Abstract
Breakthrough pain (BTP) is experienced by approximately 65% of children and adults with chronic pain. Undiagnosed or untreated BTP produces negative emotional, physical, and economic consequences. BTP episodes have a rapid onset and short duration. Short acting oral opioids are the cornerstone of BTP management. Oral medications available to treat BTP episodes like immediate-release morphine or oxycodone have a delayed onset of action so that there is a mismatch between the episode of BTP and the effect of the oral opioids. Novel fentanyl delivery systems for BTP offer pharmacokinetic properties that match the time profile of BTP. Among the transmucosal routes, intranasal fentanyl has gained popularity due to its high bioavailability, rapid onset of action, high potency, short duration, and ease of administration. Its efficacy and safety have been demonstrated in adults who are opioid tolerant. Although children with chronic cancer pain also experience BTP, there is paucity of data on the use of intranasal fentanyl for BTP in this age group.
Introduction
Breakthrough pain (BTP) is commonly defined as a transient exacerbation of significant pain superimposed on a background of well-controlled chronic pain. This term has no literal translation in many languages, and even in English, different authors use the term BTP in different ways, which is why some experts have suggested using the words episodic or transient to avoid confusion.1,2 BTP can occur spontaneously or in relation to predictable or unpredictable triggers (eg, walking, voiding). It can also arise when the concentration of the baseline medication drops below analgesic levels, such as occurs near the end of a dosing interval. 1 The origin of BTP may be nociceptive (somatic or visceral), neuropathic, or both.
BTP is characterized by its sudden onset, severe intensity, and episodic recurrence. These episodes of severe pain usually peak within 3 minutes and are of short duration; they are typically self-limited to less than 45 minutes. The number of episodes varies among patients but the average is 4 to 6 episodes daily. The incidence of BTP in cancer patients has been reported to be between 33% and 95% with an average of 65%.1–3 Patients with undiagnosed or untreated BTP are more prone to depression, more severe baseline pain, impaired function, and poor quality of life. BTP also makes the treatment of persistent pain more difficult 4 and may exacerbate comorbidities. BTP adds stress to caregivers4 and increases the cost of health care. 3
BTP can be treated pharmacologically with non-steroidal anti-inflammatory drugs (NSAIDS) or with opioids. NSAIDS are especially useful in pain from bone metastases as well as mucosal and skin lesions. They have been proven to improve basal analgesia and reduce episodes of BTP.1,2 When a rescue dose is required, sublingual or parenteral formulations can be used.
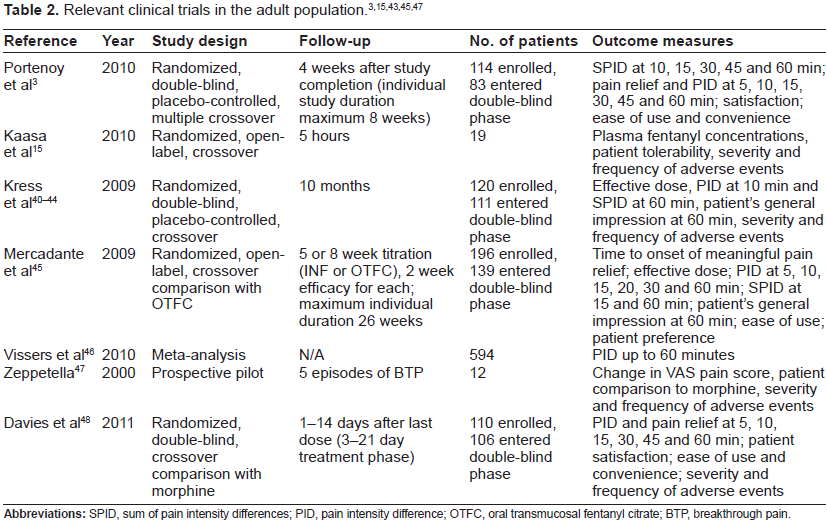
Opioids are considered the cornerstone of moderate to severe BTP management. Oral, parenteral, and rapid-onset formulation opioids are available to treat baseline and BTP. 2 Many opioids, including morphine and hydromorphone, are available in both oral and parenteral preparations for the treatment of BTP. However the lipophilicity and high potency of fentanyl make it the only opioid currently available in transmucosal, rapid-onset formulations. 2 Fentanyl products for the treatment of BTP include oral transmucosal fentanyl citrate (OTFC), fentanyl buccal tablet, sublingual fentanyl, intranasal fentanyl (INF), and fentanyl buccal soluble film (Fig. 1A and B). In recent years, INF has gained popularity due to its ease of administration and rapid absorption through the highly vascular nasal tissues. This allows rapid onset of action, a peak effect within 5 minutes, and optimal duration to cover the entire episode of BTP. 3 These characteristics are ideally suited to those of BTP.
Mechanism of Action, Metabolism, and Pharmacokinetic Profile
Fentanyl(orN-phenyl-N-[1-(2-phenylethyl)piperidin-4-yl]propanamide) is a lipophilic, short-acting, synthetic opioid with a piperidine chemical structure and a low molecular weight (Fig. 2). It has predominant agonist effects on Mu1 and Mu2 opioid receptors with an analgesic effect 75 to 125 times that of morphine. The higher potency, faster onset of action, and shorter duration of action are due to its greater lipid solubility, which facilitates rapid passage across the brain-blood barrier with an effect site equilibration time of 6.4 minutes. 5 Its short duration of action following a single dose results from rapid redistribution to inactive tissues such as fat and skeletal muscle thus decreasing the effective plasma concentration. Like most other opioids, fentanyl is cleared by hepatic metabolism, primarily through activity of the cytochrome P450 3A (CYP3A) system. Fentanyl has a high hepatic extraction ratio making it resistant to changes in metabolism due to CYP3A inhibition; however, drug interactions are possible and inhibition of the CYP3A4 enzymes may reduce the clearance of fentanyl, 6 prolonging or potentiating its effects.6,7 Repeated doses or continuous fentanyl infusions saturate the inactive sites, delaying the decrease in plasma concentration and prolonging both the analgesic and respiratory depressive effects.7,8
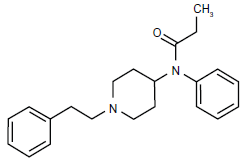
Chemical structure of fentanyl.
Fentanyl is administrated in a wide range of doses depending upon the setting, indications, and patient factors. Fentanyl shares its effects with other opioids, the most common being analgesia, sedation, brady-cardia, respiratory depression, nausea and vomiting, pruritus, spasm of the biliary smooth muscle, urinary retention, delay in gastric emptying, constipation, immunosuppression in susceptible individuals, tolerance, and physical dependence.9,10 A rare but potentially serious side effect of opioids is chest wall rigidity. This is typically reported after intravenous (IV) administration of fentanyl such as occurs with induction of anesthesia or in the neonatal intensive care unit where fentanyl infusions are used for sedation (Table 1).11,12 Histamine release and hypotension, commonly seen with morphine, do not occur with fentanyl administration. 13
Fentanyl effects on organ systems, and potential side effects.

Fentanyl can be administered by the traditional IV parenteral route but also by intrathecal or epidural, transdermal, and transmucosal routes. The intranasal delivery of fentanyl has been found to be effective and reliable. It is convenient, familiar to patients, and allows for self-administration. 7 The large surface area of the nasal mucosa, its high blood flow per cubic centimeter of tissue, and its direct connection to the central nervous system via the olfactory mucosa make this route ideal for BTP treatment. Drug absorption through the nasal mucosa also depends on lipid solubility, drug ionization, and the mucociliary clearance. First-pass liver metabolism is avoided by intranasal administration, allowing for less drug use.7,14 The onset of intranasal fentanyl is rapid, occurring between 6 and 8 minutes after administration, and its effect lasts less than 1 hour, which is an ideal profile for a medication to treat episodic pain.
Different technologies have been developed to deliver fentanyl by noninvasive routes. Transmucosal oral fentanyl can be given as a buccal tablet, sublin-gual soluble film, or lollipop. Intranasal fentanyl can be administered by direct application of the standard intravenous solution or sprayed using an atomizing device. For the specific treatment of breakthrough pain, two formulations of intranasal fentanyl spray have been approved: intranasal fentanyl spray and fentanyl-pectin nasal spray. Intranasal fentanyl spray is available in three different concentrations, 50, 100, and 200 µg per 100 µL spray. Fentanyl-pectin nasal spray is available in two concentrations, 100 and 400 µg per 100 µL. The pharmacokinetics of intranasal fentanyl spray (INFS) has been studied in a prospective randomized study using varying concentrations of fentanyl in patients with cancer and episodes of BTP. 15 This study found that the mean Tmax was 15 minutes for 50 µg, 12 minutes for 100 µg, and 15 minutes for 200 µg. The Cmax increased in proportion with the dose: 351 pg/mL for the 50 µg dose, 595 pg/mL for the 100 µg dose, and 1195 pg/mL for the 200 µg dose. When compared with IV fentanyl, intranasal fentanyl was 89% bioavailable and its duration of action was slightly longer. 16 Higher doses increased the duration of action with both routes but doubling the dose did not double the duration of action. Cmax was slightly lower with intranasal fentanyl than IV fentanyl (1.2 ng/mL vs. 2.0 ng/mL).
Pharmacokinetic data over 96 hours from healthy volunteers who received INFS and OTFC revealed that INFS had a shorter Tmax, greater Cmax, and larger AUC than OTFC. 17 Three different nasal fentanyl formulations containing pectin, chitosan, or chitosan-poloxamer 188 were compared with OTFC in a single-dose, open-label, four-way crossover study. 18 The authors concluded that all intranasal formulations were rapidly absorbed and had a biphasic decline in drug levels and terminal half-life while OTFC had triphasic elimination kinetics and a longer T1/2. The Tmax was shorter and the AUC was greater for all the intranasal formulations compared with OTFC.
When the pharmacokinetics in young adults and elderly subjects were compared, both groups had equivalent Cmax and Tmax. 19 Conditions causing nasal congestion such as seasonal allergic rhinorrhea and the common cold do not appear to alter the pharma-cokinetics of INFS.20,21 When the topical vasoconstrictor oxymetazoline was applied intranasally, Cmax was reduced and Tmax was prolonged. 20
Clinical Studies: Children and Adults
Intranasal fentanyl has been tested in a variety of populations and clinical settings. Intranasal fentanyl is widely used in children, most frequently for acute pain in the emergency department or perioperative settings. Its safety and efficacy in this population have been demonstrated in multiple prospective observational studies as well as randomized controlled trials (both comparative and placebo-controlled) using a variety of concentrations including the standard 50 µg/mL concentration.22–25 The efficacy of INF was found to be equivalent to that of both intramuscular (IM) and IV morphine for the relief of pain associated with bone fractures.26,27 INF administration was better tolerated than IM morphine, and the intranasal route allowed for more rapid analgesia after initial patient presentation than was achieved by analgesics requiring IV access.26,28,29 Intranasal fentanyl was also found to be an effective and safe alternative to the use of intranasal diamorphine in the emergency department in the management of severe pain in children with limb fractures, burns, crushed digits, and abdominal pain. 30 The implementation of protocols using intra-nasal fentanyl in the emergency department reduced the time to analgesic administration when compared with protocols dependent upon the IV route, increasing the quality of care in addition to patient and family satisfaction. This observation was more notable in younger children who were brought to the emergency department with fractures and burns, most likely due to the ease of administration when IV access has not been established.28,29 Intranasal fentanyl has been shown to be a safe, effective, and tolerable alternative to parenteral morphine for the management of acute pain in the emergency department. 31
When intranasal fentanyl was used in combination with high-concentration inhaled nitrous oxide in a prospective observational study of children receiving procedural sedation in the emergency room, minimal to moderate sedation was obtained in 80% of the patients. No serious adverse events were reported, but higher incidences of vomiting and deep sedation were seen when compared with published data on nitrous oxide alone. This finding alludes to the potential serious complications of potent opioid use. 32 INF was shown to be equivalent to oral morphine for analgesia during dressing changes in pediatric patients with burns. 33
The intraoperative administration of INF leads to reduced postoperative pain and emergence agitation scores compared with placebo.34,35 Indeed, Voronov et al 36 reported no difference in postoperative pain scores between children who received intranasal fentanyl versus a regional nerve block with bupivacaine for myringotomy and tube placement. When administered in the postoperative care unit, INF was shown to be as effective as IV fentanyl for pain control; however, time to onset of analgesia was longer, and total fentanyl doses were higher for the intranasal route. 37 Of note, the precise method of administration appears to affect the time to analgesia: Galinkin et al 34 reported that peak serum concentrations of fentanyl are reached after 10 ± 4 minutes following rapid (over 2-5 seconds) administration of a dose equally divided between both nostrils, while Manjushree et al 37 reported a longer time to a significant difference in pain scores following a slower (drop-by-drop over 60 seconds) delivery. This variation in technique may account for the finding that INF was not statistically different than placebo when used for analgesia 10 minutes prior to catheterization for voiding cystourethrography in a double-blind randomized controlled trial. 38
To the best of our knowledge, there are no randomized controlled trials conducted in children evaluating intranasal fentanyl specifically for the treatment of BTP, even though approximately 60% of children with chronic pain experience episodes of BTP and there is a need for more effective treatment strategies in this patient population. 39
Intranasal fentanyl has been studied in adults with BTP within other clinical scenarios with several different formulations. Many authors have evaluated INF for BTP in patients who are receiving opioids for the management of cancer pain. Study protocols initially begin with fentanyl titration to determine the appropriate dose to relieve BTP, and it has repeatedly been found that patients taking a lower dose of morphine or other opioid require lower doses of INF.40–45 INFS was superior to placebo when pain intensity difference was evaluated and was highly tolerable with a low rate of adverse effects. Nausea, vomiting, constipation, and epistaxis were some of the most common adverse effects related to INFS administration, and these were usually mild to moderate in severity.41–43 In 2010, Portenoy et al 3 published the results of a placebo-controlled, double-blind, multiple-crossover study using a pectin spray formulation of intranasal fentanyl. As in other studies of breakthrough cancer pain, eligibility criteria included the use of at least 60 mg of morphine or equivalent per day with fewer than four episodes of BTP daily. Primary outcomes were the pain intensity difference at various time points and the sum of pain intensity differences at 30 minutes. Fentanyl pectin nasal spray reduced the sum of pain intensity differences at 10, 30, and 60 minutes when compared with placebo. Pain intensity scores were noted to be different as early as 5 minutes. Seventy percent of patients were satisfied with the ease of administration, and 87% continued using the pectin-based INFS in an open-label longitudinal study. 3
Mercadante et al
45
compared INFS with another rapid-onset opioid, oral transmucosal fentanyl citrate in an open-label crossover trial of patients with breakthrough cancer pain. Initial titration to an effective dose using both preparations was performed. The effective dose, defined as the dose at which meaningful pain relief was experienced, was the primary outcome. Other outcomes included the pain intensity difference at 10 and 20 minutes, the sum of pain intensity differences at 15 and 60 minutes, the ease of administration, patient preference, and the relationship between the effective INFS dose and the base opioid requirement. The authors found a statistically significant difference in the time to meaningful pain relief for INFS when compared with OTFC (11 minutes compared with 16 minutes,
Vissers et al 46 reviewed the efficacy of INF and OTFC, fentanyl buccal tablet (FBT), and oral morphine. A Bayesian method was used to calculate probability of differences. The authors reported a 99% probability that INFS reduced pain intensity to a greater extent at 15 minutes than FBT, OTFC, and morphine. At 15 minutes, INF achieved a greater pain intensity difference than FBT, and both were more effective than oral morphine or OTFC. At 30 and 45 minutes, INFS reduced pain intensity to a greater extent than OTFC and oral morphine. At 45 minutes, INFS was still more effective than FBT. At 60 minutes INFS was superior to OTFC Table 2.
Efficacy and Safety
Clinical studies have shown intranasal fentanyl to be effective in the treatment of BTP in adults.3,40,42,47 Efficacy is usually measured by significant improvement in pain intensity, the sum of pain intensity differences, clinically significant pain relief, and total pain relief at different time points in comparison with placebo. Significant differences have been reported as early as 5 minutes and have persisted for 60 minutes when intranasal fentanyl is compared with placebo. 3 When intranasal fentanyl has been compared with other opioids such as oral short-acting formulations of morphine and oxycodone, intranasal fentanyl has also shown efficacy for the treatment of BTP and has a more rapid onset of action. Intranasal fentanyl has also been compared with other fentanyl formulations for BTP, including FBT and OTFC. Intranasal fentanyl was better than immediate-release morphine sulfate, FBT, and OTFC at reducing pain intensity.8,46,48
The efficacy, safety, and acceptability of intranasal fentanyl citrate was reported by Zepella et al 47 in a pilot study in which 66% of patients graded INFS efficacy as very good to good and 75% preferred INFS as a rescue medication over their previous opioid.
Short-term and long-term studies using intranasal fentanyl in BTP have reported a low incidence of mild to moderate adverse effects. Due to its lack of preservatives, it is considered safe to administer fentanyl close to the ethmoid sinus. The results of numerous studies agree on the safety of intranasal fentanyl for the treatment of BTP. The most common adverse effects are nausea, vomiting, dizziness, headache, somnolence, and constipation, all of which are commonly found with other opioids. Other adverse effects related with the intranasal administration of fentanyl are epistaxis, nasopharyngitis, and dysgeusia.3,4,41 Several studies using intranasal fentanyl for BTP in cancer reported that adverse effects were related to the progression of the disease and not related to the fentanyl itself.42,43 However, it is important to note that rapid-onset opioids are only recommended in opioid tolerant patients.2,8
Fentanyl is metabolized by the cytochrome P450 3A4 liver isoenzyme system. These drug-metabolizing enzymes are involved in the elimination of more than 50% of all currently used prescription drugs. Clinically relevant drug-drug interactions may occur (Table 3), and opioid adverse effects can be increased or prolonged with the concomitant use of inhibitors of this isoenzyme system.7,8,49,50
Potential drug-drug interactions.
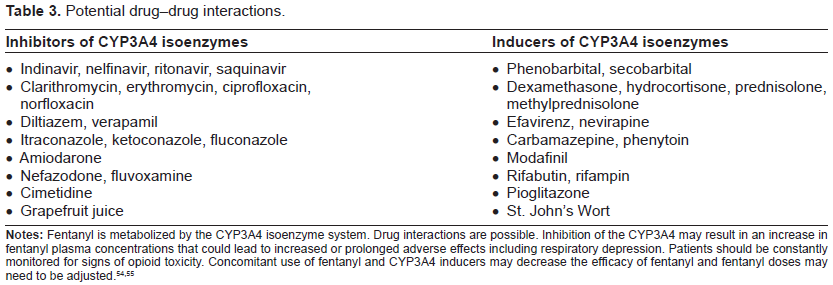
There is an increased risk of sedation and respiratory depression if other CNS depressants such as benzodiazepines are also used. Fentanyl is not recommended in patients who have taken monoamine oxidase inhibitors in the preceding 14 days, and caution should be used when fentanyl is administered concomitantly with selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors, tricyclic antidepressants, and other serotonergic medications, as all of these interactions may precipitate serotonin syndrome.4,7,51,52
Studies to measure efficacy and safety of intranasal fentanyl specifically in the treatment of BTP were designed to include adult patients who were opioid tolerant and on an opioid regimen consisting of at least 60 mg of morphine or its equivalent per day and who were having at least three or four episodes of BTP per day. A titration phase to find the appropriate dose for BTP treatment is necessary prior to beginning therapy with INF. In children, intranasal fentanyl can be safely used in non-opioid tolerant patients to treat acute and procedural-related pain, but it has not been evaluated for safety and efficacy in the treatment of breakthrough pain.
Patient Preference
To evaluate patient acceptability, patients are asked to evaluate overall satisfaction, speed of pain relief, reliability and ease of use of administration. Portenoy 3 and Davies 48 among others have reported high rates of patient satisfaction with INF administration. More than 70% of patients chose to continue using INF as their BTP medication in open-label protocols. 47 Among children and parents, intranasal fentanyl also has great acceptability, especially when IV access is not immediately available. Patients prefer INF over other rapid-onset opioids such as OFTC. 45
Place in Therapy
The benefits of intranasal fentanyl in adults with BTP and in children with acute and procedural-related pain are well elucidated. Due to its rapid onset of action, ease of administration, efficacy, safety, and low incidence of adverse effects, intranasal fentanyl has had great acceptability among patients and caregivers. In addition, intranasal administration offers a viable alternative to the oral route in patients with oral problems such as mucositis, mouth ulcers, and xerostomia, or if oral administration is otherwise difficult.
Conclusion
Breakthrough pain is common in patients treated with longer-actingorsustained-releaseanalgesics.Intranasal fentanyl is a rapid-onset opioid with proven safety and efficacy for the management of breakthrough pain in opioid-tolerant patients, and its pharmacokinetic profile is well matched to the rapid-onset and brief duration of BTP episodes. INF provides more rapid analgesia than oral opioids, but avoids the complications of parenteral medication administration for acute pain exacerbation. Many patients find INF acceptable or even preferable to other opioids for the treatment of BTP. This is due to the familiarity, ease of use, and tolerability of intranasal administration in addition to the quick onset and low incidence of adverse effects seen with fentanyl. However, rapid-onset opioids are not safe to use in an unmonitored setting in patients who are opioid naïve. Although its use for the treatment of acute pain is well established, the efficacy and safety of INF remains to be proven in pediatric patients with breakthrough pain, despite a high prevalence of this condition in children.
Author Contributions
Analysed the data: CC, RRL, UC, JG. Wrote the first draft of the manuscript: CC. Contributed to the writing of the manuscript: RRL, UC, JG. Agree with the manuscript results and conclusions: CC, RRL, UC, JG. Jointly developed the structure and arguments for the paper: CC, RRL, UC, JG. Made critical revisions and approved final version: CC, RRL, UC, JG. All authors reviewed and approved of the final manuscript
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributor-ship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.