric tests such as Kruskal Wallis-test and Mann Whitney U-test and Spearman's rank correlation coefficients (rs) were calculated, p-values < 0.05 were considered statistically significant.
Results
Concentrations of TRP were decreased in all trauma patients at all time compared to the normal range in healthy controls, whereas KYN/TRP and concentrations of KYN were increased in patients (Table 1). Also plasma concentrations of neopterin, TNF-α and IL-6 were increased in patients compared to healthy controls, whereas TNF-α production after ex vivo LPS stimulation was decreased (Table 1). However, TNF-α was detectable in less than half of the specimens. 23
Concentrations of Interleukin-6, constitutive tumor necrosis factor-α (TNF-α), TNF-α production after ex vivo lipopolysaccharide (LPS) stimulation, tryptophan, kynurenine and neopterin and of kynurenine to tryptophan ratio (KYN/TRP) (mean ± S.D., range in brackets; data from all patients from all groups) in patients post-trauma and/or with sepsis (18 patients, 84 measurements) and laboratory reference values of healthy controls (Ref. 13 and 23).
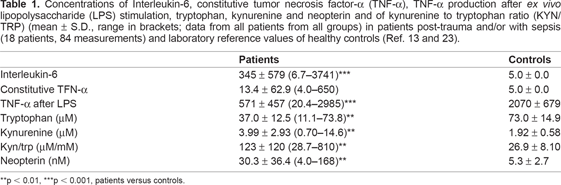
p < 0.01,
p < 0.001, patients versus controls.
Patients who died during follow-up did not differ from survivors regarding age, gender, main diagnoses, length of stay in ICU, their Apache II scores or trauma scores ISS. However, those who died presented with significantly higher KYN/TRP at several time points (Fig. 2).
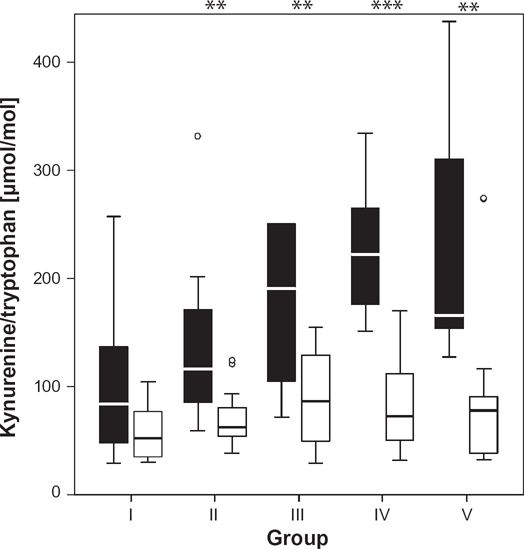
Serum kynurenine to tryptophan ratio in 18 patients with sepsis after trauma or surgery. Data are presented as box plots with median values and interquartile ranges. The black boxes show non-survivors and the white boxes show survivors. Circles indicate extreme values, asterisks indicate significance differences between survivors and non-survivors. **p < 0.01, ***p < 0.001.
Discussion
In patients with sepsis after trauma or surgery there occured significant alterations of TRP metabolism as compared to healthy controls: TRP concentrations were decreased whereas KYN concentrations and KYN/TRP were elevated in patients. Moreover, a higher rate of KYN accumulation and a higher KYN/TRP and were predictive for a bad outcome. Also neopterin, TNF-α and IL-6 concentrations were higher in non-survivors compared to survivors at several time points during follow-up. Ex vivo TNF-α production after LPS-stimulation was significantly decreased in non-survivors compared to survivors. In the whole data set, there existed a positive correlation between KYN concentrations and neopterin as well as KYN/TRP and neopterin. KYN/TRP correlated also with TNF-α (all p < 0.001). TNF-α correlated with neopterin and IL-6 (both p < 0.001; data not shown). These results agree well with earlier observations.13,23
Low TRP concentrations are unlikely to be related to a reduced dietary intake of this essential amino acid, because patients receiving standardized enteral or parenteral nutrition at the ICU. Increased IDO activity may indeed account for lowered TRP concentrations, and seems likely to be induced by pro-inflammatory stimuli, of which IFN-γ together with TNF-α are considered to be the strongest inducer of IDO in macrophages and dendritic cells. Immunocompetent cells respond against non-self structures and produce cytokines including IFN-γ and TNF-α which are aimed at halting growth of pathogens. Thus, an overwhelming Th1-type immune activation may be the cause for the enhanced IDO activity (Fig. 3). In our data several strong correlations were observed between concentrations of pro-inflammatory cytokines and of neopterin and the increase of KYN/TRP which supports our conclusion.
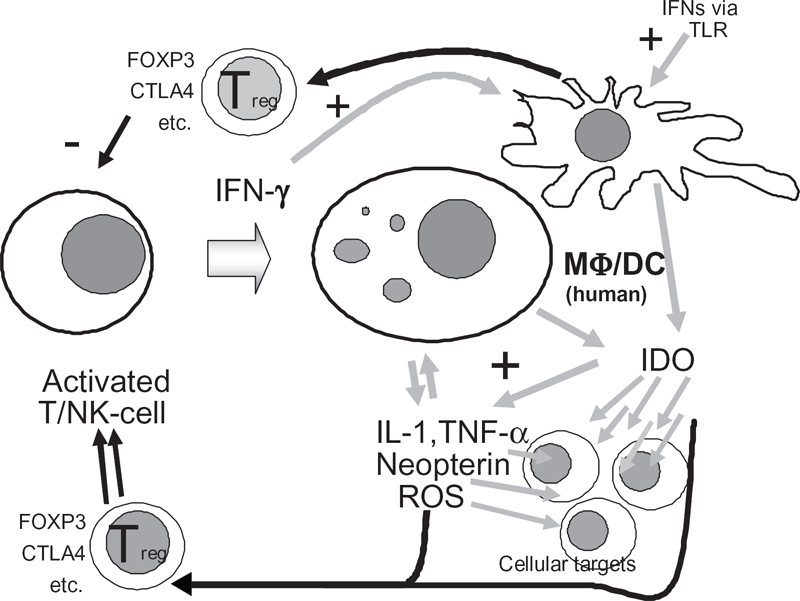
Within the systemic inflammatory response syndrome (SIRS) and sepsis pro-inflammatory cytokine interferon-γ (IFN-γ) is released by mainly T-cells and natural killer (NK) cells. Among other pro-inflammatory stimuli, IFN-γ strongly stimulates production of additional cytokines like interleukin-1 (IL-1) and tumor necrosis factor-α (TNF-α), neopterin and reactive oxygen species (ROS) in monocyte-derived macrophages (MΦ) and dendritic cells (DC), aimed at suppressing growth of infectious agents (gray arrows). In parallel antiproliferative enzymes like indoleamine 2,3-dioxygenase (IDO) are expressed. In turn, anti-proliferative action also affects immunoresponsiveness and thus a suppressed immune system status is reached. Abbreviations: FOXP3, forkhead box P3; CTLA4, Cytotoxic T-Lymphocyte Antigen 4.
Activation of IDO is one out of several important antimicrobial mechanisms triggered by IFN-γ. However activated IDO does not only affect microbes, it potentially may also counteract growth and development of T-cells. 26 TNF-α was shown earlier to enhance TRP degradation and neopterin production in monocyte-derived macrophages in a synergistic way to IFN-γ. 18 In our patients we found a significant correlation between KYN/TRP and TNF-α concentrations, and an even stronger correlation was found between KYN/TRP and neopterin. The accelerated TRP degradation and the increased production of neopterin and pro-inflammatory cytokines were found to be associated with a reduced TNF-α production of monocytes upon ex vivo stimulation with LPS. Probably the permanent stimulation and cytokine production during disease progression renders these cells refractory to further stimulation. 8 In fact, activation of antiproliferative biochemical pathways as part of the effector functions of a Th1-type immune response (Fig. 3) would well fit to the common finding of an immunosuppressed state in patients suffering from chronic inflammatory conditions.
TNF-α was below detection limit in more than half of the patients, and circulating IFN-γ concentrations in serum/plasma of patients are usually even less detectable than TNF-α. That is why we refrained from analyzing IFN-γ concentrations in our patients. These cytokines rapidly stick to their specific receptors on target cells or to their shed soluble forms, so their low concentrations in serum/plasma are limiting the diagnostic application. TNF-α and IL-6 concentrations were higher in non-survivors compared to survivors already at baseline confirming earlier results of others.14,15 TNF-α concentrations predicted mortality throughout the later post-traumatic course, whereas IL-6 concentrations were less predictive. Still, IL-6 concentrations correlated strongly with TRP degradation. In addition, a rather strong relationship existed between TNF-α and neopterin concentrations. Overall, the changes of KYN/TRP and of KYN, neopterin, TNF-α and IL-6 concentrations post trauma showed similar courses in all non-survivors, and they seemed to reflect the pro-inflammatory cascades quite well, which were related to overwhelming production TNF-α and probably also of IFN-γ. Thus, pro-inflammatory cytokines seemed to trigger those critical events probably leading to death, and TRP degradation via IDO could indeed represent one of the most important players. This is a surprising result, because at first glance one might expect that a higher degree of immune response should be in favor of increased patients' survival, when, e.g. clearance of pathogens might be enhanced by the antimicrobial mechanisms, but obviously the opposite is the case.
A kind of equilibrium might develop between the activation degree of immunocompetent cells, their suppressive effect on microbes and also the effect on themselves. In some patients the bactericidal effect of IDO will be able to stop the infectious process, in other patients immunocompetent cells are suffering more from IDO activity than microbes. Furthermore, depletion of T-cells and immunosuppression is a consequence of the host's immune response against invading pathogens. In fact we also observed a negative relationship between the capacity of monocytes to respond to LPS stimulation and KYN/TRP. This data is supporting the concept that immunocompetent cells of patients after trauma and sepsis are somewhat exhausted from their chronic activation in patients rather than being per se refractory. But certainly this study is too preliminary to give a final answer to this question.
From in vitro studies it was concluded that not only the decline of TRP concentrations but also the increase formation of toxic TRP catabolites could be involved in the development of T-cell unresponsiveness.26,27 Interestingly in our study, only the increase of KYN concentrations but not the decline of TRP was significantly associated with the outcome of patients. Supplementation of TRP via standard enteral or parenteral nutrition was initiated early in all our patients. This may have counteracted to some degree the loss of TRP despite further accelerating degradation of the amino acid and further increase of KYN concentrations. Still, data may favor a role of TRP catabolites, 26 rather than TRP lowering in the immune deviations which might be of relevance for the fatal outcome of some of our patients. KYN and its downstream metabolites are able to modulate redox chemistry: Recently, KYN was found to influence the lipid biosynthesis in rat liver cells in a rather pro-oxidative way. In fact, the influence was similar to macrophage product neopterin which shifts biosynthesis away from unsaturated towards saturated fatty acids. 29 The property of TRP catabolites to shift redox equilibria has also been demonstrated when the chemoluminescence output from H2O2- and hypochlorite-containing aqueous solutions was compared between samples with and without kynurenine and other tryptophan catabolites. 30 Such findings could relate also to the pro-apoptotic nature of these chemicals. 27
Conclusion
Accelerated TRP degradation and higher concentrations of neopterin and TNF-α are prognostic factors for shortened survival in patients post trauma, which confirms and extends earlier data. Tryptophan degradation may represent one important aspect in the development of the post traumatic failure of the immune system to respond appropriately. More extended studies are needed to examine the impact of TRP degradation and its relationship with cellular immune activation and the potential prognostic value of TRP metabolism in patients suffering from trauma, which should be helpful to define new therapeutic intervention strategies.
Finally, these observations on TRP catabolism in patients with trauma and sepsis may just reflect the special role of pro-inflammatory cascades which contribute to the fate of such patients and may be regarded as one out of many antiproliferative effector functions which are initiated in such clinical conditions.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
Footnotes
Acknowledgements
We thank Miss Astrid Haara and Miss Maria Gleinser for excellent technical assistance.
