Abstract
Evidence has accumulated indicating that proinflammatory cytokines play critical roles in the pathogenesis of RA. Recent clinical studies demonstrate that blockade of IL-6 signaling with tocilizumab, a recombinant humanized anti-interleukin-6 receptor antibody, is a new therapeutic option for the treatment of patients with RA refractory to conventional DMARD therapy and anti-TNF-α therapy. This paper discusses possible mechanisms of action, reviews the results of clinical trials, and discusses the place of tocilizumab in RA treatment.
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic, progressive autoimmune inflammatory disease with unknown etiology that particularly affects synovial joints of the hands and feet. The synovial tissue of affected joints is infiltrated by inflammatory cells such as macrophages and lymphocytes, leading to hyperplasia with neovascularization, which causes joint swelling, stiffness and pain. The final results are cartilage destruction and bone resorption in the joints, and many patients suffer permanent disability. RA patients also develop multiple systemic symptoms including fever, fatigue, anemia, anorexia, osteoporosis, weight loss and muscle weakness, and their lungs, skin and liver may be affected. Patient lifespan is reduced by up to 10 years due to the effects of chronic inflammation on major organs. 1
RA afflicts 0.3%-1.0% of the adult population worldwide, and women are two to three times more susceptible to the disease than men.2,3 Onset usually occurs at ages between 30 and 50 years, and the percentage of patients unable to work 5 and 10 years after diagnosis is 40% and >50%, respectively. 1
Although the etiology of RA is not fully understood, recent research has demonstrated that a proinflammatory cytokine, IL-6, plays important roles in the pathogenesis of RA.
Several international clinical studies have clearly indicated that blockade of IL-6 signaling by tocilizumab (TCZ), a humanized anti-human IL-6 receptor (IL-6R) antibody, is a highly effective treatment approach for RA. This article reviews the evidence for the clinical usefulness of TCZ and discusses its place in RA treatment.
Tocilizumab: Its Origin, Mechanism of Action and Pharmacokinetic Profile
Origin
TCZ (formerly MRA) is a humanized anti-human IL-6R antibody that blocks the IL-6 signaling pathway. It was created by using complementarity-determining region grafting techniques to humanize a mouse monoclonal antibody that neutralizes IL-6 function by binding to human IL-6R, 4 and is manufactured in Chinese hamster ovary cells. TCZ is an IgG1 subclass antibody and is a κ light chain glycoprotein with a molecular weight of 148 kDa. The IL-6 blocking activity of TCZ is equivalent to that of the original mouse antibody: its dissociation constant (Kd value) is 2.5 × 10-9 M. 5
Mechanism of action
TCZ inhibits the binding of IL-6 to IL-6R and thus blocks IL-6 signaling in cells. A number of published papers have shown that TCZ inhibits various biological functions of IL-6, both in vitro and in vivo.6,7
IL-6 is a multi-functional inflammatory cytokine that plays a number of important roles in RA pathogenesis. Synovial cells produce IL-6 when stimulated by IL-1 and TNF-α, both of which are elevated in RA patients. 8 The serum IL-6 level is closely related to disease activity in RA patients, 9 and many of the local and systemic symptoms and signs of RA can be attributed to the overproduction of IL-6. Animal studies have demonstrated that IL-6 is also critical in the development of experimental arthritis.10,11 The development of collagen-induced arthritis (CIA) is strongly inhibited in IL-6 deficient mice and in mice treated with anti-mouse IL-6R antibody (MR16-1).12,13 IL-6 blockade also inhibits the development of antigen-induced arthritis in mice, 14 and the spontaneous development of autoimmune arthritis in SKG mice.15–17 Hence, it is not surprising that IL-6 blockade is an effective approach for the treatment of RA patients.
IL-6 itself does not promote acute inflammation, so IL-6 transgenic mice do not develop arthritis, whereas TNF transgenic mice do develop arthritis. 18 This may mean that TNF-α acts as a typical proinflammatory cytokine, whereas IL-6 plays a pivotal role at an earlier stage of RA etiology. This suggests that IL-6 blockade treats RA at a deeper level than TNF blockade. There are clinical indications of this in the finding that serum IL-6 gradually decreased during treatment, normalizing in some patients, even though TCZ is not known to directly influence IL-6 production (Fig. 1). 19 The cause of the decrease in serum IL-6 with TCZ treatment is unclear, but inhibition of Th17 cell differentiation by IL-6 blockade is one possibility. IL-17 from Th17 cells plays an important role in recruiting inflammatory cells, including macrophages, into synovial tissues, and these cause hyper-production of proinflammatory cytokines such as TNF, IL-1 and IL-6. Since IL-6 is pivotal for Th17 cell differentiation, IL-6 blockade may eventually inhibit IL-17 production and so suppress IL-6 production in inflamed synovial tissues.
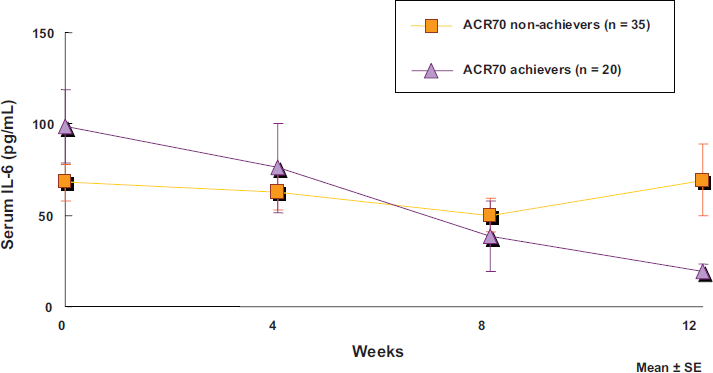
Decreased IL-6 levels in patients treated with tocilizumab.
Our recent papers support this hypothesis by demonstrating that inhibition of Th17 cell differentiation by anti-IL-6R antibody caused inhibition of the development of arthritis in collagen- and GPI-induced arthritis models in mice (Fig. 2).20,21 This means that inhibition of Th17 differentiation is a possible mechanism for the efficacy of TCZ in RA. Importantly, the timing of the IL-6 blockade was critical: Th17 cell development was not inhibited when anti-IL-6R antibody was injected more than 3 days after primary immunization with collagen, and antibody treatment was not effective against established disease. Th17 cells play an important role in CIA because IL-17 secreted from Th17 cells recruits other immune cells to peripheral tissues, leading to increased joint inflammation. TGF-β is an essential cytokine for Th17 cell differentiation. IL-6 induces Th17 cell differentiation, and anti-IL-6 antibody almost completely inhibits this induction.22–25 Interestingly, TGF-β induces regulatory T cell differentiation, but IL-6 inhibits TGF-β-induced regulatory T cell differentiation.23,24 These findings suggest that IL-6 may be a crucial polarizing factor that increases Th17 cells and inhibits regulatory T cells, leading to autoimmune inflammatory response.
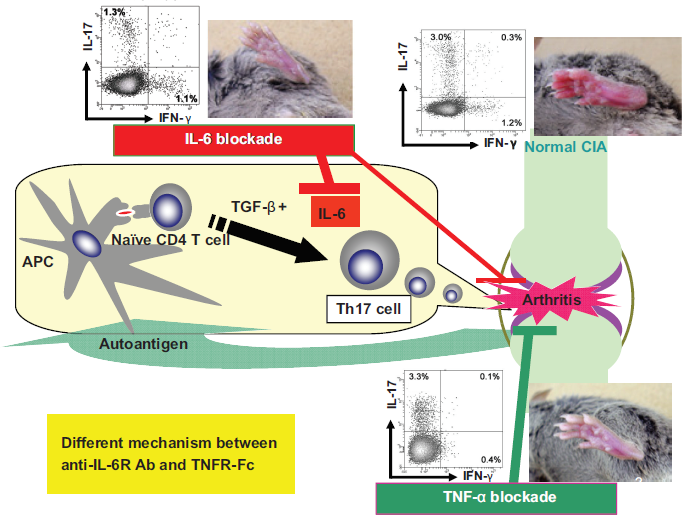
Different mechanism between anti-IL-6R Ab and TNFR-Fc. Kindly provided by Dr. T. Naka. See Ref. # 20 and 21.
Combined with the animal study results mentioned above, this may mean that IL-6 blockade by TCZ not only suppresses inflammation, it also acts to normalize the balance of Th17 and regulatory T cells in RA patients, thereby disrupting the etiology of the autoimmune response at an early and deep level.
Inhibition of VEGF synthesis and angiogenesis is another possible mechanism for the efficacy of IL-6 blockade by TCZ in RA. Angiogenesis supplies oxygen and nutrition to proliferating synovial cells in RA, and VEGF is essential for angiogenesis. VEGF levels are elevated in the serum and synovial fluids of RA patients and correlate with disease activity and the radiographic progression of joint damage. Exogenous IL-6 increases VEGF synthesis in cultured synovial cells. TCZ inhibits this increase strongly, while IL-1 and TNF-α blockers do so only weakly. 26 In TCZ-treated RA patients, serum VEGF decreases in parallel with improvement of clinical symptoms. 26
IL-6 blockade by TCZ may prevent joint destruction by inhibiting the differentiation of osteoclasts from synovial macrophages. Osteoclast differentiation is regulated by macrophage colony-stimulating factor and receptor activator of nuclear factor-κB ligand (RANKL), a TNF-family cytokine. Some synovial cells, including Th17 cells, express RANKL on the cell surface, indicating that osteoclastic bone resorption is influenced by the immune system. 27 In the presence of sIL-6R, IL-6 induces osteoclast differentiation in mice. 28 We recently found that IL-6 increased RANKL expression on fibroblast-like synovial cells obtained from RA patients. (unpublished data), and this finding has been confirmed by our colleagues. 29 IL-6 blockade by TCZ may inhibit such RANKL expression and thus inhibit osteoclast differentiation. TNF-α also induces RANKL on synovial cells, but this is an indirect action via the induction of IL-6. 29
The action of TCZ is not limited to the joints; TCZ also improves systemic signs and symptoms. For example, TCZ improves anemia, potentially improving patient quality of life (QOL). It has been recently clarified that IL-6 plays a critical role in the development of chronic anemia of inflammation: IL-6 induces hepcidin production in the liver, and this regulates the recycling of iron by macrophages and the absorption of iron from the intestine. Excessive IL-6 therefore causes hypoferremia, leading to anemia of chronic inflammation.30–33 Therefore the mechanism by which TCZ improves anemia may be that IL-6 blockade interrupts this chain of events.
It is also possible that the improvement of anemia by TCZ may be attributable to the recovery of erythropoietin signal transduction. The erythropoietin receptor and IL-6R complex share the JAKSTAT signaling pathway. 34 Excessive IL-6 signaling induces expression of intracellular factors that inhibit the JAK-STAT pathway. Hence, IL-6 blockade by TCZ may result in increased erythropoietin signaling and hematopoiesis. Interestingly, in a CIA model in monkeys, erythrocyte count correlated negatively with serum IL-6, but was not related to serum TNF-α. This suggests that IL-6 may be more heavily involved than TNF-α in the chronic anemia of RA patients. 35
IL-6 blockade by TCZ promotes appetite in RA patients. This makes sense because IL-6 enhances the function of the appetite-suppressing hormone leptin, resulting in anorexia, a symptom commonly experienced by patients with chronic inflammatory diseases.
IL-6 blockade by TCZ improves patient QOL by decreasing pain. An intriguing recent study has linked negative emotions about pain with elevated serum IL-6. The press release noted that higher levels of IL-6 were associated with decreased pain thresholds and elevated pain intensity ratings. (Poster 250: The American Academy of Pain Medicine's 25th Annual Meeting, January 29-30, 2009; Coral Ballroom Foyer, Hilton Hawaiian Village).
RA affects the levels of several plasma proteins. For example, RA patients have a decreased level of albumin and increased levels of acute phase proteins such as CRP, fibrinogen, serum amyloid A (SAA) and haptoglobin. These changes can be explained by hyperproduction of IL-6, which increases synthesis of acute phase proteins and decreases synthesis of albumin in the liver.36–39 The role of IL-6 in these changes has been confirmed in animal studies: IL-6 is essential for the induction of acute phase reaction in IL-6 knockout mice, 40 and injection of recombinant human IL-6 in cynomolgus monkeys elevates serum CRP. 41 Symptoms such as fever, fatigue, anemia and anorexia can also be explained by hyperproduction of IL-6. So it is not surprising that all of these signs and symptoms of RA are effectively treated by IL-6 blockade by TCZ.
Pharmacokinetic profile
In single-dose pharmacokinetic studies in rats and monkeys, the half-life of TCZ was about 6 to 9 days, and tissue transferability was low due to a low volume of distribution. The pharmacokinetic profile of TCZ is linear in rats and nonlinear in cynomolgus monkeys. This reflects species specificity because TCZ is cross-reactive to monkey but not to rat. In weekly dosing studies in monkeys, steady state was reached after the fifth or sixth administration (unpublished data).
In distribution studies, TCZ did not accumulate in specific tissues. In monkeys, the concentration was highest in the adrenal gland, lung, kidney and liver, and in target tissues of TCZ, such as synovium, bone marrow and spleen. The majority of radio-labeled TCZ was excreted in the urine as low-molecular-weight entities, so it is thought that TCZ is catabolized by endogenous protein pathways (unpublished data).
The clinical pharmacokinetics of TCZ was investigated in a phase 1/2 study in Japan. 42 Within a dose range of 2 to 8 mg/kg, serum TCZ decreased in a nonlinear manner over time. The half-life of serum TCZ increased with repeated dosing and with increased dose. After the third 8 mg/kg dose, the half-life was 242 ± 71 hours, similar to that of human IgG. The mean area under the serum concentration/time curve increased with dose, reaching 10.7 ± 4.1 mg·hr/mL (mean ± SD) in the 8 mg/kg groups. Serum TCZ was detectable in all patients at all times in the 8 mg/kg group. Serum sIL-6R was completely (>90%) saturated when serum TCZ was >1 μg/mL.
Clinical Studies
Efficacy
In clinical studies in RA patients conducted in Japan and other countries, including the US and Europe, TCZ has blocked IL-6 signaling with striking efficacy (Table 1).43–52 The consistent efficacy of TCZ in all clinical trials strongly supports the critical role of IL-6 in the pathogenesis and progression of RA. Promisingly, even as monotherapy, TCZ effectively treated patients refractory to conventional DMARDs. TCZ also effectively treated patients refractory to TNF blockers in an international study. 52
Brief summary of Phase 2 and Phase 3 clinical trials.
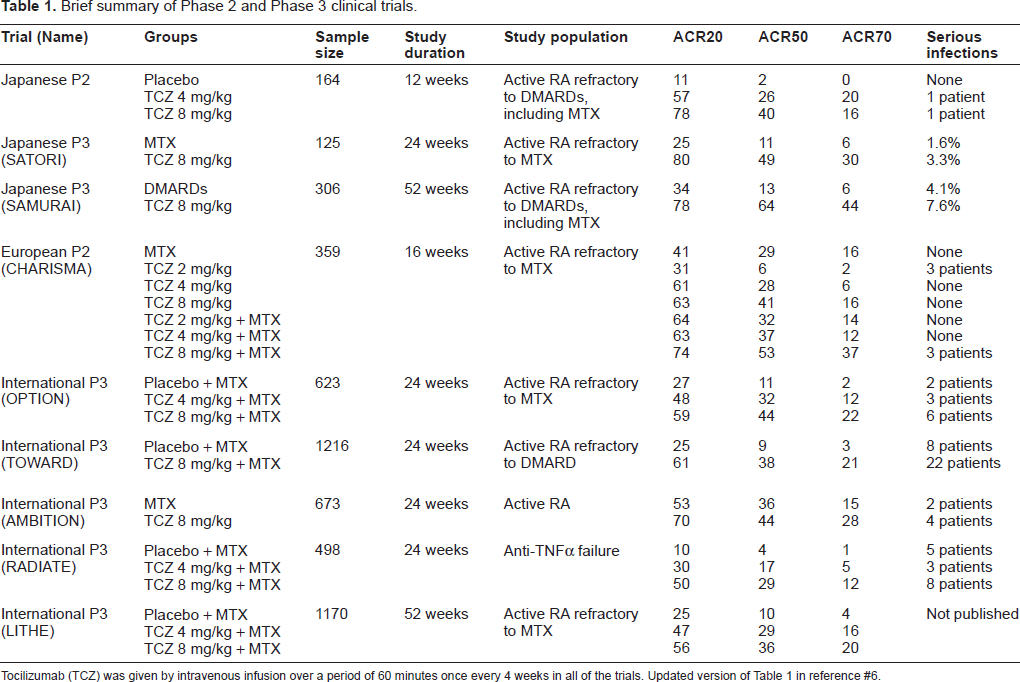
Tocilizumab (TCZ) was given by intravenous infusion over a period of 60 minutes once every 4 weeks in all of the trials. Updated version of Table 1 in reference #6.
Phase I/2 studies
TCZ or placebo was administered to 15 patients in a Japanese study and 45 patients in a UK study.42,43 In both studies, erythrocyte sedimentation rate (ESR), serum CRP and serum SAA levels normalized and remained normal as long as TCZ was detectable in the serum.
Phase 2 studies
Multicenter, double-blind, randomized, placebo-controlled phase 2 trials of TCZ were conducted in Japan and Europe.44,45 In the Japanese study, 44 164 active RA patients refractory to at least one DMARD or immunosuppressant received 4 or 8 mg/kg of TCZ or placebo intravenously every 4 weeks (q4w) for 12 weeks. TCZ significantly improved all measures of disease activity in the American College of Rheumatology (ACR) core set a (Fig. 3), and the ACR20 response rate was 78% with 8 mg/kg TCZ, versus 11% with placebo. The 28-joint disease activity score (DAS28) b decreased 91% and 72% with 8 and 4 mg/kg TCZ, respectively, versus 19% with placebo. Serum CRP, fibrinogen, SAA, albumin, RF, hemoglobin and platelets also improved.

ACR definitions of improvement in rheumatoid arthritis.
SJC: Swollen joint count, TJC: Tender joint count, GH: General health DAS remission rate: Percentage of patients with DAS28 remission (DAS28 < 2.6).
In the European CHARISMA study, 45 359 active RA patients refractory to MTX received 2, 4 or 8 mg/kg TCZ or TCZ placebo q4w plus 10-25 mg/week of MTX or MTX placebo for 12 weeks. There was significantly greater improvement in DAS28 from baseline with 8 mg/kg TCZ, with or without concomitant MTX, than with MTX alone.
Phase 3 studies
In a recent pivotal study in Japan, 46 125 RA patients refractory to MTX (8 mg/wk, the recommended dose in Japan) received either 8 mg/kg TCZ plus MTX placebo or MTX 8 mg/wk plus TCZ placebo. At Week 24, the ACR20 response rate was 80% with TCZ and 25% with MTX (p < 0.001). ACR50/70 response rates were 49% and 30% with TCZ, and 11% and 6% with MTX (p < 0.001). Mean DAS28 fell by 3.95 with TCZ and by 1.07 with MTX. Clinical remission (DAS28 remission: DAS28 < 2.6) was achieved by 43% with TCZ and by 2% with MTX (p < 0.001).
In the “SAMURAI” study, 47 302 patients with RA for ≤ 5 years who were refractory to at least one DMARD or immunosuppressant (excluding leflunomide and biologics) received either TCZ (8 mg/kg) q4w or DMARDs. TCZ monotherapy was more effective than DMARDs at inhibiting radiographic progression of joint destruction. At 52 weeks, TCZ gave significantly better improvements in total Sharp score c (2.3 vs. 6.1, p < 0.01), erosion score (p < 0.001), and joint space narrowing score (p < 0.05). ACR20/50/70 response rates were also significantly better with TCZ (78%, 64% and 44% with TCZ vs. 34%, 13% and 6% with DMARDs; p < 0.001).
The following five recent multinational phase 3 studies have supported the efficacy and safety of TCZ in RA:
The ‘OPTION’ study (623 patients, 73 sites, 17 countries): 48 A three-arm, randomized, doubleblind, controlled trial in patients with moderate-to-severe RA refractory to long-term MTX treatment. Patients received MTX plus 8 or 4 mg/kg TCZ or placebo. At Week 24, there were significantly higherACR20 response rates with TCZ (59% and 48% with 8 and 4 mg/kg TCZ, respectively; 27% with placebo; p < 0.0001), and both 4 and 8 mg/kg TCZ gave significantly higher ACR50/70 response rates than placebo (p < 0.0001). TCZ significantly decreased DAS28 from baseline and improved EULAR response. d It also gave clinically meaningful improvement in health-related QOL: the Health Assessment Questionnaire disability index score, physical and mental components of the Medical Outcomes Short Form 36, and the Functional Assessment of Chronic Illness Therapy-Fatigue Scale all improved in the TCZ groups.
The “TOWARD” study (1216 patients, 130 sites, 18 countries including the US): 50 A 24 week, randomized, double-blind, controlled study in patients with moderate-to-severe active RA refractory to at least one traditional DMARD, including MTX. Patients received either 8 mg/kg TCZ or placebo q4w plus stable antirheumatic therapy (traditional DMARDs, not biologics). TCZ significantly improved disease symptoms: at 24 weeks, ACR20/50/70 response rates were 61%, 38% and 21% for DMARDs plus TCZ, and 25%, 9% and 3% for DMARDs plus placebo.
The “AMBITION” study (673 patients, 252 sites, 18 countries): 51 A 24-week, randomized, doubleblind, double dummy study to compare TCZ with MTX in RA patients who had not previously failed treatment with MTX or biologics. Patients received either TCZ (8 mg/kg q4w) plus MTX placebo, or MTX (7.5 mg/wk titrated to 20 mg/wk over 8 wk) plus TCZ placebo. At Week 24, the rate of ACR20 response (the primary endpoint) was higher with TCZ than with MTX (70% vs. 53%), as was the DAS28 remission rate. TCZ rapidly normalized mean serum CRP; MTX did not.
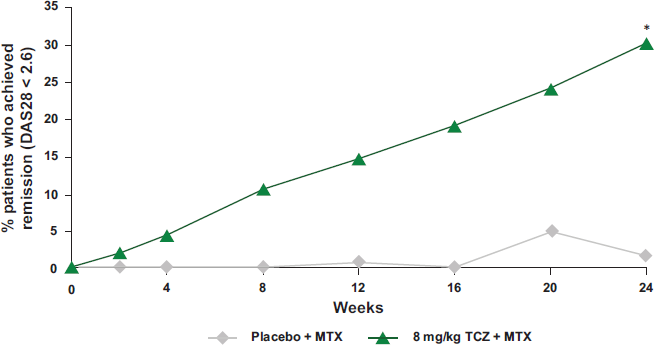
The “RADIATE” study (498 patients, 128 sites, 13 countries including the US): 52 A 24-week, randomized, double-blind study to compare MTX plus TCZ with MTX plus placebo with respect to symptom reduction and safety in patients with moderate-to-severe active RA refractory to anti-TNF therapy (considered a particularly difficult-to-treat population). Patients received TCZ (4 or 8 mg/kg) or placebo q4w, plus a stable dose of 10-25 mg/wk MTX. At week 4, the rate of ACR20 response (the primary endpoint) was higher with 8 mg/kg TCZ than with placebo. At Week 24, the ACR20 response rate was 50%, 30% and 10% in the 8, 4 and 0 mg/kg group, respectively, and the DAS28 remission rate was higher with 8 mg/kg TCZ than with placebo (30.1% vs. 1.6%) (Fig. 5).
EULAR response criteria based on disease activity score. Clinical remission (DAS28 < 2.6) following tocilizumab treatment in patients refractory to TNF blockers: *p < 0.0001 vs. placebo. Modified from P Emery et al, Ann Rheum Dis. 2008;67:1516-23.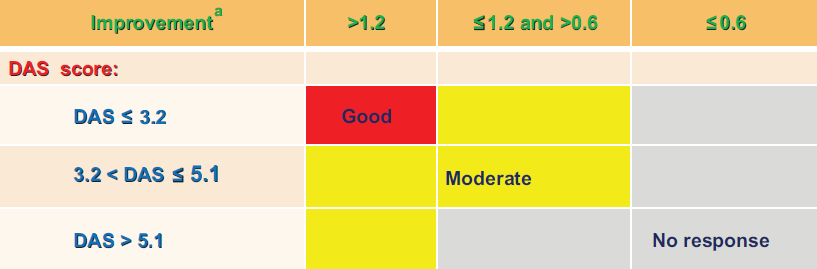
The LITHE study (1170 patients, 137 sites, 15 countries, on-going) (J. Kremer, et al EULAR 2009, Abstract No. OP-0157): A 2-year, randomized, double-blind, placebo-controlled trial to investigate the efficacy of adding TCZ to MTX treatment in patients with MTX-refractory RA. Patients received stable MTX treatment plus either TCZ (4 or 8 mg/kg) or placebo q4w. At Week 52, the 8 mg/kg TCZ group showed significantly less radiographic progression (by Genant-modified Total Sharp Score) than the control (MTX + placebo) group, and both TCZ groups had significantly more patients with no radiographic progression. Both TCZ groups also had greater improvement in physical function, higher ACR20 response rates at Week 24, and higher ACR20/50/70 response rates at Week 52 than the control group.
Long-term efficacy
The results of a long-term efficacy and safety study have recently been published. 19 This was an open-label, long-term extension study after an initial 3-month phase 2 study. Out of 163 patients in the initial study, 143 were enrolled in the extension study. Of these, 94 (66%) received 8 mg/kg TCZ monotherapy q4w for 5 years. There were 32 (22%) withdrawals due to adverse events (AEs), one withdrawal (0.7%) due to poor response, and 14 withdrawals due to patient request or other reasons. Among 88 patients receiving concomitant steroids, 78 (89%) had the dose decreased, and 28 (32%) successfully discontinued steroid use. At 5 years, ACR20/50/70 response rates were 84%, 69% and 44%, respectively, and the DAS28 remission rate was 55.3%. All of the above results clearly indicate the benefits of using TCZ to block IL-6 signaling in the treatment of RA.
Safety
In the two phase 1/2 studies,42,43 TCZ was well tolerated and no serious adverse events (SAEs) occurred. In the Japanese phase 2 study, 44 the overall incidence of AEs was 56%, 59% and 51% in the 0, 4 and 8 mg/kg TCZ groups, respectively, so there was no dose-response curve. One patient received a single dose of 8 mg/kg TCZ and then died from reactivation of a chronic active Epstein-Barr virus (EBV) infection with consequent hemophagocytosis syndrome. Retrospectively, it was found that this patient suffered from Hodgkin's disease and had increased EBV DNA in her plasma before enrollment, so she should have been excluded from the study. 53 Mild-to-moderate increase in serum liver enzymes was observed in 14 of 109 TCZ patients (13%), however none withdrew due to abnormal liver enzyme level.
In the SATORI study, 46 TCZ was well tolerated and the safety profile was similar to that seen in the phase 2 studies. In the SAMURAI study, SAEs occurred in 18% of TCZ (8 mg/kg) patients and 13% of control (DMARDs) patients, respectively. There were 12 incidents of serious infection in the TCZ group (pneumonia, upper respiratory tract infection, cellulitis, gastroenteritis, herpes zoster, herpes simplex, perianal abscess) and 8 in the DMARDs group (gastroenteritis, pneumonia, upper respiratory tract infection, herpes zoster and sepsis). All SAEs improved with appropriate treatment. Mild, transient rise in serum liver enzyme levels was common in both groups. Total cholesterol (TC), triglyceride and low-density lipoprotein cholesterol (LDL), respectively, increased in 38%, 17% and 26% of TCZ patients. Most of these events were Grade 1 according to the National Cancer Institute Common Toxicity Criteria, and as high-density lipoprotein cholesterol (HDL) also increased, the atherogenic index (TC-HDL/HDL) remained unchanged. Twenty seven patients received HMG-CoA reductase inhibitor and their cholesterol levels improved during the study. In the TCZ group, 11 patients had mild drug-related infusion reactions, but none withdrew from the study. Anti-TCZ antibodies were detected in 4 patients (2.5%). Of these, 1 patient showed a skin eruption at the 3rd injection, but the other 3 patients were asymptomatic.
In the OPTION study, TCZ was generally well tolerated, and there was a similar overall frequency of AEs in all three groups (MTX + 0, 8 or 4 mg/kg TCZ). Serious infections occurred in 6 patients in the 8-mg/kg TCZ group, 3 patients in the 4-mg/kg TCZ group, and 2 patients in the placebo group. 48
In the TOWARD study, 50 AEs leading to withdrawal occurred in 4% and 2%, SAEs occurred in 6.7% and 4.3%, and serious infections occurred in 2.7% and 1.9% of TCZ and control patients, respectively. Alanine aminotransferase (ALT) elevation from normal to >3 times the upper limit of normal occurred in 4% and 1%, and TC elevation occurred in 23% and 6% of TCZ and control patients, respectively. Among TCZ patients, 16 started lipid-lowering therapy during the study, and 3.7% developed grade 3 neutropenia.
In the AMBITION study, 51 SAEs occurred in 3.8% and 2.8%, and serious infections occurred in 1.4% and 0.7% of TCZ and MTX patients, respectively. Among TCZ patients, more patients developed reversible grade 3 neutropenia (3.1% vs. 0.4%) and more developed TC > 240 mg/dL (13.2% vs. 0.4%), but fewer developed ALT > 3 times the upper limit of normal (1.0% vs. 2.5%).
In the RADIATE study, 52 AEs occurred in 84.0%, 87.1% and 80.6% of patients in the 8, 4 and 0 mg/kg TCZ groups, respectively; most were mild or moderate.
In the long-term study, 19 the SAE rate was 27.5 events per 100 patient-years; the most common type was “infection” (5.7 per 100 pt-yr).
In summary, the most common AEs included upper respiratory tract infections, headache, nasopharyngitis and hypertension. As with other biological DMARDs, serious infections occurred in some patients. Mean TC increased initially, but with repeated dosing it stabilized near the upper limit of normal (6.18 mmol/L). These lipid-related effects may be related to the action mechanism of TCZ because IL-6 decreases serum cholesterol in cancer patients,54,55 and suppression of inflammation by infliximab has similar lipid effects.56,57 In fact, decreased TC and HDL levels are often seen in patients with severe RA, possibly because of cytokine-induced activation of the reticuloendothelial system. 58 It has recently been found that IL-6 induces LDL receptors on the cell surface, leading to increased consumption of LDL. Although increases in liver function variables were reported, these normalized during the treatment period in many patients. However, it is important to monitor and to manage transaminase elevations and interrupt hepatotoxic drugs or/and TCZ.
Place of TCZ in RA Therapy
Patients with RA are usually treated with DMARDs, including MTX. However, about 30% to 40% of patients do not respond adequately to currently available DMARDs.59–62 Such patients are treated with biologics, with or without concomitant MTX. 62 Biologics targeting TNF-α, a proinflammatory cytokine, have markedly increased the disease remission rate. However, up to 50% of patients treated with TNF blockers fail to improve significantly, 61 and TNF blockers are associated with a significantly increased risk of infection. 63 They may also increase the risk of certain malignancies, although this is controversial. 63 New therapeutic agents with new mechanisms of action, targeting other molecules, are therefore highly desirable. TCZ is a recently developed anti-rheumatic drug that targets IL-6, a multi-functional inflammatory cytokine that plays important roles in the pathogenesis and progression of RA.64,65
In Japan, TCZ was approved as an orphan drug for the treatment of patients with Castleman's disease in June 2005,66–68 and subsequently as a therapeutic drug for RA, polyarticular-course juvenile idiopathic arthritis (JIA) and systemic-onset JIA in April 2008.69–72 Recently (January 2009), it was approved by the European Medicines Agency (EMEA) for treatment of moderate-to-severe active RA in adult patients refractory to, or intolerant of, prior DMARD or TNF antagonist therapy. A biologics license application (BLA) for TCZ was submitted to the US Food and Drug Administration (FDA) in November 2007 for the indication, “improvement of signs and symptoms in adults with moderate-to-severe RA” and is under review.
TCZ has shown consistent efficacy in several independent phase 2 and 3 studies in Japan, the US, the EU and other countries (Table 1). This has strongly confirmed that IL-6 is deeply involved in the pathogenesis and progression of RA. The advent of TCZ has greatly improved the therapeutic options for RA. A long-term follow-up study has demonstrated sustained efficacy with relatively few SAEs and high tolerability. TCZ had not been directly compared with other biologics, but based on the above results, it can be considered a first-line drug for treatment of DMARD-resistant RA patients.
Indeed, TCZ is the only biologic demonstrated to have clinical efficacy that is statistically superior to that of MTX, the current gold standard in RA therapy. In the AMBITION study, 51 almost one third of TCZ-treated patients achieved either DAS28 remission or ACR70 at Week 24, whereas these two endpoints were achieved by only 12% and 15% of MTX-treated patients, respectively. In contrast, TNF blockers (etanercept, adalimumab, etc.) have shown efficacy similar to that of MTX in trials such as the ERA, TEMPO and PREMIER studies.
Some trial results suggest that TCZ not only strongly inhibits inflammation in RA, it may also improve the underlying disease etiology. TCZ does not directly suppress IL-6 production, but serum IL-6 does decrease with TCZ treatment (Levi M. et al, [poster no. THU0176] 2008 Annual European Congress of Rheumatology: 2008 Jun 11-14, Paris; Nishimoto N. et al, [abstract no. OP-0135] Ann Rheum Dis. 2008;67 Suppl II:90). Possible mechanisms for the suppression of IL-6 levels are discussed in under “Action Mechanism” above. In the SATORI study, 46 most (14) of the 23 patients with normal serum IL-6 at Week 24 achieved DAS28 remission, indicating that IL-6 normalization is closely related to clinical remission. Further support comes from the fact that rates of ACR50/70 response and DAS28 remission increase as the duration of treatment increases. 19 This tendency was even more marked in the RADIATE study in a particularly difficult-to-treat population, TNF-blocker-refractory patients, although for some unknown reason it took longer for these patients to achieve ACR70 (Fig. 5). It is even possible that immune-mediated inflammation has virtually ceased in patients with normalized serum IL-6, so it will be of vital importance to clarify whether TCZ treatment can be stopped after such patients have achieved remission. If so, serum IL-6 may be an essential biomarker for judging when remission has occurred and when TCZ treatment can be stopped.
The SAMURAI study results suggest that the earlier TCZ treatment is started, the better the clinical response, in terms of both stopping disease progression and achieving complete remission. 47 Joint destruction progressed more slowly in the second 6 months than in the first 6 months of TCZ treatment. This may indicate that TCZ takes time to achieve full efficacy in some patients, however a similar trend was also seen in the control group.
TCZ improves not only local signs and symptoms but also systemic ones, including anemia, anorexia, fever and fatigue, thereby potentially improving patient QOL. This is a major advantage of TCZ because systemic efficacy is difficult to achieve with other therapies, including anti-TNF therapies. TCZ also improves hypoalbuminemia and ESR, which may also contribute to improving patient QOL.
TCZ almost completely normalizes serum CRP and SAA, which may improve secondary amyloidosis (seen in 5% of RA patients in Europe),73–79 and also counter the increased risk of cardiovascular disease caused by high CRP in RA patients. 80 These effects may be related to IL-6 blockade because, according to recent reports, IL-6 is deeply involved in the regulation of gene expression for SAA and CRP synthesis.81–83 To improve secondary amyloidosis, serum SAA needs to be kept below 10 μg/mL, 79 and TCZ treatment is the only RA therapy that can decrease SAA to this extent.
Significantly, 88% of patients took lower doses of steroid and 41% discontinued steroid treatment during the long-term study. 19 It may also be possible to decrease the dose of MTX in patients receiving TCZ plus MTX who have achieved ACR70.
There were no increases in antinuclear or anti-DNA antibodies. This may be an advantage for TCZ because TNF blockers induce anti-dsDNA autoantibodies in 11% to 16% of patients.84–86
An advantage of IL-6 blockade over TNF blockade was demonstrated in a murine model of tuberculosis infection. After inoculation with Mycobacterium tuberculosis, mice treated with anti-mouse TNF antibody all died much earlier than the control mice, whereas mice treated with an anti-mouse IL-6R antibody (MR16-1) did not. The reason for this difference is suggested by the finding that production of interferon-γ in spleen cells in response to a purified protein derivative was suppressed more strongly by anti-TNF treatment than by anti-IL-6 treatment (Okada M, et al, unpublished data). Interferon-γ is essential for the formation of granulomas that keep mycobacteria inside macrophages, and TNF is known to have a critical role in granuloma formation, whereas IL-6 has no such activity.
Another advantage of TCZ over TNF blockade is that TNF blocker monotherapy often induces production of anti-drug antibodies, whereas anti-TCZ antibodies have been detected in only 3%-5% of patients, even though no immunosuppressants have been used. The CHARISMA study results show that monotherapy with a low dose of TCZ (<4 mg/kg) may have a higher risk of anaphylaxis. 45 This would suggest that higher doses of TCZ actually suppress the production of anti-drug antibodies and is one reason for recommending a dose of 8 mg/kg Finally, and importantly, TCZ does not interfere with influenza immunization. 87
Conclusion
Clinical studies in patients with moderate-to-severe RA, conducted in Japan, and worldwide including the EU and the US, have demonstrated that TCZ improves the signs and symptoms of RA. Furthermore, two phase 3 studies, one in Japan and the other multinational, have shown that TCZ prevents the progression of joint destruction. Importantly, TCZ monotherapy is an effective treatment for RA, and TCZ is the only biologic with efficacy superior to MTX. TCZ has shown efficacy in patients who are refractory, not only to conventional RA therapies, but also to TNF blockers. The rates of ACR70 response and DAS28 remission, and the persistence of efficacy and patient participation during long-term treatment are all high. TCZ is safe and generally well tolerated, with frequencies of AEs similar to those caused by biologic DMARDs. The advent of the IL-6R blocker, TCZ, therefore provides a new therapeutic option in RA treatment, and TCZ should be considered as a first-choice treatment for RA.
Conflicts of Interest
YO is an employee of Chugai-pharmaceutical Co. Ltd., a developer of tocilizumab. TK is a patent holder for tocilizumab.
Abbreviations
ACR, American College of Rheumatology; AE, adverse event; ALT, alanine aminotransferase; CIA, collagen-induced arthritis; CRP, C-reactive protein; DAS, disease activity score; DMARD, disease modifying anti-rheumatic drug; EBV, Epstein-Barr virus; ESR, erythrocyte sedimentation rate; EULAR, European League Against Rheumatism; HDL, high-density lipoprotein cholesterol; IL-6, interleukin-6; IL-6R, interleukin-6 receptor; JAK-STAT, Janus kinases and signal transducers and activators of transcription; LDL, low-density lipoprotein cholesterol; MTX, methotrexate; mIL-6R, membrane-bound IL-6 receptor; OPTION, tOcilizumab Pivotal Trial in methotrexate Inadequate respONders; q4w, every 4 weeks; QOL, quality of life; RA, rheumatoid arthritis; RADIATE, Research on Actemra Determining effIcacy after Anti-TNF failurEs; RANKL, Receptor Activator of Nuclear factor-κB Ligand; SAA, serum amyloid A; SAE, serious adverse event; sIL-6R, soluble IL-6 receptor; TC, total cholesterol; TNF, tumor necrosis factor; TCZ, tocilizumab; TOWARD, Tocilizumab in cOmbination with traditional DMARD therapy; VEGF, vascular endothelial growth factor.
Footnotes
Aknowledgment
We thank Drs. K. Karsten, E. Alecock, K. Watanabe and O. Okuda for their reviewing of the manuscript.
