Abstract
In 2006 a third echinocandin, anidulafungin, was approved in the USA for the treatment of candida esophagitis, candidemia, and invasive candida infections such as intra-abdominal abscesses and peritonitis in the non-neutropenic patient. Two years later it was approved in the EU for invasive candidiasis in non-neutropenic patients. Like other echinocandins, it is fungicidal against Candida species and fungistatic against Aspergillus species. It does not need adjustment for renal or hepatic insufficiency, and has no known drug interactions. Its administration is by the intravenous route only, and it is well tolerated. A steady state concentration can be achieved on day two by giving twice the maintenance dose on day one.
Keywords
Introduction
Invasive fungal infections have been on the rise throughout the world and are associated with significant morbidity and mortality. In the United States during the period of 1980–1997, there was an increase in invasive fungal infections listed as the cause of death were the majority of these infections were due to Candida and Aspergillus species. 1 From March 1995 through September 2002, 24,179 cases of nosocomial blood stream infections (BSI) from 49 US hospitals were analyzed in the SCOPE (Surveillance and Control of Pathogens of Epidemiological Importance) study. One-half of these infections occurred in the intensive care unit where prolonged stays are the most important risk factor for invasive candidiasis. Candida species were the fourth most common blood stream infection at 4.8 BSI per 10,000 admissions. This was an increase from 8% in 1995 to 12% in 2002. 2 More than 90% of cases of invasive candidiasis are caused by C. albicans, C. glabrata, C. parapsilosis, C. tropicalis, and C. krusei. Although C. albicans remains the number one cause, the number of cases decreased 10% in the ARTEMIS DISK Surveillance Program from 1997–2003. 3 In a retrospective cohort analysis of patients with candidemia, high Acute Physiology and Chronic Health Evaluation II (APACHE II) scores, prior antibiotics, and administration of antifungal treatment 12 hours after the first positive blood culture were independent determinants of hospital mortality. 4
During a five year period of a Canadian population-based surveillance of 207 patients with Candida cerebral spinal fluid and bloodstream infections, the use of appropriate empirical therapy was independently associated with a reduced risk of death [OR 0.46 (95% confidence interval 0.22–1.000); P = 0.05]. 5 The introduction of the echinocandins, with their favorable safety profile, broad spectrum of activity against Candida species which include those that are polyene- and azole-resistant, and once daily dosing have made this class of drugs a first choice in treating many candida infections as outlined in the most recent guidelines published by the Infectious Diseases Society of America in March 2009. 6
Chemistry
In 1974 a metabolite of Aspergillus nidulans var echinulatus was isolated and named echinocandin b. This discovery led to the polypeptide antimicrobial known as anidulafungin. 7 Echinocandins are amphiphilic cyclic hexapeptides with an N-linked acyl lipid side chain. This side chain is believed to intercalate with the cell membrane's phospholipid bilayer. Each echinocandin has a unique side chain. Caspofungin's is an aliphatic, micafungin's is a complex aromatic, and anidulafungin's is a terphenyl (alkoxytriphenyl). 8 The initial formulation of anidulafungin required an ethanol diluent, but the newer formulation uses water as the diluent.8,9
Anidulafungin is 1-[(4R,5R)-4,5-dihydroxy-N 2 -[[4″-(pentyloxy)[1,1′:4′,1″-terphenyl]-4-yl] carbonyl]-L-ornithine]echinocandin B whose empirical formula is C58H73N7O17. Its molecular weight is 1140.3 Daltons. 9 The molecular structure of anidulafungin can be seen in Figure 1.
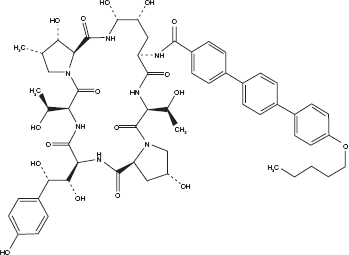
Molecular structure of anidulafungin. 9
Mechanism of Action
Anidulafungin, like the other echinocandins, is a noncompetitive inhibitor of 1,3-β-D-glucan synthase which catalyzes the synthesis of the major glucan component of the fungal cell wall. This enzyme complex is composed of the catalytic subunit and the presumed binding site of the echinocandins, Fksp, and a regulator protein Rho1. By blocking this enzyme complex there is a disruption of the formation of glucan polymers which are a component of the cell wall. This causes osmotic instability and the eventual death of susceptible cells. 8 Human cells do not contain this enzyme complex and are therefore not affected by this mechanism. Additionally, this mechanism of action differs from the antifungal activity of other agents that act at the cell membrane or inhibit DNA synthesis. Therefore, this unique mechanism of action should prevent cross-resistance with other agents that are not in this class. 10
Mechanism of Resistance
Although reported, echinocandin resistance is not common. Decreased susceptibility has been associated with mutations in two highly conserved regions (“hot spots”) of the FKS genes. 11 A naturally occurring change of proline-to-alanine in the highly conserved hot spot1 region of Fks1p in C. parapsilosis, C. orthopsilosis, and C. metapsilosis is responsible for the decreased in vitro susceptibility to the echinocandins. 12
The first reported case of multiechinocandin resistant Candida parapsilosis developed in a patient being treated for prosthetic valve endocarditis. The initial isolate had minimum inhibitory concentrations (MIC's) of 1 μg/mL for anidulafungin, 2 μg/ mL for caspofungin, and 8 μg/mL for micafungin. Higher MIC's developed as treatment progressed where micafungin and caspofungin increased to >16 μg/mL but anidulafungin increased to only 2 μg/mL. 13
In an HIV-infected patient being treated with micafungin for esophagitis caused by azolerefractory C. albicans, initial susceptibility testing showed MIC's of 0.03 μg/mL for anidulafungin and micafungin and 0.06 μg/mL for caspofungin. At weeks 6 and 36 the MIC's increased to 2 μg/ml for micafungin and caspofungin and 1 μg/mL for anidulafungin. Mutations were acquired in FKS1 14 weeks prior to clinical failure. 14 More recent research suggests that other genes are involved in echinocandin resistance. One gene, RER1 (regulator of echinocandin resistance), encodes a fungal-specific Zn(2)Cys(6) transcription factor. When this gene is overexpressed it confers resistance to echinocandins but not to other antifungal agents. This overexpression does not result in upregulation of FKS1 or efflux pumps. 15
Susceptibility testing of isolates of C. parapsilosis obtained from patients in a burn unit showed increased MIC90 values for caspofungin and micafungin at 8 μg/mL and 16 μg/mL, respectively, while anidulafungin was 2 μg/mL. No mutations in the FKS1 gene were found that could explain the differences in susceptibilities between the echinocandins. 16
Paradoxical growth of some Candida isolates has been demonstrated at concentrations above the MIC for echinocandins. This phenomenon is similar to the Eagle effect seen for other agents, such as the penicillins, that are active at the level of the cell wall. In order to assess this paradoxical activity, bloodstream isolates from cancer patients which included C. albicans, C. parapsilosis, C. krusei, C. tropicalis, C. glabrata were studied. Paradoxical growth was more frequent with caspofungin than with the other echinocandins and was unrelated to the MIC. It was absent in the C. glabrata isolates. 17 Further research is needed to understand the differences in the echinocandins which are not only specific to the drugs themselves but also how they interact with different Candida species.
In Vitro studies
Anidulafungin, like other echinocandins, is active against Candida and Aspergillus species but not Cryptococcus neoformans, Glomeromycetes, Fusarium or Trichosporon species. 8 It has demonstrated in vitro activity against molds such as Bipolaris spicifera, Exophiala jeanselmei, Madurella mycetomatis, Paecilomyces spp., Penicillium marneffei, Pseudallescheria boydii, Sporothrix schenckii, and Wangiella dermatitidis. 18
From 2001 to 2006 medical centers worldwide provided 5346 candida isolates from patients that were used to test the in vitro activity of all three echinocandins. Using a standardized broth microdilution method endorsed by the Clinical and Laboratory Institute (CLSI) Antifungal Subcommittee, 98%–100% of all isolates were susceptible to all echinocandins at an MIC ≤ 2 μg/mL. This resulted in the ‘susceptible only’ breakpoint for the echinocandins. Resistance could not be clearly defined because there were few isolates with higher MIC's. 19
Infections associated with indwelling devices are often very difficult to cure because many antimicrobials do not penetrate the micro organism biofilms that reside on these devices and removal is often necessary. Anidulafungin was tested against 30 isolates of C. albicans obtained from sterile-site infections. The planktonic MIC50 and MIC90 were ≤0.03 and 0.125 μg/mL (MIC range ≤0.03 and 2 μg/mL), respectively, and the sessile MIC50 and MIC90 were ≤0.03 and ≤0.03 μg/mL, (MIC range ≤0.03 to >16 μg/mL), respectively. These results indicate that anidulafungin is active against Candida biofilms. 20 Similar results were seen when other antifungals were tested against C. albicans and C. parapsilosis biofilms as well as their corresponding planktonic cells. Notably, the planktonic MIC's of all Candida isolates were susceptible to voriconazole, posaconazole, caspofungin, and anidulafungin, but the azoles had elevated MIC's against Candida isolates from the biofilms, unlike the echinocandins which retained their activity against both the planktonic and sessile forms. 21
Animal Studies
A model using neutropenic rabbits with disseminated candidiasis was used to evaluate the pharmacokinetics, safety, and efficacy of anidulafungin. At the end of the study histopathology was done to assess the clearance of candida from the tissues. A dose-dependent clearance was seen in the liver, spleen, kidney, lung, vena cava, and brain. 22
In another study of immunosuppressed rabbits with esophagitis caused by fluconazole-resistant Candida albicans treated with anidulafungin, dose-dependent clearance of the organism from the tongue, oropharynx, esophagus, stomach, and duodenum was observed. The concentration of drug in the esophagus was dose-dependent and surpassed the MIC for all doses. 23
A neutropenic mouse model was used to study the pharmacodynamics of anidulafungin in disseminated candidiasis where a post-antifungal effect was observed in infected tissues over at least 96 hours. 24 In another disseminated candidiasis murine model the in vivo activity of anidulafungin and treatment success was associated with the ratio of the maximum serum drug concentration to the MIC (Cmax/MIC) and the area under the concentration curve (AUC) to the MIC (AUC/MIC). 25
Penetration into the CSF is poor, hence there is no current role for the echinocandins in meningitis. A murine model was developed to evaluate the activity of anidulafungin against Candida central nervous system infection. Mortality and fungal burden in brain tissue were reduced suggesting that anidulafungin may have a role in brain abscesses caused by Candida. 26 Further research is needed in this area.
Pharmacokinetics and Pharmacodynamics
Anidulafungin was initially developed as an oral preparation, but it was found to have poor oral bioavailability. When rights to the compound were purchased by Versicor of King of Prussia, PA in 1999, an intravenous preparation was developed. 27 It has been studied in healthy individuals and patients with invasive fungal infections. In a phase I study of healthy volunteers, linear pharmacokinetics with dose-independent plasma clearance and dose-proportional increases in the area under the curve (AUC) were demonstrated using single doses of 35, 50, 70, and 100 mg intravenously. 28 A steady state concentration can be achieved on day two by administering double the maintenance dose on day one. 29 It's half-life of elimination is approximately 24 hours which is the longest of the currently marketed echinocandins, and its protein binding is 99%.9,29 With a volume of distribution of 30–50 L, which corresponds to total body water, it is well-distributed into body tissues. 29 Tissue concentrations can reach or exceed concentrations in the plasma. 30 Penetration into cerebral spinal fluid is minimal due to its large molecular weight and high protein binding. 31
Anidulafungin does not need adjustment in those with renal or hepatic insufficiency as it is neither renally cleared nor hepatically metabolized. It is unique in that it is chemically degraded to an open-ring product in the blood which is then further degraded to inactive products by nonspecific peptidases. 32 This is in contrast to caspofungin and micafungin which undergo some hepatic metabolism in addition to degradation.33,34 More than 90% of the dose of anidulafungin is eliminated in the stool of healthy volunteers aged 16–65 years of age as degradation products and the remaining 10% is excreted as intact drug.28,35 Anidulafungin does not act as a substrate, inducer or inhibitor of the cytochrome P450 system and therefore, there is a lack of interaction with such drugs as rifampin, tacrolimus or voriconazole.28,35,36 Caspofungin, on the other hand, needs a dose adjustment when given with inducers of the P450 system such as rifampin, phenytoin, dexamethasone, or efavirenz. 37
Data from four clinical Phase II/III trials was used to analyze the pharmacokinetics in subjects with fungal infections. A two-compartment model with first order kinetics best fit the data. The pharmacokinetics in this analysis were similar to those of healthy volunteers who received anidulafungin at similar doses. Although the clearance of anidulafungin was affected by gender, body weight, and being in the invasive candidiasis study (older subjects who were more acutely ill), these covariates were felt to have little clinical relevance as they accounted for less than 20% of the intersubject variability of anidulafungin's clearance. 29 Table 1 is a summary of anidulafungin's pharmacokinetic properties.
Pharmacokinetic properties of anidulafungin. 9
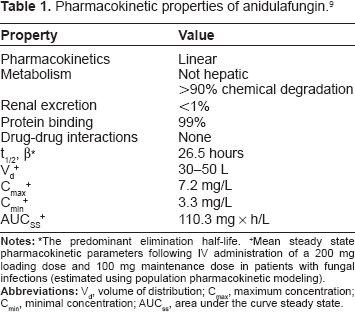
+Mean steady state pharmacokinetic parameters following IV administration of a 200 mg loading dose and 100 mg maintenance dose in patients with fungal infections (estimated using population pharmacokinetic modeling).
A pediatric study to determine the safety, tolerability and pharmacokinetics in neutropenic children ages 2–12 years at doses of 0.75 or 1.5 mg/kg/day showed similar pharmacokinetics to adults who received 50 or 100 mg/day, respectively. The clearance and volume of distribution at steady state were affected by body weight but not by age. The half-life was slightly less than that seen in adults (20 vs 26.5 hours) but still supported once daily dosing. 38
Drug Interactions
The lack of drug interactions exhibited by anidulafungin is probably related to its unique mechanism of elimination. Its lack of hepatic metabolism results in it not interacting with substrates, inducers or inhibitors of the cytochrome P450 system.32,35 Pharmacokinetic parameters were similar when coadministered with voriconazole or liposomal amphotericin B.39,40 Mild elevations in transaminases occurred in subjects enrolled in a Phase I clinical trial of anidulafungin coadminstered with cyclosporine, but these abnormalities were not felt to be clinically relevant. 41 No disulfiram reactions have been reported when the alcohol containing preparation of anidulafungin was administered to subjects receiving metronidazole.42,43
Clinical Efficacy
Candida esophagitis
The efficacy and safety of anidulafungin were compared to oral fluconazole in subjects with endoscopically and microbiologically proven esophageal candidiasis. This multicenter randomized, double-blind, double-dummy noninferiority study enrolled 601 who were randomized to receive intravenous anidulafungin (loading dose of 100 mg on day 1 followed by 50 mg daily) and a daily oral placebo or an intravenous placebo and oral fluconazole (200 mg loading dose on day 1 followed by 100 mg daily). The duration of treatment was 7 days beyond resolution of symptoms where the total duration was 14–21 days. Clinical, microbiologic, and endoscopic evaluations were done at baseline, at the end of therapy (EOT), and at a follow-up (FU) visit two weeks after the EOT. Seventy-percent of the subjects had AIDS but only ten were on antiretroviral therapy (three in the anidulafungin arm and seven in the fluconazole arm). During antifungal treatment more subjects in the fluconazole arm started antiretroviral therapy (58 versus 26 subjects). Prior antifungal use was predominately nystatin. Only nine subjects previously had fluconazole (four in the anidulafungin arm and five in the fluconazole arm). 44
The primary analysis of efficacy was defined as a comparison of endoscopic response at the EOT in evaluable subjects. Within the group of 504 subjects who were evaluable, 242 (97.2%) of the 249 subjects in the anidulafungin arm had endoscopic success defined as cure or improvement compared with 252 (98.8%) of 255 (95% CI of −4.1–0.8) subjects that were in the fluconazole arm. As a result, the pre-defined criteria for noninferiority was met. In an intent-to-treat analysis similar results for endoscopic response at the EOT were found for anidulafungin at 86.7% and fluconazole 88.0% (95% CI, −6.7–3.9). The symptomatic or clinical success rate was high at 97% for anidulafungin and 98% for fluconazole. The mycologic success at the EOT was 86.7% for anidulafungin and 90.9% for fluconazole. 44
Endoscopy was performed at the 2-week follow-up on 462 evaluable subjects. Sustained endoscopic response was seen in 150 (64.4%) of the 233 subjects who received anidulafungin versus 205 (89.5%) of 229 subjects who received fluconazole (95% CI, −325% to −17.8%; P < 0.001). Overall more than 70% of the study subjects had AIDS where the recurrence of Candida esophagitis is high. More subjects in the fluconazole arm were on HIV antiretroviral therapy (65 vs. 29). The study drugs were well tolerated. Serious events that possibly could be related to their administration were low in both arms (less than one percent in each arm). Laboratory abnormalities, which were mainly hepatic or hematologic, were similar for both drugs. 44
Azole-refractory mucosal candidiasis
The safety and efficacy of anidulafungin for treatment of azole-refractory mucosal candidiasis was evaluated in a phase 2 open-label noncomparative study. Subjects were eligible if they were 12 years of age or older and continued to have active oropharygeal (OPC) and/or esophageal candidiasis (EC) within one month of completing a 14 day course of fluconazole or voriconazole. Those with OPC were enrolled if a clinical diagnosis was confirmed by culture and/or microscopy. Those with EC were enrolled if grade I or higher plaques and/or friable mucosa were documented endoscopically. Subjects who met the criteria for azole-refractory mucosal candidiasis received a loading dose of anidulafungin at 100 mg on day 1 followed by a maintenance dose of 50 mg daily for a maximum of 21 days. The primary efficacy end point was clinical response for OPC and endoscopic and clinical response for EC. These end points were assessed at the EOT in the modified-intent-to-treat (MITT) population which was defined as all subjects in the intent-to-treat population who had Candida growing in cultures at baseline. Clinical response at follow-up (10–14 days after the EOT or earlier if another antifungal was started because of failure or if there was a relapse) and microbiologic responses at the EOT and follow-up were the designated secondary end-points. 45
A total of 19 subjects received study drug and 18 remained in the study. Of these, 17 had HIV where the median CD4 was 9 cells/mm3 and the majority had severe disease. At the EOT, clinical success was observed in 18/19 subjects. In those with OPC the success was 94% (17/18) of subjects and in 92% (11/12) of those with EC. There was only one treatment failure in a patient who previously failed oral amphotericin B and itraconazole and who eventually required GM-CSF and intravenous amphotericin B to control his disease. At the follow-up visit, clinical success was achieved in 14 (47%) subjects (eight with OPC and six with EC). The microbiological success was 37% at the EOT and 32% at the follow-up visit. Anidulafungin was well tolerated with the most common side effect being nausea and/or vomiting in 4/19 subjects (21%). Two subjects experienced hypokalemia, one experienced mild flushing during infusion, and one had a maculopapular rash that resolved with discontinuation of anidulafungin. 45
Candidemia and other forms of invasive candidiasis
In a phase 2, randomized, dose-ranging study the efficacy and safety of anidulafungin was evaluated in subjects with candidemia and other forms of invasive candidiasis. Subjects were eligible if they were 18 years of age or older, expected to survive for more than 72 hours, and had invasive candidiasis defined as positive blood or tissue culture and had the presence of a least one symptom or sign of infection within 4 days of starting treatment. A total of 123 subjects were randomized to 50 mg, 75 mg or 100 mg daily. The primary measure of efficacy was global response (both clinical and microbiological) at a follow-up visit 14 days after the EOT in evaluable subjects. A secondary efficacy analysis included global response at EOT. 46
Of the 120 subjects who received at least one dose of study drug, and were thus included in the intent-to-treat (ITT) population, similar baseline characteristics were seen among the three different dosage groups except those in the 75 mg and 100 mg groups had slightly higher APACHE II scores. APACHE II scores of ≥20 were seen in approximately 25% of the study participants. Sixty-eight (55%) completed the study and there were 33 deaths (fourteen deaths occurred in those while on the study drug and the most common causes of death were reported as multiorgan failure, cardiac arrest, and nonfungal sepsis). There were 116 subjects in the MITT group; 83 were evaluable at the EOT and 68 at follow-up two weeks after the EOT. 46
Fifty-three percent of subjects were infected with C. albicans, 31% with C. glabrata, 9% with C. parapsilosis, 9% with C. tropicalis, 4% with C. krusei, and 3% with other species. The majority (93%) had candidemia; 22 of the 47 subjects with an intravascular catheter when infection was identified had the catheter changed or removed prior to receiving anidulafungin. 46
At the EOT the global response for the 50 mg, 75 mg, and 100 mg arms was 84%, 90%, and 89%, respectively. The global response at follow-up two weeks after the EOT, the primary efficacy endpoint, was 72%, 85%, and 83% in the 50 mg, 75 mg, and 100 mg groups, respectively. The rates for microbiological success at the EOT and FU were also higher at the higher doses and hence, the 100 mg dose following a 200 mg loading dose was chosen in subsequent studies. Approximately 5% of subjects in each dose arm had adverse events that were thought to be related to the study drug with the exception of hypokalemia that occurred in four (10%) of subjects in the 50 mg arm. There were no infusion-related events, anaphylactic reactions, or dose-related relationships between serum chemistries or hematologic values. 46
In a randomized double-blind, phase 3 trial anidulafungin was compared to fluconazole for the treatment of candidemia and other forms of invasive candidiasis. Subjects 16 years of age or older with candidemia or a culture positive for Candida species from a sterile site were randomized to either anidulafungin (200 mg on day 1 followed by a maintenance dose of 100 mg daily) or fluconazole (800 mg on day 1 followed by 400 mg daily. The fluconazole dose was adjusted for those with a creatinine clearance of less than 50 mL/minute and those on hemodialysis). Subjects were stratified according to APACHE score and absolute neutrophil count. Subjects with endocarditis, osteomyelitis, and meningitis were excluded. The drugs were administered for at least 14 days beyond the first documented negative blood culture and improvement in signs and symptoms of infection. After 10 days of intravenous study drug, and if afebrile for at least 24 hours with clinical improvement and negative blood cultures, all subjects could be transitioned to oral fluconazole at the discretion of the investigator. Although the trial was designed to show noninferiority of anidulafungin, the predefined statistical analysis was a two-step process. If noninferiority was not met in the first step, in the second step, if the lower limit of the 95% confidence interval was greater than 0, then anidulafungin was considered superior to fluconazole. 47
Two hundred and sixty-one subjects were enrolled; 256 who received at least one dose of study drug were included in the intent-to-treat (ITT) group and 245 were in the MITT group where they received at least one dose of study drug and grew a Candida species from culture within 96 hours of being enrolled. Most of the subjects had candidemia only. After BSI, the second most frequent site of infection was intra-abdominal. The primary efficacy analysis of global response, which was defined as both clinical (resolution of signs and symptoms of candida infection) and microbiologic (negative repeat culture or presumed eradication of infection if no culture available) success was assessed at the end of intravenous therapy in the MITT population. The global response at the end of all therapy and at follow up visits at weeks 2 and 6 was also assessed. Both study groups had similar demographics, Candida species isolated, prior treatment with oral fluconazole, duration of study drug, and switch to oral fluconazole. 47
At the end of intravenous therapy in the MITT population, a successful global response occurred in 75.5% of subjects who received anidulafungin versus 60% in the fluconazole group (95% CI, 3.9–27) thus demonstrating superiority of anidulafungin over fluconazole. After adjusting for prior azole therapy, diabetes mellitus, catheter removal, immunosuppressive therapy, and infection with C. glabrata inmultivariate logistic-regression mode, only treatment allocation was significant. 47
At the end of intravenous therapy the success rate in those with candidemia only (the majority of subjects at 89%) was 75.6% (88 of 116) in the anidulafungin group and 61.2% (63 of 106) in the fluconazole group (95% CI, 2.5–26.9; P = 0.02). The higher response to anidulafungin was also seen in the subset of those with other forms of invasive candidiasis where the success rate for anidulafungin was 72.7% versus 53.3% in the fluconazole group. At the EOT and at the 2 week follow up anidulafungin was statistically more efficacious than fluconazole. Although there were more treatment successes in the anidulafungin group at the 6 week follow up, the difference was not statistically significant and hence only non-inferiority was met at that endpoint. 47
In subjects who received anidulafungin, the global response was higher for every Candida species (all candida species 77% in those who received anidulafungin versus 61% who received fluconazole, P = 0.01) except C. parapsilosis (64% for anidulafungin versus 83% for fluconazole, P = 0.37) although this was not statistically significant. The success rate was highest in those with C. albicans (81.1% in the anidulafungin group versus 62.3% in those who received fluconazole, P = 0.02), and similar for C. glabrata (56.3% for anidulafungin and 50.0% for fluconazole, P = 0.75). The difference in per-pathogen success rates was not a function of fluconazole resistance, since the majority of the isolates, including those of C. glabrata, were susceptible. Persistent infection at the end of intravenous therapy was documented in eight subjects in the anidulafungin group (6.3%) and 17 in the fluconazole group (14.4%) (P = 0.06). 47
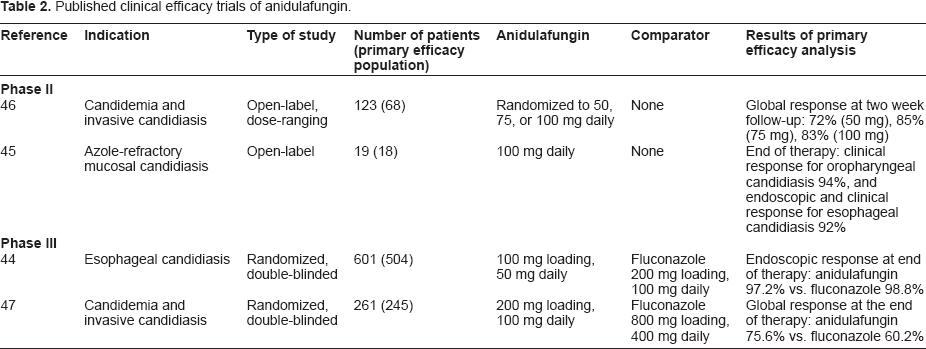
Both drugs were well tolerated and adverse events that were treatment-related were similar between the two groups. Deaths in the MITT were greater in those who received fluconazole (31.4%) than in those who received anidulafungin (22.8%), P = 0.13. 47 Table 2 summarizes the published clinical efficacy trials.
Published clinical efficacy trials of anidulafungin.
Pharmacoeconomic Analysis
The economic impact of anidulafungin was analyzed retrospectively in hospitalized subjects with candidemia and other forms of invasive candidiasis who received this drug or intravenous fluconazole in the previously sited randomized phase 3 trial comparing the two drugs. The total cost-adjudication was $44,781 for anidulafungin versus $42,588 for IV fluconazole (P = 0.70) and the total cost-regression was $47,658 versus $44,977 (P = 0.66), respectively. Thus, the superior response in those treated with anidulafungin did not result in a higher cost when compared with IV fluconazole. 48
Safety and Tolerability
Nausea, vomiting, fever, and hypokalemia have been observed at rates similar to other echinocandins and fluconazole.9,49 In early studies, infusion-related reactions such as urticaria, rash, and hypotension were felt to be histamine related, and slowing the infusion rate to 1.1 mg/mL led to resolution of these reactions and the recommendation not to exceed this rate.9,49,50 In studies with rats, anidulafungin crossed the placenta and was found in the milk of lactating animals.9,49 To date, no studies have been performed in pregnant women. As a result anidulafungin, like the other echinocandins, is considered a category C drug and caution should be used in pregnant and lactating women.
Administration and Dosage
Lyophilized anidulafungin was previously reconstituted in 20% dehydrated alcohol and then further diluted in 0.9% sodium chloride or 5% dextrose. It is now reconstituted with sterile water for injection and further diluted with 5% dextrose or 0.9% sodium chloride. Once it is in solution it can be stored at 2° C–8° C but it should be administered within 24 hours. For candidemia and invasive candidiasis the recommended dose is to load with 200 mg on day one followed by a maintenance dose of 100 mg daily. In general, the duration of therapy is 14 days beyond the first negative culture and clinical improvement. For esophageal candidiasis, it is recommended to load with 100 mg on day one followed by a daily maintenance dose of 50 mg for a minimum of 14 days and at least 7 days following resolution of symptoms. No dose adjustments are required for hepatic or renal insufficiency or hemodialysis.9,49
Summary
As suggested by the most recent guidelines of the Infectious Diseases Society of America, the echinocandins have become the first choice in treating many serious infections caused by Candida species because they have excellent efficacy, they are fungicidal, and they have a very favorable safety profile. Additionally, they have a broad range of activity that includes polyene- and azole-resistant species. Anidulafungin does not need dose adjustment for renal or hepatic insufficiency or for gender, age, or weight. A steady state concentration can be achieved on day two by administering a loading dose of twice the maintenance dose on day one, and it has no known drug interactions. It has demonstrated clinical efficacy in the treatment of oropharyngeal and esophageal candidiasis, although the effect is not durable in severely immunocompromised hosts, as well as candidemia and other forms of invasive candidiasis. Since most clinical trials of anidulafungin and the other echinocandins have studied candidemia and intra-abdominal infections (peritonitis and abscess) almost exclusively, there is a paucity of data for most other forms of invasive candidiasis. The echinocandins demonstrate extensive distribution into tissues such as liver, lung, spleen, and kidney but minimal penetration into urine, CSF, brain, and ocular fluid. They should not be used to treat meningitis or endophthalmitis. With low concentrations achieved in urine, anidulafungin is not expected to eradicate Candida urinary tract infections. However, case reports have been published in which another echinocandin was efficacious in the treatment of complicated and ascending C. glabrata infection, even when that echinocandin also does not achieve high concentrations in urine. There is a significant and increasing number of case reports and case series that document favorable outcomes when echinocandins have been used to treat Candida endocarditis, such that the latest version of the IDSA guidelines acknowledges this new information. Further studies to address the differences among the drugs in this class as well as combining the echinocandins with other classes of antifungals for serious fungal infections should expand our understanding of their clinical use.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Dr. Reboli has been a consultant, investigator, and lecturer for Merck and Pfizer. She has performed clinical trials involving caspofungin and anidulafungin. Dr. de la Torre has been a coinvestigator in clinical studies for Merck and Pfizer.
