Abstract
Interleukin-6 (IL-6), produced by a variety of cells, is a typical cytokine featuring redundancy and pleiotropic activity. IL-6 is promptly and transiently synthesized in response to infections or injuries, and participates in host defense by inducing immune responses, hematopoiesis, and acute-phase reactions. However, since its abnormal persistent production of mostly unknown etiology plays an important pathological role in the development of various immune-mediated diseases, a humanized anti-IL-6 receptor monoclonal antibody, tocilizumab, was developed and is now used as an innovative biologic for rheumatoid arthritis in more than 90 countries. Several factors strongly suggest that a IL-6 blockade strategy may have a broad application for the treatment of various immune-mediated diseases. These factors include favorable results of pilot or case studies with off-label use of tocilizumab, pathological analyses of the contribution of IL-6 to the development of immune-mediated diseases, and the potential capability of tocilizumab to both repair an imbalance of effector T cell subsets and to suppress pathologic autoantibody production. However, clinical trials to evaluate the efficacy and safety of tocilizumab for these diseases are essential. Furthermore, clarification of the cell source of IL-6 production and of the mechanisms through which dysregulated continuous IL-6 synthesis is induced constitutes an important issue for future studies into the pathogenesis of diseases.
Interleukin-6
In the early stages of research into the soluble factor interleukin-6 (IL-6), its various and distinct functions were being studied and each research group had its own name for this cytokine. One was B cell stimulatory factor 2 (BSF-2) because it induces B-cell differentiation into antibody (Ab) producing cells. Another was hepatocyte-stimulating factor (HSF) because it induces synthesis of acute phase proteins in hepatocytes. Yet another was hybridoma growth factor (HGF) due to its promotion of growth of fusion cells with myeloma, or IFNβ2 due to its IFN anti-viral activity. Later, it was discovered that these diverse functions reflect the wide range of target organs for IL-6. 1 In 1986, two groups independently reported on the molecular structures of IFNβ2 and BSF-2.2,3 Since these two molecules were found to be identical, the cytokine was renamed IL-6. Human IL-6 (GenBank: X04602) consists of 212 amino acids, includes a 28-amino-acid signal peptide, and its gene has been mapped to chromosome 7p21. The structure of IL-6 includes four helix bundles, which are arranged in an up-up-down-down topology (Fig. 1: A to D), and three loops (two long ones, A–B and C–D, and a short one, B–C). 4
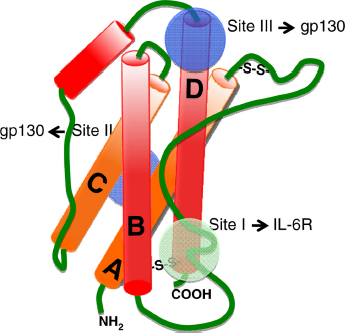
Schematic representation of the domain structure of human IL-6.
IL-6 mediates inflammatory signals from infected or injured lesions to other parts of the body in order to generate protection against emergent events. There are many cytokines that are involved in and regulate inflammation, but IL-6 is the most imoprtant for mediating systemic inflammation. The clinical function of IL-6 was discovered first by observing and examining patients with cardiac myxoma or Castleman's disease in whom IL-6 was persistently produced from myxoma tissues or involved lymph nodes, respectively.5,6 These patients presented with symptoms of systemic inflammation and autoimmunity (eg, fever, arthralgia, fatigue) and laboratory findings of inflammation such as elevated C-reative protein (CRP), serum amyloid A (SAA), and fibrinogen levels, as well as thrombocytosis. As a result of chronic inflammation, anemia, autoantibody production, hypergammaglobulinemia and hypoalbuminemia were also detected. The fact that all of these signs and laboratory data became normalized after surgical removal of cardiac myxoma suggested that IL-6 played a role in systemic inflammation. An IL-6 deficient mouse model was thus compared with its IL-6 transgenic expression counterpart. The IL-6 deficient mice could develop almost normally and were fertile, 7 indicating that IL-6 is not essential for organ development, whereas mice with persistent IL-6 expression showed chronic inflammation accompanied by swollen lymph nodes and spleen. 8 Similarly, healthy human individuals feature almost undetectable serum IL-6 levels, whereas in those with infection, trauma or burn IL-6 levels increase and inflammatory symptoms appear. 9 These findings indicate that production of IL-6 provides an SOS signal to indicate occurrence of an emergent infection or injury in an organ.
In infectious stress, IL-6 is promptly produced by monocytes and macrophages through the stimulation of Toll-like receptors (TLRs) by microbial motifs. 10 IL-6, when acting on hepatocytes, induces a wide spectrum of acute phase proteins, such as CRP, SAA, fibrinogen, and hepcidin, and inhibits the synthesis of albumin (Fig. 2). 11 High levels of hepcidin cause increased internalisation of iron transporter ferro-portin molecules on cell membranes, which inhibits release from iron stores and leads to anemia of chronic disease. 12 Induction of zinc transporter Zip14 by IL-6 in liver increases zinc uptake into hepatocytes, resulting in hypozincemia. 13 IL-6 in the bone marrow promotes maturation of megakaryocytes, which leads to the production of platelets. 14 These changes in acute phase serum protein levels and red blood cell and platelet counts are in fact used in clinical practice for the diagnosis and evaluation of inflammatory severity. After the infiltration of neutrophils into the focus of acute infection, IL-6 orchestrates the switch in leukocyte recruitment from neutrophils to more sustained monocytic cells that develop acquired immune responses. 15 IL-6 thus performs an important function in acquired immunity. In lymphocytes, CD4-positive T-helper cells display distinct effector functions after their differentiation. 16 IL-6, together with TGF-β, has been shown to be essential for Th17 differentiation from naive CD4-positive T cells. 17 On the other hand, IL-6 inhibits the generation of regulatory T cells (Treg) induced by TGF-β.16,17 The resultant Th17/Treg imbalance leads to a rupture in immunological tolerance and is thus essential for the development of autoimmune or chronic inflammatory diseases. 18 IL-6 also acts on CD8-positive T cells to induce cytotoxic T cells, 19 and on activated B cells for differentiation into Ab producing cells. 20 Besides its effects on hepatocytes and lymphocytes, IL-6 has various effects that are associated with inflammatory symptoms. For example, IL-6 production in bone marrow stromal cells induces the receptor activator of NF-kappaB ligand (RANKL), which is an essential factor for the differentiation and activation of osteoclasts and bone resorption.21,22 IL-6 production by bone marrow stromal cells is suppressed by 17 beta-estradiol and in mice the loss of estrogen due to ovariectomy increased the number of osteoclasts in trabecular bone. Conversely, the administration of 17 beta-estradiol or anti-IL-6 Ab prevented such an increase, 23 suggesting that estrogen loss results in an IL-6-mediated bone resorption in postmenopausal osteoporosis and affects the development of rheumatoid arthritis (RA) in postmenopausal women. 24 Furthermore, enhanced angiogenesis and increased vascular permeability are pathological features of chronic inflammatory sites such as synovial tissue of RA; these pathological changes are due to the excess production of vascular endothelial growth factor (VEGF), which is induced by IL-6. 25 Finally, IL-6 has been demonstrated to promote the proliferation of keratinocytes 26 and collagen production 27 in dermal fibroblasts.
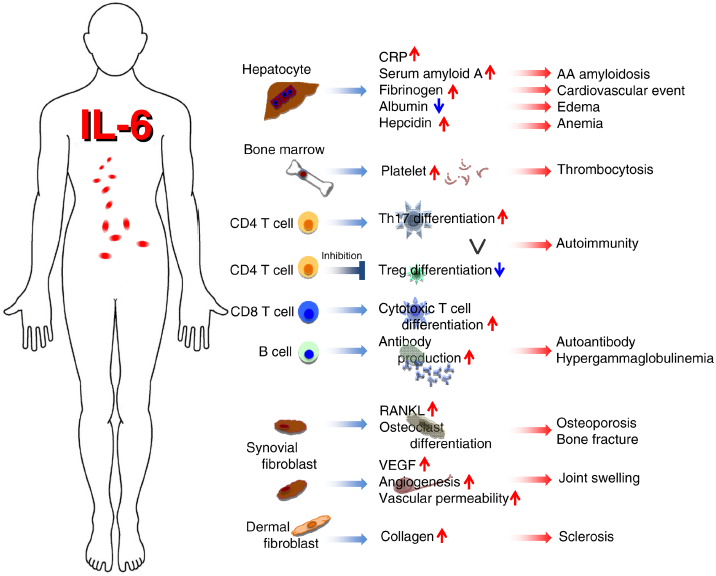
Pleiotropic activity of IL-6.
The IL-6 receptor (IL-6R) system consists of two chains, IL-6-binding chain IL-6R and gp130, both of which belong to the cytokine receptor family. 28 The latter transduces the IL-6 signal into cells. 29 The broad range of expression of gp130 on various cells suggests that IL-6 has pleiotropic effects because the soluble form of IL-6R (sIL-6R) is present in human serum. 30 The IL-6/sIL-6R complex can transduce the IL-6 signal on gp130-expressing cells even in cells lacking transmembrane IL-6R. The IL-6/IL-6R complex in turn induces homodimerization of gp130 to trigger downstream events in cytoplasm (Fig. 3). The activated IL-6 receptor complex is formed as a hexameric structure comprising two molecules each of IL-6, IL-6R, and gp130. 31 In this complex, IL-6 provides one IL-6R binding site (site I) and two gp130 binding sites (sites II and III) (Fig. 1). IL-6R is a unique binding receptor for IL-6 and its expression is restricted to certain cells, including leukocytes and hepatocytes, whereas signal-transducing chain gp130 is expressed in almost all cells and is shared by members of the IL-6 family of cytokines. 32 These members include leukemia inhibitory factor (LIF), oncostatin M (OSM), ciliary neurotrophic factor (CNTF), IL-11, cardiotrophin 1 (CTF1), cardiotrophin-like cytokine (CLC), IL-27, and IL-35. 33 These cytokines bind to their unique binding receptors but use their common gp130 to transduce their signal. The only exception is virus-encoded IL-6 (vIL-6), which is the product of Kaposi's sarcoma-associated herpes virus (also known as human herpes virus 8) and directly binds to and activates gp130. 34 It is known that the activities of the IL-6 family of cytokines often overlap with those of IL-6. The model in which IL-6 family members use the common signal-transducing chain makes it clear why different cytokines show functional redundancy. Activated gp130 triggers activation of the JAK (Janus kinase)-STAT3 (signal transducer and activator of transcription 3) pathway and the JAK-SH2-domain containing protein tyrosine phosphatase-2 (SHP-2)-mitogen-activated protein (MAP) kinase pathway. In this context, STAT3 is the transcriptional factor that regulates various sets of IL-6 responsive genes, including acute phase proteins. 28
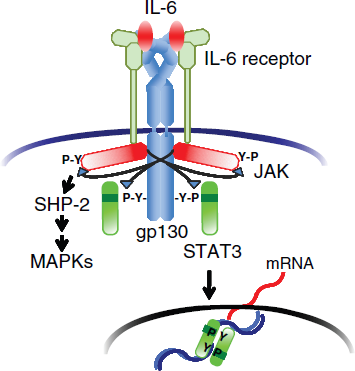
IL-6 receptor system.
IL-6 is produced from inflammatory lesions. The synthesis of IL-6 is strictly regulated both through levels of gene transcription and the stability of mRNA. The transcriptional factors NF-kB and NF-IL-6 (also known as C/EBPβ) promote transcription of IL-6 mRNA in response to various stimuli.35,36 Some RNA binding proteins or micro-RNAs bind to the 3‘-untranslated regions of the IL-6 mRNA so as to control the stability of mRNA.37,38 However, dysregulated and persistent expression of IL-6 has been implicated in the development of various autoimmune diseases, chronic inflammatory diseases, and even cancers; the IL-6 signal pathway could therefore be an appropriate target for the treatment of such diseases. 39 Inhibition of this function of IL-6 consists of four strategies–-inhibition of any of IL-6, IL-6R, gp130, or the cytoplasmic signaling cascade via gp130. The humanized anti-IL-6R monoclonal Ab, tocilizumab (TCZ), 40 is the first drug that successfully blocks the IL-6 signal and has been approved worldwide for the treatment of RA.
Tocilizumab, a Humanized Anti-IL-6 Receptor Monoclonal Antibody
TCZ blocks IL-6-mediated signal transduction by inhibiting IL-6-binding to transmembrane and soluble IL-6R. If concentration of free TCZ is maintained at more than 1 μg/mL, CRP remains negative 41 and the serum CRP level becomes a hallmark for in vivo confirmation of whether IL-6 activity is completely blocked. TCZ is currently approved for the treatment of RA in more than 90 countries, for the treatment of systemic juvenile idiopathic arthritis (JIA) in Japan, India, the EU, and the USA, and of polyarticular JIA and Castleman's disease in Japan and India.42–44 For RA, the recommended TCZ posology is 8 mg/kg once every 4 weeks in Japan and the EU. The recommended starting dose in the USA is 4 mg/kg once every 4 weeks, followed by an increase to 8 mg/kg, depending on clinical response. TCZ is used for the treatment of Castleman's disease and systemic JIA at 8 mg/kg once every 2 weeks in Japan, while in the USA the recommended TCZ dosing for systemic JIA patients weighing less than 30 kg is 12 mg/kg and 8 mg/kg for those weighing more than 30 kg, intravenously once every 2 weeks.
Efficacy and Safety of Tocilizumab for Rheumatoid Arthritis
RA is an inflammatory arthritis which causes joint destruction. Inflammatory cytokines including IL-6 play an important role in the pathogenesis of RA; treatment options which inhibit IL-6 activity for patients with RA have advanced significantly over the past decade. Among these, TCZ is the first approved IL-6 signaling inhibitor.
The key mechanism by which TCZ affects RA is thought to be interference with systemic inflammation, which can normalize imbalance in the effector CD4-positive T cell subsets that generate autoimmune reactions. Interestingly, IL-6 blockade inhibits arthritis in type II collagen-induced arthritis (CIA), which requires the presence of CD4-positive T cells, and leads to the production of antitype II collagen IgG. 45 It does not, however, inhibit the development of antitype II collagen Ab-induced arthritis (CAIA), which skips the priming phase of T cell-dependent Ab generation. 46 These observations suggest that IL-6 is required primarily for the activation of T cell responses, as well as the production of Abs specific to joint components (the priming phase). 47 TCZ has little effect on spondyloarthritides such as psoriatic arthritis and ankylosing spondylitis,48,49 for which the autoimmune process appears not to be required. Additionally, the onset of the anti-arthritic effect of TCZ is slow compared with that of TNF inhibitors. These observations suggest that TNF contributes to local inflammation, a direct cause of joint destruction, and IL-6 mainly contributes to immune activation, synovitis, and systemic inflammation (Fig. 4).
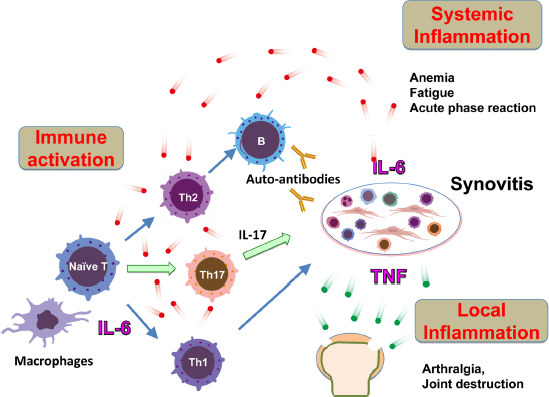
Pathological role of IL-6 in rheumatoid arthritis.
The efficacy of TCZ on RA was evaluated in seven major phase III clinical trials. Its efficacy was found to be outstanding for RA patients either as monotherapy or in combination with disease-modifying antirheumatic drugs (DMARDs).42,43 The efficacy of TCZ administered alone or in combination with methotrexate (MTX) was also confirmed for RA patients of various backgrounds. The results of these trials are summarized in Table 1. The findings of three phase III trials of TCZ monotherapy have been reported. The AMBITION trial was performed with MTX- or biologic-naïve patients, 50 the SAMURAI trial with patients with an inadequate response to DMARDs, 51 and the SATORI trial with patients with an inadequate response to MTX. 52 These studies showed that TCZ monotherapy is better than MTX and DMARD monotherapy for patients who are naïve to MTX and other biologics and that it is also effective for those who are resistant to MTX and other DMARDs. All studies showed that after 24 weeks patients treated with TCZ showed superior ACR20 (American College of Rheumatology) responses and reduction in disease activity score 28 (DAS28) when compared to controls treated with MTX or other DMARDs.50–52 In addition, four phase III trials of TCZ combination therapy have been performed. The OPTION trial was designed to evaluate the efficacy of TCZ in combination with MTX and showed that TCZ combination therapy was effective for and well tolerated by patients with moderate-to-severe active RA and with an inadequate response to MTX.53,54 The TOWARD trial, with an enrollment of 1,220 RA patients, demonstrated that TCZ combined with DMARDs such as MTX, chloroquine, gold, sulphasalazine, azathioprine, and leflunomide was effective for reducing RA disease activity in patients with an inadequate response to monotherapy with any one of the DMARDs. 55 The RADIATE trial proved that TCZ plus MTX was effective for achieving rapid and sustained improvements in signs and symptoms of patients with RA refractory to TNF inhibitors; it also showed that it had a manageable safety profile. 56 The LITHE trial, which was designed to evaluate not only disease activity but also structural joint damage, demonstrated that TCZ plus MTX was effective for the suppression of disease activity and symptoms. The LITHE trial also showed that at 52 weeks TZ plus MTX significantly inhibited radiographic progression for patients with an inadequate clinical response to MTX.57,58 In short, all these studies enrolled patients with an inadequate response to all previous treatments–-including MTX, TNF inhibitors, and other DMARDs–-and all showed that TCZ combination therapy was effective for these populations. In addition, the OPTION, RADIATE, and LITHE studies reported that the efficacy of TCZ was dose dependent (4 mg/kg and 8 mg/kg).53,56,57
Summary of phase III clinical trials of tocilizumab in rheumatoid arthritis.
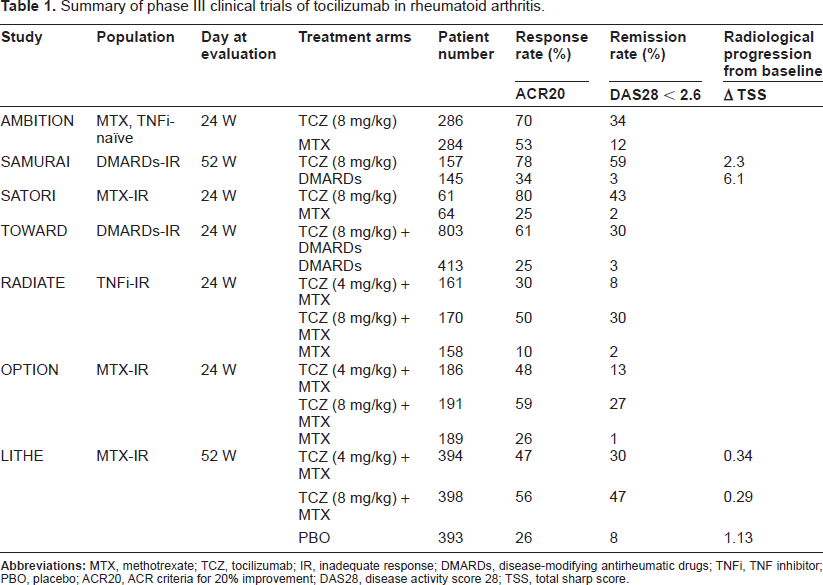
Recently many phase IIIb/IV studies have been conducted in clinical settings as well. The ACT-RAY trial compared TCZ plus MTX therapy with TCZ monotherapy in a setting that closely resembled real-life clinical practice. 59 The trial was a double-blind, 2-year study in which active RA patients being treated with MTX were randomly assigned either to continue MTX with the addition of TCZ 8 mg/kg every 4 weeks or to switch to TCZ plus placebo. Of 556 randomly assigned patients, 512 (92%) completed 24 weeks. ACR20/50/70 response rates were 71.5%/45.5%/24.5% for the TCZ plus MTX group and 70.3%/40.2%/25.4% for the TCZ plus placebo group. TCZ plus MTX combination therapy was therefore shown not to be clinically superior to TCZ monotherapy. The ACT-SURE trial was a phase IIIb, open-label, single-arm, 6-month study which was conducted with a patient population resembling one to be expected in clinical practice. 60 Patients, who were categorised as TNF inhibitor-naïve (never received TNF inhibitor), TNF inhibitor-previous (washout: TNF inhibitor therapy discontinued for more than 2 months) or TNF inhibitor-recent (TNF inhibitor discontinued for less than 2 months), received open-label TCZ 8 mg/kg every 4 weeks, with or without DMARDs, for 24 weeks. Overall, 1,681 (976 TNF inhibitor-naïve, 298 TNF inhibitor-previous, and 407 TNF inhibitor-recent) patients were treated. ACR20/50/70 rates by week 24 were 70.5%/51.9%/31.8%, 60.7%/35.2%/17.8% and 62.7%/42.3%/19.7% for TNF inhibitor-naïve patients, TNF inhibitor-previous, TNF inhibitor-recent patients, respectively. The ACT-STAR study was a 24-week, prospective, open-label study conducted in the USA. 61 Of 886 patients enrolled, 163 were assigned to TCZ 8 mg/kg monotherapy. The remaining 723 patients were assigned to combination therapy, with 363 randomized to receive a starting dose of TCZ 4 mg/kg plus DMARDs and 360 to receive a starting dose of TCZ 8 mg/kg plus DMARDs. In the initial TCZ 4 mg/kg plus DMARDs group, 152 patients (41.9%) at week 8 and 68 patients (18.7%) after 8 week escalated to TCZ 8 mg/kg plus DMARDs. Overall, the 24-week study was completed by 82.5% of patients. By week 24, 49.7%, 27.1%, and 10.3% of the patients in the 8 mg/kg TCZ plus DMARDs group and 47.9%, 24.5%, and 7.4% of the patients in the 8 mg/kg TCZ monotherapy group had attained ACR20/50/70 responses. The safety profiles and efficacy were similar for the TCZ monotherapy and the TCZ plus DMARD groups. Therefore, the efficacy of TCZ monotherapy and combination therapy with MTX or DMARDs was similar, demonstrating that TCZ monotherapy can provide patients who cannot use MTX with a good quality of life (QoL).
In addition, TCZ showed a good continuation rate and could induce drug-free remission. Reduced efficacy is one of the issues when using a treatment for RA that includes biologics. In the Danish DANBIO registry, the drug adherence rate after 48 months was 64%. 62 In another multicentre registry, the Japanese REAL, the continuation rate for TCZ at 2.5 years was 67%. 63 The DREAM study reported findings on the maintenance of TCZ-free remission in TCZ monotherapy; 64 remisson rates following the cessation of TCZ were 35.1% and 13.4% at 24 and 52 weeks, respectively. Even if recurrence was noted after TCZ cessation, TCZ re-administration could re-induce remission. 65 The OPTION trial reported on maintenance of TCZ-free remission for combination therapy with MTX; efficacy after the cessation of TCZ was 52.5% at 12 months. 66 Relapses occurred in 47.5% patients, half of which were noted during the first 3 months after the final TCZ administration.
The systemic effects of TCZ enhance the satisfaction of RA patients; health-related QoL (HRQoL) was reportedly improved with TCZ therapy.67,68 Depression, fatigue, and quality of sleep in patients treated with TCZ also improved.69–71
The safety profile of TCZ monotherapy was first published in 2010. 72 The overall incidence of adverse events (AE) and serious AE (SAE) rates were 465.1/100 patient-year (PY) and 6.22/100 PY, respectively. As for the risk of AEs for RA patients treated with TCZ, pooled odds ratios (ORs) indicate a statistically significantly increased risk of AEs (OR = 1.53; 95% confidence interval (CI) = 1.26–1.86) in addition to a heightened risk of infection (OR = 1.30; 95% CI = 1.07–1.58). The safety profile of TCZ combination therapy was first published in 2011. 73 Overall AE and SAE rates were 278.2/100 PY and 14.4/100 PY, respectively. These events included serious infections (4.7/100 PY), opportunistic infections (0.23/100 PY), and gastrointestinal perforations (0.28/100 PY). Pooled ORs revealed a significantly increased risk of AEs (OR = 1.53; 95% CI = 1.26–1.86) and infection (OR = 1.30; 95% CI = 1.07 = 1.58) for patients treated with 8 mg/kg TCZ plus MTX compared with that for controls.
TCZ blocks IL-6 function and has been shown to have excellent efficacy and tolerability when used in the treatment of RA. The efficacy of TCZ monotherapy appears to be similar to that of combination therapy with DMARDs or MTX. In addition, the recent development of the option of subcutaneous administration of TCZ may also be beneficial for patients with RA. Because of the outstanding efficacy of TCZ, other new biological agents that target IL-6 are also being developed. These IL-6 inhibitors can be expected to constitute a new class of antirheumatic drugs in the future.
The clinical efficacy of TCZ in RA is mediated by the inhibition of IL-6-induced RANKL induction followed by osteoclastogenesis and suppression of IL-6-induced production of matrix metalloproteinases. 74 Moreover, it has been shown that TCZ corrects Th17 (CD4+IL-17+)/Treg (CD4+CD25highFoxp3+) imbalance in RA patients. 75 Other studies have found both that TCZ induced a significant reduction in the peripheral pre-switch and post-switch memory B cells of RA patients 76 and that TCZ but not TNF inhibitors significantly reduced somatic hypermutation in immunoglobulin gene rearrangements in pre-switch memory B cells. 77 These findings thus indicate that modulation of memory B cells may constitute a potential target for TCZ.
Additional Applications of Tocilizumab for other Immune-Mediated Diseases
In addition to the diseases for which it has been approved, IL-6 blockade strategy is expected to come into extensive use for the treatment of various other immune-mediated diseases; several clinical studies to evaluate the efficacy and safety of TCZ for such diesases are in progress.
Systemic sclerosis
Systemic sclerosis (SSc) is a connective tissue disease, characterized by skin and tissue fibrosis, vasculopathy, and immune abnormalities. 78 Numerous studies have analyzed the pathogenic mechanisms of SSc, but no effective treatment has been established yet. 79 IL-6 is a potential target for SSc therapy for several reasons.80,81 IL-6 levels in the sera of SSc patients are elevated and these levels correlate well with skin severity scores.82–86 Indeed, the culture supernatants of peripheral blood mononuclear cells and skin tissues from patients with SSc were found to contain higher concentrations of IL-6 than did those from controls.87–89 Furthermore, in vitro studies indicate that IL-6 may contribute to fibrosis by inducing collagen production 27 by dermal fibroblasts and induce their differentiation into myofibroblasts. 90 Moreover, anti-IL-6 Ab suppressed procollagen type 1 production in vitro in fibroblasts derived from SSc patients. 91 Another reason for targeting IL-6 in SSc is that SSc serum mediated largely by IL-6 was found to induce endothelial cell activation and apoptosis in endothelial cell-neutrophil co-cultures. 92 Loss of IL-6 expression led to amelioration of skin and lung fibrosis in a mouse model immunized with DNA topoisomerase 1 and Freund's complete adjuvant. 93 Meanwhile, IL-6 deficiency produced by either administration of anti-IL-6R Ab or gene knockout in a bleomycin-induced mouse model of SSc suppressed fibroblast activation, which resulted in a reduction of dermal thickness and hardness. 94
We reported our clinical findings for two patients with SSc who responded well to TCZ. 95 The skin became softer as indicated by the modified Rodnan total skin score and a novel device known as Vesmeter, which can measure viscosity, elasticity, and hardness of the skin. 96 Histological analysis showed that collagen fibre bundles had become thinner and activated myofibroblasts reduced in the dermis after a 6-month treatment with TCZ.94,95 These findings indicate that IL-6 blockade strategy is a promising approach for the treatment of SSc. A phase II/III, multicenter, randomized, double-blind, placebo-controlled study is in progress to assess the efficacy and safety of TCZ as compared with a placebo for patients with SSc.
Polymyositis
Inflammatory myopathies encompass a group of acquired muscle disorders that includes polymyositis (PM), dermatomyositis, and inclusion body myositis; these disorders share the clinical features of progressive symmetrical muscle weakness and mononuclear inflammatory cell infiltrating muscle tissue. 97 PM appears to be another suitable target disease for TCZ for three reasons. Firstly, excessive IL-6 expression has been found in the sera and in the infiltrating mononuclear cells in the muscles of PM patients.98–101 Secondly, infiltrating cytotoxic T cells are thought to be involved in muscle fiber damage and IL-6 reportedly functions as a helper factor in the induction of cytotoxic T cells. 19 Thirdly, in a model of myosin-induced experimental myositis it was shown that control mice developed clinically manifest muscle damage, whereas IL-6-deficient mice showed no clinical or histological signs of muscle damage. 102 In another model of PM, known as C protein-induced myositis, intraperitoneal administration of anti-IL-6R Ab reduced the severity of myositis. 103
We therefore administered TCZ to two PM patients who had been refractory to corticosteroids and immunosuppressive drugs for more than 5 years. Creatine phosphokinase levels of both patients normalized and magnetic resonance images showed that high-intensity zones in the thigh muscles had disppeared, 104 indicating that TCZ administration may also be an option for the treatment of refractory PM.
Vasculitis syndrome and polymyalgia rheumatica
Vasculitis syndrome refers to a heterogeneous group of disorders featuring inflammation and damage of blood vessel walls, leading to tissue necrosis.105,106 According to the 2012 revised international Chapel Hill Consensus Conference nomenclature of primary vasculitis syndrome, large vessel vasculitis includes giant cell arteritis (GCA) and Takayasu arteritis (TA), medium vessel vasculitis comprises polyarteritis nodosa and Kawasaki disease, and small vessel vasculitis includes anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis, immune complex small vessel vasculitis, and hypocomplementary urticarial vasculitis. The pathological significance of IL-6 for large vessel vasculitis has been well documented.107,108 It was found that the serum concentrations of IL-6 were elevated at the onset and during clinical relapse and correlated with the disease activity of GCA and TA,109–115 while tissue-infiltrating cells reportedly produced major quantities of IL-6 in patients with GCA and TA.116–119
Nishimoto et al were the first to report that TCZ treatment for a 20 year old woman with refractory active TA improved clinical manifestations and abnormal laboratory findings. 120 It was subsequently reported that TCZ treatment induced a rapid remission in five patients with GCA and two patients with TA, so that cumulative corticosteroid doses could be reduced substantially. 121 Corticosteroid treatment is currently recognized as the mainstay therapy for GCA, but interestingly, TCZ as monotherapy without corticosteroids was effective for two of the patients with GCA in the study. Furthermore, recent case reports and case series of off-label use of TCZ have demonstrated its outstanding efficacy for refractory GCA and TA.122–132 It has been reported to date that the treatment had rapid beneficial effects on 22 patients with GCA and 12 patients with TA, resulting in the successful tapering of corticosteroids, with the exception of one GCA patient. Instrumental examination of the arteries involved by determining the vascular 18 F-fluorodeoxyglucose (FDG) uptake on positron emission tomography by means of CT and ultrasonographic resolution, showed improvement in all cases evaluated. These results strongly suggest that IL-6 inhibition may become a novel therapeutic strategy for large vessel vasculitis. As of this study, a phase II, randomized, double-blind, placebo controlled study of TCZ for patients with GCA has just been initiated.
In addition to the clinical effect of TCZ on large vessel vasculitis, the treatment reportedly improved clinical symptoms in a patient with a chimeric anti-CD20 antibody rituximab-refractory cryoglobulinemic vasculitis. 133 A patient with myeloperoxidase-ANCA-associated crescentic glomerulonephritis complicated by RA also showed improvements. 134
Polymyalgia rheumatica (PMR) is a chronic inflammatory disorder that affects the elderly and is characterized by aching and morning stiffness in the shoulders, neck, and pelvic girdles. 107 PMR can occur in its isolated form or may be associated with GCA. Although the pathogenesis of this disorder remains unknown, IL-6 has been identified as the only cytokine occurring at a consistently high level in patients with the active form of the disease; it is recognized as the most sensitive indicator of disease activity and course.107,109–111,135 IL-6 was also reported to be elevated in the synovial fluid 136 and in the muscle interstitial concentrations of cytokines. 137 IL-6 inhibition with TCZ may thus constitute a novel strategy for the treatment of PMR. Low doses (15–20 mg/day) of corticosteroids are effective for PMR, but 43%–65% of patients with this disease reportedly experienced at least one steroid-related adverse event such as osteoporosis, fragility fracture, or arterial hypertension.138,139
We found that TCZ had a significantly beneficial effect on a patient with long-standing steroid-refractory PMR complicated by diabetes, osteoporosis, and hypertension. 140 After five injections of TCZ, all symptoms such as pain and morning stiffness improved and the patient was judged to be in remission; improvements were such that the prednisolone dose could be reduced. In addition, TCZ treatment was found to produce a disease-free status for four patients with PMR complicated with GCA. 121 As mentioned before, two patients with PMR treated with TCZ but not with corticosteroids also attained remission. Moreover, recent studies have demonstrated the ameliorative and steroid-sparing effects of TCZ for patients with PMR,126,130 while a phase IIa clinical trial of tocilizumab for the treatment of polymyalgia rheumatica is in progress.
Systemic lupus erythematosus
Systemic lupus erythematosus (SLE) is a systemic autoimmune disease of unknown etiology that mainly affects young women. 141 While pathogenesis of SLE remains unclear, cytokine dysregulation is pervasive in SLE and its expression profiles may serve as a marker of disease activity and severity. Recent findings have highlighted the presence of the type I interferon pathway 142 or the activation of Th17 cell 143 in the pathogenesis of SLE. IL-6 has also been shown to play a role in the development of SLE, 144 since it was found that serum IL-6 levels of SLE patients were elevated,145–147 urinary excretion of IL-6 had increased in SLE patients with active proliferating lupus nephritis,146,148 and IL-6 levels were elevated in the cerebrospinal fluid of SLE patients with central nervous system (CNS) involvement. 149 A comparison with healthy controls showed that SLE patients have significantly more IL-6 secreting peripheral blood mononuclear cells. 150 Moreover, lymphoblastoid cells isolated from SLE patients produced higher levels of IL-6 and blocking of IL-6 inhibited anti-double-stranded DNA (dsDNA) production in vitro.151,152 In murine SLE models, IL-6 administration exacerbated glomerulonephritis,153,154 while IL-6 blockade by means of anti-IL-6R or anti-IL-6 Ab prevented the onset and progression of the disease.155,156 Mice with epidermal loss of JunB reportedly developed an SLE phenotype associated with enhanced epidermal IL-6 secretion. 157 Facial skin biopsies of SLE patients displayed low levels of JunB protein expression and high levels of IL-6 and activated STAT3 within lupus lesions. 157
An open-label phase I dosage-escalation study (2 mg/kg, 4 mg/kg or 8 mg/kg of TCZ, every 2 weeks for 12 weeks) with an enrollment of 16 SLE patients with mild-to-moderate disease activity showed significant improvement in disease activity in 8 of the 15 evaluable patients, with a median reduction of 47% in levels of anti-dsDNA antibodies. 158 The percentage of CD38highCD19lowIgDnegative plasma cells in the peripheral blood, which was higher for SLE patients than for normal controls (mean 5.3% vs. 1.2%), had been significantly reduced to 3.1% after 6 weeks. Moreover, according to two case reports, TCZ was efficacious for a patient with intractable SLE complicated by RA and treated in combination with tacrolimus. 159 TCZ was also efficacious for a multiple drug-refractory SLE patient with cutaneous lupus and urticarial vasculitis. 160 These results indicate that TCZ represents a promising therapeutic biologic for SLE.
Relapsing polychondritis
Relapsing polychondritis is a very rare disease, characterized by recurrent inflammation and cartilage destruction;161,162 the cartilaginous structures of ear, nose, joints and respiratory tract are especially affected. 163 Autoimmune reactions to antigens present in cartilage, for example, type II, IX and XI collagen and matrilin and excess generation of proinflammatory cytokines and chemokines, are thought to evoke the disease symptoms. 164 The involvement of laryn-gotracheal cartilage causes severe airway destruction and thus requires vigorous treatments with corticosteroids and immunosuppressive drugs. Biologics including TNF inhibitors, rituximab, and IL-1R antagonist anakinra, have been successfully used for refractory relapsing polychondritis. 165 Two patients with relapsing polychondritis, who had been refractory to conventional regimens, were treated with TCZ. 166 The treatment ameliorated clinical symptoms related to the upper and lower airways. In one patient, airway narrowing of the bronchi was improved within one year of treatment while in the other patient Gallium citrate uptake in the involved cartilages had disappeared 21 months after the treatment. The TCZ treatment for these two patients has continued for more than five years without any signs of relapse so that the prednisolone dose could be reduced. Moreover, in a patient with relapsing polychondritis, tocilizumab caused a complete resolution of all clinical symptoms, which had been refractory to conventional therapy and adalimumab, a TNF inhibitor. 167 Because of the very rare occurrence of the disease it is difficult to perform a randomized controlled trial and in order to evaluate its efficacy for this disorder there is therefore an urgent need for reports on clinical experience with the use of TCZ for relapsing polychndritis.
Neuromyelitis optica
Neuromyelitis optica (NMO) is a chronic inflammatory CNS disorder predominantly affecting the spinal cord and optic nerves, in which anti-aquaporin-4 (AQP4) autoantibodies perform a pathologic function.168,169 AQP4, which belongs to the aquaporin family of integral membrane proteins that conduct water through the cell membrane, is constitutively expressed in astrocytes and is upregulated in response to direct insult to the CNS. 170 Several studies have reported a marked increase of IL-6 in cerebrospinal fluid of patients with NMO.171–174 Moreover, it has been found that the population of plasmablasts showing the CD19intCD27highCD38highCD180negative phenotype was selectively increased in the peripheral blood of NMO patients and that anti-AQP4 Abs were mainly produced by the plasmablasts. 175 IL-6 enhanced the survival of plasmablasts, as well as anti-AQP4 Ab secretion, whereas anti-IL-6R Ab diminished their survival and, indeed, clinical improvement and reduced serum levels of anti-AQP4 Abs were observed in a patient with NMO following therapy with TCZ. 176 Moreover, the prominent positive effect of TCZ was reported in a patient with NMO who had failed to respond to numerous immunosuppressive interventions, including high-dose corticosteroids, mitoxantrone, plasma exchange, rituximab and anti-CD52 Ab, alemtuzumab. 177
Crohn's disease
Crohn's disease (CD) is a chronic inflammatory bowel disease of unknown etiology. However, both Th1 and Th17 cells are believed to play a central role in its development.178,179 IL-6 also contributes to its development, since elevated levels of IL-6 have been detected in the blood and in the cultures of colonic mucosal specimens from CD patients.180–182 In a colitis mouse model generated by transfer of CD45RBhighCD4positive T cells into SCID mice, anti-IL-6R Ab prevented the occurrence of signs and symptoms of colitis. 183
A randomized pilot trial of TCZ for 36 patients with active CD demonstrated that 80% of the patients given 8 mg/kg every 2 weeks showed a clinical response, as compared with only 31% of placebo-injected patients. 184 Colitis-associated intestinal perforation is one of the most serious complications of inflammatory bowel diseases, but there was no event of gastrointestinal perforation in TCZ- or placebo-injected patients in the clinical trial for CD.
Adult-onset still's disease
Adult-onset Still's disease (AOSD) is a chronic inflammatory disease characterized by four cardinal symptoms, namely spiking fever, evanescent maculopapular rash, arthritis, and leukocytosis.185,186 Pathologically it resembles systemic JIA; IL-6 levels are elevated and correlate with disease activity in patients with AOSD.187–190 Although the efficacy and safety of TCZ for AOSD have not yet been evaluated in a randomized controlled trial, numerous case and pilot studies have shown that TCZ treatment improved clinical symptoms and signs of AOSD in patients who had been refractory to conventional treatment regimens and biologics, including TNF inhibitors and anakinra.191–207 These findings indicate that TCZ may become a first-line biologic for the treatment of AOSD.
Amyloid A amyloidosis
Amyloid A amyloidosis is a serious complication of chronic inflammatory diseases in which amyloid fibril deposition causes progressive deterioration in various organs. 208 SAA, an acute phase protein produced in the liver, is an amyloid fibril precursor protein and sustained high concentrations of SAA have been found to correlate with progression of renal amyloid diseases. 209 Several therapeutic strategies have been proposed for the treatment of amyloid A amyloidosis, but the inhibition of SAA appears to be the most suitable approach, since long-term suppression of its level (less than 10 mg/L) was found to lead to a regression or stabilization of the amyloid load. 210 Activation of the SAA1 gene depends primarily on IL-6,211,212 and indeed, TCZ administration was found to cause a marked reduction of serum SAA concentrations, irrespective of the underlying diseases.53,213–215 Case studies of amyloid A amyloidosis complicated with RA, JIA, or latent tuberculosis reported that TCZ had an ameliorative clinical effect on gastrointestinal symptoms and renal function.216–222 Surprisingly, amyloid A fibril deposits were found to have disappeared in two cases after only three injections of TCZ.217,219 This suggests that IL-6 blocking may be an innovative strategy for amyloid A amyloidosis complicated with chronic inflammatory diseases. 223
Behcet's disease and uveitis
Behcet's disease (BD) is a systemic inflammatory disease of unknown etiology, characterized by relapsing episodes of oral aphthous ulcers, genital ulcers, skin lesions, ocular lesions, and other manifestations that include neurological, gastrointestinal, and vascular involvement. 224 The involvement of IL-6 has been demonstrated in the pathological development of BD.225–228 We administered TCZ in the treatment of one BD patient with posterior uveitis who had been suffering from BD for 10 years and had been treated with colchicine, prednisolone, and ciclosporin. 229 Treatment with infliximab used for recurrent posterior uveitis brought the uveitis attacks under satisfactory control. When a severe relapse of posterior uveitis occurred 16 months later, however, TCZ was initiated and continuous treatment for 1 year reduced the number of ocular attacks and the BD Current Activity Form score. 229 IL-6 may well be the most important inflammatory cytokine involved in both acute and chronic progressive neuro-BD,230–232 and a case report of off-label use with TCZ noted outstanding and beneficial effects in a patient with neuro-BD refractory to all previous treatment regimens. 233
Autoimmune and inflammatory uveitis refers to a group of potentially blinding intraocular inflammatory diseases that arise without a known infectious trigger and which are often associated with immunological responses to unique proteins; they are also known to frequently occur in conjunction with systemic diseases. 234 IL-6 is known to be elevated in the vitreous body of patients with active intermediate and posterior uveitis,235–239 and both Th1 and Th17 cells have been found to play a pathologic role in experimental autoimmune uveoretinitis, an animal model of uveitis. 240 Blockade of IL-6 signaling reportedly suppresses autoimmune uveoretinitis and its protective effect is mediated by Treg induction and by Th17 and Th1 inhibition.240–242 This indicates that TCZ may constitute a therapeutic option for uveitis; a recent case report on the clinical efficacy of TCZ for two patients with anti-TNFα refractory uveitis has suggested this possibility. 243 In this context, an open-label trial has just been started to assess the efficacy and safety of TCZ in the management of JIA-associated vision-threatening uveitis refractory to other modes of systemic immunosuppression.
Other candidate diseases
Case series or reports of off-label use of TCZ have also raised the possibility that TCZ might be applicable for the treatment of other diseases. These include graft-versus-host disease,244,245 acquired hemophilia A, 246 autoimmune hemolytic anemia,247–249 remitting seronegative, symmetrical synovitis with pitting edema, 250 pulmonary arterial hypertension,251–253 tumor necrosis factor receptor-associated periodic syndrome, 254 atopic dermatitis, 255 interstitial granulomatous dermatitis, 256 and sciatica. 257 Moreover, some studies have reported the efficacy of TCZ for spondyloarthritides such as ankylosing spondylitis258–262 and reactive arthritis.263,264 However, recent clinical trials of TCZ 49 and sarilumab, 265 a fully human anti-IL-6R Ab, could not detect any favorable effect on ankylosing spondylitis, nor did our study of the clinical effect of TCZ on skin and joint lesions in two patients with psoriasis. 48 Observational studies of RA patients complicated with type 2 diabetes mellitus treated with TCZ found a reduction in hemoglobin A1c (HbA1c) levels. 266 Moreover, a study of 11 non-diabetic patients with RA detected, after 3 months of tocilizumab treatment, a significant decrease in insulin resistance indexes such as the homeostasis model assessment of insulin resistance (HOMA-IR) and the leptin-to-adiponectin ratio. 267 Serum levels of reactive oxygen metabolites also decreased in RA patients treated with TCZ. 268 It can thus be expected that long-term TCZ treatment may offer protection against the progression of atherosclerosis leading to cardiovascular events. 269 A randomized, open-label, parallel-group, multicenter study is in progress to determine and assess the rate of cardiovascular events for patients with RA following treatment with TCZ, with a comparison to treatment with etanercept, a TNF inhibitor, as a control.
Conclusions and Future Prospects
Currently TCZ is approved worldwide for the treatment of RA, Additionally, in several countries it is approved for treatment of Castleman's disease and systemic and polyarticular JIA. In view of the favorable results for off-label use of TCZ and the pathologic role of IL-6 in various immune-mediated diseases, TCZ is expected to be widely used in the treatment of such diseases. However, further clinical trials are required to achieve this goal.
The mechanisms through which TCZ exerts its therapeutic effects on various phenotypically different diseases are not yet well understood. It is possible that TCZ can rectify the imbalance of Th17/ Treg in various diseases, 18 as seen in treatment of RA. 75 On the other hand, TCZ could exert its beneficial clinical effect by inhibiting pathological autoantibody production, since TCZ treatment was found to lead to a reduction in the pathologic CD38highCD19lowIgDnegative plasma cells of SLE patients 158 and to diminish the survival of plasmablasts–-that is, CD19intCD180negativeCD27+CD38+ cells, which produce the anti-AQP4 Ab seen in NMO.175,176
Finally, as already mentioned, IL-6 synthesis is regulated by transcriptional levels and post-transcriptional mechanisms. It has already been shown that some viral products derived from Kaposi's sarcoma-associated herpes virus, human T lymphotropic virus-1, human immunodeficiency virus-1, or human hepatitis virus B, could constitutively activate transcriptional activation of the IL-6 gene and/or inhibit its mRNA degradation.270–276 For these reasons, clarification of the cell source and the mechanism of dysregulated persistent IL-6 production is expected to aid and facilitate the investigation of the pathogenesis of a wide range of diseases.
Author Contributions
All authors contributed to the writing of the manuscript, reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
TT has received grant and payment for lectures including service on speakers bureaus from Chugai Pharmaceutical Co, Ltd. AO has received a consulting fee as a medical adviser, grant and payment for lectures including service on speakers bureaus from Chugai Pharmaceutical Co, Ltd. MN has received grant and payment for lectures including service on speakers bureaus from Chugai Pharmaceutical Co, Ltd.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
Footnotes
Acknowledgements
The authors gratefully acknowledge the valuable discussion of Professor Tadamitsu Kishimoto, Professor Atsushi Kumanogoh, and Professor Kazuyuki Yoshizaki.
