Abstract
The display of the reproductive behavior in most noctuid Lepidoptera follows a diel periodicity and is limited to a precise period of either the day or the night. These behavioral traits and the sex pheromone chemistry can be species specific and thus might be linked to the phylogeny. The objective of this study was to test the relationship of these reproductive traits with phylogeny. The study was undertaken using eight closely related species of noctuid stem borers, which are easy to rear under artificial conditions, namely,
Keywords
Introduction
Reproductive activities in Lepidoptera follow a diel periodicity and are limited to a precise period of either the day or the night. They can differ among species and somehow can show some species' specificity. Most of the noctuid moths live for a few days and display a diel periodicity in emergence time, calling behavior, and oviposition period; this determines the onset of reproductive activities. These traits can affect the reproductive isolation between population variants feeding on different host plants as larvae, but sharing habitats as adults at their reproductive stage, and they can therefore be involved in the reinforcement of ecological speciation.
In addition to these behavioral parameters, the sex pheromone composition both within and between species is involved in the reproductive isolation of Lepidoptera species.1,2 In Lepidoptera, sex pheromone compositions are mostly species specific and the pheromone evolution processes can support systematic positions,3–8 and participate in the complex processes of reproduction and gene flow 9 by driving mate localization, recognition, and acceptance. Chemical cues play an essential role in specific mate recognition systems 10 and they maintain the reproductive isolation between closely related species. The reproductive isolation, preventing interbreeding between different species, is at the origin of the “biological species” concept developed by Mayr in 1942. 11 Sex pheromones consist of a precise blend of compounds with different chemical structures, and the resulting chemical signature of a species induces in the conspecific partner of the opposite sex a specific behavior. 12 Between closely related species, related or identical molecules are used, and the chemical signature is the result of different ratios of the same compounds. Males are very sensitive to both the ratio and the total amount of the pheromone components.13,14 For the distinction of closely related species, sex pheromone chemistry can be used in addition to temporal patterns to confirm phylogeny. Such information is essential for all studies in chemical ecology.
Lepidopteran maize stem borers (including noctuids, pyralids, and crambids) are the most important pests in sub-Saharan Africa.
15
Among the noctuids,
Materials and Methods
Lepidoptera species used
The following eight species of noctuid stem borers were used in this study:
Another Lepidoptera species,
All insects collected in the field were maintained in the laboratory for two-to-three generations at 24.4 ± 0.03°C and 58.4 ± 0.2% Relative Humidity (means ± standard errors [SEs]) on an artificial diet developed by Onyango and Ochieng'-Odero. 25 The pupae and adults were maintained at 25.3 ± 0.9°C and 68.6± 12.8% h.r. (means ± SEs) and 12 hours light: 12 hours darkness cycle. One room was kept under a reversed photoperiod, with the scotophase lasting from 7.00 to 19.00 h, herewith referred to as night, and another under normal conditions with the photophase lasting from 7.00 hours to 19.00 hours. This allowed all observations to be carried out during the day time. For the reversed photoperiod, the observations were made using a red 80 W fluorescent tube as a light source.
Pupae were sexed and, males and females were kept separately until emergence in a plastic box (30 cm length, 12 cm width, and 10 cm height) for molecular analyses, experiments, and sex pheromone collection.
Molecular data sets
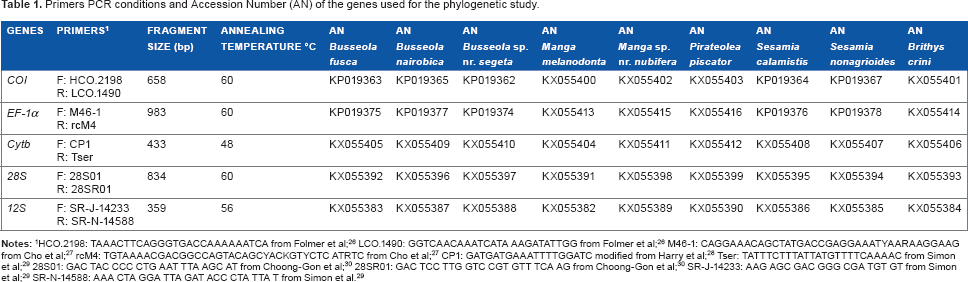
For all species, the DNA was extracted from the hind legs of the adult, experienced to be the muscle-rich part of the insect, using a Macherey-Nagel NucleoSpin® tissue kit (Macherey-Nagel GmbH & Co. KG). Polymerase chain reaction (PCR) amplifications were conducted for three mitochondrial genes (Table 1),
Primers PCR conditions and Accession Number (AN) of the genes used for the phylogenetic study.
Phylogenetic analyses
The data set consisted of a total of eight Lepidoptera stem borer species using 3267 bp
Temporal patterns measured
Adult emergence period
For each species and each sex, emergence was recorded hourly from 7.00 hours to 19.00 hours in both experimental rooms through the aforementioned plastic box. The experiment was run for 10 consecutive nights and days.
Mating time
To determine the mating time, one-day-old females were placed with one-day-old males in a mosquito net cage (30 X 30 X 30 cm). Mating postures were observed at hourly intervals. Because no mating was observed during the photophase, the experiments started at the onset of the scotophase and ended at the onset of the photophase.
Oviposition period
None of the females laid eggs the night it mated. Thus, all the females used for determining the temporal pattern of oviposition had mated the night before the observations started.
Gravid females were placed individually in transparent plastic jars (10 cm diameter X 20 cm height) together with an oviposition substrate consisting of a wax paper cut rectangularly (15 cm X 6 cm) and rolled helicoidally from top to bottom to form a cylindrical surrogate stem. 33 No eggs were laid on surrogate stems during the photophase. Thus, oviposition experiments started at the onset of the scotophase and ended with the onset of the photophase.
The oviposition time was estimated by monitoring at hourly intervals during the first two nights after mating. The surrogate stems were renewed every hour.
In a separate experiment, to evaluate the oviposition rate per night, the number of eggs was counted each night over five successive nights, renewing the surrogate stem each night.
After each experiment, each female was dissected to verify the presence of spermatophore(s) in the
Sex pheromone analysis
For all Lepidoptera species, the pheromone glands of calling virgin females were extruded with a thin forceps, and the gland tegument area was gently rubbed for five minutes with the adsorbent part of a Carbowax™–Divinylbenzene (Supelco) 65 μm solid-phase microextraction (SPME) fiber assembly.
34
The SPME collection of each female was analyzed by gas chromatography (GC) using a Varian 3400 CX gas chromatograph with a split–spitless injector heated at 240°C and a flame ionization detector detector at 260°C. The fiber was subjected to thermal desorption in the injector. A 30 m long ∼0.32 mm internal diameter (i.d.) Rtx®-Wax column (Restek®, France) was used with the oven temperature being ramped from 60°C to 100°C at 15°C/min, then to 245°C at 5°C/min. Helium was the carrier gas (15 psi). GC–mass spectrometry (GC-MS) analyses were carried out on a Varian ion trap Saturn II mass spectrometer coupled to a 3400 CX gas chromatograph. The MS conditions were as following: electron impact (IE) mode, 70 eV, 40–330 amu. The GC injector split–splitless was held at 250°C, and the RTX-5 Sil-MS (Restek®), 30 m ∼0.32 mm i.d. column temperature ramped from 50°C to 300°C at 8°C/min. Pheromone compounds were identified by comparing retention times and mass spectra of SPME samples collected from the gland with synthetic reference compounds. For each Lepidoptera species, at least three females were analyzed (n = 3). For
Statistical analyses
To test the phylogenetic signal for temporal pattern parameters and sex pheromone compounds, a phylogenetic Bayesian tree was assembled based on the Lepidoptera species used in this study (Fig. 1). The data of this phylogenic tree were then transformed to an ultrametric form and a multivariate phylogenetic comparative method (‘mvMORPH’ package R of Clavel et al
35
) was used to test the phylogenetic inertia of the different variables evaluated (ie, the temporal pattern parameters, including the pheromone components). This method allows the testing of the phylogenetic signal using various statistics; Abouheif's
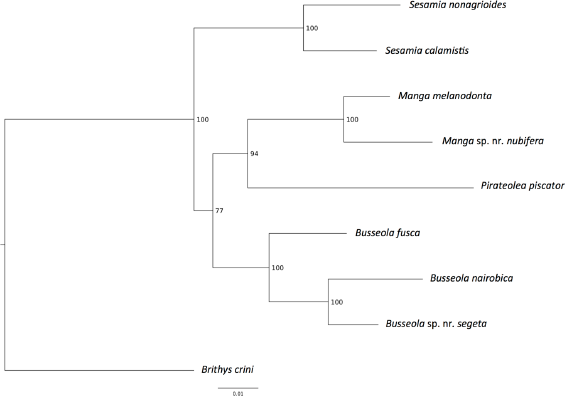
Phylogenetic Bayesian tree using 1896 bp (
Results
The phylogenetic analysis confirmed the monophyly of the genera,
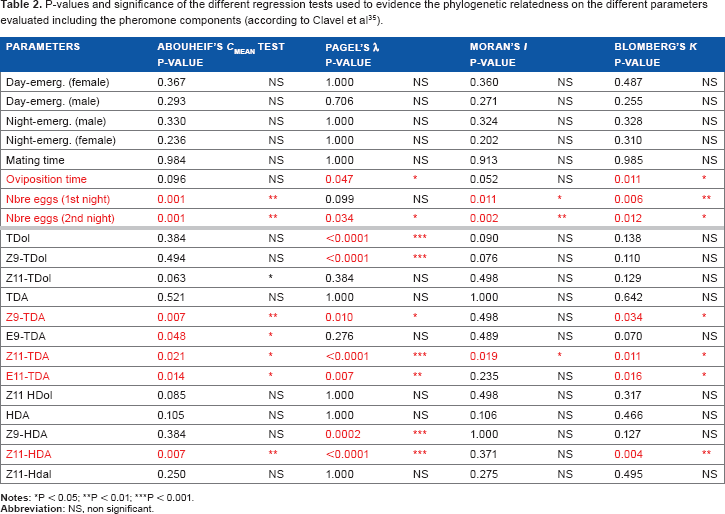
The different statistics of phylogenetic inertia produced congruent results (Table 2). Pagel's λ was significant only for oviposition time and the number of eggs during the second night, as well as for several pheromone components: tetradecenol (TDol), Z9-TDol, Z9-tetradecenyl acetate (Z9-TDA), Z11-TDA, E11-TDA, Z9-hexadecenyl acetate (Z9-HDA), and Z11-HDA. However, TDol, Z9-TDol, and Z9-HDA were not significant for the other regression tests. The number of eggs during the first night was not significant for Pagel's λ but significant for all other tests (Morans'
P-values and significance of the different regression tests used to evidence the phylogenetic relatedness on the different parameters evaluated including the pheromone components (according to Clavel et al 35 ).
P < 0.05;
P < 0.01;
P < 0.001.
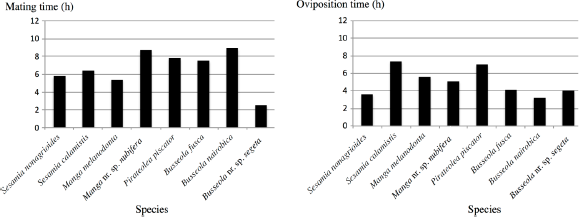
Mean mating times (left) (n = 20–50) and oviposition times (right) (n = 15–30) of different Lepidoptera species.
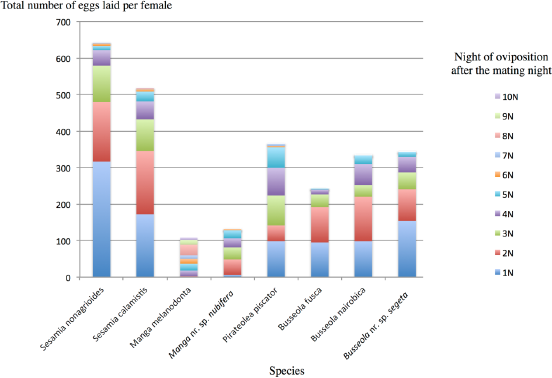
Mean total number of eggs laid per female (n = 10–20) of different Lepidoptera species during several nights (1N, 2N … 10N: first night, second night … tenth night) after the night of mating.
For all species, males and females emerged at the last two hours of the photophase period and at the first 2–4 hours of the night (Fig. 4). In the context of the mating time, some species such as
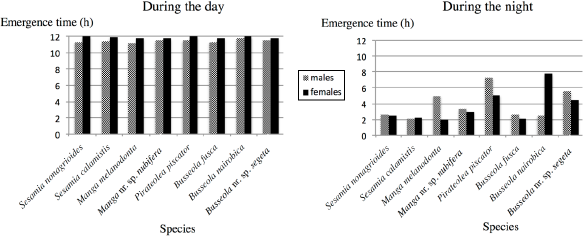
Mean emergence times (n = 20–70) of males and females during the day (left) and during the night (right) of different Lepidoptera species.
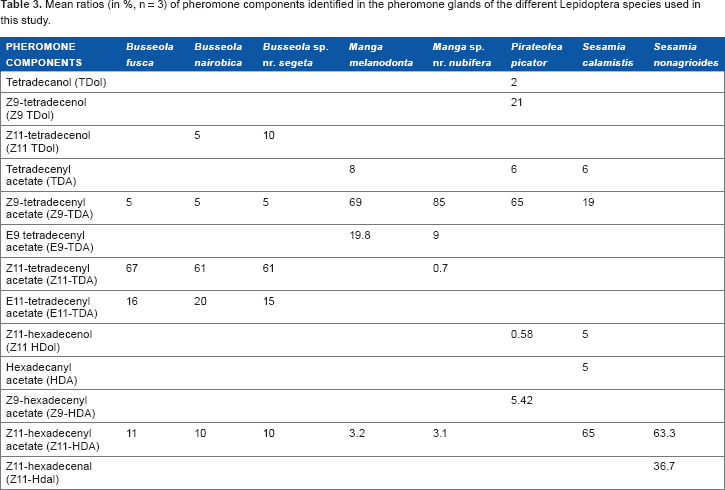
For all stem borer species, 13 sex pheromone components were identified, and only four of them, Z9-TDA, Z11-TDA, E11-TDA, and Z11-HDA were significantly correlated with the phylogeny (validation by most of the regression tests used; Table 2). Z9-TDA was more abundant in the
Mean ratios (in %, n = 3) of pheromone components identified in the pheromone glands of the different Lepidoptera species used in this study.
Discussion
As also reported for other noctuids, for all species used in this study, both males and females started to emerge at the last hours of the photophase period and emerged mostly at the beginning of the night. 17 ,37–39 As reported by the authors of these studies, light duration and transition play an important role in promoting the emergence of moths.
In many insects, there is a general tendency for males to emerge before the females, which is known as protandry. This was only observed for
Among the moth species that emerged earlier at night, such as
The results on sex pheromone components indicated that all the noctuid species exhibited C14 and C16 acetate components in their sex pheromones. The presence of both C16 and C14 components shows that these species share the same biosynthesis pathway, namely, oxidizing (ie, double bond appearance by loss of hydrogen) C16 compounds into C14 compounds. Among the 13 components identified, only four, namely, Z9-TDA, Z11-TDA, E11-TDA, and Z11-HDA, were mostly correlated with the phylogeny of the stem borer moth species studied. These pheromone components were shown to be present in a large number of Lepidoptera species, including several families such as Crambidae, Gelechiidae, Noctuidae, Pieridae, Pyralidae, Tortricidae, and Yponomeutidae. 43 However, some differences in their abundances occur between Lepidoptera families. For example, E11-TDA is more frequently found in Tortricidae and Z9-TDA is more represented in Noctuidae whereas E11-TDA is more rare (B. Frérot, personal communication).
Conclusion
Some traits measured in this study contribute to premating isolation and reinforcement of reproductive isolation in stem borer noctuids. Accelerated evolution of those traits can occur for host races that differ in their host specialization as larvae, but that share a habitat as adults. Host races would pay a postmating fitness cost in the absence of premating reproductive barrier. 44 Pheromone components and mating time are generally good candidates for premating selected traits in host race formation. Other traits such as emergence time are more constrained by environmental adaptation. They are more likely to be under conservative selection and exhibit phylogenetic inertia. Our study indicated that most pheromone components (nine out of 13) as well as mating time do not exhibit any phylogenetic signal. This might be due to a rapid evolution of these traits. As discussed, traits linked to oviposition showed phylogenetic signal, suggesting that inertia can be due to conservative selection. Alternatively, the absence of a phylogenetic signal can be due to the little power of the test when little variation is present on the trait. This is the case for emergence time characterizing the noctuid family and for the majority of pheromone components (particularly true when the component was detected in a single or in two insect species only). Therefore, although it is difficult to conclude on the validation of the hypothesis that all traits evaluated in this study are under diversifying selection related to speciation reinforcement, some of them–-such as oviposition time, egg loads per night, and four pheromone components (Z9-TDA, Z11-TDA, E11-TDA, and Z11-HDA)–-are better candidates to contribute to such diversifying selection.
Author Contributions
Conceived and designed the experiments: P-AC. Analyzed the pheromone components: BF and GG. Conducted the experiments: P-AC and PA. Conducted the molecular analyses and the phylogeny: CC-D. Analyzed the data: SD. Wrote the first draft of the manuscript: P-AC. Contributed to the writing of the manuscript: P-AC, SD, BF, CC-D, and BLR. All authors reviewed and approved of the final manuscript.
Footnotes
Acknowledgment
The authors thank Fritz Schulthess for his critical review of the manuscript and English corrections.
