Abstract
Kidney cancer is the most lethal of the genitourinary malignancies. Prior to the advent of cytokine-based immunotherapy 25 years ago, surgery was the only viable treatment for advanced renal cell carcinoma (RCC). Adjuvant therapy with interferon and interleukin-2 revolutionized the treatment paradigm, although responders were too often uncommon. The mid-portion of the last decade saw born a new class of drugs aimed at the angiogenesis/VEGF pathway, common to most clear-cell RCC. The latest FDA-approved compound in this class has promised to yield equal or better first-line efficacy as its predecessors while, perhaps, providing better tolerability. Here we review the available data at present regarding pazopanib.
Keywords
Introduction
Kidney cancer accounts for 3–5% of all cancers in the United States. 1 The 2013 estimated incidence and mortality for kidney cancer are 65,150 and 13,680 cases, respectively. 2 Of the primary tumors occurring within the kidney, renal cell carcinomas (RCCs) of epithelial origin are responsible for 80–85% of these pathologies. RCC is predominantly a disease of the sixth to eighth decade of life with median age of diagnosis of 64. 1 Risk factors for sporadic disease include smoking, hypertension, obesity, and end-stage renal disease.3–5 Among the various histologic subtypes, clear-cell renal cell carcinomas (ccRCCs) account for 75–85% of RCC. 6 Compared to other urologic malignancies, RCC stands as perhaps the most aggressive and lethal – 20–30% of patients present with metastatic disease at the time of diagnosis 7 and approximately 25% of patients with early stage disease relapse after nephrectomy. 8
Advances in the understanding of ccRCC cellular biology have begun to explain much of what had been known at a macroscopic level–-that is high vascular density helps promote growth. 9 This is believed to be associated with increased risk of metastasis, recurrence, and adverse prognosis. Although most commonly sporadic, it is through identification and intense examination of the hereditary forms of the disease that have led to the greatest degree of understanding of its varied and detailed biology. There are several hereditary forms of RCC, each with different mechanisms, phenotypes, and pathophysiologies. The study of patients with von-Hippel Lindau (VHL) disease, also presenting with kidney cancer, has revolutionized our understanding and treatment of all patients with both sporadic and hereditary forms of RCC. The field of study into the relationship of molecular derangement of the VHL protein leading to overexpression of vascular endothelial growth factor (VEGF) and other angiogenic proteins 10 has culminated most recently with the approval of several new small-molecule and angiogenesis pathway-targeting agents. Among these agents, pazopanib is one of the newer pharmacotherapies of promise with a better tolerability profile.
VHL and Molecular Pathogenesis
Linkage studies in the 1980s mapped VHL to the short arm of chromosome 3 (3p25). 11 In the 1990s, VHL was successfully isolated and identified. 5 The gene coding sequence spans three exons to create a 4.5 kb mRNA product encoding two proteins: a full-length 213 amino acid protein (pVHL30) and a small protein lacking the first 53 amino acids (pVHL19). 12 In hereditary disease, the largest group of germline mutations involves deletions ranging from 0.5 to 250 kb. The remaining mutations include mis-sense substitutions and mutations leading to truncated protein.13–15
VHL-associated neoplasms exhibit increased vasculature secondary to overproduction of angiogenic peptides within the hypoxia-inducible pathway. 16 Hypoxia-inducible mRNAs include VEGF, platelet-derived growth factor-β (PDGF-β), transforming growth factor-α/β (TGF-α and TGF-β), cyclin D1, carbonic anhydrase IX, glucose transporter, among others. 17 In concert with our understanding of VHL, pVHL30 inhibits accumulation of these mRNAs by degrading the α-subunits of hypoxia-inducible factor-1/2 (HIF-1 and HIF-2) transcription factors. By acting as part of a ubiquitin ligase protein complex, pVHL binds the α-subunits to target HIF-1 and HIF-2 transcription factors for ubiquitin-mediated proteosomal degradation. 18 Of note, pVHL binding requires modification of proline hydroxylase enzymes by oxygen. 19
Under normoxic conditions, HIF-α-subunits are rapidly degraded. However, in conditions of either hypoxia or absent/inactive pVHL, HIF-1 and HIF-2 become stabilized. 20 This activates the hypoxic gene response, leading to angiogenesis and proliferation. HIF accumulation can also occur with activation of mammalian target of rapamycin (mTOR) through the phosphoinositide 3-kinase/Akt pathway; mTOR phosphorylates and activates p70s6 kinase to enhance translation of HIF. Activated mTOR also phosphorylates 4E binding protein-1 to promote dissociation of the complex and allow eukaryotic initiation factor-4 subunit E to stimulate increase in translation of cell-cycle regulators such as cyclin D1 and c-Myc.21–23
VEGF and Tumorigenesis
Inactivation of VHL tumor suppressor gene in RCC allows for VEGF to serve as an important target for therapy. VEGF is a family of signaling proteins derived from alternative splicing of VEGF gene, which consists of eight exons. 24 These ligands modulate endothelial vasculature by binding to structurally similar receptor tyrosine kinases with various affinities: VEGFR1 (FLT1), VEGFR2 (KDR), and VEGFR3 (FLT4).25–29 Activation leads to downstream pathways mediating tumor proliferation. Tumor angiogenesis occurs by increasing blood flow to promote endothelial cell proliferation and survival, as well as by increasing permeability of existing cellular transport channels to develop an environment for endothelial cell migration by boosting chemotaxis of bone marrow-derived endothelial precursor cells.30,31 Therefore, inhibition of VEGF signaling may interfere with tumor growth. Current VEGF-targeted agents for metastatic renal cell carcinoma (mRCC) include bevacizumab, a humanized monoclonal antibody that inhibits VEGF-A, as well as receptor tyrosine kinase inhibitors (TKIs) such as sorafenib, sunitinib, axitinib, and pazopanib.
PDGF Receptor (PDGFR) and Tumorigenesis
There is accumulating evidence exploring the role of PDGFR signaling in tumor angiogenesis. As mentioned previously, VEGF has been shown to act as both an angiogenic factor and a survival factor for endothelial cells. But, susceptibility of tumor vessels to interference by VEGF/VEGFR-2 signaling may be restricted to only the subpopulation of vessels lacking colocalization with pericytes – in other words, restricted to more immature vessels. 32 Contact between endothelial cells and periendothelial support cells (pericytes, smooth muscle cells) helps to support new blood vessels, promotes endothelial survival, and inhibits endothelial cell proliferation. 33 Therefore, targeting pericyte–-endothelial cell interaction may enhance tumor regression by destruction and interfering with both VEGF and PDGFR.
The molecular mechanism of how pericytes confer vessel stability remains elusive. However, angiopoietin-1 (Ang-1/Tie2) and PDGF-B/PDGFR-β have been implicated; transgenic mouse models lacking Ang-1/Tie2 or PDGF-B/PDGFR-β fail to recruit pericytes in vessel formation resulting in a disturbance of blood vessel stabilization and maturation.34–36 Furthermore, in vivo studies with human tumor xenografts demonstrate that by inhibiting VEGFR-2 and PDGFR-β, resistance to tumor vessel regression can be prevented. This results in tumor cell apoptosis, blood vessel destabilization, and regression. 37 38 Thus, a PDGFR-β kinase may be a valuable interference tool in tumor angiogenesis and metastasis. 39
Pazopanib: Therapy for mRCC
mRCC is resistant to usual cytotoxic chemotherapy (floxuridine, 5-fluorouracil, and vinblastine), is inadequately addressed by hormonal agents (medroxyprogesterone acetate, tamoxifen, and toremifene), and responds relatively poorly to cytokine therapy such as high-dose interleukin-2 (IL-2) or interferon-alpha (IFN-α). 40 Response rates (RRs) to standard immunotherapies range between 5 and 30% and carry high, albeit short-term, toxicity to the patient.41–43 The development of molecular agents targeting VEGF or mTOR ligands and receptors to inhibit angiogenesis and cell proliferation has revolutionized the treatment of advanced RCC.
Pazopanib (Votrient; GlaxoSmithKline) is an orally available and potent competitive inhibitor of VEGFR-1, VEGFR-2, and VEGFR-3 with similar activity against PDGFR-α, PDFGR-β, and cytokine receptor (c-kit). It has modest activity against fibroblast growth factor receptor-1 (FGFR1), FGFR3, and transmembrane glycoprotein receptor tyrosine kinase (c-FMS).44–46 Approved by the United States Food and Drug Administration (USFDA) and European Medicines Agency (EMA) in 2009 and 2010, respectively, pazopanib is an approved first-line therapy for mRCC in both treatment naïve and previously treated patients who have progressed on cytokine therapy.
Phase I
VEG10003 (2002–2006) was an open-label, non-randomized, multiple-dose-finding phase I study to determine clinical activity, pharmacokinetics, and safety of pazopanib. 47 Studies of pazopanib in vivo inhibited growth of several models of human tumor xenografts in mice. Further, it was determined that steady-state concentration ≥40 μmol/L of pazopanib is required for maximal inhibitory in vivo activity. 47
Sixty-three adult patients with solid tumors refractory to standard therapy and without further standard therapy options were treated with various doses of drug in sequential dose-escalating cohorts (50 mg three times weekly to 2000 mg once daily and 300–400 mg twice daily). Clinical activity response was evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines, 48 and observed in the form of partial response and prolonged stable disease. Adverse events (AE) and laboratory values were graded according to National Cancer Institute (NCI) common terminology criteria. 49
A peak concentration of pazopanib was achieved within two to four hours with bioavailability between 13 and 39%. Plateau in steady-state concentration was observed at doses equal to and greater than 800 mg daily. Half-life of the drug was 31.1 hours. This study did not find a maximum tolerated dose; 800 mg daily was used for subsequent trials. 47
The most frequent drug-related toxicities included hypertension (33%), diarrhea (33%), hair depigmentation (32%), and nausea (32%). Uncommon toxicities include drug-related rash, epistaxis, mouth ulceration and stomatitis, and asthenia. Hand–foot syndrome was not observed as with other TKIs. 47 As with many other angiogenesis inhibitors, hypertension is the most frequent grade 3 toxicity reported (25%). Liver transaminases (aspartate aminotransferase (AST)/alanine aminotransferase (ALT)) and bilirubin elevation were other AE (all grades: 38, 24, 13%). 47
Phase II
The phase II clinical trial VEG102616 (2005–2008) was a multi-center study assessing the safety profile and efficacy of 800 mg pazopanib once daily administered orally to patients with mRCC or locally recurrent, predominantly ccRCC. 50 Originally designed as a randomized discontinuation trial, this study enrolled 225 patients (69% were treatment naïve; 31% had received one prior cytokine- or bevacizumab-containing regimen) but changed to an open-label trial after a planned interim analysis gave early indication of promising activity (RR 38%). The primary end point of the study was overall RR defined according to RECIST. Secondary endpoints included duration of response and progression-free survival (PFS).
The overall RR was 34.7% (95% CI, 28.4–40.9%) with median duration of response of 68 weeks as determined by independent review. This was similar whether patients were treatment naïve (RR 34%; 95% CI, 26–41%) or had one previous line of therapy (RR 37%; 95% CI, 26–49%). Median PFS was estimated to be 51.7 weeks (95% CI, 44–60 weeks). AE included diarrhea (63%), fatigue (46%), hair depigmentation (43%), nausea (42%), and hypertension (41%). Most common grade 3 or grade 4 AE include hypertension (8%), elevated AST/ALT (4–6%), diarrhea (4%), and fatigue (5%). 50
Phase III: Pazopanib vs. Placebo
VEG105192 (2006–2008) was a phase III randomized, double-blind, placebo-controlled study to evaluate efficacy and safety of pazopanib monotherapy in treatment-naïve and cytokine-pretreated patients with advanced renal cell carcinoma. At the time of study design, limited use of TKIs sunitinib and sorafenib precluded the use of either as a control arm. 51
The study included 435 adult patients with measurable, locally advanced mRCC. The patients were randomly assigned in the ratio of 2:1 to receive 800 mg oral pazopanib once daily or placebo (pazopanib n = 290; placebo n = 145). If placebo patients were found to have progressive disease, there was the option of receiving pazopanib in the open-label study. The primary end point was PFS. Secondary endpoints included overall survival (OS) and RR according to RECIST. Subject quality of life (QOL) was also assessed using questionnaires. 51
Baseline population characteristics of study subjects included similar proportions of treatment-naïve and cytokine-pretreated subpopulations (53 and 54%; 47 and 46%), median age of pazopanib-treated and placebo-treated subpopulations, and nephrectomy status (89 and 88%) between experimental arms. 51
Pazopanib significantly prolonged PFS as compared with placebo in the overall study population (median, 9.2 and 4.2 months; hazard ratio (HR), 0.46; 95% CI, 0.34–0.62; P < 0.0001), similar for both treatment-naïve (median, 11.1 and 2.8 months; HR, 0.40; 95% CI, 0.27–0.60; P < 0.0001) and cytokine-pretreated subpopulations (median, 7.4 and 4.2 months; HR, 0.54; 95% CI, 0.35–0.84; P < 0.0001). Tumor RR for all patients by independent review for pazopanib-treated patients in the overall study population was significantly improved compared to placebo (30 vs. 3%; P < 0.001) with a median duration of response of 58.7 weeks. Similar RR was seen in both pazopanib-treated (32%) and cytokine-pretreated (29%) subpopulations. 51 However, the difference in OS between pazopanib and placebo-treated patients was not statistically significant (median, 22.9 vs. 20.5 months; HR, 0.91; CI, 0.71–1.16; P = 0.224). 52
Most AE were limited to grade 1 and grade 2. AE included diarrhea (52%), hypertension (40%), hair color changes (38%), nausea (26%), anorexia (22%), vomiting (21%), and fatigue (19%). Clinical chemistry aberrancy includes elevated liver transaminases (53%) as well as hyperglycemia (41%) and increase in bilirubin (36%). Proportion of patients experiencing grade 3 and grade 4 AE was 33 and 7%, respectively, in the pazopanib arm as opposed to 14 and 6% in the placebo arm.
Summary of key trials in treatment of mRCC.
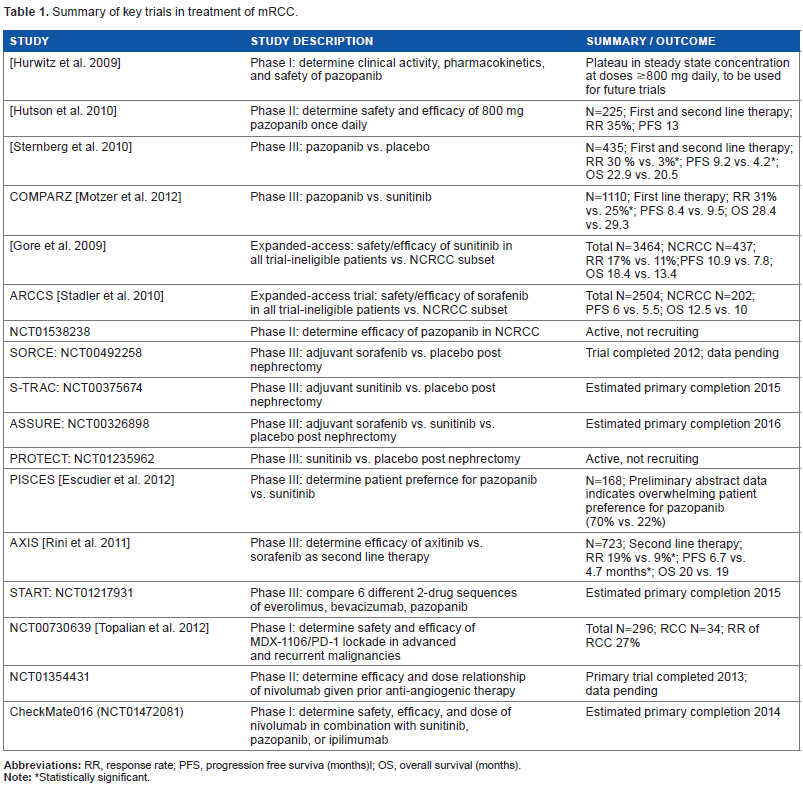
Statistically significant.
The most common grade 3 and grade 4 AE in the pazopanib arm included hypertension and diarrhea (4 and 4%). Mixed-model repeated-measures analysis of QOL questionnaires did not show statistical differences between pazopanib and placebo at any of the time points assessed. 51
Phase III: Pazopanib vs. Sunitinib in Treatment-naïve Patients (COMPARZ)
Another phase III trial (2008–2012) later compared the safety and efficacy of pazopanib against sunitinib in patients with mRCC as the first-line treatment. At the time of study design, the USFDA had approved both pazopanib and sunitinib among the TKIs as the first-line treatment options. Sunitinib had previously been compared to IFN-α in treatment-naïve patients. 53 Pazopanib had previously been compared to placebo in treatment-naïve and previously treated patients. Cross-trial analysis of these previous studies suggested similar PFS with pazopanib and sunitinib. Safety comparison, however, suggested that pazopanib would be associated with a lower incidence of fatigue, hand–foot syndrome, stomatitis, and myelosuppression but higher incidence of liver function abnormalities than sunitinib. 54 Direct comparisons from a controlled study are yet to occur.
COMPARZ was a randomized, open-label trial enrolling 1110 patients with clear cell mRCC. In a 1:1 randomization, subjects received continuous pazopanib (800 mg once daily; 557 patients) or sunitinib in six-week cycles (50 mg once daily for four weeks followed by two weeks off-therapy; 553 patients). The primary endpoint was PFS; secondary endpoints included OS, safety, and QOL. 55
Pazopanib proved non-inferior to sunitinib for PFS in independent review (median, 8.4 vs. 9.5 months; HR, 1.05; 95% CI, 0.90–1.22). OS was similar in both groups (median, 28.4 and 29.3 months; HR, 0.91; 95% CI, 0.76–1.08). RR, as determined by independent review, was significantly higher for pazopanib compared with sunitinib (31 vs. 25%; P = 0.03). Patients treated with sunitinib had a higher incidence of fatigue (63 vs. 55%), hand–foot syndrome (50 vs. 29%), and thrombocytopenia (78 vs. 41%). Conversely, patients treated with pazopanib had a higher incidence of change in hair color (30 vs. 10%), weight loss (15 vs. 6%), as well as elevated AST (all grades: 61 vs. 60%; grades 3 and 4: 12 vs. 3%) and elevated ALT (all grades: 60 vs. 43%; grades 3 and 4: 17 vs. 5%). Answers to peer-reviewed QOL questionnaires (Functional Assessment of Cancer Therapy Anemia and Fatigue scales, Renal Cell Carcinoma-Symptom Index, Functional Assessment of Cancer Therapy–Kidney Symptom Index, Cancer Therapy Satisfaction Questionnaire) in the first 6 months of treatment favored pazopanib. 55
Role of TKIs in the Treatment of Non-ccRCC (NCRCC)
It was stated earlier that approximately 85% of RCC are composed of the clear cell subtype. Of the remaining histologic subtypes, connecting tubule/collecting duct cell types (chromophobic, oncocytic, and duct Bellini-type tumors) comprise 11% of the tumor burden whereas chromophilic, metanephroid, and transitional cell types comprise less than 1% each of tumor burden. 6
There is lack of prospective and randomized data evaluating the efficacy of TKIs in the treatment of NCRCC. However, data from expanded access programs suggest that TKIs may have some activity in this subgroup of patients. NCT00130897 (2005–2010) is one of these expanded-access trials, designed to provide sunitinib on a compassionate-use basis to trial-ineligible patients with RCC from countries where regulatory approval had not been granted. Sunitinib was shown to have a tolerable safety and toxicity profile but decreased PFS and OS in the NCRCC subgroup when compared to all study patients (total patients n = 3464; NCRCC histology n = 437 patients; PFS of all patients (median, 10.9 months; 95% CI, 10.3–11.2); PFS of NCRCC subgroup (median, 7.8 months; 95% CI, 6.3–8.3 months); OS of all patients (median, 18.4 months; 95% CI, 17.4–19.2 months); OS of NCRCC subtype (median, 13.4 months; 95% CI, 10.7–14.9)). RR of all patients was 17% compared to 11% for NCRCC subtype. 56
Similarly, the Advanced Renal Cell Carcinoma Sorafenib (ARCCS) study (NCT00111020; 2005–2010) was a nonrandomized open-label expanded program exploring the safety and efficacy of sorafenib before regulatory approval. Efficacy and safety results were similar across subgroups with sorafenib (total patients n = 2504; NCRCC histology n = 202 patients; PFS of all patients (median, 24 weeks; 95% CI, 22–25); PFS of NCRCC subgroup (median, 22 weeks; 95% CI, not available); OS of all patients (median, 50 weeks; 95% CI, 46–52 weeks); and OS of NCRCC subtype (40 weeks; 95% CI, not available)). 57 The efficacy of TKI against NCRCC may be due to the tumor's expression of c-kit. Given that pazopanib is a small molecule inhibitor of c-kit tyrosine kinases, there is an ongoing phase II open-label trial evaluating the efficacy of pazopanib in NCRCC (NCT01538238; 2012–ongoing).
Role of TKIs as Adjuvant Therapy in the Treatment of RCC
To date, there are no data supporting the use of adjuvant therapy post-nephrectomy for the treatment of RCC. IFN-α and IL-2 adjuvant therapy have failed to demonstrate clinical benefit.58–60 However, several ongoing trials are evaluating the role of TKIs as adjuvant therapy: the SORCE, ASSURE, S-TRAC, PROTECT trials.
The SORCE trial (NCT00492258; 2007–2012) is a randomized phase III double-blind trial studying sorafenib vs. placebo in treating patients at risk of relapse after surgical removal of kidney cancer. Although the study has concluded, published data are pending. Similarly, the S-TRAC trial (NCT00375674; 2006–ongoing) is a randomized double-blind phase III study to compare disease-free survival time and safety of sunitinib vs. placebo in adjuvant treatment patients at high risk of recurrent kidney cancer after surgery. Secondary outcome includes OS, safety, and tolerability. The estimated primary completion date is 2015. Seemingly a combination of the SORCE and S-TRAC trials, the ASSURE trial (NCT00326898; 2006–ongoing) is a randomized phase III trial studying sorafenib vs. sunitinib vs. placebo in treating patients at risk of relapse post-nephrectomy. The primary outcome measures include PFS, and secondary outcome measures include OS, disease-free survival in patients with clear cell histology, and QOL. The estimated primary completion date is 2016. Finally, pazopanib is also being evaluated in the PROTECT trial (NCT01235962; 2010–ongoing). Randomized phase III study was conducted to evaluate whether pazopanib compared with placebo can prevent or delay in treating patients with moderately high or high risk of developing recurrence post nephrectomy. The estimated primary completion date is 2016. We eagerly await the results of these trials to see whether TKIs have a role as adjuvant therapy in the treatment of RCC.
Future Directions
Pazopanib is a powerful TKI exploiting the therapeutic benefits of VEGFR/PDGFR signaling inhibition to disrupt tumor angiogenesis. It is USFDA and EMA approved for first-line treatment of mRCC and advanced RCC. It proves to be an effective agent in both treatment-naïve and cytokine-pretreated patients, and non-inferior to sunitinib in the COMPARZ trial while having a better tolerability profile.
In an attempt to further characterize tolerability, the PISCES (Patient Preference Study of Pazopanib Versus Sunitinib in Advanced or Metastatic Kidney Cancer) trial (NCT01064310; 2010–2013) probes patient preference between pazopanib and sunitinib. It is a randomized, double-blind phase III study enrolling 168 treatment-naïve patients in a cross-over design. Patients are randomized to receive standard doses of pazopanib or sunitinib in a blinded fashion for 10 weeks with two-week washout period before cross-over to the opposite drug for an additional 10 weeks. The trial is expected to conclude in November 2013, but data presented at ASCO 2012 revealed an overwhelming preference for pazopanib (n = 114 given attrition; 70 vs. 22%; P < 0.001). Patients indicated that pazopanib resulted in a significantly better health-related QOL with less fatigue than sunitinib. 61 The caveat is that patients were surveyed at trial week 22, which coincides with the crescendo period of AE in sunitinib-treated patients. 62
Another area of great interest surrounds the timing and order of treatments. Current treatment algorithms support the use of a second TKI after disease progression. The AXIS (Axitinib As Second Line Therapy for Metastatic Renal Cell Cancer) trial (NCT00678392; 2010–2015) was a phase III randomized study that enrolled 723 subjects with RCC who progressed despite first-line therapy containing sunitinib, bevacizumab and IFN-α, temsirolimus, or cytokine-based therapy. 63 Subjects were randomized 1:1 to either axitinib (5 mg twice daily) or sorafenib (400 mg twice daily). The primary endpoint measured PFS; secondary endpoints included OS and QOL. PFS proved to be significantly improved with axitinib compared to sorafenib (median, 6.7 vs. 4.7 months; HR, 0.66; 95% CI, 0.54–0.81). Axitinib had a significantly improved RR compared with sorafenib (19 vs. 9%) as well as a higher rate of stable disease >20 weeks (27 vs. 20%). Treatment was discontinued due to toxicity in 4% of patients treated with axitinib as opposed to 8% of patients treated with sorafenib. The major AE (grades 3 and 4) for patients treated with axitinib included hypertension (16%), diarrhea (11%), fatigue (11%); major AE for sorafenib included hand–foot syndrome (16%), hypertension (11%), and diarrhea (7%). 55 In a later update, there was no difference reported in OS between the two arms (median, 20 vs. 19 months, HR, 0.96, 95% CI, 0.80–1.17). 64 Based on these findings, and prior approvals of six other targeted agents, axitinib has been promoted as a viable second-line strategy for progressive disease, even in patients previously treated with TKI therapy.
Building on the results of this trial and others with focus on sequential therapy, the START (Sequential Two-agent Assessment in Renal Cell Carcinoma Therapy) trial (NCT01217931; 2010–ongoing) is a currently accruing trial that aims to compare six different two-drug sequences (group 1: pazopanib and possible bevacizumab; group 2: pazopanib and possible everolimus; group 3: everolimus and possible bevacizumab; group 4: everolimus and possible pazopanib; group 5: bevacizumab and possible pazopanib; group 6: bevacizumab and possible everolimus) in the setting of mRCC. The primary endpoint is time to overall treatment failure. The estimated completion date is June 2015. One recent study examining the role of salvage-targeted kidney cancer therapy in patients progressing on high-dose IL-2 immunotherapy demonstrated promising results for the use of TKIs. This retrospective nonrandomized study analyzed data from 2003 to 2010 of a cohort of 286 patients who had all undergone cytoreductive nephrectomy. A total of 21 patients received salvage-targeted therapy after progression on high-dose IL-2, whereas 111 patients received targeted therapy alone. The remaining 15 patients had other treatment combinations or experimental therapy only. The results demonstrated a significantly higher disease-specific survival in patients who received salvage-therapy after progressing on high-dose IL-2 compared with that of patients treated with targeted therapy alone (median, 30 months; P = 0.004 with median not reached). Disease-specific survival was comparable for patients treated with salvage-targeted therapy after progression on high-dose IL-2 (median, 34 months) vs. first-line targeted therapy (median, 26 months; P = 0.175). 65 This study reiterates the role of immunotherapy in the treatment of mRCC.
A renaissance surrounding immunotherapy is occurring in the treatment of cancer. Kidney cancer has long stood as a tumor model that has potential to respond to immune modulation.66,67 The most recent iteration of immune modulation has borne molecules targeting two significant checkpoints involved in T-cell priming and effector mechanisms. One of these checkpoints involves the interaction of programmed death-1 (PD-1) and PD-1 ligand (PD-1 L). PD-1 in an inhibitory receptor expressed on T-cells. In murine tumor models, ligand B7-H1 confers immune resistance; disrupting PD1/B7-H1 interactions yield anti-tumorigenic effects.68–70 Nivolumab (MDX-1106) (BMS-936558/ONO-4538) is a genetically engineered human immunoglobulin G4 monoclonal antibody specific for PD-1. It binds PD-1 with high affinity to prevent interactions with B7-H1. An open-label phase I dose-escalation study of MDX-1106 (NCT00730639; 2008–ongoing) in advanced and recurrent malignancies (metastatic castration-resistant prostate cancer; RCC; metastatic melanoma; non-small cell lung cancer) evaluated the safety, clinical activity, and pharmacodynamics of PD-1 blockade. 71 Subjects (n = 39) with refractory disease were found to tolerate nivolumab well with some suggestion on efficacy (RR = 27%; 95% CI, 13–46). 72
The promising results have led to two noteworthy ongoing clinical trials. First, there is a phase two randomized, double-blind trial (NCT01354431; 2011-ongoing) studying nivolumab in the setting of patients specifically with RCC who have received before anti-angiogenic therapy. The primary outcome measures include PFS. Secondary outcome measures include tumor RR. The primary completion date was May 2013; however, the estimated study completion date is June 2014.
Second, CheckMate 016 is a phase I randomized, blinded study of nivolumab (NCT01472081; 2012–ongoing) in subjects with progressive, advanced/metastatic ccRCC to receive nivolumab in combination with sunitinib, pazopanib, or ipilimumab. The purpose is to study the safety, efficacy, and the best dose of nivolumab to use in combination with the above drugs for treatment of mRCC. The primary end points include safety and tolerability; secondary outcome measures include antitumor activity. The primary completion date is estimated to be May 2014.
Conclusions
The treatment of kidney cancer has undergone some of the most revolutionary changes in the field of genitourinary oncology. The advent and approval of TKIs and small molecules in this space continues to serve as a testament to the importance of research focused on the understanding and modulation of basic scientific pathways involved in cellular tumorigenesis and its selective advantage.
Pazopanib is one of the latest fruits of this labor. Much future work remains involving our better understanding of the proper sequencing of therapies. This includes tolerability pro-filing also as it potentially involves detailed genomic sequencing and true personalization of care. The multispoke model of cancer research is beginning to witness the convergence of seemingly unrelated therapeutics. The better we understand that cancer is not the simple derangement of one cell that remains immortalized, as we once thought, the closer we will be to winning this war.
Abbreviations
adverse events
alanine aminotransferase
angiopoietin-1
aspartate aminotransferase
clear cell renal cell carcinoma
cytokine receptor
European Medicines Agency
fibroblast growth factor receptor
hypoxia-induced factor
hazard ratio
interleukin
interferon
metastatic renal cell carcinoma
mammalian target of rapamycin
National Cancer Institute
non-clear-cell renal cell carcinoma
overall survival
programmed death-1
PD-1 ligand
platelet-derived growth factor
progression-free survival
platelet-derived growth factor receptor
quality of life
renal cell carcinoma
Response Evaluation Criteria in Solid Tumors
response rate
tyrosine kinase with immunoglobulin-like and EGF-like domains
transforming growth factor TKI: tyrosine kinase inhibitor
United States Food and Drug Administration
vascular endothelial growth factor
vascular endothelial growth factor receptor
von-Hippel Lindau
Author Contributions
Analyzed the data: TL and ER. Wrote the first draft of the manuscript: TL and ER. Contributed to the writing of the manuscript: TL and ER. Agree with manuscript results and conclusions: TL and ER. Jointly developed the structure and arguments for the paper: TL and ER. Made critical revisions and approved final version: ER. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. this article was subject to blind, independent, expert peer review. the reviewers reported no competing interests. provenance: the authors were invited to submit this paper.
