Abstract
In previous studies we found expression of the protein colligin 2 (heat shock protein 47 (HSP47),
We demonstrate overexpression of
A wide range of physiological and pathological stresses trigger the heat shock gene transcription. Cells respond to elevated temperatures and to chemical and physiological stresses by an increase in the synthesis of heat shock proteins. HSP are a highly conserved family of proteins which function as molecular chaperones or proteases.1,2 Molecular chaperones form a class of proteins that control the proper folding of nascent polypeptides into the correct 3D structure. During stress responses the role of HSPs is critical in preventing the appearance of intermediates that lead to misfolding or otherwise damaging molecules. 3 HSPs assist in the recovery from stress either by repairing damaged proteins (protein refolding), or protein degrading, thus restoring protein homeostasis and promoting cell survival. The regulation of heat shock proteins is mediated by heat shock transcription factors (HSF). Under normal conditions, HSFs reside in the cytoplasm, but are activated upon stress and relocalize to the cell nuclei. 4 Activated HSFs form a trimmer with high-affinity binding to DNA; it binds to heat shock elements (HSE) in the promoters of the heat shock genes. 4 The activation results in the expression of heat shock proteins (HSPs). In vertebrates and plants, there are at least four members of the HSF gene family (HSF1–4), 1 while in human cells, three HSFs (HSF-1, -2, and -4) have been characterized.2,5 The expression of HSF1 and HSF2 is ubiquitous. However, the factors that induce their activation differ. While HSF1 is activated by heat shock and other forms of stress, HSF2 activity has been associated with development and differentiation. The expression of HSF4 appears to be tissue-specific and is restricted to heart, skeletal muscle or brain. 5 The simultaneous expression of the different HSFs in particular tissues would enable differential responses to various forms of stress. In the context of tumors, HSPs may be tumor-specific and therefore, they may well become therapeutic targets.6–8
In previous studies, we found specific overexpression of colligin 2 in glioma neovasculature as compared to the normal vasculature of the brain.9,10 Here we investigate whether there is a correlation between the expression of colligin 2 and any of the HSF genes (HSF1, HSF2 and HSF4) at the mRNA level in low- and high-grade glioma. We isolated RNA from human frozen samples by using RNeasy Micro kit (Qiagen BV, Venlo, The Netherlands). We measured the relative transcription levels of colligin 2 (
qRT-PCR revealed a significant 5.6-fold increase in mRNA levels for colligin 2 in glioblastoma and mildly elevated levels in low-grade glioma (Fig. 1). In parallel, significant increases in mRNA levels of collagen type I was found in the gliomas. Of the heat shock factors,
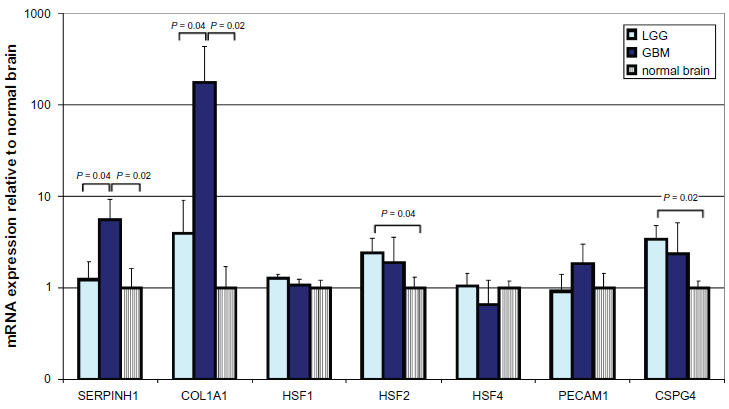
mRNA expression of colligin 2, HSF1, 2 and 3, collagen 1, CD31 and NG2 in low- and high-grade glioma and normal control brain. Data in this figure are the average ± SD of one representative experiment with 4 tissues in each group. expression data are presented relative to the average mRNA expression levels measured in total RNA isolated from normal brain tissues (n = 4). Prior to isolation, all tissues were assessed by a qualified pathologist to ensure the origin and quality of the tissues. Total RNA was isolated with the RNeasy Micro kit (Qiagen BV, Venlo, The Netherlands). cDNA was prepared by use of the RevertAid H Minus First Strand cDNA synthesis kit (Fermentas, St Leon-Rot, Germany). The resulting cDNA preparations were analyzed by real-time PCR with TaqMan gene expression assays and TaqMan Universal PCR Master Mix (Applied Biosystems, Nieuwerkerk a/d IJssel, The Netherlands). pCRs were performed in a 20 µL reaction volume in an Applied BioSystems 7900HT Fast Real-Time PCR system. Negative controls included minus RT and H2O-only samples, which showed to be negative in all cases. The most stable mRNA set for our 4 tissue groups were calculated with NormFinder
19
with the Datan Framework GenEx Pro package version 4.3.2 and was shown to be a combination of
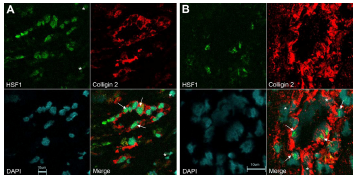
Expression of HSF1 and colligin 2 in blood vessels of glioblastoma. A) Double immunolabeling for HSF1 and colligin 2 in small blood vessels of glioblastoma. The endothelial cells express both colligin 2 = red and HSF1 = green (arrows). Colligin 2 is exclusively expressed in the blood vessels while HSF1 is expressed in some cells that surround the blood vessel (asterix). B) Double immunolabeling for HSF1 and colligin 2 in hypertrophied blood vessels of glioblastoma. Some of the endothelial cells and the pericytes in the blood vessels co-express colligin 2 = red and HSF1 = green (arrows). Some cells in the blood vessel wall express colligin 2 exclusively (asterix). The co-expression of colligin 2 and HSF1 in blood vessels of glioblastoma depends on the blood vessel type and in the activity level of a blood vessel. We used normal brain samples that do not express colligin 2 as a negative control. For the quality control we used adjacent slides from the same glioblastoma samples stained with the secondary antibodies only. We obtained negative controls for both single and double staining.
Several studies showed the transcription of HSP genes requires the activation and translocation to the nucleus of heat shock factors (HSF's).
13
It has been shown that HSF1 induces the mRNA expression of colligin 2 in rats
14
and in young zebrafish.
15
This is the first report on the parallel upregulation of colligin 2 and heat shock factors in human glioma. Heat shock responses appear to be implicated in a broad range of pathological conditions including heat shock, oxidative stress, ischemia and reperfusion, inflammation, tissue damage, exposure to heavy metals and infection.
1
In addition, tumor neovascularization is associated with the heat shock response. Mammals have three different HSFs which are considered to be functionally distinct: HSF1 is essential for the heat shock response and is also required for developmental processes; HSF2 and HSF4 are important for differentiation and development.
16
Although deletion of
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
