Abstract
Although tryptophan (Trp) and its metabolites, such as serotonin (5-HT) and kynurenines (KYNs), are strong modulators of emotional behavior, the metabolic pathway(s) responsible for this physiological modulation is not fully understood. Two of the initial rate-limiting enzymes of the kynurenine pathway for Trp metabolism are known: tryptophan 2,3-dioxygenase (TDO) and indoleamine 2,3-dioxygenase (IDO). Based on our comparison of tdo-deficient (Tdo–/–) mice with their wild-type littermates, we report that TDO is the physiological modulator of systemic Trp, brain Trp and serotonin (5-HT), and, therefore, anxiety-related behavior. Tdo–/– mice showed increased plasma concentrations of Trp (about 10-fold) and its metabolites 5-hydroxyindoleacetic acid (5-HIAA) and kynurenine, as well as increased levels of Trp (about 20-fold), 5-HT and 5-HIAA in the hippocampus and midbrain. The Tdo–/– mice also showed anxiolytic modulation in the elevated plus maze and open field tests, and increased neurogenesis during adulthood, as evidenced by double staining with 5-bromo-2‘-deoxyuridine (BrdU) and neural progenitor/neuronal markers. TDO also plays a role in the maintenance of brain morphology in adult animals by regulating neurogenesis in the hippocampus and subventricular zone. Collectively, our results in Tdo–/– mice indicate a direct molecular link between Trp metabolism and mental status in mice. Tdo–/– mice will likely prove useful both in identifying the physiological role of Trp metabolism in normal brain function and in psychiatric disorders and in developing new therapeutic interventions for mental disorders. In addition, the potential role(s) and molecular mechanisms of TDO in metabolic mental disease(s) and in emotional behavior are discussed.
Keywords
Introduction
Mood and its dysregulation, anxiety-related disorders in particular, have become increasingly important medical and social issues. 1 Although some have proposed that tryptophan (Trp), an essential amino acid, plays a role in mood disorders, the molecular mechanisms linking Trp and anxiety-related behavior are yet to be fully understood. The association of Trp and its metabolites with these conditions suggests that regulation and dysregulation of enzyme(s) in the Trp metabolic pathways may play a key role in the molecular mechanisms responsible for mood disorders.
Since the isolation of kynurenine (Kyn) by Kotake and his colleagues in 1927, Trp metabolism has been extensively studied (Fig. 1). Three major Trp metabolic pathways have been described. One is the so-called serotonin pathway, which includes serotonin (5-hydroxytryptamine, 5-HT) 2 and 5-hydroxyindoleacetic acid (5-HIAA) (Fig. 1A). The first and rate-limiting enzyme in this pathway is tryptophan hydroxylase (TPH; tryptophan 5-monooxygenase) (EC 1.14.16.4). Fujisawa and his colleagues purified two types (isoforms) of TPH from the brain and mastocytoma (and the pineal gland), TPH2 (brain-specific isoform of tph 3 ) and TPH1, respectively, and these two types showed slightly different characteristics.4,5 Loss-of-function-type tph2 gene mutations reportedly are associated with depression, and this variation could influence personality traits and disorders related to emotional dysregulation.6,7 The regulatory mechanisms of TPH by aquayamycin and phosphorylation of TPH by Ca2+/calmodulin-dependent protein kinase II (CaM-kinase II), cAMP-dependent protein kinase (PKA) and protein kinase C (PKC) have been extensively studied by Hayaishi, Fujisawa and his colleagues.4,8,9 However, further elucidation of the basic contribution of this pathway and its regulatory mechanism(s) might be crucial for understanding the physiological and pathophysiological role of TPH on mood and emotion. The second Trp metabolic pathway is the transamination pathway, which includes indolelactic acid (ILA) and indoleacetic acid (IAA) (Fig. 1B). Only a small proportion of dietary Trp is used in protein synthesis or in the 5-HT and transamination pathways, and most Trp (>95%) is metabolized via the Kyn pathway predominantly in the liver under normal physiological conditions (Fig. 1C). 10 The initial and rate-limiting enzyme in the pathway, which converts Trp to N-folmylkynurenine, was initially isolated by Kotake et al from rabbit liver, and was termed tryptophan pyrrolase (EC 1.13.11.11). 11 The enzyme was later called tryptophan 2,3-dioxygenase (TDO) after Hayaishi et al discovered its ability to incorporate atmospheric molecular oxygen into N-formylkynurenine.12–15 Mammalian full-length tdo cDNA was initially cloned from the rat liver cDNA library as an open reading frame of 1218 bp consisting of 406 amino acids by Nakamura and his colleagues. 16 The tdo gene is predominantly expressed in the adult liver by terminally differentiated hepatocytes. In addition to TDO, indoleamine 2,3-dioxygenase (IDO) (EC 1.13.11.52) from rabbit small intestine, which converts Trp to N-formylkynurenine (Fig. 1), 17 was cloned in 1990. 18 IDO is thought to function outside the liver including sensitive responses to immune signals under highly specific conditions, such as pregnancy and autoimmune diseases (see reviews:13,19,20), and the contribution of IDO in controlling systemic Trp concentrations has not been well defined.

A schematic diagram of Tryptophan (Trp) metabolic pathways. Three major Trp metabolic pathways are also known. The conversion of Trp to 5-hydroxytryptamine (serotonin, 5-HT) and 5-hydroxyindoleacetic acid (5-HIAA) via the serotonin (5-HT) pathway (
By comparison with the 5-HT pathway, the physiological/pathophysiological role of the Kyn pathway in emotional/psychiatric behavior and the role of Trp metabolism is poorly understood. However, in addition to the Trp metabolites 5-HT, Kyn are neuronal modulators in the central nervous system (CNS), 10 and the Kyn pathway is now considered a potential target for the development of novel drugs for neurological diseases.21,22 In comparisons with IDO, TDO has long been considered a more important enzyme in the physiological regulation of Trp in the body, and antidepressants have been postulated to act by directly inhibiting the activity of TDO, while TDO inhibitors are considered useful for the treatment of depression when co-treated with Trp.23,24 Thus, we used studies of tdo-deficient mice (Tdo–/– mice) to assess the role of TDO under physiological conditions in systemic Trp, brain Trp and 5-HT synthesis, brain development/morphology, neurogenesis (all of which has been suggested to relate to emotional/psychiatric behaviors), and anxiety-related behavior in vivo.
Materials and Methods
Construction of the targeting vector and disruption of the tdo locus
Construction of the targeting vector and disruption of the tdo locus were performed as previously described. 25 Briefly, a 12.5-kb tdo genomic fragment containing exons 1 to 3, was used to construct a tdo targeting vector (Fig. 2) by replacing exons 1 and 2 of the tdo fragment that contained the translation initiation site with the PGK-neomycin (Neo) cassette. After backcrossing with wild-type C57BL/6 mice for five generations, homozygous tdo mutants and wild-type animals (Tdo+/+ mice) were obtained by intercrossing heterozygotes.

Generation of tdo-deficient (Tdo–/–) mice. A targeting strategy for tdo gene disruption. Exons are represented as numbered boxes (coding regions; black boxes). Neo, PGK-neomycin resistant cassette; DT-A, diphtheria toxin-A.
Measurement of amino acids in plasma and brain
Plasma was deproteinated with 5% sulfosalicylic acid followed by centrifugation. Hippocampus and midbrain were rapidly micro-dissected after perfusion with ice-cold Hanks’ balanced salt solution, homogenized in a solution containing 0.5 M HClO4 and 0.025% EDTA, incubated on ice for 10 min, and centrifuged at 12,000 × g and 4 °C for 20 min. The collected supernatants were filtered and either used in analysis of amino acid concentrations or stored at −80 °C until use in other analyses. 25 The amino acid concentrations in the plasma and brain samples were immediately analyzed using automated ion-exchange chromatography with lithium-based buffers on a high-speed amino acid analyzer (L8500, Hitachi).
Quantitation of Trp metabolites
Plasma and brain samples were prepared and concentrations of Trp, Kyn, KYNA, ILA, IAA, 5-HT, and 5-HIAA were determined using HPLC-FD and HPLC-UV systems with a reverse-phase C18 column, as previously described.25–27
Histological and immunohistochemical analyses
Male mice were deeply anesthetized and transcardially perfused with PBS (pH 7.4), followed by perfusion with 4% paraformaldehyde (PFA) in PBS, and processed for H&E staining and immunostaining as previously described. Primary antibodies to the following proteins were used: Ki67 (DAKO), nestin (BD Pharmingen), GFAP (Glial fibrillary acidic protein; Chemicon), DCX (doublecortin; Santa Cruz), NeuN (Neuronal Nuclei; Chemicon), βIII-tubulin (TuJ1; Covance Research), MAP2 (Microtubule-associated protein 2; Sigma), and PSA-NCAM (Poly-Sialated Neural Cell Adhesion Molecule; AbCys S.A.). After washing with PBS, either Alexa Fluoro 488- or Alexa Fluoro 546-conjugated secondary antibodies and the nuclear counter-staining reagent TO-PRO-3 iodide (Molecular Probes) in PBS were used. The slides were visualized using an LSM 510 PASCAL confocal microscope (ZEISS).
Administration of BrdU and staining
BrdU injections and subsequent analyses were performed as previously described25,28 using an antibody specific for BrdU (Oxford Laboratory). In brief, to assess the dividing progenitor cells, 13-week-old male mice were intraperitoneally administered BrdU (4 × 75 mg/kg) every 2 h and were then sacrificed 24 h after the last BrdU injection. To determine the fate of BrdU-labeled cells, 9-week-old male mice were administered BrdU (4 × 75 mg/kg) and were allowed to survive for 28 days after the last BrdU injection. Brain sections were prepared as described above. For BrdU immunohistochemistry, sections were fixed in acetone, then were treated with 1 N HCl for 30 min at 60 °C to denature the DNA, followed by rinsing in PBS. Subsequent processes were identical to those described above.
Cell quantitation
Four weeks after the injection of BrdU, sections were prepared and the number of cells immunopositive for NeuN, TuJ1, PSA-NCAM, DCX, Nestin, GFAP, BrdU, and/or Ki67 in the hippocampus of 13-week-old Tdo+/+ and Tdo–/– male mice (n = 3 for each genotype) were quantified as previously described.25,28 In these experiments, cells were totaled for the surfaces of the GCL and SGZ in the DG in one of at least five sections per animal (n > 3 per group) between 1.7 and 2.06 mm caudal to the bregma, according to the method of Franklin and Paxinos. 29
Elevated plus maze (EPM), and open field (OFT) test
EPM
Tdo+/+ and Tdo–/– male mice (13–15 weeks old, n = 16 and n = 11, respectively) were tested in an EPM according to Lister 29 with slight modifications. In brief, the plus maze consisted of two open (30 × 6 cm) and two wall-enclosed arms (30 × 6 × 15 cm) connected by a central platform (6×6 cm). The apparatus was elevated 40 cm above the floor. Each mouse was placed in the central zone facing an open arm, which the animal usually would enter first. Exploratory behavior was monitored during the 5-min test period.
OFT
The OFTs for 13~15-week-old Tdo+/+ and Tdo–/– male mice (n = 22 and n = 20, respectively) were performed according to Paylor et al, 30 as follows. The open field consisted of four adjacent activity chambers (40 × 40 × 40 cm) surrounded by walls with the field lit by overhead lightning. Mice were released into the center of the field and allowed to roam the open field for 30 min. The total distance moved, time spent and the distances moved in the margins of the field (within 10 cm of the walls) and in the center zone (area >10 cm from the walls) were measured. The ratio of the distance moved in the center to the total distance moved was calculated and used as a measure of anxiety-related behavior.
Statistical analysis
Statistical analysis was carried out using StatView software version 5.0.1 (SAS Institute). A Student's t-test was used for the amino acid and histological analyses. The other data were analyzed by one-way factorial analysis of variance (ANOVA). When the P-value by ANOVA was less than 0.05, statistical significance was determined using a Fisher's protected least significant difference (PLSD) test (post-hoc test). All behavioral tests (EPM, and OFT) were conducted between 9:30 AM and 1:00 PM and all experiments were monitored by an automated video motility system and were analyzed with EthoVision Ver. 2.3.19 software (Noldus), as previously described. 24
Results
Generation of mice with targeted disruption of the tdo gene locus
Homologous recombination was used to disrupt the tdo gene in mice. 25 The targeting vector was constructed by replacing genomic tdo exons 1 and 2 (containing the translational initiation site) with the PGK-neomycin (Neo) cassette (Fig. 2). Heterozygous mice were crossed with C57BL/6 mice for five generations. Interbreeding of the resultant heterozygotes produced wild-type (Tdo+/+), heterozygote (Tdo+/–), and homozygote (Tdo–/–) mice. 25 Disruption of the tdo gene was verified by the absence of tdo mRNA transcripts and TDO protein in the liver, and the null mutation of tdo was also verified by assay of enzyme activity in liver extracts. 25 The mutant mice were born at ratios that followed Mendelian inheritance, and they matured for at least one year with no apparent gross abnormalities.
Role of TDO in systemic (plasma) Trp and its metabolites
By comparing Tdo–/– mice to their wild-type littermates (Tdo+/+ mice), we demonstrated that, in the presence of intact IDO, plasma Trp concentrations were increased approximately 10-fold in Tdo–/– compared with Tdo+/+ adult mice with no obvious differences in the concentrations of other essential amino acids (Fig. 3). 25 By contrast, no difference was seen in plasma Trp concentrations between ido-knock-out mice, 31 which possess one of the two enzymes that convert Trp to formylkynurenine17,20 (Fig. 1), and their wild-type littermates. 25 The plasma concentrations of the Trp catabolites (5-HIAA, ILA, and IAA) were also elevated in Tdo–/– mice relative to Tdo+/+ mice. 25 These results clearly demonstrated that TDO is largely responsible for controlling the systemic concentrations of Trp and, in turn, indirectly contributes to the plasma levels of Trp catabolites from both the 5-HT and transamination pathways, even in the presence of IDO under physiological conditions.

Effect of tdo deletion on systemic and brain Trp. Plasma amino acid composition and Trp metabolites in 18- to 20-week-old Tdo+/+ and Tdo–/– mice (n = 9 each). Plasma Trp concentration (
Role of TDO in brain Trp levels and brain 5-HT synthesis in the presence of intact TPH
Given that large neutral amino acids (LNAA) compete for transport across the blood-brain barrier (BBB), 10 the elevation in plasma concentrations of Trp, but not the other LNAA, and the consequent increase in the Trp/LNAA ratio in Tdo–/– mice suggested increased transport of Trp across the BBB. Indeed, based on HPLC analysis, Trp concentrations in the brain were more than 20-fold higher in Tdo–/– than in Tdo+/+ mice. By contrast, there were no obvious differences in the other amino acids, except a slight modulation in methionine concentration (Fig. 3). 25 We therefore predicted that 5-HT synthesis in the brain would be altered in Tdo–/– mice. Levels of 5-HT in the brain were higher in Tdo–/– than in Tdo+/+ mice, and those of the 5-HT metabolite 5-HIAA were nearly 5-fold higher, indicating accelerated 5-HT synthesis and turnover in these mice (Fig. 4). Because tryptophan hydroxylase (TPH) is believed to be the rate-limiting enzyme for 5-HT biosynthesis, we asked whether the mRNA content or enzyme activity of tph2 would be modulated by TDO deletion. However, the mRNA level and enzyme activity of TPH were not modified in Tdo–/– mice, indicating that TDO, rather than TPH, was the dominant regulator of 5-HT biosynthesis in vivo under physiological conditions (Fig. 4). 25
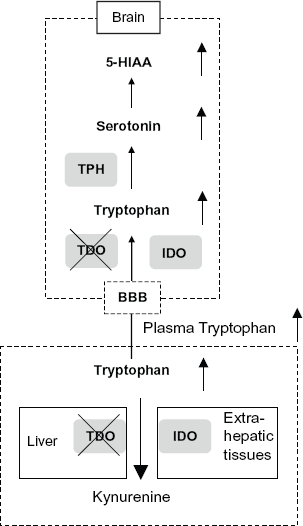
Modulation of brain Trp, 5-HT and its metabolite 5-HIAA by tdo disruption. The alteration of Trp, 5-HT and its metabolite 5-HIAA in the brain, hippocampus and midbrain of 18- to 20-week-old Tdo–/– mice. In Tdo–/– mice, the concentrations of Trp in the liver and plasma were markedly elevated by tdo disruption, and elevated plasma Trp passes into the brain through an amino acid transporter at the blood-brain barrier (BBB), thus increasing the levels of Trp in the brain. Subsequently, elevated brain Trp is metabolized to 5-HT by TPH and then to 5-HIAA.
Role of TDO in anxiety-related behavior: TDO as a molecular link between mood (anxiety) and Trp in the mice
We next assessed whether TDO deletion modulated anxiety-related behavior by analyzing anxiety-related behavior using an elevated plus maze test (EPM) and an open-field test (OFT). 25 In the EPM, adult Tdo–/– mice spent significantly more time in the open arms of the maze than the Tdo+/+ mice with no obvious differences in locomotor activity or time spent in the center zone (Fig. 5A). Moreover, in the OFT, Tdo–/– mice showed increased center locomotion and more time spent in the center zone, but there was no difference in total movement in the open field (Fig. 5B). 25 These results clearly indicate that depletion of TDO had an anxiolytic effect without affecting locomotor activity or the behavioral phenotype of Tdo–/– mice.

Modulation of anxiety-related behavior in Tdo–/– mice compared with Tdo+/+ mice.
Collectively, the results of the present study suggest that TDO is a molecular link between mood (anxiety) and Trp in mice (Fig. 6). Whether TDO(s) is a possible molecular link between mood and Trp and its metabolites in human is the next issue to be examined. The role of IDO in mood is another important issue that has yet to be examined.

TDO(s) as a possible molecular link between mood and Trp in the mice. Our results in Tdo–/– mice indicate that TDO (and its variants 33 ) is a molecular link between mood, including anxiety, and Trp and its metabolites 5-HT and Kyn under both physiological and pathophysiological conditions in mice.
Increased neurogenesis in the dentate gyrus (DG) of the hippocampus of Tdo–/– mice
To assess the mechanism by which TDO modulates anxiety-related behavior, we examined neurogenesis in the hippocampus of Tdo–/– mice. It was previously reported that the effects of stress and antidepressant treatment on hippocampal neurogenesis is parallel to behavioral changes in animal models. 32 Moreover, ablating hippocampal neurogenesis renders antidepressants inactive in behavioral paradigms used to model antidepressant response and anxiety-like behavior in mice. 28 Tdo–/– mice showed a greater number of deeply stained cells with neurite-like spines, resembling neurogenic cells, in the subgranular zone (SGZ) and granular cell layer (GCL) of the hippocampus (Fig. 7). 25 Some of these cells were immunopositive for PSA-NCAM, which is a marker for migrating neuroblasts. This result suggests that TDO modulates neurogenesis in the hippocampus. To further examine whether these cells are involved in neurogenesis, we injected mice with BrdU with/without the neurogenic markers nestin (a neural stem cell marker), GFAP (a neural stem cell marker), DCX and PSA-NCAM (neural progenitor/migrating neuroblast markers), and NeuN (a neuronal marker). Although no significant difference in the total number of cells in the SGZ and the GCL was observed between genotypes, the total number of BrdU-labeled cells was markedly higher in Tdo–/– than in Tdo+/+ mice. These results suggest enhanced proliferation in Tdo–/– mice and raise the concern that the survival rate of newly generated cells may differ between wild-type and Tdo–/– mice. Furthermore, a significant increase in BrdU-positive cells co-labeled with neural stem cell markers (nestin and GFAP), neural progenitors (DCX and PSA-NCAM) and a neuronal marker (NeuN) in the SGZ of Tdo–/– mice was detected, indicating the promotion of both neural stem and progenitor cell proliferation in the SGZ of these mice (Fig. 7). Intriguingly, these increases were greater than those observed following antidepressant injection, 28 suggesting a marked acceleration of de novo neurogenesis in Tdo–/– mice, which might be a mechanism by which anxiety-related behavior was altered in Tdo–/– mice. Of note, when we examined another brain region where neurogenesis occurs in adult mice, Tdo–/– mice showed increased proliferation of neural progenitors in the subventricular zone (SVZ), a marked reduction in the size of the lateral ventricles, and accelerated adult neurogenesis in the GCL of the olfactory bulb. These results demonstrated that neurogenesis was also accelerated in the SVZ of adult Tdo–/– mice. 25 Thus, TDO modulates neurogenesis not only in the hippocampus, but also in the SVZ.

Increased neurogenesis in the hippocampus of Tdo–/– mice compared with wild-type littermates. Paraffin-embedded brain sections (5 μm) were stained with H&E, and immunostained with Nestin/BrdU, PSA-NCAM/BrdU and NeuN/BrdU. A schematic view of the hippocampus is illustrated. The boxed area shows a magnified view of the DG of the hippocampus. Arrows illustrate the acceleration of neurogenesis in 13-week-old Tdo–/– mice at the levels of neural stem cells, neuroblasts and new neurons as revealed by Nestin/BrdU, PSA-NCAM/BrdU, and NeuN/BrdU immunostaining, respectively. Cells were counted in >3 matched sections from the DG of each mouse (n = 3 for each genotype).
Discussion
Here, we present evidence that Tdo–/– mice showed a marked increase in Trp levels in systemic circulation as well as increased Trp and 5-HT levels in the brain, even in the presence of intact TPH. Therefore, our results indicate that TDO is the key regulatory enzyme that modulates systemic Trp concentrations, ie, hepatic TDO plays an essential role in the regulation of Trp in systemic circulation (blood) (Fig. 8). In addition to Trp, 5-HT and 5-HIAA levels were also elevated in the brain. Although precise mechanisms have to be determined, our findings raise the possibility of the following working model in mice, as shown in Figuer 8. Trp in the blood passes through the BBB by competition with neutral amino acids. In the brain, Trp is metabolized to 5-HT and 5-HIAA. Therefore, TDO regulates both systemic and brain Trp and 5-HT, and its metabolite 5-HIAA (Fig. 4). Taken together, these results suggest that TDO, which is expressed predominantly in the liver, plays an essential and dominant role in the in vivo regulation of brain levels of 5-HT, even in the presence of TPH1 (in the periphery and pineal gland) and TPH2 (in the brain) (Fig. 8).

Working model of the role of TDO in modulating anxiety-related behavior and neurogenesis via regulation of Trp metabolism in the mice. Increased consumption of dietary Trp increases plasma Trp, which is predominantly metabolized to Kyn via hepatic TDO. Up-regulation of TDO activity in the liver (or locally expressed in the brain 33 ) by stress or stress-induced glucocorticoids would decrease systemic Trp levels, Trp transport to the brain through the BBB, and 5-HT biosynthesis in the central nervous system, even in the presence of intact TPH. In contrast, a decrease in TDO activity via gene knockout or genetic mutation would increase systemic and brain Trp/5-HT levels. These changes in Trp metabolism by TDO regulation/dysregulation would modulate anxiety-related behavior and adult neurogenesis, and may be largely involved in the modulation of mood by stress and environment under physiological conditions or psychiatric diseases. In addition, changes in the level of Kyn by TDO may also contribute to anxiety-related behavior. Because TDO and its novel variants are expressed in the brain in vivo 33 and in primary human neurons in vitro, 54 it seems likely that brain TDO(s) may play a role, and we intend to investigate the role of brain TDO(s) in a future study.
Because the role of the 5-HT transporter in anxiety-related behavior has been reported, 34 and 5-HT/5-HT1A receptor-mediated neurogenesis is critically involved in the anxiolytic effects of anti-depressant fluoxetine in adult animals, 28 a likely mechanism is that the deletion of hepatic TDO modulates plasma Trp and subsequently increases brain Trp and 5-HT, which in turn accelerates neurogenesis in the hippocampus and modulates anxiety-related behavior (Fig. 8). It should be noted that mRNA for TDO and its variants is expressed in various regions of both the developing and adult brain, 33 suggesting that local expression of TDOs modulates specific regions of the brain and is responsible for subsequent behavioral changes. Moreover, altered immunoreactivity against TDO has been reported in patients with schizophrenia and depression. 34
We also used Tdo–/– mice to evaluate the role of TDO in anxiety-related behavior. Although the mechanism remains to be elucidated, TDO deletion had clear anxiolytic effects, as revealed by two classical behavioral tests. In agreement with our data, Yamasaki et al reported a marked reduction in the level of tdo mRNA in the hippocampus of alpha-CaM-Kinase II deficient mice (alpha-CaMKII+/–), which show an anxiolytic phenotype. 35 In addition, Kyn levels are associated with the regulation of behavior in insects, and increased plasma Kyn concentrations are positively associated with endogenous anxiety in humans. Therefore, we cannot exclude the possibility that anxiety-related behavior is also modulated by TDO-induced changes in Kyn, and possibly in other kynurenines as well.36,37 If the contribution of Kyn to anxiety-related behavior is indeed important, then TDO appears to be a key modulator of behavior under physiological conditions via the control of both 5-HT and Kyn. This possible role of TDO differs from that of TPH, which has been considered a rate-limiting enzyme in the synthesis of 5-HT, but not of Kyn.
The role of stress and stress-induced glucocorticoids in affecting mood and anxiety is well known. Administration of glucocorticoids to rats elevates tryptophan-metabolizing enzymes and TPH in vivo, and administration of dexamethasone phosphate regulates TDO activity in the cells of control and adrenarectomized mice.38,39 In addition, a variety of substances including hormones, such as glucagon, insulin and glucocorticoids, have been used to regulate both the activity and mRNA levels of TDO in rat liver40–43 in vitro 44 in isolated primary hepatocytes.45–49 Indeed, stresses such as forced running, immobilization and exposure to cold increases rat liver TDO activity. 50 Taken together, the results of the present study suggest that TDO might modulate mood and anxiety-related behavior, and possibly the psychiatric response to stress and the environment (Fig. 8).
In summary, the present study has resulted in the first evidence that TDO plays an essential role in the homeostasis of systemic and brain Trp metabolism, including a dominant role in the regulation of the serotonergic pathway, under physiological conditions. TDO also plays a role in adult neurogenesis and modulation of anxiety-related behavior, indicating a role in higher brain functions. Collectively, our results with Tdo–/– mice indicate a direct molecular link between Trp metabolism and mental status. Because of the association of tdo2 (human tdo) gene polymorphism with attention deficit hyperactivity disorder (ADHD), Tourette's syndrome and autism,51–53 further elucidation of the role of TDO and its metabolites, such as Kyn, may identify the molecular mechanisms responsible for not only systemic Trp metabolism, but also higher brain functions and diseases, including emotional behavior(s) and mental and neurodegenerative disease(s). Identification of the molecular mechanisms provides a foundation for the development of new therapeutic interventions for these disorders.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
We are grateful to Prof. Emeritus Osamu Hayaishi for giving us the opportunity to present at the ISTRY 2009 meeting. We are also grateful to Profs. F. Moroni, A. A-B Badawy, K. Goda, G. Guillemin, and the members of the ISTRY2009 organizing committee for their kind and warm hospitality during ISTRY2009, Florence. We are also grateful to Dr. Hisaaki Takahashi. This work was supported in part by research grants from COE to Osaka University and by grants from the Ministry of Health and Welfare of Japan to H.F.
