Abstract
The thrombopoietin receptor agonists (TPO-RAs) are a class of platelet growth factors used to treat immune thrombocytopenia (ITP) in children and adults. Romiplostim is a peptide TPO-RA approved for over a decade to treat adults with ITP but was just recently US Food and Drug Administration approved to manage ITP in children 1 year of age and older who have had an inadequate response to corticosteroids, intravenous immunoglobulin, or splenectomy. Like the small molecule TPO-RA eltrombopag, romiplostim offers a high clinical response rate in pediatric patients with ITP, but requires use over an extended, and possibly indefinite, duration. This review is a critical appraisal of the role of romiplostim in pediatric ITP, discussing the safety and efficacy of this agent in clinical trials of children and adults and defining the patients most likely to benefit from romiplostim treatment. The treating hematologist is additionally provided guidance with treatment goals, dosing strategies, toxicity management, and indications for discontinuation.
Keywords
Introduction
Thrombopoietin (TPO) is the major hematopoietic growth factor responsible for the production of platelets. 1 Initial studies with first-generation recombinant thrombopoietic agents [pegylated recombinant human megakaryocyte growth and development factor (PEG-rHuMGDF) and recombinant human TPO (rhTPO)] were halted in the West due to the development of neutralizing antibodies against PEG-rHuMGDF that were capable of cross-reacting with and neutralizing endogenous human TPO. This led to the development of second-generation thrombopoietic agents, the TPO receptor agonists (TPO-RAs), that had improved function and lacked antigenicity that might result in similar anti-TPO antibody formation. TPO-RAs mimic endogenous TPO, promoting growth and development of megakaryocyte precursors and megakaryocytes, thereby increasing the platelet count. 2 There are currently four TPO-RAs available in the United States: the peptibody romiplostim (Nplate, Amgen, Thousand Oaks, CA) and the small molecule agents eltrombopag (Promacta, Novartis, Basel, Switzerland), avatrombopag (Doptelet, Dova, Durham, NC), and lusutrombopag (Mulpleta, Shionogi, Florham Park, NJ). In addition to management of immune thrombocytopenia (ITP) in adults and children, the TPO-RAs have been approved or are under investigation in numerous other thrombocytopenic disorders, including perioperative thrombocytopenia,3–5 chemotherapy-induced thrombocytopenia,6,7 severe aplastic anemia, 8 and support of antiviral treatment in hepatitis C-associated thrombocytopenia. 3 Romiplostim and eltrombopag were initially developed for treatment of chronic ITP, for which they have been approved for use in adults for over a decade. In December 2018, romiplostim was approved by the US Food and Drug Administration (FDA) to manage ITP of greater than 6 months’ duration in pediatric patients 1 year of age or older. Its approved use was extended to adults with ITP of any duration following failure of corticosteroids, intravenous immunoglobulin (IVIG), or splenectomy in October 2019.
Use of the TPO-RAs in ITP is supported by the modern understanding of ITP as a disorder of both increased platelet destruction as well as inadequate platelet production. While other ITP therapeutics act to diminish platelet destruction by the reticuloendothelial system, cytolytic T cells, and complement, the TPO-RAs act to reverse the production defect, inhibiting the pro-apoptotic action of glycoprotein-specific platelet autoantibodies and lymphocytes on megakaryocyte precursors and megakaryocytes, thereby promoting their survival and platelet production.9–12
This review will describe the pharmacology and data for use of romiplostim in pediatric patients with ITP, including safety, efficacy, dosing strategies, goals of treatment, and treatment discontinuation. Updated ITP guidelines will integrate use of romiplostim in pediatric and adult ITP patients.13–15 While the vast majority of data describing clinical romiplostim use in ITP is in adult patients, some of these data may also apply to the pediatric population and will be discussed as well.
Pharmacology and dosing of romiplostim
Pharmacodynamics
Romiplostim increases the platelet count of healthy subjects and ITP patients in a dose-dependent fashion. 16 The pharmacodynamics of all thrombopoietic agents, romiplostim included, approximate physiologic thrombopoiesis. Physiologic TPO elevation occurs in the setting of diminished platelet production resulting in a reduction of TPO clearance. 17 In response to an acute increase in TPO concentration, megakaryocyte colony-forming cells differentiate into mature megakaryocytes. As this process occurs, approximately 2–5 days are required for megakaryocyte size and ploidy to peak, with the platelet count starting to increase at approximately the fifth day and to peak after 10 days. Therefore, TPO-RAs, including romiplostim, generally take a minimum of 5–7 days to produce a clinically significant rise in the platelet count, as was demonstrated in pharmacodynamic studies. 18
Pharmacokinetics
Over the dose range of 3–15 µg/kg, romiplostim has a median half-life of approximately 3.5 days, with significant variations in half-life (1–34 days) noted in pharmacokinetic studies. 18 It is bound to and recycled by the neonatal fragment crystallizable (Fc) receptor (FcRn) on endothelial cells. Peak drug concentrations were observed at a median of 14 h following a romiplostim dose. Romiplostim is eliminated by the dual action of the reticuloendothelial system and the platelet TPO receptor, cMPL. Serum concentrations of romiplostim therefore correlate with the platelet mass, rather than the dose of romiplostim administered, with higher platelet counts resulting in lower serum concentrations. 19 Pharmacodynamic-mediated drug disposition models in healthy subjects demonstrated that weekly dosing produces a sustained pharmacodynamic response as compared with dosing intervals of 2 weeks or greater, which produce considerable fluctuation in the platelet count. 20
There are no known clinically significant drug–drug interactions with romiplostim. It can be safely administered in combination with other ITP therapies, such as corticosteroids or IVIG. 19 Its effect on platelet production is not altered by deficiencies in either renal or hepatic function; thus, dose adjustment in these settings is not required.
Immunogenicity
Existence of pre-existing antibodies to romiplostim and development of antiromiplostim antibodies is not uncommon, occurring in 5% and 4% of adult ITP patients, respectively, across clinical studies including a total of 1112 patients.19,21 But neutralizing anti-romiplostim antibodies were much less common, occurring in less than 1% of adults treated with romiplostim. 21 Even when present, neutralizing antibodies were rarely associated with a loss of treatment effect. Immunogenicity was more common in children, with 8% and 3% developing non-neutralizing antiromiplostim antibodies and neutralizing antiromiplostim antibodies, respectively, across clinical studies including a total of 282 pediatric patients. 19 In patients with a documented sustained response to romiplostim who later lose this response, the treating clinician should consider the possibility of neutralizing antiromiplostim antibodies. To this end, patient blood samples can be sent to the drug manufacturer (Amgen, Thousand Oaks, CA) to evaluate for the possibility of such antibodies. There have been no reports of antibodies to romiplostim capable of cross-reacting with and neutralizing native TPO; none of the 1112 adult patients or 282 pediatric patients in the aforementioned studies developed such antibodies.
Initiation and dose escalation
The romiplostim prescribing information recommends a starting dose of 1 µg/kg/week in both adults and children with ITP. 19 The dose is uptitrated weekly by 1 µg/kg, based on the platelet count, advising an increase of 1 µg/kg for a platelet count < 50 × 109/l, continuing at the same dose for platelet counts 50–200 × 109/l, and reduction in the dose by 1 µg/kg for platelet counts > 200 × 109/l for 2 consecutive weeks. 19 Despite these dosing recommendations, a large phase III study of romiplostim in adults utilized a starting dose of 3 µg/kg/week in all patients. 22 Real-world studies of romiplostim use in adults have further demonstrated the safety of initiation of romiplostim at doses higher than 1 µg/kg/week, with very low rates of thrombocytosis. 23 In a small group of pediatric patients, the median starting romiplostim dose when used off label was 2 µg/kg/week. 24
In clinical practice, initiation at a higher dose reduces the duration of drug titration, and therefore, also the duration of profound thrombocytopenia, reducing bleeding risk; clinical trials have demonstrated that bleeding risk is highest during dose titration. 25 In a small study of hospitalized adult ITP patients, relative to patients initiated at romiplostim at 1 µg/kg/week, patients initiated on romiplostim at a median starting dose of 4.5 µg/kg/week had fewer bleeding events, a shorter hospital length of stay, and improved rates of achieving a platelet count ⩾ 50 × 109/l with no thrombotic events. 26
For these reasons, the authors routinely start most adult and pediatric patients at 3–5 µg/kg/week. Similarly, we do not hesitate to titrate the dose by greater than the 1 µg/kg/week instructed by the prescribing information to minimize the total duration of profound thrombocytopenia, the bleeding risk, and the time until an effective dose is achieved. More aggressive titration does increase the risk of thrombocytosis, which raises the concern of thrombosis risk (as discussed further below), but the bleeding risk often takes priority in ITP patients and the thrombotic risk is lower in children, than adults, with ITP. Likewise, in case of emergencies, such as life-threatening bleeding, we use 10 µg/kg/week for the first one to two doses to maximize the chances that the platelet count will adequately rise as quickly as possible; if there is no response after two doses, alternative treatments are chosen. Our approach to romiplostim dosing is summarized in Table 1.
Our approach to romiplostim titration in pediatric and adult ITP.

ITP, immune thrombocytopenia.
Attention should be paid to the ‘first-dose effect’ in some patients with ITP; by this, we mean that the platelet count may rapidly rise after the first dose. 22 The rapid rise should not lead to a reduction in the second dose, since in most patients continuation of the same dose will be accompanied by a fall in platelet count at weeks 2 and 3. This probably occurs due to the expanded bone marrow megakaryocyte pool which is now prevented from undergoing apoptosis.
Because approximately two thirds of pediatric patients eventually remit, the authors generally titrate the romiplostim dose in pediatric patients to a platelet count of 30–150 × 109/l so that continual dose decreases will allow the treating provider to know if romiplostim should be discontinued altogether to assess for remission (sustained platelet counts of >150 × 109/l off treatment).
Romiplostim is dispensed in vials of lyophilized powder for reconstitution. Standard administration is initially by a healthcare provider in a clinic setting due to the technical demands of reconstitution and injection of small volumes of reconstituted drug subcutaneously. Therefore, a significant time burden is therefore imposed upon the patient, and in the case of most pediatric patients, a parent or caregiver to take the child to the hematology clinic or their pediatrician’s practice each week for injections. There is evidence in both adult and pediatric patients that provided proper education and tools (a home administration training pack), self-administration in selected patients can be as effective as administration by a healthcare provider while alleviating much of the time and travel burden.24,28,29 The authors encourage patients and families to learn how to administer romiplostim at home to ease this burden and increase the feasibility of this treatment to patients who live far from their medical center. Self-administration of romiplostim is standard of care in most countries, but it has not yet received FDA approval in the US.
Holding doses
The romiplostim prescribing information advises withholding a dose of romiplostim for platelet counts > 400 × 109/l. 19 This guidance is not based on any clinical evidence for risk. Despite the increased rate of venous and arterial thrombosis in ITP patients, these events are not associated with any platelet count or TPO-RA use. 30 The authors do not follow this threshold due to the marked rebound thrombocytopenia (platelets dropping below their prior baseline) that occurs in 10–15% of patients when romiplostim is abruptly stopped. 31 We opt to dose reduce by one to two thirds instead to eliminate thrombocytosis and avoid the bleeding risk associated with rebound thrombocytopenia.
Laboratory monitoring on romiplostim
Platelet counts in children on romiplostim are checked weekly with dose titration and no less frequently than monthly when on a stable dose. The authors additionally advise review of the peripheral blood film every 6 months.
Efficacy of romiplostim in immune thrombocytopenia
Several randomized trials have evaluated the use of romiplostim against standard of care treatments or placebo in adult and pediatric ITP patients. There are two randomized studies of romiplostim in pediatric patients, both of which were followed by an open-label extension. The first was a phase I/II trial of ITP patients aged < 18 years that randomized patients to romiplostim (
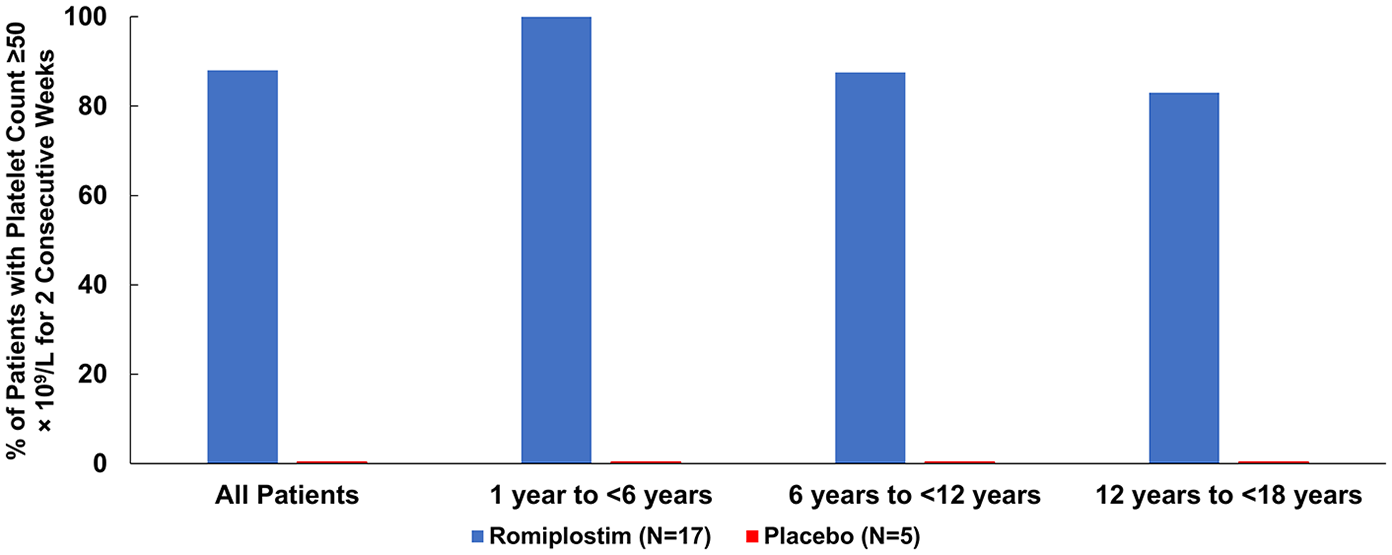
Percent with platelet count ⩾ 50 × 109/l for at least 2 consecutive weeks phase I/II study of romiplostim for pediatric ITP (prepared from data from Bussel
The second study was a phase III double-blind, placebo-controlled study randomizing 62 children with ITP (baseline platelet count < 30 × 109/l) 2:1 to romiplostim or placebo for 24 weeks. 33 The primary endpoint was a durable platelet response, defined as achieving a platelet response (platelet count ⩾ 50 × 109/l without rescue drug use in the preceding 4 weeks) in 6 or more of the final 8 weeks of the trial. The overall incidence of platelet response was significantly higher in the romiplostim group (72%) as compared with the placebo group (20%; Figure 2). A durable platelet response was achieved in 52% of the patients in the romiplostim group, compared with 10% in the placebo group. In the extension study that followed, 94% of patients had one or more platelet response, 72% had a platelet response at ⩾75% of visits, and 58% responded at ⩾90% of visits. 34 The median duration of treatment was 2.6 years.

Overall platelet response by age group in children with ITP treated with romiplostim
The effect of romiplostim on bleeding symptoms and quality of life in children with ITP in comparison with other second-line ITP treatments has been additionally demonstrated in a large, prospective, multicenter observational study of 120 children. 35 Treatments evaluated included romiplostim, eltrombopag, splenectomy, rituximab, and oral immunosuppressive agents. In addition to demonstrating that romiplostim had the most pronounced improvement on platelet counts, only patients treated with romiplostim or rituximab had significant reduction in both skin-related and nonskin-related bleeding symptoms after 1 month of treatment. Health-related quality of life also improved significantly in children treated with romiplostim or eltrombopag.
There are, additionally, several pivotal studies of adults with ITP treated with romiplostim that deserve mention given their size and role in regulatory approval of the agent. Two parallel double-blind phase III studies carried out in the United States and Europe, randomizing a total of 83 adults with ITP (platelet count < 30 × 109/l) for ⩾12 months to romiplostim and 42 to placebo found significantly higher rates of platelet response (platelet count ⩾ 50 × 109/l) in romiplostim-treated patients, with 88% of nonsplenectomized and 79% of splenectomized patients responding, compared with 14% of nonsplenectomized and 0% of splenectomized patients given placebo.
31
Patients treated with romiplostim achieved a response over a mean of 15.2 weeks (nonsplenectomized) and 13.8 weeks (splenectomized)
A large open-label phase III study carried out in North America, Europe, and Australia randomizing 157 nonsplenectomized adults with ITP (platelet count < 50 × 109/l) for ⩾12 months to romiplostim and 77 to standard of care (SOC) found the rate of platelet response (platelet count ⩾ 50 × 109/l at a clinic visit) was 2.3 times higher in the romiplostim arm than the SOC arm. 22 Additionally, there were lower rates of treatment failure (11% romiplostim, 30% SOC), splenectomy (9% romiplostim, 36% SOC), bleeding, and blood transfusions in the romiplostim arm as compared with the SOC arm. There was significantly greater quality-of-life improvement in romiplostim-treated patients compared with SOC, as well. Extension studies of romiplostim treatment of adults with ITP demonstrated continued efficacy for up to 5 years of treatment.
Phase III trials of romiplostim in ITP in children and adults are summarized in Table 2.
Phase III trials of romiplostim in ITP.

Each trial was a prospective, multicenter, randomized, placebo-controlled, double-blind study except Kuter
Platelet response defined as a platelet count ⩾ 50 × 109/l at a given assessment on treatment with romiplostim or placebo.
ITP, immune thrombocytopenia.
A large prospective 17-country open-label trial treating children with ITP for ⩾6 months with romiplostim found similar efficacy to the smaller phase III studies. 37 Although final results of this trial have not yet been published, preliminary results were recently published in abstract form. A total of 203 children (mean age of 10 years) with a median baseline platelet count of 14 × 109/l were treated for a median duration of 145 weeks, with 88% of patients having at least one platelet response (platelet count ⩾ 50 × 109/l with no rescue therapy in the preceding 4 weeks) in the first 6 months of treatment. Additionally, the median percent of time with an increase in platelet count of ⩾20 × 109/l above baseline from week 2 until the end of treatment was 79%.
Safety of romiplostim in immune thrombocytopenia
Romiplostim was overall well tolerated in clinical trials. In adult trials, the most common nonbleeding-related adverse events were headache, insomnia, myalgia, arthralgia, and mild dizziness. 22 In pediatric studies, the most commonly reported nonbleeding-related adverse events were headache, upper respiratory infection, nausea/vomiting, and oropharyngeal pain25,33; no patients suffered a thromboembolic event in the randomized pediatric studies or the open-label extension.
Consistent with the natural history of childhood ITP, bleeding events were rare overall in pediatric studies, with the highest risk of bleeding occurring in the initial few weeks of treatment during drug titration. In the initial randomized phase I/II trial, 12 patients had grade 1 bleeding and 1 patient had grade 2 bleeding (epistaxis, contusion, petechiae). 25 In the phase III study, 83% of romiplostim-treated patients had bleeding events, but nearly all were mild epistaxis, petechiae, contusion, or gingival bleeding; there were no intracranial hemorrhages or fatal bleeding. 33 Similar findings were seen in the extension study following the phase III trial 34 and have been reported from a large international prospective open-label study published in abstract form. 37
Potential adverse events of greatest concern with the long-term use of romiplostim are thromboembolic events and bone marrow fibrosis. Studies of platelet function from human ITP patients treated with romiplostim, 38 eltrombopag, 39 and avatrombopag 40 showed no evidence of spontaneous platelet aggregation or hyper-reactive platelets in the setting of TPO-RA use. In a study examining platelet function by light-transmission platelet aggregometry in romiplostim-treated adults with ITP, rather than observing hyperactive platelets, there were, instead, defects in platelet-aggregation response to adenosine diphosphate and epinephrine in the platelets of these patients. These defects are probably due to the effects of platelet autoantibodies on platelets rather than any impact of romiplostim. Since ITP is known to be a prothrombotic state with higher rates of arterial and venous thromboembolic events than the general population,41–43 it remains unclear whether the thromboembolic events seen in ITP patients on TPO-RAs are due to the underlying risks of the disease or an effect of these treatments. All randomized studies of romiplostim in ITP patients have shown no higher risk of either arterial or venous thrombosis in romiplostim-treated patients compared with placebo. 44 Furthermore, in large studies of ITP patients treated with romiplostim there was no association of the platelet count with thromboses 45 ; indeed, prior data in over 1000 ITP patients showed that the rate of thromboses increased as the platelet count decreased. 45 An international multicenter study of 203 pediatric ITP patients treated with romiplostim with a total of 428.7 patient-years of romiplostim exposure did not report any thrombotic events. 37
Bone marrow fibrosis over long-term use of TPO-RAs remains a potential concern, although evidence from adult patients receiving romiplostim for several years is reassuring, with very low risks of reversible marrow reticulin fibrosis and a near-zero risk of serious, irreversible collagen fibrosis.46,47 The risk of bone marrow fibrosis in infants or young children treated with romiplostim for long periods remains unknown, however, and is a risk that should be discussed with patients and caregivers. Randomized pediatric romiplostim studies did not require interval bone marrow biopsy to evaluate for possible fibrosis but there were no hematologic complications consistent with development of fibrosis; a large, international multicenter study including 203 patients reported bone marrow biopsy results on 75 children with ITP receiving romiplostim, finding no occurrence of collagen fibrosis or bone marrow abnormalities. A potentially significant increase in reticulin fibrosis (an increase of ⩾2 grades) occurred in a single patient. 37 Long-term effects of romiplostim and other TPO-RAs in children with chronic ITP, who may require lifelong treatment, are unknown. Finally, concerns for the possibility of leukemogenesis with romiplostim have been largely put to rest following publication of a large study of adult patients with myelodysplastic syndrome treated with romiplostim for extended periods, showing no increased risk of progression to acute myeloid leukemia after several years of follow up. 48
Selection of thrombopoietin receptor agonist in chronic pediatric immune thrombocytopenia: romiplostim versus eltrombopag
Choice of therapy in chronic ITP in children is a complex topic 49 and a subject of consensus guideline statements.13–15 Because platelet counts vary considerably in ITP and many patients are able to maintain counts ⩾ 30 × 109/l consistently, the first question to be asked in each patient is whether treatment is indicated. Prior bleeding events, current bleeding symptoms, activity level and sports participation, fatigue, patient and caregiver anxiety, and other factors play a major role in this determination beyond baseline platelet count. In patients for whom treatment is indicated, long courses of glucocorticoids are contraindicated due to effects on growth and other short and long-term complications in children; IVIG and intravenous anti-D immunoglobulin typically provide only temporary platelet count improvement. Therefore, subsequent, or second-line treatment options in pediatric ITP generally include rituximab, thrombopoietin receptor agonists, and off-label immunosuppressive treatments such as azathioprine, mycophenolate, and sirolimus, with splenectomy much less commonly selected in comparison with adults with ITP. Thrombopoietin receptor agonists are the only FDA-approved second-line treatments in pediatric ITP and offer high response rates without immunosuppression and durable responses at the cost of prolonged or indefinite treatment. The new American Society of Hematology guidelines favor TPO-RA over rituximab and splenectomy in the treatment of children with ITP. 14
In those patients for whom a TPO-RA is selected, romiplostim and eltrombopag are the two options FDA approved in children (avatrombopag’s ITP approval is limited to adults, and lusutrombopag is not approved for ITP), Table 3. Currently, it is not possible to predict response to TPO-RAs, or differential response to romiplostim or eltrombopag, in children, although there are several studies evaluating this in adults that have found an inverse relationship between baseline TPO level and likelihood of response to TPO-RAs. A study of baseline endogenous TPO levels in adult ITP patients found different TPO thresholds for prediction of nonresponse to romiplostim and eltrombopag, suggesting that romiplostim is likely to be effective in a population of ITP patients with mild elevations in TPO levels, whereas eltrombopag is less likely to be effective. 50 This may be related to agent potency; in healthy subjects, maximal doses of romiplostim increased the platelet count approximately 8–10 times that of maximal doses of eltrombopag.20,51–53
Comparison of the TPO-RAs used in ITP treatment. Lusutrombopag is not included as it is not approved for ITP.
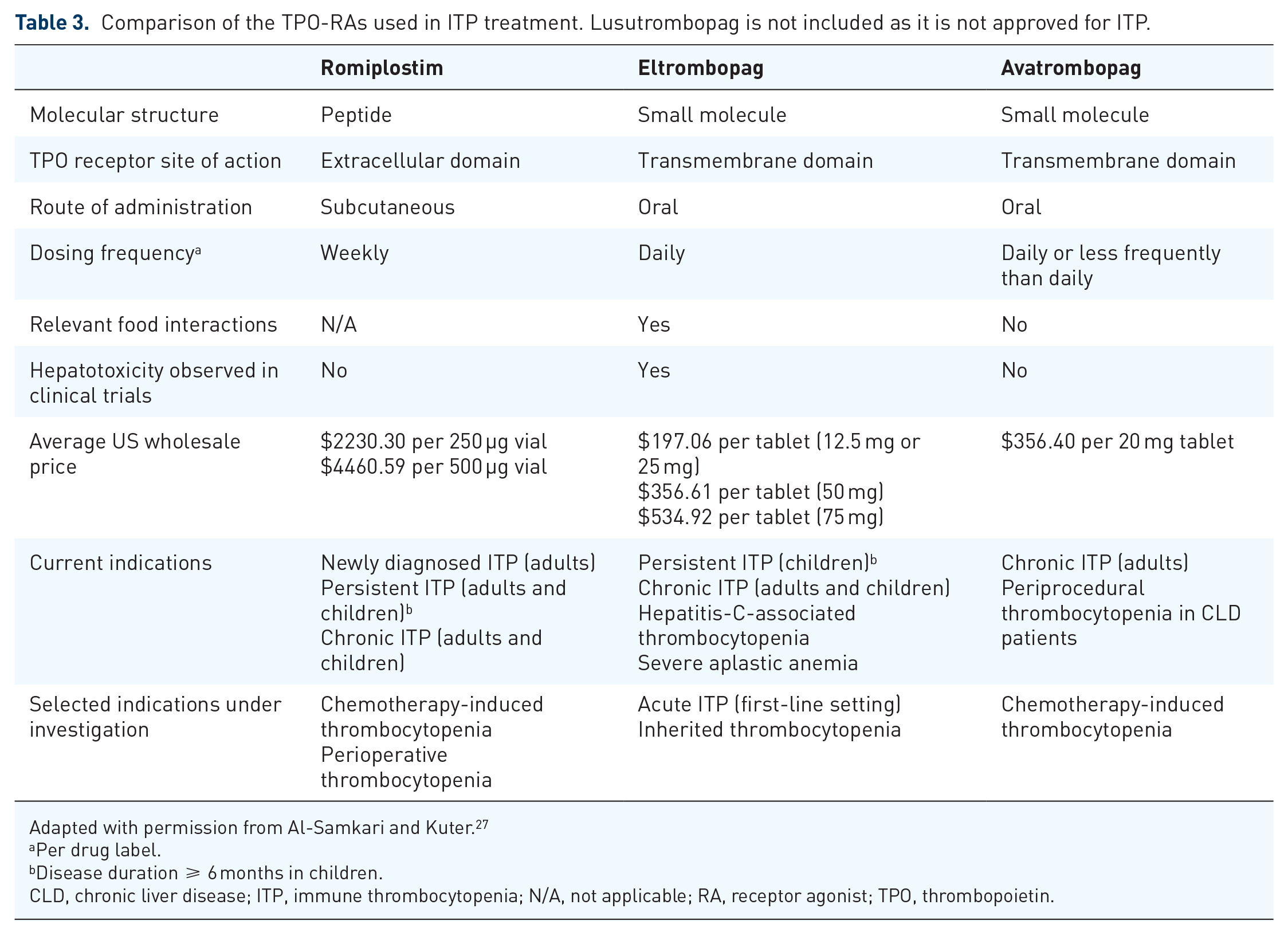
Adapted with permission from Al-Samkari and Kuter. 27
Per drug label.
Disease duration ⩾ 6 months in children.
CLD, chronic liver disease; ITP, immune thrombocytopenia; N/A, not applicable; RA, receptor agonist; TPO, thrombopoietin.
The primary advantage of eltrombopag is oral administration. This may be especially relevant in young children, who may develop needle phobia from weekly romiplostim subcutaneous injections and, initially, weekly lab monitoring. However, there are several issues with eltrombopag that can affect its effectiveness and additional safety concerns compared with romiplostim. Absorption of eltrombopag is dramatically reduced by divalent cations such as calcium or magnesium, as well as dietary fats, and the drug label advises avoidance of these for a 4–6-hour window around its administration. Because dietary fats and minerals are so common in foods and unavoidable in most standard Western diets, this functionally becomes a 4–6-hour fasted window to ensure proper absorption, which may be very challenging in children. Given this, many pediatric hematologists will accept the child taking eltrombopag at least 1 h prior to eating, or 2 h after eating and away from dairy or iron supplements. Considering the prolonged durations of treatment required, these dietary restrictions are not a minor issue; either quality of life or treatment effectiveness is compromised. Because eltrombopag has a half-life of 35 h in patients with ITP, dosing less frequently than once daily to minimize quality-of-life concerns is reasonable; this approach has been tried in adults with success, 54 but there is no evidence to support this approach in children. The major important adverse effect of eltrombopag is hepatotoxicity, which merits regular liver enzyme monitoring and is not associated with romiplostim. Finally, eltrombopag is an iron chelator and can precipitate iron-deficiency anemia in children with ITP.55,56 Avatrombopag is an oral TPO-RA with no dietary restrictions with administration, reported hepatotoxicity, or iron-chelation properties,57,58 so presents a potentially appealing oral option if approved for children in the future. Romiplostim has none of these disadvantages but typically requires clinic visits for administration, which can be burdensome and time consuming for both patient and caregiver. Occasionally, the decision may be dictated by the patient’s insurance coverage. Both romiplostim and eltrombopag are expensive, but cost-effectiveness studies of the two drugs are conflicting.59,60
Defining treatment success and failure
The goal of romiplostim treatment is bleeding prevention and optimizing patient quality of life. The target platelet count should be based on achievement of these treatment goals and is generally between 50 × 109/l and 150 × 109/l in most patients. Normalization of the platelet count is rarely necessary, although an argument for a slightly higher goal range (albeit without supporting evidence), such as between 100 × 109/l and 200 × 109/l, may be made for children participating in contact sports. Children with ITP requiring major surgery may require platelet counts ⩾ 100 × 109/l. In most patients, and especially those with difficult-to-control disease, a platelet count of ⩾20 × 109/l without bleeding symptoms is adequate.
Treatment discontinuation for futility is advised if the platelet count is not sufficiently improved after 4 weeks of treatment at maximal dose (10 µg/kg) of romiplostim. 19 Often, the addition of small amounts of prednisone (1–5 mg/day) in patients failing romiplostim treatment will convert a nonresponder to a responder. 50 If prednisone addition is unsuccessful or not tolerated, a switch to eltrombopag is not unreasonable; multiple publications report success with this approach in adults. 61 However, some patients will fail both TPO-RAs and require a transition to a different treatment modality.
Most children with ITP will spontaneously remit regardless of treatment. A minority of adult patients with chronic ITP eventually remit, and remissions after prolonged treatment with TPO-RAs in these patients have been reported.62,63 Although it is not known if a similar phenomenon occurs in pediatric ITP, we advise gradual romiplostim downtitration and discontinuation should be considered in patients with platelet counts persistently > 200 × 109/l. When treatment is discontinued for possible remission or some other reason, we perform a taper over 2–4 weeks, given that rebound thrombocytopenia with romiplostim discontinuation has been observed in approximately 10–15% of adult ITP patients. 15
Conclusion
Romiplostim is a newly FDA-approved option in persistent and chronic pediatric ITP, supported by two randomized clinical trials and two long-term extension studies demonstrating efficacy and safety. It has a high response rate with few side effects. Its primary drawback is weekly subcutaneous administration, but it is not associated with the hepatotoxicity or dietary restrictions of eltrombopag. Choice of TPO-RA in the pediatric patient with ITP is patient dependent and clinicians should consider the benefits and risks of each agent. The most common adverse event attributable to romiplostim in clinical trials was headache. While it remains a theoretical concern, particularly in adults, thrombotic events have not been elevated in romiplostim-treated
Footnotes
Acknowledgements
HA is the recipient of the National Hemophilia Foundation-Shire Clinical Fellowship Award, American Society of Hematology Scholar Award, and Harvard KL2/Catalyst Medical Research Investigator Training Award, which provide salary support.
Author contributions
HA drafted the manuscript, created the tables and figures, and contributed to concept and design, critical revision of the intellectual content, and final approval. RFG contributed to concept and design, critical revision of the intellectual content, and final approval. DJK contributed to concept and design, critical revision of the intellectual content, and final approval.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
Dr Al-Samkari reports consultancy (Agios, Dova, Moderna) and research funding (Agios, Dova). Dr Grace reports scientific advisorship (Agios), consultancy (Agios), and research funding (Agios, Novartis). Dr Kuter reports research funding (Protalex, Bristol-Myers Squibb, Rigel, Bioverativ, Agios, Syntimmune, Principia, Alnylam) and consultancy (ONO, Pfizer, 3SBios, Eisai, GlaxoSmithKline, Genzyme, Shire, Alexion, Amgen, Shionogi, Rigel, Syntimmune, MedImmune, Novartis, Bioverativ, Argenx, Zafgen).
Ethics approval statement
Ethics approval was not required for this review.
Informed consent statement
Informed consent was not required for this review.
