Abstract
We tested a global collection of Candida spp. strains against fluconazole, voriconazole, caspofungin and anidulafungin using CLSI M27-A3 broth microdilution (BMD) method, in order to to compare if there are any differences between the susceptibility data using the old and new CLSI clinical breakpoints for azoles (fluconazole and voriconazole) and echinocandins (caspofungin and anidulafungin) in a large number of Candida albicans and the non-albicans Candida species emerging in Europe (C parapsilosis and C. tropicalis) strains isolated from BSIs. During the study period, a total of 919 isolates of Candida spp. (427 C. albicans, 304 C. parapsilosis, 82 C. tropicalis and 106 isolates of other species) were obtained from over 40 Spanish hospitals. The MICs90 (in mg/L) for fluconazole, voriconazole, caspofungin and anidulafungin, respectively, for each considered species were as follows: C. albicans, 2, 0.06, 0.125, 0.125; C. parapsilosis, 2, 0.06, 2, 2; C. tropicalis, 2, 0.125, 0. 125, 0.125. Considering the effects in antifungal susceptibility of the new clinical breakpoints it seems that the new clinical breakpoints are more sensitive in the detection of cross - resistance between different azoles, but contrary to what happened on the susceptibility profile of azoles, the increase in the new clinical breakpoints MIC values decreased the rate of C. parapsilosis echinocandin resistant isolates. Therefore, the new epidemiological clinical breakpoints provided by the CLSI promises to be a more sensitive tool to detect emerging reduced antifungal susceptibility among Candida spp. as well as improve the clinical utility of antifungal in vitro testing.
Keywords
Introduction
There has been an increase in the incidence of nosocomial invasive fungal infections during last years. In fact, Candida spp. bloodstream infections (BSIs) are the fourth most common cause of nosocomial invasive infection in Northern hemisphere tertiary care hospitals.1–11 with a high morbidity and mortality rates that have converted Candida spp. BSIs in one of the most important public health associated problems.12,13
Although, Candida albicans has been the most prevalent yeast isolated from patients suffering from invasive fungal BSI, but non-albicans Candida species have emerged as etiological agents of invasive candidiasis.1,14–16 Despite this fact, there are epidemiological differences between the epidemiology of candidemia in different countries. For example, while C. glabrata is the major non-albicans Candida species that causes invasive candidemia in the United States,10,17–19 recent studies showed that C. parapsilosis and C. tropicalis are increasing in Europe, Canada and Latin American countries.10,20,21 These epidemiological changes in Candida BSI have generated a new geographically different distribution of Candida species. On the other side the emergence of fluconazole resistance, especially linked to some non-albicans Candida species such as C. krusei and C. glabrata22–27 had led to the extensive use of new antifungal azoles and echinocandins in the treatment of invasive candidemia.28–33 In spite of the fact that the epidemiology of candidemia is changing, not all these changes in epidemiology and antifungal susceptibility are homogeneous. Moreover there are geographical differences due to different matters such as risk conditions to develop a candidemia, infection control programs, therapeutic or prophylaxis approaches to the patient, etc.3–5,7,8,10,13,15 Recently some studies have focused on the evaluation of these epidemiological and susceptibility changes in invasive candidemia and showed that these changes could be detected even in different regions from the same country. This fact has been recently documented from the data obtained from two large nationwide candidemia surveillance studies that have been conducted in Spain during the period from 2008 to 2010.8,10 Although these sort of antimicrobial resistance surveillance programs have provided useful and complete data regarding resistance trends, the distribution of pathogens and types of clinical invasive infections, there is no knowledge on how the new CLSI proposed breaking points 36,37 could affect the susceptibility results obtained in Spain and if the conclusions and further recommendations made based on these surveillance studies are valid nowadays. Therefore, the aim of this study is to compare if there are any differences between the susceptibility data using the old and new CLSI clinical breakpoints for azoles (fluconazole and voriconazole) and echinocandins (caspofungin and anidulafungin) in a large number of Candida albicans and the non-albicans Candida species emerging in Europe (C. parapsilosis and C. tropicalis) strains isolated from BSIs.
Materials and Methods
Clinical isolates
Nine hundred and nineteen Candida spp. clinical isolates obtained from 40 Spanish tertiary care hospitals located across the country from June 2008 through June 2009 were tested. The collection species distribution was the following: 427 isolates of C. albicans, 304 C. parapsilosis, 82 C. tropicalis and 106 isolates of other species (C. glabrata, C. krusei and C. guilliermondii). All isolates were obtained by automated blood culture systems and subcultured onto Chromagar® Candida screening medium and tested to ensure the colony purity and viability. The Candida specie identification was performed according to their morphology, filamentation test and auxonogram using the API ID 32C system. Besides, all the isolates identified as Candida spp. or those identified (eg, C. famata, C. lusitaniae or others) but with a total distribution rate less than 1%, were not considered in this work
Ethical issues
The Ethical committee of each local institutional board reviewed the research protocol and approved the study according to the ethical and legal issues at the moment.
Antifungal compounds
Reference powders were provided by their respective manufacturers (anidulafungin, voriconazole and fluconazole by Pfizer, and caspofungin by Merck). Stock solutions for broth microdilution testing were prepared in water for fluconazole and caspofungin and dimethyl sulfoxide (DMSO) for voriconazole and anidulafungin, taking into account the potencies of the powders according to the methodology included in the reference document CLSI M27-A3.
In vitro susceptibility testing
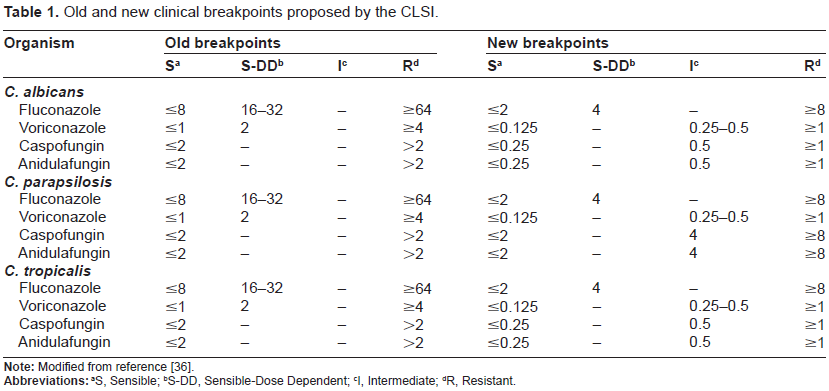
Broth microdilution was performed strictly according to the CLSI M27-A3 standard. Plates were stored at -86 °C for a maximum of 15 days before use. Micro-titer 96 plates were read visually and the MIC determined using prominent inhibition (corresponding to half percent) as endpoint. C. krusei ATCC 6258 and C. parapsilosis ATCC 22019 were used as quality control reference strains in each time that clinical isolates were evaluated as indicate the CLSI document in order to check the drug dilution accuracy and the results reproducibility. The MIC breakpoints were interpreted taking into account the recommendations made on the CLSI M27-A3 document and the new breakpoints suggested by the CLSI subcommittee as can be shown in the Table 1.36,37
Old and new clinical breakpoints proposed by the CLSI.
S, Sensible;
S-DD, Sensible-Dose Dependent;
I, Intermediate;
R, Resistant.
Statistical analysis
Categorical data were analyzed using Chi-square or Fisher's exact tests as appropriate, and t-test or Wilcoxon test were used to compare continuous variables according to the significance of the previous normality test. Spearman rank-order correlation was used to measure the relationship between the MICs of both azoles (fluconazole and voriconazole) and both echinocandins (caspofungin and anidulafungin).
The number of overlaps (NOO) and very major errors (VME) were used for the evaluation and comparison of the performance using the former and revised susceptibility breakpoints for each drug considered. These parameters were defined as follows: (1) Number of overlaps (NOO): calculated as number of isolates for which their classification was overlapping, and (2) Very major errors (VME): which is the number of isolates for which the original classification was susceptible and the actual is resistant or viceversa.
Besides the global analysis of the whole data, the Spanish territory was artificially divided in four different geographical areas (North, South, Center and East) (see Fig. 1) and each area's data was analyzed individually to asses if there were differences in the epidemiology and the antifungal susceptibility profile across the country.
Spanish geographic regions distribution.
Results and Discussion
The first remarkable finding of our study is that the total number and distribution of the strains isolated was not homogeneous across the country. The distribution of the 919 clinical isolates obtained during the study period along the four different geographical areas mentioned above is described on Table 2. Summarizing, the obtained distribution was the following: 197 isolates in the Northern part, 171 isolates in the Southern part, 214 in the Center area and 337 in the Eastern site of the country, respectively. C. albicans remains as the most frequently isolated species covering almost half (45.9%) of the global cases, but the rates of this species between the four different areas were not homogenous. In fact in the Southern part of the country the rate of C. albicans isolates (39.2%) was very similar to the proportion of C. parapsilosis (37.4%).
Species distribution of Candida isolates by geographic region in Spain.
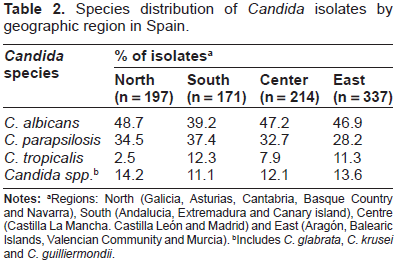
Regions: North (Galicia, Asturias, Cantabria, Basque Country and Navarra), South (Andalucia, Extremadura and Canary island), Centre (Castilla La Mancha. Castilla León and Madrid) and East (Aragón, Balearic Islands, Valencian Community and Murcia).
Includes C. glabrata, C. krusei and C. guilliermondii.
Considering the global distribution of species C. albicans was followed by C. parapsilosis (32.3%), which is concordant with the results published for other European countries where C. parapsilosis candidemia account the vast majority of non-albicans Candida species.3,8,10
The rate of incidence of C. tropicalis isolates ranged from 2,5% in the Northern area to 12,3% in the Southern area, with a global rate of 8.8%. These variations in the proportion of C. tropicalis BSIs affect the rate of other non-albicans Candida species like C. glabrata or C. krusei which are prone to multi-drug resistance compared with other Candida species isolated from BSI.15,21,23,27 In this study, C. glabrata and C. krusei together with other Candida species comprised the remaining 13% of the isolates.
These differences in the species distribution described for each area across Spain could reflect differences in the age of the patients attended, infection control procedures, transplant programs, clinical guidelines for treatment and prophylaxis of fungal invasive infections, use of central invasive devices or parenteral nutrition, cancer rate, social and economic inequalities and health policies in the Spanish regions. Maybe all these situations mentioned, could explain the different epidemiological profile recorded in our study for each Spanish region, but this point needs further clarification and is out of the scope of this study.
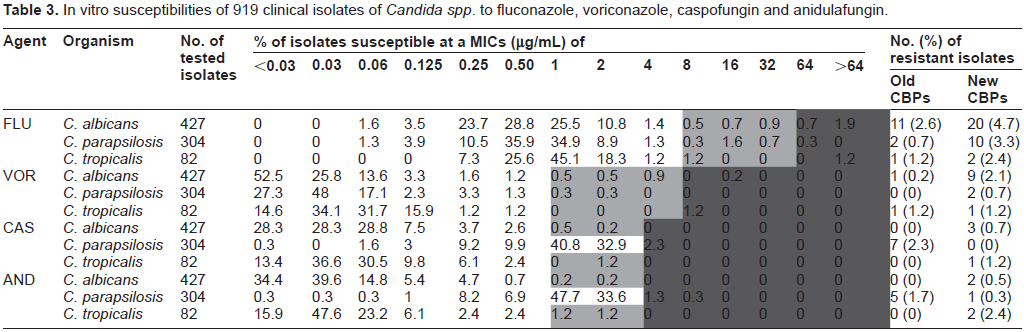
MICs values of the different Candida species for fluconazole, voriconazole, caspofungin and anidu-lafungin are summarized in Table 3. The new clinical breakpoints increase the number of C. albicans and C. tropicalis isolates which are dose dependent (SDD) or resistant to azoles and echinocandins. When we considered the C. parapsilosis isolates, the susceptibility profile for azoles was the same to the one expressed for C. albicans and C. tropicalis, but on the contrary, the echinocandins susceptibility profile was completely different because there was a decrease in the number of resistant isolates as a result of the increase in echinocandins MIC clinical breakpoints for this specie34,35,38,39 (see Table 1).
In vitro susceptibilities of 919 clinical isolates of Candida spp. to fluconazole, voriconazole, caspofungin and anidulafungin.
Effect of the old and new species specific CBPs in global and cross-resistance in azoles
Although, most Candida species analyzed were susceptible for fluconazole and voriconazole, we found that the activity of voriconazole is higher, independently of clinical breakpoints considered (Table 4).34,35
MIC 90 distribution for the different antifungal drugs and Candida species studied in this work.
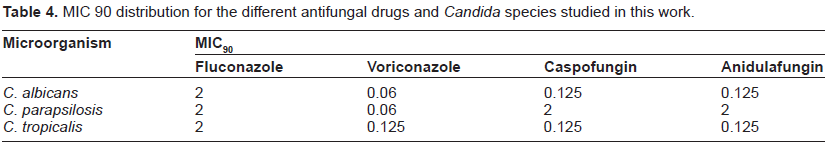
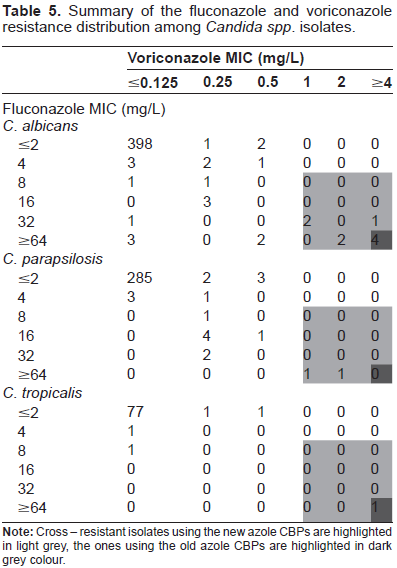
C. albicans showed the major percentage of resistant isolates to fluconazole and voriconazole followed by C. parapsilosis and C. tropicalis considering both clinical breakpoints (CBPs). When we considered the old breakpoints (MIC breakpoint ≥64 µg/mL for fluconazole and ≥4 µg/mL for voriconazole), 11 and 5 isolates of C. albicans were resistant to fluconazole and voriconazole, respectively. Eighty percent of the voriconazole resistant C. albicans isolates showed cross-resistance with fluconazole. The proposed new breakpoints for azoles (MIC ≥ 8 µg/mL for fluconazole and ≥1 µg/mL for voriconazole), did not change the proportion. Twenty isolates were resistant to fluconazole and 9 for voriconazole and the nine of them showed cross-resistance with flu-conazole. Therefore, it seems that the new clinical breakpoints are more sensitive in the detection of cross - resistance between different azoles and they could play a role of clinical interest in guiding the election of the most convenient antifungal therapy. When the effect of the new breakpoints in the susceptibility profile of C. parapsilosis isolates was analyzed, we found that the new CBP increased 5 times the number of resistant isolates to fluconazole (being 2 isolates using the old CBP and 10 isolates when the new CBP was considered). Besides, none but 2 of the fluconazole resistant C. parapsilosis isolates showed cross-resistance to voriconazole. Regarding the C. tropicalis isolates, only one isolate was resistant to fluconazole when the old and new CBPs were considered. This isolate also showed cross-resistance to voriconazole (see Tables 3 and 5).
Summary of the fluconazole and voriconazole resistance distribution among Candida spp. isolates.
Effect of the old and new species specific CBPs in global and cross-resistance in echinocandins
The MICs obtained for C. albicans and C. tropicalis were lower compared to those obtained for C. parapsilosis as it has been described along the literature.8,10,21,38,39 The overall resistance to both drugs was low. The susceptibility profile for caspofungin showed only 3 C. albicans and one C. tropicalis isolates resistant to this drug. Nearly the same situation was recorded for anidulafungin, where 2 isolates of each species were resistant. All of the resistant isolates were determined using the new breakpoints, because with the old ones we did not found any resistant isolate. Despite many clinical trials have demonstrated that any of the three echinocandins can be used for the treatment of invasive candidemia due to a sensible isolate using the old CBPs, 28 the data from in vitro enzyme kinetics experiments as well as the emergence of isolates that harbored FSK1 mutations, showed that these sort of isolates have a reduced susceptibility to these drugs and could lead to treatment failure. 39 As there are no additional mechanism of resistance to echinocandins described at the moment, the new CBPs seem to indicate those isolates that could be problematic and maybe could not respond to antifungal treatment with these drugs at the dose regimen established for them.38,39
There were 7 and 5 C. parapsilosis isolates resistant to caspofungin and anidulafungin when considered the old and new breakpoints respectively. Three of seven caspofungin resistant C. parapsilosis isolates showed cross-resistance to both drugs. Contrary to what happened on the susceptibility profile of azoles, the increase in the new clinical breakpoints MIC values decreased the absolute number of C. parapsilosis resistant isolates.
Changes in antifungal resistance distribution among Candida species isolated from different geographical areas
The vast majority of fluconazole and voriconazole resistant C. albicans isolates were found in the Northern and Southern parts of the country, whereas for the major distribution to azole resistant C. parapsilosis isolates were comprised in the Center and Eastern areas of Spain. Despite the resistance to echinocan-dins were minimum, most of the resistant isolates were located in the Northern region. These differences in the susceptibility profile of each area are reflected on Figure 2.
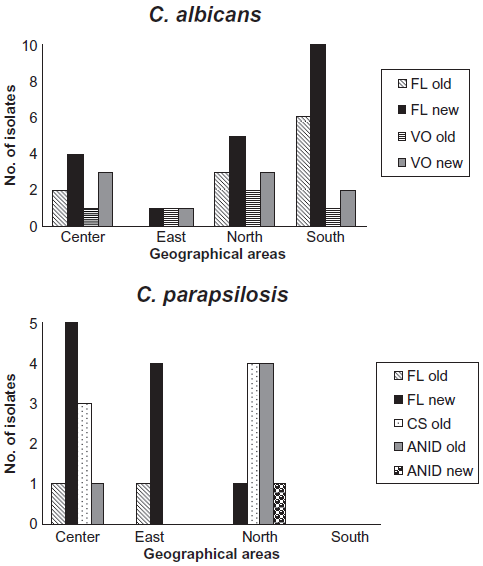
Susceptibility differences in C. albicans and C. parapsilosis isolates.
Though these results emphasize the initial idea that there are geographical variations in the susceptibility profile and different epidemiological risk factors associated with invasive Candida strains,8,10,18–20 they must be taken with care because the low proportion of resistant isolates observed in our study. In fact, when we studied statistically the differences between the three Candida species considered and the susceptibility profile of them according to the different regions we found no statistically significant differences between them.
Finally when we analyzed the number of category overlapping results we found an overall agreement of more than 95% with no very major errors (VME) for all drugs considered in this study.
Summarizing, the new epidemiological cut-off points and clinical breakpoints provided by the CLSI promises a more sensitive tool to detect emerging resistance as well as improve the clinical utility of anti-fungal in vitro testing. While there are no so many differences in the overall rate of resistance for azoles and echinocandins using the old or the new breakpoints, the new ones seems to be more sensitive in the detection of problematic strains with a reduced susceptibility to different drugs and illustrate the potential problem of cross-resistance between azoles with a direct impact in treatment failure and the outcome of the patient.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contrib-utorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
