Abstract
Danofloxacin is a fluoroquinolone antibacterial agent approved for use in veterinary medicine to treat and control bovine respiratory disease caused by Mannheimia haemolytica or Pasteurella multocida. Susceptible minimal inhibitory concentration (MIC) breakpoint (≤0.25 µg/mL) and disk diffusion interpretive criteria (≥22 mm) values for danofloxacin against M. haemolytica and P. multocida were first approved by the Clinical and Laboratory Standards Institute (CLSI) in 2003. However, intermediate and resistant breakpoint values were not established because only susceptible wild-type populations were evident at the time of breakpoint approvals. Since then, nonsusceptible isolates of M. haemolytica and P. multocida have been identified. We report danofloxacin intermediate MIC breakpoint (0.5 µg/mL) and disk diffusion interpretive criteria (18–21 mm), as well as danofloxacin-resistant MIC breakpoint (≥1 µg/mL) and disk diffusion interpretive criteria (≤17 mm), based on scattergram plots of MIC values versus disk zone diameters and calculated error-bound rates using M. haemolytica and P. multocida isolates recovered from bovine respiratory disease in North America in 2004–2014. These newly established intermediate and resistant clinical breakpoint values have been endorsed by CLSI and can be used for interpreting results from antibacterial susceptibility testing of danofloxacin against M. haemolytica and P. multocida isolated from bovine respiratory disease.
Keywords
Antibacterial susceptibility testing methods require interpretive criteria, also known as clinical breakpoints, to enable the diagnostic laboratory and veterinary clinician to interpret results for a bacterial pathogen as being susceptible, intermediate, or resistant to an antibacterial agent. 8 Danofloxacin mesylate is a fluoroquinolone antibacterial agent that was developed and approved for use in the United States, Europe, and other countries for the treatment and control of bovine respiratory disease (BRD) associated with Mannheimia haemolytica or Pasteurella multocida. 9 Susceptible (S) danofloxacin minimal inhibitory concentration (MIC; ≤0.25 µg/mL) and disk diffusion (S ≥22 mm) interpretive criteria for cattle M. haemolytica and P. multocida were approved by the Clinical and Laboratory Standards Institute (CLSI), and the establishment of the susceptible interpretive criteria was based on analysis of appropriate MICs, pharmacokinetic–pharmacodynamic (PK-PD) targets, and clinical efficacy data. 1 At the time that susceptible interpretive criteria were established, BRD pathogens with higher danofloxacin MIC values had not been identified and it was not possible to establish intermediate or resistant interpretive criteria. Since the approval of these susceptible clinical breakpoints in 2003, increased MIC and decreased disk zone diameter values for danofloxacin have been determined among M. haemolytica and P. multocida field isolates collected in North America in 2004–2014. Susceptibility data using these field isolates were plotted and analyzed in which scattergrams showed good correlation between MIC and zone diameter values with no major or very major calculated error-bound rates, thus allowing for the establishment of intermediate and resistant interpretive criteria for danofloxacin. The population MIC values, generated scattergrams, calculated error-bound rates, and proposed intermediate and resistant interpretive criteria values are presented herein for danofloxacin with M. haemolytica and P. multocida from BRD cases in North America. These newly approved CLSI interpretive criteria can be used by the diagnostic laboratory and veterinary clinician for interpreting results from antibacterial susceptibility testing of danofloxacin against M. haemolytica and P. multocida isolated from BRD.
All M. haemolytica and P. multocida isolates used in our study were recovered from diseased or dead beef or dairy cattle in 2004–2014 from U.S. and Canada veterinary laboratories. These laboratories selected isolates based on their own protocols and were requested not to use antibacterial susceptibility as a criterion for selection. Laboratories were also requested to submit no more than 32 isolates each year (8 isolates per quarter) in order to prevent over-representation from any one geographic area. The number of isolates was always ≤32 for each bacterial species per laboratory, and each participating laboratory provided no more than 1 isolate of each bacterial species from a herd each quarter-year in order to reduce over-representation of bacterial clones. Bacterial isolates were identified to the species level by each participating laboratory. Further identification and characterization of bacterial species were performed using standard biochemical tests and commercially available identification systems.a–c All isolates were stored in 1.0 mL of trypticase soy broth d supplemented with 10% glycerol and stored at approximately −70°C until tested.
In vitro susceptibility data were generated by performing MIC tests following CLSI standardized methods and quality control during susceptibility testing. 2 MIC values for all isolates were determined using a dehydrated broth microdilution system, e which conforms to CLSI standards for testing of veterinary pathogens. Direct colony suspensions were used and prepared at a final bacterial concentration of ~5 × 105 colony forming units (CFU) per milliliter. Custom-made, 96-well microtiter panels included serial doubling dilutions of danofloxacin, and the concentration ranges were chosen to encompass appropriate quality control ranges and available clinical breakpoints. CLSI standardized methods and quality control methods were also followed for disk diffusion testing in which disks containing 5 μg danofloxacin were used. 2 Direct colony suspensions were used for zone diameter testing and prepared at a final bacterial concentration of ~1 × 108 CFU/mL.
MIC values for 3,823 M. haemolytica and 4,054 P. multocida isolates were used to examine the presence of nonsusceptible (MIC values >0.25 μg/mL) isolates to danofloxacin. For M. haemolytica, the MIC50 value was 0.064 μg/mL and the MIC90 was ≥4 µg/mL in which 20% of the population was above the susceptible clinical breakpoint of 0.25 μg/mL (Fig. 1). For P. multocida, the MIC50 was 0.016 μg/mL and the MIC90 was 0.5 μg/mL in which 12.1% of the population was above the susceptible clinical breakpoint of 0.25 μg/mL (Fig. 2). After the presence of nonsusceptible populations for M. haemolytica and P. multocida were confirmed, MIC versus disk diffusion zone diameter values for M. haemolytica and P. multocida were generated and plotted in scattergrams in which error rates were calculated using CLSI-approved guidelines to determine if acceptable intermediate and resistant breakpoints could be established. 1
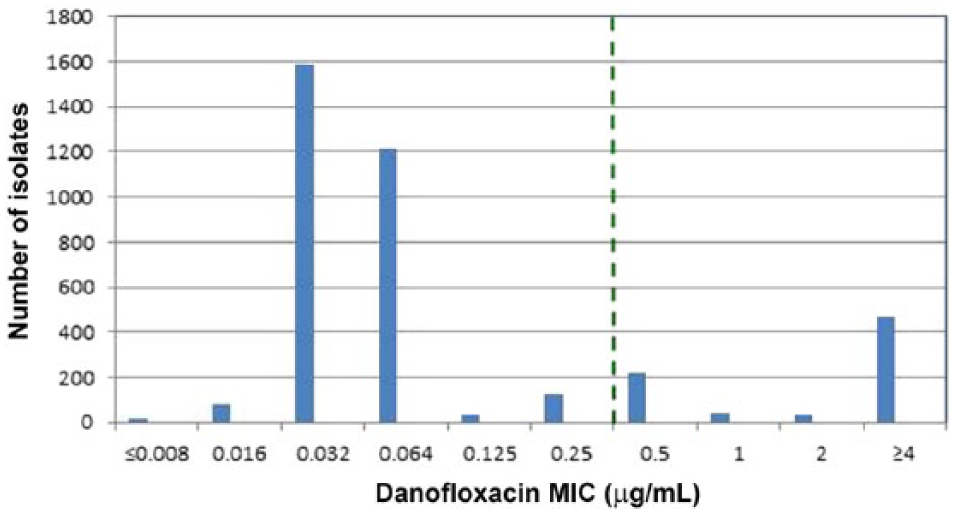
Danofloxacin minimal inhibitory concentration distributions for Mannheimia haemolytica (n = 3,823) recovered from bovine respiratory disease (BRD) cases in North America in 2004–2014. Vertical dashed line indicates the susceptible danofloxacin Clinical and Laboratory Standards Institute–approved breakpoint (≤0.25 µg/mL) for M. haemolytica BRD pathogens.
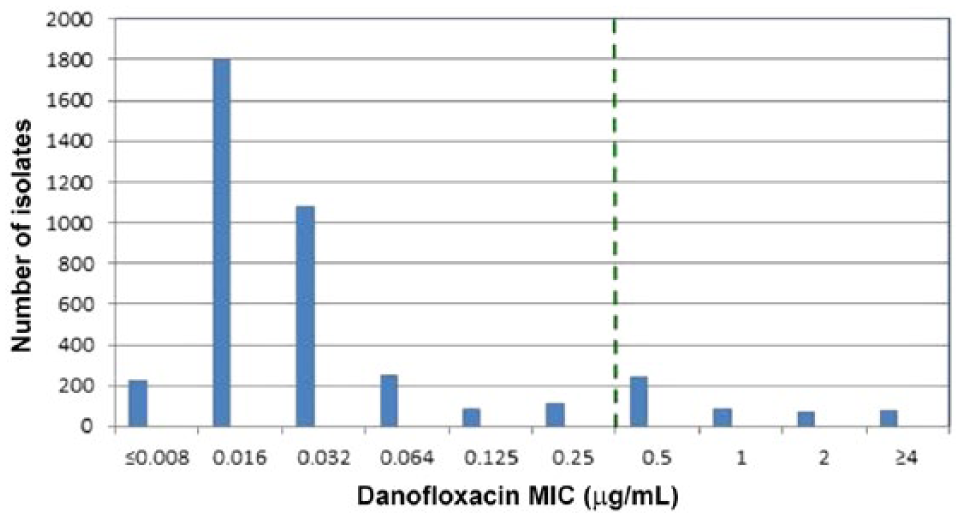
Danofloxacin minimal inhibitory concentration distributions for Pasteurella multocida (n = 4,054) recovered from bovine respiratory disease (BRD) pathogens in North America in 2004–2014. Vertical dashed line indicates the susceptible danofloxacin Clinical and Laboratory Standards Institute–approved breakpoint (≤0.25 µg/mL) for P. multocida BRD pathogens.
Scattergram plots were generated to illustrate the relationship between MIC and disk diffusion zone size using 324 M. haemolytica and 298 P. multocida isolates of bovine origin. Isolates used for these scattergrams were obtained from BRD cases in North America between 2013 and 2014 as previously described. Standardized procedures and quality control strains for generation of MIC and zone diameter values were utilized according to CLSI, and calculated error-bound rates were determined following procedures outlined by CLSI.1,2 Scattergram plots for M. haemolytica included horizontal lines for the proposed MIC breakpoints and vertical lines for the proposed disk diffusion breakpoints as well as calculated error (discrepancy) rates based on the breakpoints (Supplemental Fig. 1, available at http://vdi.sagepub.com/content/by/supplemental-data). Very major or major error rates were not identified when setting intermediate MIC and zone diameter values at 0.5 µg/mL and 18–21 mm, respectively, and for resistant MIC and zone diameter values at ≥1 µg/mL and ≤17 mm, respectively. A minor error rate of 0.9% was calculated. Scattergram plots for P. multocida also included horizontal lines for the proposed MIC breakpoints and vertical lines for the proposed disk diffusion breakpoints as well as the calculated error (discrepancy) rates based on the same breakpoints (Supplemental Fig. 2, available at http://vdi.sagepub.com/content/by/supplemental-data). Very major or major error rates were not identified when setting intermediate MIC and zone diameter values at 0.5 μg/mL and 18–21 mm, respectively, and for resistant MIC and zone diameter values at ≥1 μg/mL and ≤17 mm, respectively. A minor error rate of 3.4% was calculated.
MIC and disk diffusion zone diameter values rely on interpretive criteria to predict a potential susceptible, intermediate, or resistant outcome for an antibacterial agent in an infection. The category of “susceptible” implies that an infection caused by a bacterial pathogen may be treated with the drug dosage that is recommended for that type of infection and infecting species; the “intermediate” category implies that an infection caused by a bacterial pathogen may be treated in body sites where the drug is physiologically concentrated and serves as a buffer zone against technical factors that may cause discrepancies in interpretation; the “resistant” category implies that resistant strains are not inhibited by the achievable concentrations of an antibacterial agent and that resistance mechanisms are likely present within the pathogen. 2 Values from antimicrobial susceptibility testing that are reported as nonsusceptible or not interpretable (NI) usually indicates that only a susceptible interpretive criterion has been established for an agent because of the absence or rare occurrence of resistant strains. In the absence of danofloxacin intermediate and resistant interpretive criteria, numerous veterinary diagnostic laboratories and clinicians have been reporting NI for values above the currently established susceptible interpretive criteria or have been using enrofloxacin interpretive criteria values to predict the danofloxacin outcome. However, without further studies, enrofloxacin should not be used to predict danofloxacin outcome because other factors such as drug activity, dosage, and PK-PD parameters must be considered.
Using the currently established susceptible MIC breakpoint for danofloxacin, inspection of histograms in Figures 1 and 2 showed that nonsusceptible populations (MICs >0.25 μg/mL) were observed for danofloxacin among M. haemolytica and P. multocida pathogens isolated from BRD cases in North America collected over an 11-y period (2004–2014), necessitating a need for intermediate and resistant interpretive criteria for danofloxacin. Increases in danofloxacin MIC values may be caused by numerous mechanisms that are known to decrease fluoroquinolone activity and include efflux pumps, which decrease intracellular fluoroquinolone concentrations, plasmid-mediated resistance genes, which produce proteins that bind to DNA gyrase and protect the organism from the action of quinolones, and mutations at key sites in DNA gyrase or topoisomerase, which can decrease the binding affinity to quinolones and decrease drug activity.3–7 Although not determined, the populations of nonsusceptible M. haemolytica and P. multocida found in our study presumably contain similar resistance mechanisms for an increased MIC phenotype to danofloxacin.
MIC versus disk diffusion testing showed good correlation for M. haemolytica and P. multocida isolates (Supplemental Figs. 1, 2). Calculation of error-bound rates did not identify very major or major errors for the proposed intermediate and resistant breakpoint values. The following intermediate and resistant breakpoints are proposed, which have been endorsed by CLSI for danofloxacin susceptibility testing of M. haemolytica and P. multocida recovered from BRD: for broth or agar dilution susceptibility tests, danofloxacin MIC breakpoints for intermediate and resistant strains are set at 0.5 μg/mL and ≥1 μg/mL, respectively; in disk diffusion with the 5-μg danofloxacin disk for zone diameter testing, danofloxacin breakpoints for intermediate and resistant strains are set at 18–21 mm and ≤17 mm, respectively (Table 1). These newly established, CLSI-approved interpretive criteria can be used along with the currently established susceptible clinical breakpoint (MIC breakpoint at ≤0.25 μg/mL and disk diffusion interpretive criteria at ≥22 mm) for interpreting results from antibacterial susceptibility testing of danofloxacin against M. haemolytica and P. multocida isolated from BRD. The dose of 1.25 mg/kg is not assured to meet currently accepted PK-PD targets, and the interpretive criteria are limited to the 2 approved doses (6 mg/kg twice per day, or 8 mg/kg once per day).
Zone diameter and minimal inhibitory concentration (MIC) interpretive standards for Mannheimia haemolytica and Pasteurella multocida from bovine respiratory disease for danofloxacin.*
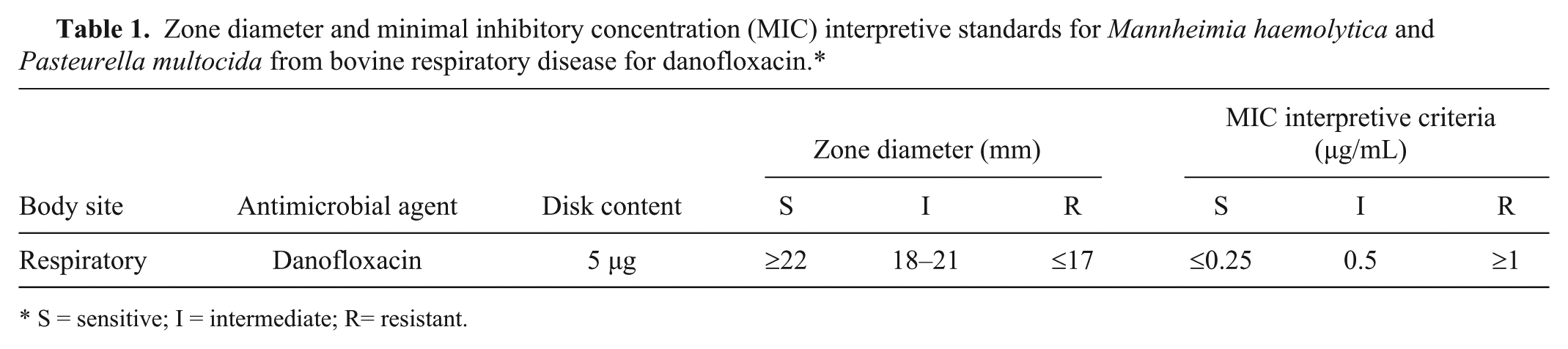
S = sensitive; I = intermediate; R= resistant.
Footnotes
Acknowledgements
We thank Don Bade, Michelle Ruiter, and Angela Sykes for their laboratory contributions.
Authors’ contributions
Michael Sweeney contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. Mark Papich and Jeffrey Watts contributed to design of the study and interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
API microbial identification kits, bioMérieux, Durham, NC.
b.
Microbial identification systems, Biolog, Hayward, CA.
c.
Matrix assisted laser desorption ionization time-of-flight mass spectrometry, Bruker, Billerica, MA.
d.
BD Biosciences, Sparks, MD.
e.
Sensititre system, Thermo Fisher Scientific, Waltham, MA.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Michael Sweeney and Jeffrey Watts are employed by Zoetis.
Funding
The author(s) disclosed receipt of the following financial support for the research authorship, and/or publication of this article: This study was funded by Zoetis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
