Abstract
Immune Thrombocytopenia (ITP) is an autoimmune disorder characterized by autoantibody-mediated destruction of platelets. In general, the goal of therapy is the prevention of bleeding complications. Since the risk of life-threatening bleeding complications (eg, intracranial hemorrhage) is extremely rare, the most optimal treatment must remain individualized on the basis of all clinical conditions related to the affected patient. Based on this finding and on our own experiences the majority of affected patients may not require treatment. If indicated, the accepted standard therapies are corticosteroid-base treatments, intravenous immunoglobulins (IVIG) or anti-D, and in severely affected and unresponsive cases, splenectomy.
Romiplostim is a 60 kDa molecule that is composed of four TPO-mimetic peptides that are attached via glycine bridges to an IgG heavy-chain Fc molecule. It effectively competes with eTPO for binding to TPO-receptor, leading to an increase in platelet counts within five to ten days after weekly subcutaneous injection. Romiplostim has been approved for the treatment of ITP in the U.S. and Europe. The aim of this review is to summarize results the current data from controlled clinical trials on the safety an efficacy of romiplostim in the treatment of ITP.
Introduction
Immune thrombocytopenia (ITP) is defined as an isolated thrombocytopenia (<100 x 10 9 /L) without any evident predisposing cause. It is an autoimmune disorder characterized by the autoantibody-mediated destruction of platelets. In addition, T-cells and an impaired megakaryopoiesis have also been implicated.1,2 The latter process is supported by the successful treatment of ITP with thrombopoietin-mimetic agents. Although the principles of ITP-therapy had recently been reviewed and concised in numerous guidelines,1,3–7 the most optimal treatment must remain individualized on the basis of all clinical conditions related to the affected patient. In general, the goal of therapy is the prevention of bleeding complications. To reach this aim, the duration of the disease and the individual environment of the patient (eg, age, lifestyle, comorbidities, potential interventions or concomitant medications with bleeding risks, tolerability of side effect should be taken into account. The risk of life-threatening bleeding complications (eg, intracranial hemorrhage) is extremely rare, except in cases where the patient has comorbidities with an increased bleeding risk.8–12 Based on this finding and on our own experiences during the last four decades, the majority of affected patients may not require treatment. If indicated, the accepted standard therapies are corticosteroid-base treatments, intravenous immunoglobulins (IVIG) or anti-D, and in severely affected and unresponsive cases, splenectomy. Alternative treatments including azathioprine, mycophenolate mofetil, cyclosporine A, vincristine, cyclophosphamide, dapsone, danazol, or interferon have unique potential toxicities, and the majority of patients may remain unresponsive.
Rituximab is not licensed for the treatment of ITP and approximately one-third of patients achieve a relatively durable response.13,14 Therefore, this drug should only be considered for patients with high bleeding risks who fail to respond to corticosteroids, IVIg, and/or splenectomy. 1
Since 1958, the existence of thrombopoietin (TPO) has been postulated. 15 It was not until 1994 that the molecule was first sequenced and cloned.15–19 During the 1990s, recombinant thrombopoietins were studied in numerous hematology and oncology trials and the stimulatory effects of theses molecules had been demonstrated. 20 However, a benefit of TPO in the treatment of leukemia or during stem cell transplantation could not be demonstrated. The first successful treatment of ITP using recombinant thrombopoietins was first described in two small studies.21,22 However, further studies with these molecules were truncated with the perception that recombinant thrombopoietins may lead to the development of neutralizing antibodies which cross-react with endogenous TPO (eTPO), resulting in profound thrombocytopenia. 23
These results led to the search for non-immunogenic molecules that mimic the effect of TPO. The first described molecules were SB-497115 (Eltrombopag; GlaxoSmithKline, Collegeville, PA, USA) and AMG 531 (Romiplostim; Amgen, Thousand Oaks, CA, USA).24,25 This review focuses on the application of romiplostim in the treatment of ITP.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
To avoid the generation of cross-reactive antibodies to eTPO, a random peptide sequence with TPO-mimetic activity, but no sequence homology to eTPO, was developed. 25 Romiplostim is a 60 kDa molecule that is composed of four TPO-mimetic peptides that are attached via glycine bridges to an IgG heavy-chain Fc molecule. 26 Due to binding of the Fc-part to the FcRn salvage receptor, the molecule undergoes endothelial re-circulation resulting in a half-life of 120-160 h, which is much longer than for the peptide alone. The romiplostim molecule itself undergoes reticuloendothelial elimination, whereas smaller proteins (<20-40 kDa) may be filtered by the glomeruli following tubular readsorption and subsequent elimination. 25 Binding antibodies to romiplostim occur in approximately 5.8% of treated patients. None of the detected antibodies showed cross-reactivity to eTPO. Although these patients demonstrated a trend to lower platelet counts, only a marginal proportion (two of 537 patients tested; 0.4%) developed neutralizing antibodies. Interestingly, pre-existing antibodies to romiplostim were detectable in approximately 8% and to eTPO, in 5.4% of the patients.27,28
Romiplostim effectively competes with eTPO for binding to normal platelets, leading to a rapid tyrosine phosphorylation of the TPO-receptor, JAK2 and STAT5. 29 In healthy volunteers, a single intravenous or subcutaneous administration of romiplostim resulted in a dose-dependent increase in platelet counts which did not occur prior to day five following administration. Platelet counts reached maximum levels between day 12 and 16 and decreased to baseline levels by day 28. In a single intravenous dose range of 0.3-10 µg/kg, the pharmacokinetics of romiplostim is nonlinear with the dose. 25 Romiplostim is available as a lyophilized powder and is to be stored at 2 °C-8 °C. A small volume of 0.72 mL sterile water is required for its reconstitution to 250 µg and a 1 µg/kg BW subcutaneous dose for a patient with 80 kg body-weight equates to an injection volume of no more than 0.14 mL. The reconstituted drug remains stable for up to 24 h. The recommended individual weekly dose is between 1-10 µg/kg BW.
Efficacy
Efficacy in adult patients with ITP
We evaluated the results of six randomized controlled trials with romiplostim in patients with chronic ITP (see Table 1). In a phase I study (ClinicalTrials.gov number NCT00111475; phase I), 24 patients with ITP who had received a least one prior treatment for ITP were assigned to cohorts of four patients with escalating doses of romiplostim or the placebo. 30 The primary objective of this study was to assess the safety and tolerability of two injections of romiplostim. Secondary objectives wore to determine the dose that would result in a platelet count within 50,000-450,000/µL. Patients from each cohort received two identical weekly doses of romiplostim (0.2-10.0 µg/kg). Patients that received 0.2, 0.5, and 1.0 µg/kg romiplostim did not reach the target platelet count (50,000-450,000 x 10 3 /µL and twice the baseline count, respectively), whereas platelet counts in seven of 12 patients who received 3.0, 6.0, and 10.0 µg/kg increased to at least 50,000 x 10 3 /µL. In three of these patients, the platelet count exceeded 450,000 x 10 3 /µL. Four of 12 patients achieved a platelet count that was within the target range. The median response time ranged from five-eight days, and the peak platelet count following a single injection of romiplostim was found to be dose-dependent (163,000 x 103/µL, 309,000 x 10 3 /µL, and 746,000 x 10 3 /µL for the 3.0, 6.0, and 10.0 µg/kg dose, respectively).
Basic characteristics of randomized trials with romiplostim in ITP.
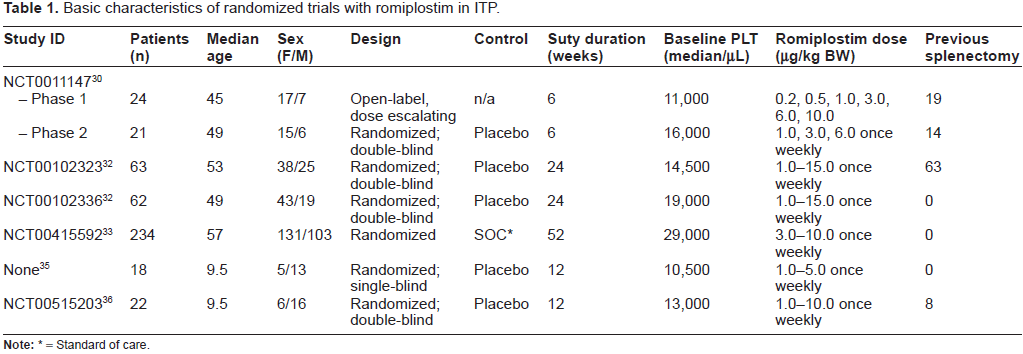
= Standard of care.
This study was followed by a double-blinded, placebo-controlled phase II study (ClinicalTrials.gov number NCT00111475; phase II) that investigated six weekly injections of 1.0, 3.0, and 6.0 µg/kg romiplostim or the placebo in 21 patients with ITP. 30 The objectives were to evaluate the safety of romi-plostim and to determine a weekly dose that would result in a platelet between 50,000 to 450,000/µL.
Four patients were assigned to the placebo group and 17 to the romiplostim groups. The 6.0 µg/kg cohort recruited only one patient, since this cohort was later eliminated by a protocol amendment, as platelets increased to 520,000 x 10 3 /µL in one patient. The target platelet range was achieved in seven of eight patients that received 1.0 µg/kg and in three of eight patients that received 3.0 µg/kg. Two patients receiving 3.0 µg/kg exceeded the target platelet range. Neither the first nor the second phase of this study assessed the incidence and severity of bleeding in relation to romiplostim treatment.
This study was followed by two parallel, double-blinded, placebo-controlled phase III trials that investigated the efficacy of romiplostim in splenectomized (n = 63; ClinicalTrials.gov number NCT00102323) and non-splenectomized patients (n = 62; ClinicalTrials.gov number NCT00102336). The primary endpoint of both studies was to evaluate the efficacy of AMG 531 in the treatment ITP as measured by durable platelet response during the last eight weeks of treatment. The patients received romiplostim for 24 months. One of the primary objectives of these studies was the efficacy of romiplostim to induce a durable platelet count of ≥50 x 10 3 /µL during six or more of the eight weeks of treatment. Twenty-five percent of splenectomized and non-splenectomized patients achieved a platelet count ≥50,000 x 10 3 /µL following one week and 50% of the patients following two to three weeks of treatment. During the last weeks of treatment (week 18-25), platelet counts for patients treated with romiplostim ranged from 56,000 to 85,000 x 10 3 /µL for splenectomized and from 63,000 to 96,000 x 10 3 /µL for non-splenectomized patients. Within the placebo group, platelets ranged from 13,000 to 21,000 x 10 3 /µL for the splenectomized and from 29,000 to 38,000 x 10 3 /µL for the non-splenectomized patients. In both studies, the durable response rate was significantly higher in patients receiving romiplostim than in those receiving the placebo (difference in proportion of splenectomized patients responding was 38% [95% CI 23.4-52.8], P = 0.0013; difference in proportion of non-splenectomized patients responding was 56% [95% CI 38.7-73.7], P < 0.0001). Furthermore, 40% of splenectomized patients and 27% of non-splenectomized patients did not achieve a durable response, but a transient platelet increase of more than four weeks. Altogether, 88% of the non-splenectomized and 79% of the splenectomized patients treated with romiplostim achieved an overall platelet response (either durable or transient), whereas within the placebo group, only 14% of the non-splenectomized and none of the splenectomized patients demonstrated such a platelet response. Thirty-nine patients across both trials (romiplostim group n = 23; placebo group n = 16) received concomitant medication (corticosteroids, azathioprine, danazol). As permitted per protocol, 52% patients treated with romiplostim discontinued within the first 12 trial-weeks in comparison to only 19% of the placebo-treated patients. An ad hoc analysis of the bleeding events of both studies that were not predefined endpoints, revealed that the number of bleeding events per 100 patients was very similar between the placebo and romiplostim group (7.9 and 7.8 events, respectively), with the median number of bleeding events amounting to one for both groups. However, 24.4% of patients in the placebo group developed four or more bleeding episodes compared to 15.5% in the romiplostim group. 31 Bleeding complications of at least grade 2 occurred in a significantly higher percentage of patients in the placebo group (placebo: 14/41, 34%; romiplostim 13/84, 15%; P = 0.018). Overall, more of the placebo-treated patients required additional medication to prevent bleeding complications than the romiplostim-treated patients (59.5% and 21.7%, respectively; P < 0.0001).31,32
Another phase III trial investigated the efficacy of romiplostim to achieve a target platelet count between 50,000 and 200,000 x 10 3 /µL in comparison to standard of care (SOC) in non-splenectomized ITP patients (ClinicalTrials.gov number NCT00415532). 33 The primary endpoint was the number of subjects undergoing a splenectomy by treatment group and the number of subjects with a treatment failure during the 52-week treatment period. A total of 234 patients were enrolled; 157 patients received weekly injections of romiplostim (starting dose of 3 µg/kg) and 77 were treated by SOC, which was selected by the investigators on the basis of standard institutional practices or therapeutic guidelines. During the 52-week study period, participation was discontinued by 12 patients in the romiplostim and 15 patients in the SOC group. In the romiplostim group, the number of patients with a platelet count 50,000 x 10 3 /µL ranged from 71% to 92%, and from 26% to 51% in the placebo group. The incidence of treatment failures and of splenectomy was significantly lower amongst patients in the romiplostim group (11% and 9%, respectively) than patients in the SOC group (30% and 36%, respectively). The overall rate of bleeding events was widely identical for both the SOC and romiplostim group (53% and 52%, respectively). However, bleeding events of greater or equal to a severity grade of 3 appeared to be more frequent in the SOC group (7%) than in the romiplostim group (3%).
The results of the aforementioned studies indicate that treatment of ITP with romiplostim has the ability to effectively increase platelet counts and reduce bleeding complications in the majority of patients with ITP. However, it appears that this effect is less pronounced in splenectomized patients. This phenomenon may be related to a deterioration in the course of the disease in patients that underwent splenectomy in comparison to non-splenectomized patients. Compared to placebo or SOC, romiplostim therapy led to a reduction of concurrent medication and splenectomy rate, and prevented bleeding complications. Analysis of a long-term extension study demonstrated a reduction of bleeding complication in patients that were treated for more than 24 weeks. 31 However, a recent meta-analysis concluded that treatment of ITP with the TPO-mimetic agents romiplostim and eltrombopag did not significantly improve the risk of bleeding complications. 34
Efficacy in children
Two randomized placebo controlled trials investigated the safety and efficacy of romiplostim in children with ITP. The first trial enrolled 18 ITP patients that were non-responsive to a prior treatment or that failed to maintain response. The patients received romiplostim for 12 weeks. 35 The second trial comprised of 22 patients with chronic ITP who also received romiplostim for a 12 week period. Previous therapy (successful or failed) was not required for study entry (ClinicalTrials.gov number NCT00515203). 36 The primary endpoint of this study was the Occurrence of one or more adverse events in the participant during the 12-week treatment period.
The results of both studies revealed that romi-plostim effectively elevates the platelet counts and prevent bleeding complications in children with ITP.
Safety
Adverse events
Adverse events which have been reported in the aforementioned trials were rated in almost all cases as mild to moderate, and included headache, fatigue, minor bleeding events (petechiae, epistaxis, gingi-val bleeding), arthralgia, myalgia, back pain, nausea and dizziness.30,32,33 The frequencies of these adverse events were widely similar between the placebo/SOC and romiplostim groups. These findings were confirmed by data of an open-label extension study that enrolled 292 adult ITP patients that received romi-plostim for an average of 78 weeks (1-277 weeks). 37
A potential risk is an abrupt loss of response to romiplostim. It has been demonstrated that TPO-mimetic agents such as romiplostim or eltrombopag do not sufficiently prevent adverse platelet fluctuations and subsequent bleeding complications during infections. 38 Furthermore, an uncontrolled discontinuation of romiplostim should be avoided, since a rebound exacerbation of thrombocytopenia including a therapy requiring bleeding tendency may occur.26,30
A further potential complication of romiplostim treatment is the occurrence of thromboembolic events. During the described controlled phase I/II and phase III trials, thromboembolic events occurred in the romiplostim (n = 2) as well as in the placebo groups (n = 2). An accumulation of thromboembolic events in the romiplostim groups was not observed.26,30 During the open label long-term extension study, eight thromboembolic events occurred in four patients. All patients who experienced thromboembolic events during the phase III and open-label extension studies had pre-existing risk factors for cardiovascular events or thrombosis. 31 In addition, ITP patients may be at an increased risk of thromboembolic events.39–41
Treatment with romiplostim resulted in similar frequencies of serious adverse events of grade 3 or higher (grade 3 vertigo, asthma, subdural hemorrhage, rebound worsening of thrombocytopenia with bleeding complications after discontinuation of romiplostim, deep vein thrombosis, arterial thromboembolism, pulmonary embolism, cerebrovascular accident, reversible increase in reticulin deposition) compared to the placebo group.30,32,34
During thephase III and long-term extension studies, bone marrow reticulin deposition has been reported in a minority of patients treated with romiplostim.27,30,32,33 No bone marrow reticulin deposition was reported in children treated with romiplostim.35,36 Reticulin is a normal bone marrow component and detectable in about 5% (grade 2, Bauermeister scale) 42 and 70% (grade N to 1, Bauermeister scale) of bone marrow samples from healthy subjects.42–44 Normal samples do not demonstrate grade 3 and 4 reticulin deposition by the Bauermeister scale. A similar pattern of reticulin staining has been reported in a retrospective analysis of patients with ITP. 45
The presence of bone marrow reticulin in ITP patients treated with ITP was associated with prior splenectomy, exposure to prior multiple ITP treatment courses, a relatively high dose of romiplostim, and a minimal platelet response. 27 Although there was no evidence that suggested a progression to a clonal disorder and the reticulin deposition tended to improve following discontinuation of romiplostim, its real frequency, clinical significance, and reversibility remain speculative. Recent analysis of 25 ITP patients treated with TPO-mimetics (romiplostim, n =15; eltrom-bopag, n = 9; AKR 501 [EE501], n = 1) revealed a statistically relevant increase in bone marrow reticulin deposition, but no cytogenetic or morphologic abnormalities were detected. In addition, a decreased Procollagen III N-propeptide was observed, which may suggest a decreased reticulin production and an elevated hepatocyte growth factor. 46 These novel findings require further investigation. Two ongoing clinical trials are currently investigating the clinical relevance of bone marrow transformation in ITP patients undergoing treatment with TPO mimetic agents (romiplostim, NCT00907478; eltrombopag, NCT01098487). However, intermediate results are not yet available.
Adverse events in children
Most adverse events in children were graded as mild to moderate. The most common reported adverse events listed were headache, epistaxis, cough and vomiting. The overall incidence of adverse events was comparable within the romiplostim and placebo groups. Severe adverse events (adverse events of grade 3 or higher) were only reported in two patients. One patient received romiplostim and the other the placebo. Neither event was considered to be related to the course of treatment. 36 Thromboembolic events neither occurred in the romiplostim nor in the placebo groups.
Quality of Life
In addition to the limitations related to bleeding risks, ITP patients may suffer from fatigue.47,48 Furthermore, the health-related quality of life (HRQoL) may not only be reduced due to the symptoms associated with disease, but also by its treatment procedures. Therefore, it could be hypothesized that effective treatment of ITP may improve the symptoms of fatigue and the HRQoL as well as preventing bleeding complications. 49
Two studies used an ITP patient assessment questionnaire (ITP-PAQ), which comprised of 50 questions, to investigate the HRQoL in adult ITP patients treated with romiplostim.49,50 Both studies revealed that effective treatment of ITP with romiplostim led to a significant improvement in HRQoL. Furthermore, Kuter and colleagues described significantly greater improvements in HRQoL in patients treated with romiplostim in comparison to SOC treatment. 33 However, a subgroup-analysis of such patients with a maintained response to SOC compared to romiplostim was not performed.
Another study investigated the HRQoL in children with ITP that were treated with romiplostim. 51 The questionnaire in this study consisted of a child self-report component, a parent-impact component and a proxy-impact component. While the children of the placebo group had a significant decrease from the baseline in HRQoL, the romiplostim group demonstrated an increase, which was not significant. However, romiplostim significantly reduced the parental burden.
Altogether, treatment of ITP with romiplostim may not only result in a reduction in bleeding complications, which is highly associated with an increase in platelet counts, but also in an improvement in the HRQoL. However, further studies are necessary to compare the quality of life of ITP patients with a comparable maintained response to romiplostim and SOC.
Patients are required to be trained in the preparation and injection of the drug. Otherwise, weekly consultations are inevitable. These treatment modalities, storage recommendations and the potential side effect of the subcutaneous injection (eg, hematoma, skin irritation, pain) may have an impact on the quality of life and on the patients’ preference.
Conclusions
Romiplostim has been licensed for the treatment of chronic ITP of adult patients in North America, Europe, and Australia. However, current guidelines still recommend treatments that focus on decreasing the destruction of platelets as first-line therapy (eg, Corticosteroids, IVIg, or Anti-D),1,6 followed by splenectomy in unresponsive cases. Although it has been demonstrated that early treatment with romiplostim may avoid the necessity of splenectomy, 33 splenectomy provides a durable remission of ITP in more than 60% of treated patients.14,52
TPO mimetic agents like romiplostim may in fact reflect a major improvement in the management of severely affected patients with ITP. However, this treatment cannot invariably be justified, and the effects of long-term treatment remain unclear. Ultimately, the effect of this treatment is largely based on the uncontrolled stimulation of platelet production.
Further long-term data on the safety profile (risk of thromboembolic events, relevance, clinical significance of bone marrow reticulin, and induction of malignancies), overall survival data, 34 and superiority compared to SOC are needed to finally rank the drug for the treatment of ITP.
Author Contributions
Conceived and designed the experiments: OM, AS. Analysed the data: OM. Wrote the first draft of the manuscript: AS. Contributed to the writing of the manuscript: OM, AS. Agree with manuscript results and conclusions: OM, AS. Jointly developed the structure and arguments for the paper: AS. Made critical revisions and approved final version: AS. All authors reviewed and approved of the final manuscript.
Competing Interests
OM has received a consulting fee or honorarium from Amgen and sits on its Advisory Board, and is a member of the GSK Advisory Board and has also received payment for lectures including service on speakers bureaus from GSK. AS has received consulting fees or honorarium from Amgen and sites on its Advisory Board, and is a member of the GSK Advisory Board and has also received payment for lectures including service on speakers bureaus from GSK.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
Disclosures
A.S. and O.M. conduct clinical studies on behalf of Amgen and GlaxoSmithKline and received consultation fees from Amgen and GlaxoSmithKline.
Footnotes
Acknowledgement
We thank Dr. Sundrela Kamhieh-Milz for editing and drafting the manuscript.
