Abstract
Taxol is recognized as one of the most potent anticancer agents used in the treatment of breast and ovarian cancers, which are common cancers in women. To overcome its shortcomings, that is, its low water-solubility that reduces drug loading capacity of DDS carriers when incorporating taxol, chemo-enzymatic synthesis of ester-linked taxol-glucose conjugate, i.e., 7-propionyltaxol 2′-O-α-D-glucoside, as a water soluble taxol prodrug was achieved by using a-glucosidase as a glucosylation catalyst. The water-solubility of 7-propionyltaxol 2′-O-α-D-glucoside (25 mM) was 63 fold higher than that of taxol (0.4 mM). The pre-S1 peptide which displays on the surface of bio-nanocapsules, which are nanoparticles composed of the hepatitis B virus surface antigen, was replaced with the antibody affinity motif of protein A. Conjugation of such bio-nanocapsules with anti-human epidermal growth factor receptor antibody gave hybrid bio-nanocapsules. The hybrid bio-nanocapsules were effective for delivering 7-propionyltaxol 2′-O-α-D-glucoside to human brain glioma cells. 7-Propionyltaxol 2′-O-α-D-glucoside was effectively hydrolyzed to give taxol in 95% by human glioma cells. The drug loading capacity of hybrid bio-nanocapsules incorporating 7-propionyltaxol 2′-O-α-D-glucoside was 120 times higher than that incorporating taxol itself.
Introduction
Taxol (paclitaxel), which is a taxane diterpenoid isolated from Taxus Brevifolia, shows cytotoxic activity against leukemia cells and inhibitory action against a variety of tumors such as ovarian cancer. It is recognized as one of the most potent anticancer agents used in the treatment of breast and ovarian cancers. It presents disadvantages such as low solubility in water and toxicity to normal tissues despite its effective pharmacological activities. Many efforts have been made to modify taxol chemically in order to create a more soluble and more easily delivered drug.1–4 Taxol derivatives that incorporate acids or amino acids have attracted considerable attention, because ester and amide linkages improve the solubility of taxol and can be hydrolyzed by hydrolytic enzymes in the living body to release taxol.1–4 However, acid or amino acid conjugates lack tumor selectivity. In order to improve drug selectivity toward tumor cells, many efforts to chemically synthesize taxol prodrugs designed containing a transport system have been made. Saccharide-based-transporters are useful, and ester-linked taxol-glycoside conjugates as water-soluble taxol derivatives, which employ enzymatic cleavage by hydrolytic enzymes including esterases and glycosidases as their mode of activation, have been synthesized. 5 On the other hand, a convenient drug delivery system (DDS) using hepatitis B virus surface antigen L particles (bio-nanocapsules) has been recently reported. 6 This DDS system is not effective for delivering taxol, because the present nano-particles can incorporate only soluble drugs. The bio-nanocapsules have been reported to be able to incorporate water-soluble ester-linked taxol-glycoside conjugates. 5
On continuing the study to develop the technology for delivering taxol to target brain tumors, we report the chemo-enzymatic synthesis of glycolyl-ester-linked taxol-sugar conjugate; i.e., 7-propionyltaxol 2′-O-α-D-glucoside, and its new delivery system to human brain glioma cells using hybrid bio-nanocapsules as the drug carrier.
Experimental
General
Taxol was a gift from Ensuiko Sugar Refining Co. Ltd. The 1 H and 13C nuclear magnetic resonance (NMR), H-H correlation spectroscopy (COSY), C-H COSY, and heteronuclear multiple-bond correlation (HMBC) spectra were recorded in CD3OD using a Varian XL-400 spectrometer (Varian Inc.). The chemical shifts were expressed in S (ppm) referring to tetramethylsilane. The fast atom bombardment mass spectrometry (FABMS) spectra were measured using a JEOL MStation JMS-700 spectrometer (JEOL Ltd.). High performance liquid chromatography (HPLC) was carried out on Crestpak C18S column (4.6 × 150 mm, JASCO) [solvent: MeOH-H2O (2:3, v/v); detection: UV (228 nm); flow rate: 1.0 mL/min].
Synthesis of 7-propionyltaxol 2′-O-α-D-glucoside
Propionic acid was glucosylated by a-glucosidase as follows. 7 To a solution of maltose (0.2 mol) and propionic acid (0.02 mol) in dimethyl sulfoxide (DMSO)-H2O was added a-glucosidase (500 U). The mixture was stirred for 24 hours (h) at 40°C and then was extracted with n-butanol. The organic layer was concentrated and purified by column chromatography on silica gel to afford carboxyethyl α-D-glucopyranoside (1a).
Synthesis of 7-propionyltaxol 2′-O-α-D-glucoside was carried out as follows. Carboxyethyl α-D-glucopyranoside was added to a solution of BnBr/NaH (0.15 mol) in dimethylformamide (DMF) (1a). The mixture was stirred at room temperature (rt) for 12 h, followed by stirring with aq. KOH (1.5 equiv.). The reaction mixture was quenched with saturated aq. NaHCO3 and extracted with ethyl acetate. The ethyl acetate layer was concentrated in vacuo and purified by silica gel column chromatography to give carboxyethyl 2,3,4,6-tetra-O-benzyl-α-D-gluctopyranoside (2a). Chlorotriethylsilane (0.1 mol) was added to a solution of taxol (0.03 mol) and imidazole (0.12 mmol) dropwise at rt in dry DMF. The reaction mixture was stirred at rt for 2 h and diluted with ethyl acetate. The mixture was washed with water and brine, dried over MgSO4, and concentrated in vacuo. Column chromatography of the residue on silica gel gave 2′-TES ester of taxol. 2a (1.2 equiv) was added to a mixture of 2′-TES ester of taxol (0.015 mol) in the presence of EDCI/DMAP (0.022 mol) in CH2Cl2 (10 mL). The mixture was stirred at rt for 12 h. The reaction mixture was extracted with ethyl acetate. The organic layer was concentrated in vacuo and purified by column chromatography on silica gel to give 3a. 3a was added to a solution of Pd black (0.001 mol) in HOAc-H2O (9:1, v/v). The suspension was stirred at room temperature for 24 h. Extraction of the reaction mixture with n-butanol followed by column chromatography on silica gel yielded 7-propionyltaxol 2′-O-α-D-glucoside (4).
Spectral data of 7-propionyltaxol 2′-O-α-D-glucoside are as follows.
7-Propionyltaxol 2′-O-α-D-glucopyranoside (4): high resolution fast atom bombardment mass spectroscopy (HRFABMS): calcd for C56H65NO21Na [M+Na] + m/z 1110.3502, found 1110.3510;1H-NMR (400 MHz, CD3OD, d in ppm): d 1.08 (3H, s, H-16), 1.15 (3H, s, H-17), 1.75 (3H, s, H-19), 1.82 (1H, m, H-6b), 1.88 (3H, s, H-18), 2.02 (1H, dd, J = 15.6, 9.0 Hz, H-14a), 2.15 (3H, s, CH3 in 10Ac), 2.25 (1H, dd, J = 15.6, 9.0 Hz, H-14b), 2.37 (3H, s, CH3 in 4Ac), 2.58 (1H, m, H-6a), 3.25–3.88 (10H, m, H-2′, H-3′, 2a, 3a, 4a, 5a, 6a), 3.90 (1H, d, J = 7.2 Hz, H-3), 4.15 (1H, m, H-7), 4.18 (2H, m, H-20), 4.75 (1H, d, J = 5.1 Hz, H-2′), 4.90 (1H, d, J = 8.0 Hz, H-1a), 5.00 (1H, d, J = 9.0 Hz, H-5), 5.62 (2H, m, H-2, 3′), 6.15 (1H, t, J = 9.0 Hz, H-13), 6.20 (1H, s, H-10), 7.27 (1H, t, J = 7.6 Hz, p-H in Ph), 7.39–7.58 (9H, m, m-H in NBz, p-H in NBz, m-H in OBz, o-H in Ph, m-H in Ph), 7.65 (1H, t, J = 7.6 Hz, p-H in OBz), 7.85 (2H, d, J = 8.0 Hz, o-H in NBz), 8.10 (2H, d, J = 8.0 Hz, o-H in OBz); 13C-NMR (100 MHz, CD3OD, d in ppm): d 11.3 (C-19), 14.6 (C-18), 20.6 (CH3 in 10Ac), 22.0 (C-16), 23.0 (CH3 in 4Ac), 26.7 (C-17), 34.1 (C-6), 36.0 (C-14), 44.5 (C-3, C-15), 57.0 (C-3′), 57.7 (C-8), 62.5 (C-6a), 68.0 (C-3′), 70.0 (C-2′), 71.5 (C-7, C-13), 72.1 (C-4a), 73.5 (C-5a), 74.1 (C-2a), 74.8 (C-2′), 75.1 (C-3a), 75.5 (C-2), 76.6 (C-10), 77.0 (C-20), 78.8 (C-1), 82.0 (C-4), 85.1 (C-5), 100.5 (C-1a), 128.3 (o-C in NBz, o-C in Ph), 128.8 (p-C in NBz), 129.5 (m-C in OBz, m-C in Ph), 131.1 (m-C in NBz, q-C in OBz), 132.7 (o-C in OBz, p-C in Ph), 134.1 (C-11), 134.5 (q-C in Ph), 135.1 (p-C in OBz), 140.2 (q-C in NBz), 142.1 (C-12), 167.3 (C = O in OBz), 170.0 (C = O in NBz), 170.6 (C-1′), 171.2 (C = O in 4Ac), 172.0 (C = O in 10Ac), 174.3 (C-1′), 203.2 (C-9).
Water-solubility of 7-propionyltaxol 2′-O-α-D-glucoside
Water-solubility of 7-propionyltaxol 2′-O-α-D-glucoside was examined as follows. The compound was stirred in water for 24 h at 25°C. The mixture was centrifuged at 100000 g for 30 minutes (min) at 25°C. The concentration of test compounds was estimated on the basis of their peak areas using calibration curves prepared by HPLC analyses of authentic samples.
Preparation of hybrid bio-nanocapsules
The pre-S1 region of L protein was replaced with an IgG Fc binding motif (ZZtag), which is the 2-helix domain of protein A from Staphylococcus Aureus. Bio-nanocapsules were incubated with rhodamin B isothiocyanate (RITC, 1 mg/mL) at room temperature for 15 min and the mixture was further reacted at 4°C overnight in phosphate buffered saline (PBS). After incubation, bio-nanocapsules were incubated with ethanolamine (2.5 mM) for 2 h at 4°C. The buffer solution of the bio-nanocapsules was replaced by PBSE. The RITC-conjugated bio-nanocapsules were mixed with monoclonal anti-epidermal growth factor receptor (EGFR) antibodies for 1 h at 4°C to give hybrid bio-nanocapsules. 8
In vitro cytotoxicity assay
The sensitivity of human glioma cell line Gli36 to 7-propionyltaxol 2<′-O-α-D-glucoside or 7-propionyltaxol 2′-O-α-D-glucoside electroporated into hybrid bio-nanocapsules was determined according to a previously reported method. 6 Cells were diluted with culture medium to the seeding density (105 cells/mL), suspended in 96-well tissue culture plates (100 mL/well), preincubated at 37°C for 4 h, and then treated for 24 h with 7-propionyltaxol 2′-O-α-D-glucoside or 7-propionyltaxol 2′-O-α-D-glucoside electroporated into hybrid bio-nanocapsules at various concentrations to obtain a dose-response curve for each compound. After incubation, 20 mL MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, yellow tetrazole) solution (2.5 mg/mL) was added to each well and the plates were further incubated for 4 h. Absorbance at 570 nm was measured with a microplate reader model 450 (BIO-RAD). Dose-response curves were plotted on a semi-log scale as percentage of the cell numbers in control cultures not exposed to test compounds.
Hydrolysis of 7-propionyltaxol 2′-O-α-D-glucoside (4) by human glioma cells
To a 5-mL vial containing 20 mg of human glioma cells was added 5 mmol of compound 4. The mixture was incubated at 37°C for 24 h. The cells and medium were separated by centrifugation at 10000 g for 5 min. The cells were extracted with MeOH. MeOH extract was concentrated, and the residue was partitioned between H2O and CH2Cl2. The medium was extracted with CH2Cl2. The CH2Cl2 fractions were combined, concentrated, and analyzed by HPLC. The yield of taxol was calculated on the basis of the peak area from HPLC using a calibration curve provided by HPLC analyses of authentic taxol.
Results and Discussion
The water soluble taxol derivative; i.e., 7-propionyltaxol 2′-O-α-D-glucoside (4), was synthesized from taxol by chemo-enzymatic procedures as shown in Figure 1. First, the 2′-hydroxyl group of taxol was protected with triethylsilyl (TES) group to give 2′-TES ester of taxol. Incubation of the reaction mixture including propionic acid, a-glucosidase, and maltose in DMSO-H2O at 40°C for 24 h gave carboxyethyl α-D-glucoside. The carboxyethyl α-D-glucoside was benzylated with BnBr/NaH in DMF at room temperature for 12 h, followed by stirring with KOH (1.5 equiv) to give carboxyethyl 2,3,4,6-tetra-O-benzyl-α-D-glucoside. The coupling of 2′-TES ester of taxol with carboxyethyl 2,3,4,6-tetra-O-benzyl-α-D-glucoside (1.2 equiv) in the presence of EDCI/DMAP in CH2Cl2 at room temperature for 12 h afforded 2′-TES-7-propionyltaxol 2′′,3′′,4′′,6′′-tetra-O-benzyl-2′-O-α-D-glucoside. The deprotection of both TES and benzyl groups with Pd black in HOAc-H2O (9:1, v/v) yielded 7-propionyltaxol 2′-O-α-D-glucoside (4).

Synthesis of 7-propionyltaxol 2′-O–α-D-glucopyranoside (
The water-solubility of 7-propionyltaxol 2′-O-α-D-glucoside (4) was examined (Table 1). The water-solubility of 7-propionyltaxol 2′-O-α-D-glucoside (4) was 25 mM, which was 63-fold higher than that of taxol (0.4 mM). The glucosyl conjugation effectively improved the water-solubility of taxol.
Water-solubility of 7-propionyltaxol 2′-O-α-D-glucoside.

Water-solubility was measured at 25°C.
Hepatitis B virus is a human liver-specific virus, the genome of which harbors 3 overlapping envelope genes in a single open reading frame. Hepatitis B virus envelope large (L) protein was produced in yeast cells as hollow particles with no hepatitis B genome inside and the particle was used as immunogens in hepatitis B vaccines that were proven safe for humans. The N-terminal amino acid residues 108–119 of the L protein displayed on the surface of L particles functions as the specific ligand for receptor binding on human hepatocytes (pre-S1 region). The pre-S1 peptide was replaced with the antibody affinity motif of protein A-binding anti-human EGFR antibody, according to the previously reported procedures to give hybrid bio-nanocapsules. 8 Next, taxol-prodrug 7-propionyltaxol 2′-O-β-D-glycoside was electroporated into hybrid bio-nanocapsules. The mixture of taxol-prodrug (final concentration of 2.1, 4.2, 8.3, 17, and 33 mg/mL) and 500 mL of hybrid bio-nanocapsules solution was electroporated in a 4-mm gap cuvette for 20 min. To clarify the efficient incorporation of taxol-prodrug in the hybrid bio-nanocapsules, the filtrate of hybrid bio-nanocapsules solution was analyzed by HPLC and no taxol-prodrug was detected. After adding the same volume of water as the filtrate, the cytotoxicity of taxol derivative 4 incorporated in hybrid bio-nanocapsules toward human glioma cells was examined. Human glioma cells were diluted with RPMI/10% FBS (105 cells/mL), suspended in 96-well tissue culture plates (100 mL/well), preincubated at 37°C for 12 h, and treated for 3 days (d) with 7-propionyltaxol 2′-O-α-D-glucoside (4) and 7-propionyltaxol 2′-O-α-D-glucoside (4) incorporated in hybrid bio-nanocapsules. After incubation, 20 mL MTT solution (5 mg/mL) was added to each well and the plates were further incubated for 5 h. Absorbance at 570 nm was measured with a microplate reader. The Dose-response curves were shown in Figure 2. The cytotoxicity of both 7-propionyltaxol 2′-O-α-D-glucoside (4) and 7-propionyltaxol 2′-O-α-D-glucoside (4) incorporated in hybrid bio-nanocapsules was increased dose-dependently. At each concentration tested, the cytotoxic activity of 7-propionyltaxol 2′-O-α-D-glucoside (4) incorporated in hybrid bio-nanocapsules was higher than 7-propionyltaxol 2′-O-α-D-glucoside (4). The drug concentration of 7-propionyltaxol 2′-O-α-D-glucoside in hybrid bio-nanocapsules was 12 w% and that of taxol in hybrid bio-nanocapsules was 0.1 w%. Additionally, 7-propionyltaxol 2′-O-α-D-glucoside (4) was hydrolyzed to give taxol in 95% by human glioma cells.
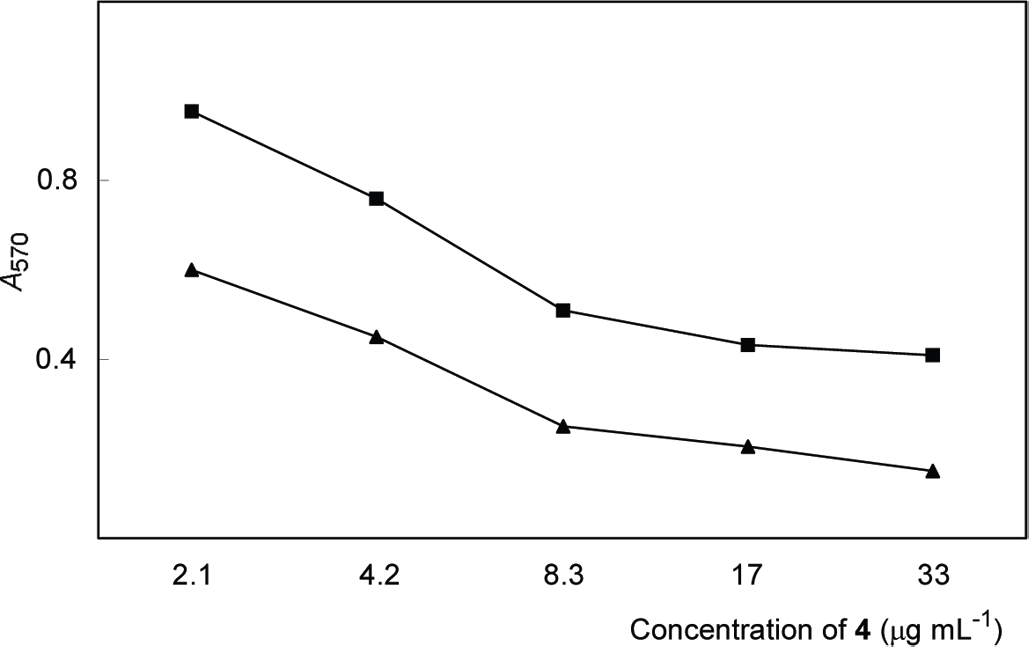
dose-response curves in MTT assay of 7-propionyltaxol 2′-O-α-D-glucoside (▪) and 7-propionyltaxol 2′-O-α-D-glucoside incorporated in hybrid bio-nanocapsules (▴). Absorbance at 570 nm was measured with a microplate reader model 450 (BIo-RAD).
Thus, a water-soluble taxol derivative; i.e., 7-propionyltaxol 2′-O-α-D-glucoside, was successfully synthesized by chemo-enzymatic procedures. The drug delivery system using hybrid bio-nanocapsules was effective for delivering 7-propionyltaxol 2′-O-α-D-glucoside to human glioma cells. EGFR is over-expressed in a variety of human malignancies of epithelial origin (cancers of the brain, lung, colon, and head). Gliomas often express EGFRvIII, a constitutively active genomic sequence deletion variant of EGFR. The variant of EGFR strongly activates the phosphatidylinositol 3′ kinase (PI3K) signaling pathway, which provides critical information for cell proliferation and motility. The bio-nanocapsules conjugated with anti-human EGFR antibody that recognizes EGFRvIII would be capable of targeting brain tumors in vivo. The drug loading capacity of hybrid bio-nanocapsules incorporating 7-propionyltaxol 2′-O-α-D-glucoside was 120 times higher than that incorporating taxol; that is, use of water-soluble taxol prodrug much improved the drug concentration in hybrid bio-nanocapsules compared with the case of taxol itself. Further studies on in vivo thrapeutic values of taxol derivative, which is incorporated in hybrid bio-nanocapsules, are now in progress.
Footnotes
Author Contributions
Conceived and designed the experiments: HH and KS. Analyzed the data: HH, KS, and MS. Wrote the first draft of the manuscript: HH and KS. Contributed to the writing of the manuscript: HH and KS. Agree with manuscript results and conclusions: HH, KS, and MS. Jointly developed the structure and arguments for the paper: HH, KS, and MS. Made critical revisions and approved final version: HH and KS.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
