Abstract
Objectives
To evaluate the anti-inflammatory and memory-enhancing effects of memantine (MEM) in complete Freund’s adjuvant (CFA)-induced rheumatoid arthritis (RA) in Wistar rats. The effect of MEM on cognition in CFA-induced RA was also evaluated.
Materials and Methods
The rats were randomly assigned to one of five groups: sham control (SC), disease control (DC)—CFA 0.1 mg, positive control (PC)—methotrexate (MTX) 1 mg/kg, low dose MEM—20 mg/kg, and high dose MEM (HD-M)—50 mg/kg. All groups received a subcutaneous injection of CFA in the left paw on day 0, except for the SC group, which received a distilled water (DW) injection. The study drugs, MEM and MTX, were administered orally from days 15 to 28. The SC group received DW orally from days 15 to 28. Variables for physical (paw volume, paw surface temperature, and ankle joint diameter) and behavior (open field test, grip strength test, and water corn test) were performed on days 1, 0, 7, 14, 21, and 28. On day 28, blood was collected for serum inflammatory markers, tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-6. Ex vivo radiological changes in the ankle joint were recorded using a digitalized X-ray machine. Histopathology of the ankle joint was performed.
Results
When compared to the DC group, the HD-M group showed a significant improvement in all physical and behavioral characteristics (p < 0.001). When compared to the DC group, TNF-α, IL-6 levels, and histopathological scores were significantly lower in the HD-M group (p < 0.01).
Conclusion
MEM reduces inflammation and improves memory in Wistar rats with CFA-induced RA. MEM can be used as an anti-inflammatory drug in elderly patients at risk of dementia.
Introduction
The chronic autoimmune disease, rheumatoid arthritis (RA), is characterized by extra-articular involvement and inflammatory arthritis. 1 Around 24.5 million people worldwide suffer from RA, with women being affected 2.5 times more often than men. 2 RA patients have a lower quality of life than the general population. 3
The pathophysiology of RA is complicated and involves both genetic and environmental factors leading to adaptive immune system responses. Dendritic cells, macrophages, and activated B cells are examples of antigenic antigen-presenting cells that deliver arthritic disease-associated antigens to T cells. Tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1, and IL-6 are some of the proinflammatory cytokines released by activated T cells. Clinical and laboratory manifestations of RA result from these cytokines. In addition, cytokines also cause cartilage and bone damage by activating (mainly via the receptor activator of nuclear factor kappa beta ligand [RANKL]-RANK system) chondrocytes and osteoclasts.4, 5
In RA treatment, reducing inflammation and delaying the onset of new symptoms are the primary goals. First-line therapies known as disease-modifying anti-inflammatory drugs (DMARDs) have a variety of side effects, including nausea, abdominal discomfort, and significant liver and lung toxicity. Anti-TNF drug treatment used in RA must be administered parenterally, which is inconvenient and increases the risk of infection. Not only do these drugs fail to achieve complete long-term remission, but they also lead to withdrawal due to their negative effects. Therefore, it is necessary to investigate new therapy options.6, 7
Memantine (MEM) is a non-competitive, low-affinity, voltage-dependent N-methyl-D-aspartate receptor antagonist. It also blocks nicotinic, cholinergic, and serotonergic receptors. In addition to these effects, recent studies support MEM’s ability to reduce inflammation. 8 According to a study by Azarbaijani et al., MEM is a powerful anti-inflammatory drug as it reduces paw edema, leukocyte infiltration, myeloperoxidase (MPO) activity, and malondialdehyde levels. 9 MEM also reduces synovial fibroblast proliferation, which may have a disease-modifying effect on rheumatoid synovial proliferation. 10 Inhibition of TNF-α and IL-6 prevents RA symptoms and joint degeneration and may also have a slightly delayed effect on upstream events of inflammatory pathogenesis.11, 12
Along with anticholinesterases, MEM is one of two drug classes clinically approved for the treatment of Alzheimer’s disease (AD). 8 MEM decreases neuroinflammatory biomarkers, including TNF-α, and protects against neuroinflammation and neuronal death. It has been found that people with RA are more likely to have dementia than people in the general population. Due to the high levels of systemic inflammatory markers in RA patients, dementia progresses rapidly, as it is characterized by inflammatory processes. 13 Recent research suggests that drugs used to treat inflammatory RA may be beneficial in dementia, as it is also associated with neuroinflammation.
Thus, the present study was planned to evaluate the anti-inflammatory effect of MEM in complete Freund’s adjuvant (CFA)-induced rheumatoid arthritis in Wistar rats. We also examined the effect of MEM on cognition in CFA-induced RA.
The inducing agent most commonly used in RA models is CFA. Several of the clinical and pathological features of human RA, such as edema, joint erosions, and ankylosis, are mimicked in the CFA-induced RA model. The expression of inflammatory cytokines, including TNF, IL-6, and activated T cells, is also observed in this model. 14
Materials and Methods
After approval by the Institutional Animal Ethics Committee, the study was conducted according to the Committee for the Purpose of Control and Supervision of Experiments on Animals guidelines. The animals were housed in polypropylene cages with stainless steel grills and were fed pellet-based chow and clean water. The bedding was made from rice husks. The temperature was between 18°C and 29°C, the humidity was between 30% and 70%, and there was a 12-h cycle of light and dark.
Sun Pharmaceutical Industries Ltd. in Mumbai provided MTX and MEM as free samples. CFA was purchased from Krishgen Biosystem. A total of 46 Wistar rats of either sex, 6–8 weeks old and weighing 150–200 g, were divided into five groups, with six rats in the sham control (SC) group and 10 rats in the remaining groups. On day –1, basic behavioral assessments were completed. On day 0, after administration of anesthesia with 50 mg/kg ketamine, the rats were given a subcutaneous injection of 0.5 mg (0.1 mL) of CFA (Krishgen Biosystem) in the left hind paw (0.2 mL). Physical parameters were assessed at baseline (day –1), 0, 7, 14, and 28 days. MEM 20 mg/kg and 50 mg/kg (low-dose of MEM [LD-M] and high-dose of MEM [HD-M]) and methotrexate (MTX) 1 mg/kg (positive control group) were administered from day 15 to day 28 (see Figure 1). MEM doses were selected from the previous study conducted by Motaghi et al. In this study, MEM showed an anti-inflammatory effect by attenuating plasma IL-6 and colonic TNF-α levels 11 . MTX was used as a positive control in previous studies by Chen et al. because of its immunosuppressive and anti-inflammatory effects. 15 In addition, the study by Komaya et al. demonstrated that daily low-dose oral administration of MTX prevented adverse effects and had a greater anti-rheumatic effect in collagen-induced arthritic rats. Hence, MTX 1 mg/kg dose was used as a positive control. 16 The study groups were as follows:
Brief Overview of Methodology.
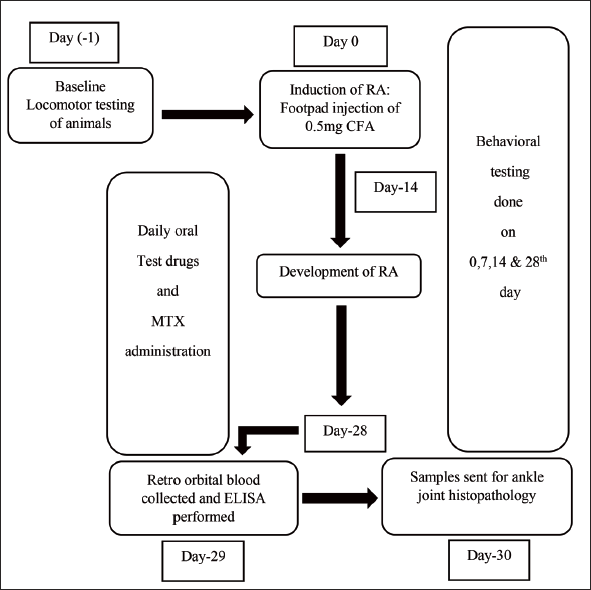
Group 1: Sham control (SC): prick of needle in left hind paw on day 0; distilled water (DW) daily orally from day 15 to day 28.
Group 2: Disease control (DC): Inj. CFA 0.5 mg in left hind paw on day 0; DW daily orally from day 15 to day 28.
Group 3: Positive control (PC): Inj. CFA 0.5 mg in left hind paw on day 0; MTX 1 mg/kg daily orally from day 15 to day 28.
Group 4: LD-M, Inj. CFA 0.5 mg in left hind paw on day 0; MEM 20 mg/kg orally (PO) from day 15 to day 28.
Group 5: HD-M, Inj. CFA 0.5 mg in left hind paw on day 0; MEM 50 mg/kg PO from day 15 to day 28.
The following physical and behavioral parameters were assessed at baseline, that is, days −1 and 0, 7, 14, 21, and 28 days (refer to Figure 1).
Physical Variables
(1) A mercury plethysmometer was used to measure the volume of the paw in millimeters of mercury. The amount of mercury in millimeters that corresponded to the displaced mercury column at the opposite end was noted. (2) Vernier calipers were used to measure the ankle joint‘s diameter. To measure the diameter of the ankle, vernier caliper screws were positioned just 1–2 mm above the rat paw’s ankle joint. (3) A touch thermometer was used to gauge the temperature of the paw surface. A thermometer was placed on the surface of the rat paw for 1–2 min, and the temperature it indicated was recorded.17, 18
Behavioral Variables
(1) Musculoskeletal strength of rat paw19: the grip strength meter (Orchid Scientific TM Innovative India Pvt. Ltd.) was used to measure the muscular and skeletal strength of the rat paw. Three readings were averaged for each rat, and the results were represented in Newton Force (N).
(2) Open field test (OFT)18: the number of lines crossed in 5 min was counted using an open field equipment connected to a video tracking system. Decreased locomotor activity, an indication of inflammatory discomfort, is indicated by fewer lines being crossed.
(3) Morris water maze test20: the water maze is an aversively motivated exercise based on the spatial learning and memory of animals. Memory is reflected in the shorter latencies to escape and the decrease in the length of the path to find the platform. Escape latency to find the platform was measured in seconds (S) within 5 min. In order to do this, we used the same video tracking system (VTS) as was used for the open field test.
Biochemical Variables
After behavioral testing on day 28, retro-orbital blood (2 ml) from rats was taken and centrifuged to produce clear serum for TNF-α and IL-6 measurement using ELISA kits (KrishgenBiosystems)
Radiology of Rat Paw
On day 29, rats were sacrificed with a lethal dose of sodium pentobarbital (50 mg/kg). The left hind paw of each rat was amputated and placed over a radiographic cassette containing standard X-ray film. The hind paws were kept at a distance of 90 cm from the X-ray source for a 0.1-s exposure time to obtain an axial view and were scanned using a digitizer. The digitized images were stored for further analysis. Radiological analysis was performed by a radiologist who was blinded to the groups for soft tissue swelling or deformity in ankle joints. Findings were recorded based on the soft tissue swelling and deformity at the ankle joints.
Histopathology of Ankle Joint
These samples were descaled in 10% EDTA. Each ankle, including the distal tibia and talus, was bisected in the mid-coronal plane, one anterior and one posterior. The two resulting pieces of tissue (anterior and posterior halves) were then both embedded in a single block of paraffin with the cut surfaces facing down. Ten adjacent sections were collected at distances of 0 m, 100 m, and 200 m. Sections were stained with hematoxylin and eosin (H&E) and were read by the pathologist blind to the study groups. The pathologist examined the slides under 100× magnification using a compound microscope. For histopathological changes in the ankle, the composite scoring system of Brenner et al. is used.17, 21
Statistical Analysis
Results are expressed as mean ± SD. A p < 0.05 was considered statistically significant. Parametric data, that is, physical variables, behavioral variables, and biomarkers, were compared using a one-way ANOVA followed by a post hoc Tukey’s test, while non-parametric data, that is, histopathology scores, were analyzed using the Kruskal–Wallis test followed by a post hoc Dunn’s test. Statistical analysis was performed using the GraphPad InStat software version 3.06.
Results
Physical Variables
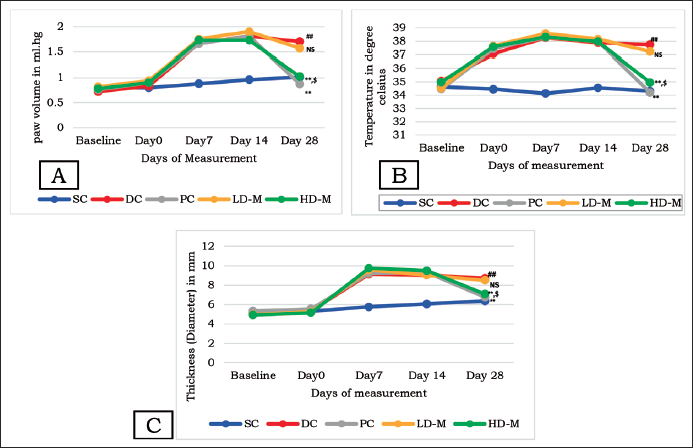
When comparing the DC group to the SC group, there was a statistically significant increase in all physical characteristics, including paw volume, paw surface temperature, and ankle joint diameter (p < 0.001). At day 28, there was a substantial decrease in all physical measurements (p < 0.001) in the HD-M group compared to the DC group, indicating less arthritic damage and inflammation. The decrease in physical variables observed in the HD-M group was equivalent to PC (p > 0.05). Compared to DC, the decrease in the LD-M group was not statistically significant (refer to Figure 2).
Behavioral Variables
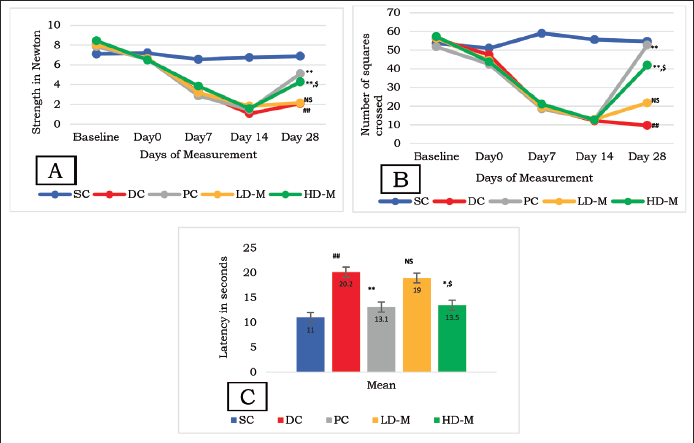
In the DC group compared to the SC group, there was a significantly lower amount of force used by the hind paw, fewer lines were crossed, and a decrease in escape latency (p < 0.001). When compared to the DC group, the HD-M group showed a statistically significant increase in all of these behavioral indicators by day 28 (p < 0.001). The improvement observed in HD-M was comparable to PC (p > 0.05). When compared to DC, the improvement in the LD-M group was not statistically significant (refer to Figure 3A–C).
Biomarkers
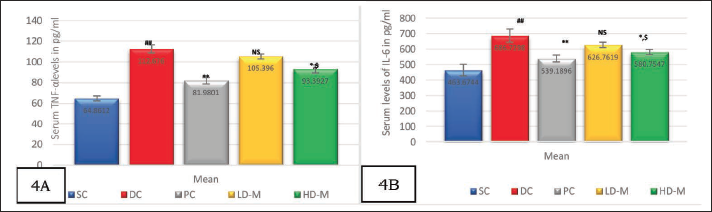
TNF-α and IL-6 levels were significantly lower in the HD-M group compared to the DC group (p < 0.01). The reduction in biomarker levels seen with HD-M was comparable to PC (p > 0.05). The improvement in the LD-M group was not statistically significant compared to DC (see Figure 4B and C).
Radiology: X-ray of Rat Paw
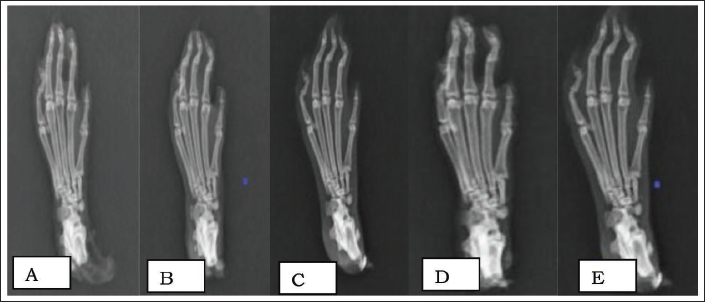
Compared to the SC, an X-ray of a left rat paw in the DC shows severe deformities at the intertarsal-phalangeal and metacarpal joints, as well as soft-tissue swelling. In the HD-M group, the deformity was less pronounced, and the soft-tissue edema on the radiograph was less (see Figure 5A–E).
Radiological Findings of Rat Paw in all Study Groups. (A) Sham Control: No Soft Tissue Swelling, the Interphalangeal, Metacarpel Joints are Normal. No Deformity. (B) Disease Control: Deformity at Distal Interphalangeal (1–4). Generalized Soft Tissue Swelling Along the Digits of the Entire Paw. Distal and Interphalangeal (1–4) Joint Space Reduced. (C) Positive Control: Normal Interphalangeal Joints and No Soft Tissue Swelling. Normal Joint Space in Distal and Middle Interphalangeal Joint Space. (D) Low-Dose Memantine: No Soft Tissue Swelling; Interphalangeal Joint Space is Reduced Minimally Compared to Disease Control. Mild Deformity at 2–4 Distal and Middle Interphalangeal Joints. (E) High-Dose Memantine: Minimal Soft Tissue Swelling. Mild to Moderate Deformity.
Histopathology: Ankle Joint
Compared to the DC group, the histopathological scores of the HD-M group were significantly lower (p > 0.001). This indicates fewer joint injuries and inflammations. HD-M and PC both had similar histopathological scores (p > 0.05). Compared to DC, the values in the LD-M group were not statistically significant (see Figure 6).
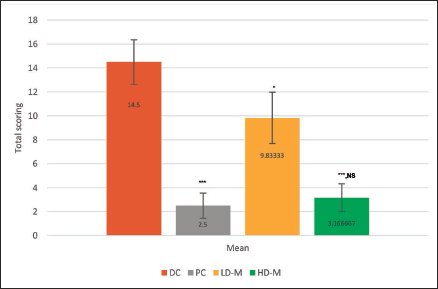
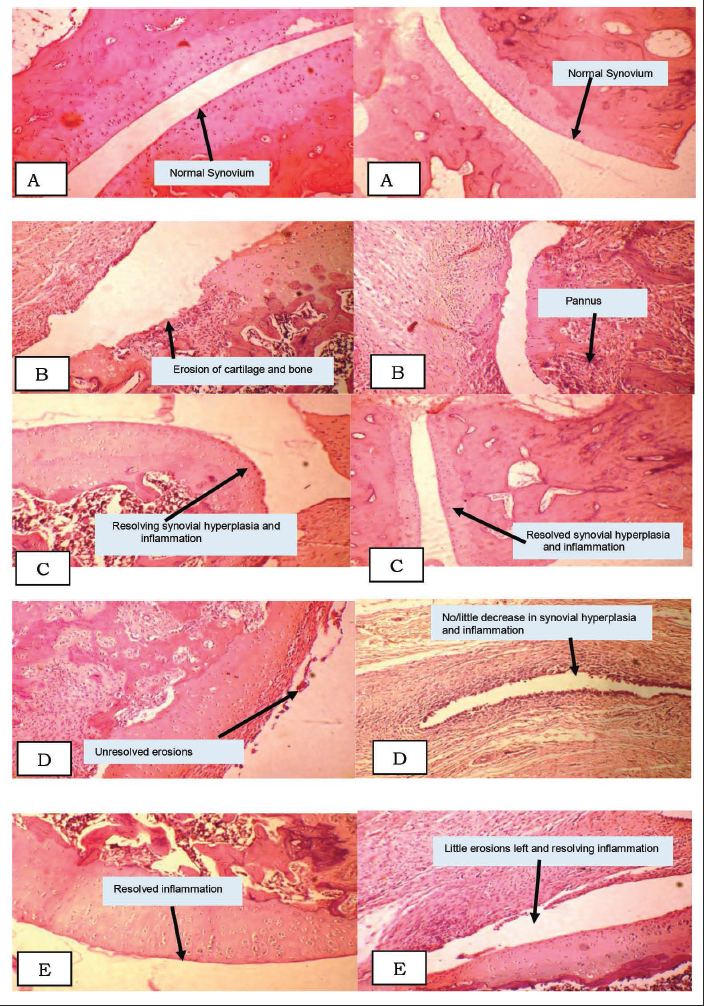
Figure 7 refers to the ankle joint histopathology slides, 28 days post-induction, with the articular surface of the bone: SC group: normal synovium. As rats in this group were pricked just with a needle, there were no rheumatic changes found in this group. DC group: erosion of cartilage and bone and pannus formation. PC group: resolving/resolved synovial hyperplasia and inflammation. LD-M group: unresolved erosions and no/little decrease in synovial hyperplasia and inflammation. HD-M group: resolved inflammation, little erosion left, and resolving inflammation (refer to Figure 7A–E).
Histopathology of Rat Ankle Joint in All Study Groups. (A) Sham Control (Magnification 1000×) Showing Normal Synovium; (B) Disease Control (Magnification 1000×) Showing Erosion of Cartilage, Bone, and Pannus Formation; (C) Positive Control (Magnification 1000×) Showing Resolved and Resolving Synovial Hyperplasia and Inflammation; (D) Low-Dose Memantine (Magnification 1000×) Showing Unresolved Erosions and No/Little Decrease in the Synovial Hyperplasia and Inflammation; and (E) High-Dose Memantine (Magnification 1000×) Showing Resolved Inflammation and Little Erosion Left and Resolving Inflammation.
Discussion
The major component contributing to cartilage degradation in RA is synovial inflammation. More than 80% of people with bone loss also have growing and persistent inflammation. TNF-α and IL-1 and 6 inhibitors are of crucial importance in the clinical therapy of RA since these two cytokines are crucial in the pathogenesis of the disease.
Several preclinical models of MEM have demonstrated its anti-inflammatory properties. In an experimental in vitro model of the blood–brain barrier (BBB), it suppressed the nuclear factor (NF-) pathway by inhibiting the proinflammatory cytokine TNF-α. 22 MEM significantly reduced pulmonary inflammation in a study by Cheng et al. by attenuating elevated cytokine release, such as TNF-α, IL-6, and IFN-β. 23 MEM prevented the growth of synovial fibroblasts in an earlier in vitro investigation. 24 However, these studies are all in vitro and did not evaluate behavioral tests. MEM has the potential to exert neuroprotection independent of RA due to its anti-inflammatory action. It has therefore been suggested to be used for dementia caused by AD.
One of the first DMARDs used to treat RA was MTX. MTX leads to an adenosine-mediated reduction in inflammation by reducing the production of proinflammatory cytokines, including TNF-α and IL-6. 25 MTX was used as a positive control in a previous study (Chen et al.) because of its immunosuppressive and anti-inflammatory effects. 15 This led to the choice of MTX as a positive control. Because they are inexpensive, easy to handle, and have a homogeneous genetic background, mouse animal models of RA are often used. 26 The joint damage in the injected paw and other systemic aspects observed in mouse models are consistent. 27 A single subplantar application of CFA causes acute inflammation, extensive bone resorption, cartilage degradation, and bone erosion in Wistar rats. This model has been claimed to accurately reproduce the systemic and local inflammatory symptoms of RA observed in humans. It is, therefore, considered a suitable model for evaluating drugs with anti-inflammatory and disease-modifying effects. This model is also commonly used to assess anti-TNF activity due to increases in levels of several inflammatory cytokines, including TNF-α and IL-6, which play key roles in the development of arthritis and other systemic features of RA. 28 In our study, these clinical (decreased locomotor activity), biochemical (elevated TNF levels), and histological (bone erosion) characteristics were also found in the disease control group.
Ankle joint diameter, paw surface temperature, and paw volume were chosen as the three physical variables. Because of their simplicity and low cost, researchers most commonly use these three variables, which represent inflammation and early-stage arthritis.
In our study, the HD and PC groups showed improvement in physical variables. These results are consistent with previous MEM research using these factors in experimental designs. In a study, Azarbaijani et al. found a significant reduction in hind paw thickness at MEM doses of 10 and 20 mg/kg. The increase in paw surface temperature observed in our study is consistent with the work of Snekhalata et al., who studied the course of arthritis in CFA-induced RA in Wistar rats and monitored inflammatory activity using thermal imaging. 27
Behavioral indicators for HD MEM also experienced significant improvement. Oral MEM treatment in a previous study in transgenic mice had no effect on paw grip durability. 29 However, in patients with complicated pain syndrome, MEM increased muscle strength as assessed by a dynamometer. 30 The results of the OFT used to show how MEM affected motor activity were similar to those obtained from previous experimental models in which MEM affected the OFT.31, 32 In our investigation, both the HD-M and PC groups showed improved musculoskeletal strength and a number of lines crossed, implying improved functional capacity with MEM.
Cognitive impairments are at risk because of chronic pain. Meade et al.’s systematic review found a significant correlation between RA and cognitive decline. Cognitive impairments caused by chronic pain in RA patients can have a significant negative impact on their quality of life. 33 It is crucial that chronic diseases like RA receive medications that target both pain and cognitive impairment. Therefore, it was decided to study the effects of MEM on spatial memory in an experimental model of RA. The improvement in escape latency observed in our study is consistent with other studies using MEM in the water Maorris test in an experimental model of monoarthritis pain in SLE. 34 Several cytokines, such as TNF-α and IL-17A, are thought to play a role in neuroinflammation and cognitive impairment, as their levels are elevated in RA. MEM’s anti-IL-17A property could reduce oxidative stress and neuroinflammation, which would reduce cognitive impairment.
According to a literature review, TNF-α is the most commonly assessed biomarker in the induced arthritis paradigm. 35 Compared to the DC group, the biomarker levels in the HD-M and PC groups were significantly reduced. Similar results were reported by Lindblad et al., who found that mice treated with MEM had significantly reduced levels of TNF-α and IL-6. In this study, CD4+ and CD25 levels were much lower in the MEM-treated group, explaining the activated regulatory T cells. 24 Treatment with paroxetine or rivastigmine significantly reduced serum levels of IL-6 and TNF-α compared to the RA control group in a study by Shafiey et al. 36 These results led the researchers to conclude that the study drug MEM has anti-inflammatory properties because it inhibits TNF-α and IL-6.
In order to assess the effectiveness of research drugs, it is crucial to consider the histology of the affected joints in any arthritis. It was decided to use the composite scoring system of Brenner et al., for histological assessment covering synovial inflammation, synovial hyperplasia, pannus development, synovial fibrosis, cartilage erosion, and bone erosion. In our study, the histology of the rat joints showed that the HD-M and PC groups had less inflammation, cartilage wear, and bone erosion. In the study by Shafiey et al., the RA group displayed severe alterations as evidenced by the proliferation of synovial membrane, pannus development, inflammatory exudate in the joint area, and focal necrosis of the cartilage for histological assessment. 36 These results are consistent with our histopathological features. We can confirm that high doses of MEM reduce joint injury and inflammation. As already mentioned, a study by Lindblad et al. showed that MEM reduced synovitis and bone loss in a mouse model of rheumatoid arthritis.
Based on the results of the above behavioral tests, biomarker analysis, and histological examination, a high dose of MEM has the potential to be an anti-rheumatic drug by reducing joint damage and TNF production. In addition, it could slow the progression of RA-related memory loss.
Limitations
Instead of H&E stain, bone-inflammation-cartilage (BIC) staining might have been more accurate, allowing accurate visualization and assessment of bone erosion, articular cartilage loss, and inflammation in a single stain. Rigor mortis in the tissues may have influenced our results, as ex vivo ankle X-ray imaging was performed.
Conclusion
MEM has anti-rheumatic and memory-promoting effects in CFA-induced RA in Wistar rats. These benefits of MEM may be related to its anti-inflammatory properties and its ability to inhibit the essential cytokines TNF-α and IL-6. In older people with persistent RA, MEM may be helpful in halting the progression of both dementia and RA. However, additional animal testing in various animal models of RA, as well as clinical investigations, are needed to support all of these claims for MEM.
Footnotes
Acknowledgments
The authors thank Sun Pharmaceutical Industries Ltd, Mumbai for providing gift sample of study drugs, Memantine and Methotrexate.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from Institutional Animal Ethics Committee.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
