Abstract
Atazanavir (ATV) is an HIV protease inhibitor (PI) that was approved as antiretroviral agent in year 2003. Ritonavir (RTV) enhances ATV plasma exposure and increases its barrier to resistance. Antiretroviral therapy with ATV plus RTV (ATV/r) has demonstrated high potency for achieving virological suppression in antiretroviral-naive patients and in simplification strategies. In rescue interventions, ATV/r-based combinations have shown to be equivalent in terms of viral response to other PI/r-containing regimens. In contrast with all other PIs, ATV has also demonstrated efficacy when given unboosted with RTV, an option attractive in special situations. Other benefits of the drug are its good metabolic and gastrointestinal profile. Its main adverse event is hyperbilirubinemia, since ATV inhibits the hepatic uridin-glucoronyl-transferase. A signature mutation at the protease gene, I50L, confers loss of susceptibility to the drug whereas it may confer hypersusceptibility to other PIs. Many ATV studies have provided data until or beyond week 96, supporting the efficacy and safety of the drug in the long-term.
Introduction
Atazanavir (ATV) is a potent inhibitor of the HIV protease enzyme that prevents the formation of mature virions. ATV offers the benefits of having very good tolerance and pharmacokinetic profile, allowing once daily administration. Moreover, it is the only protease inhibitor (PI) that may be administered without rito-navir (RTV) boosting.1,2
In year 2003 ATV was approved to be used as part of antiretroviral therapy (ART) by both FDA and EMEA. However, while in the United States the approval was for both ARV-experienced and ARV-naïve patients, in Europe ATV was firstly approved for use in ARV-experienced patients. It was not until year 2008 when the approval was extended to ARV-naïve patients. After nearly one decade of ATV commercial use, a substantial amount of information has been produced for the drug. Herein, we update the current information on the pharmacological profile and drug resistance patterns of ATV as well as long-term experience in different clinical scenarios.
Pharmacology
ATV is an azapeptide inhibitor of the HIV-1 protease. As other PIs, it prevents the virus-specific processing of viral Gag and Gag-Pol poliproteins. 3 The inhibition constant of the drug is 66 nmol/L with a 50% effective concentration (EC50) or inhibition of 50% of viral replication and EC90 of 2.6-5.3 and 9-15 nmol/L, respectively. 4 Its intrinsic antiretroviral potency is generally greater than that of indinavir (IDV), nelfinavir (NFV), RTV, saquinavir (SQV) and amprenavir (APV). In contrast, ATV has showed less potency than other PIs against HIV-2 and therefore it should not be considered of choice in HIV-2 carriers. 5
ATV is rapidly absorbed with a peak plasma concentration (Cmax) occurring at approximately 2.5 h. The extent of absorption is highly dependent on gastric pH and increases when taken together with a light meal. ATV is 86% bound to human serum proteins to both alpha-1-acid glycoprotein and albumin to a similar extent. ATV is a substrate for the P-glycoprotein (P-gp), an efflux transporter that acts limiting tissue compartment distribution. Like other PIs, ATV is extensively metabolized by the hepatic cytochrome P450, primarily by CYP3A4/CYP3A5 isoenzymes. ATV and its metabolites then undergo biliary and urinary excretion (79% and 13% of an administered dose, respectively). The steady state is achieved between days 4 and 8, with an accumulation of ≈2.3-fold. Finally, ATV scarcely enters the cerebrospinal fluid (CSF) and semen, with a CSF/plasma ratio ranging between 0.0021 and 0.022 and a seminal fluid/plasma ratio ranging from 0.11 to 4.42.6,7 RTV at low doses (100 mg/day) inhibits the CYP3A enzyme and P-gp to a lesser extent. Consequently it enhances ATV exposure, allowing lower dosing of ATV (300 mg/day instead of 400 mg/day).
In patients with moderate to severe hepatic impairment (Child-Pugh class B/C), exposure to ATV increases up to 42% and half-life nearly 2-fold compared to healthy volunteers. 6 Therefore, ATV should be used with caution in patients with mild-to-moderate liver insufficiency. In patients with Child-Pugh Class B with no prior virologic failure, ATV dosing should be reduced to 300 mg/day. ATV should be avoided in patients scored as Child-Pugh Class C. ATV/r has not been studied in subjects with hepatic impairment and is not recommended. In contrast, no dose adjustments are needed for patients with severe renal insufficiency not managed by hemodialysis.
Drug interactions
The solubility of ATV decreases as pH increases, and plasma concentrations of the drug may be reduced when co-administered with proton pump inhibitors, H2-receptor antagonists, antiacids or buffered drugs. Co-administration of ATV plus RTV partially attenuates this effect. ATV/r should be administered at least 10 h after H2-receptor antagonists. Proton pump inhibitors are not recommended in patients taking ATV;6,7 however, when unavoidable, ATV should always be administered boosted with RTV and the dose increased to 400 mg/day. Omeprazole should not exceed 20 mg/day (or equivalent when using other proton pump inhibitors) and therapeutic drug monitoring (TDM) would be desirable. In patients with prior history of antiretroviral failure, the co-administration of ATV and proton pump inhibitors should always be avoided. When other antiacids or buffered drugs might be administrated, ATV should be given 2 hours before or 1 hour latter.
ATV is both a substrate and inhibitor of CYP3A4 and P-gp. The drug also inhibits CYP2C8 and UGT1A1. Therefore, co-administration of ATV and drugs primarily metabolized by all these enzymes may result in increased plasma levels of the concomitant drug. On the other hand, drugs that induce CYP3A4 and/or P-gp may decrease ATV exposure, potentially compromising its therapeutic effect. Table 1 records the main drug interactions of ATV.
Main drug interactions with atazanavir.
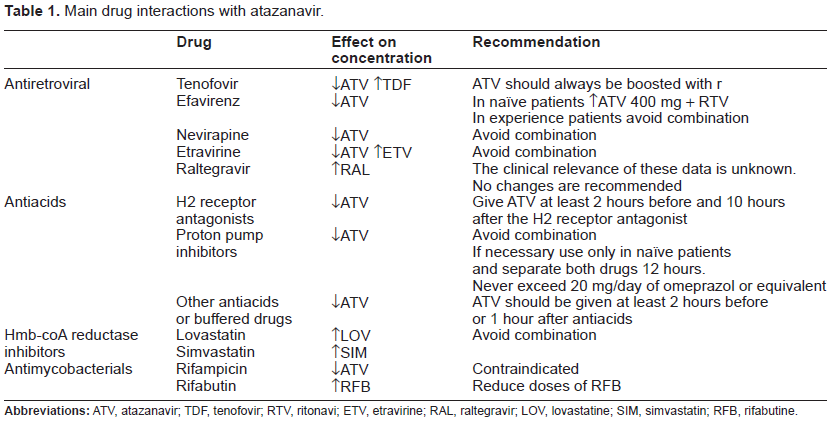
Drug interactions between ATV and other ARV agents are limited. However, the co-administration of ATV with other PIs is not recommended. Likewise, the use of ATV with nevirapine (NVP) and efavirenz (EFV) is neither recommended. However, in naive patients EFV might be given with ATV/r 400/100 mg. Co-administration of ATV/r with etravirine (ETV) is contraindicated, because ETV significantly decreases ATV Cmin. The mechanism of interaction between tenofovir (TDF) and ATV is unknown, but there is a 20% reduced ATV exposure. Therefore, ATV should always be boosted when given along with TDF.
PI pharmacokinetic enhancers other than RTV are being developed. Cobicistat (COBI) is a specific inhibitor of CYP3A with no antiretroviral activity. Moreover, COBI exhibits fewer clinically significant drug interactions than RTV. In a phase II trial, COBI was compared with RTV, both combined with ATV and fixed-dose TDF/FTC. 8 Comparable rates of viro-logical suppression and CD4 gains were obtained, being the tolerance of both combinations similar and satisfactory.
Therapeutic drug monitoring
There is large inter-individual variability in ATV disposition, even in the presence of RTV. This fact together with a low intra-individual variability supports the value of monitoring plasma concentrations of the drug. The definition of a therapeutic range for ATV plasma concentrations has been pursued in an attempt to minimize side effects, mainly hyperbilirubinemia, while ensuring maximal efficacy. 1 Both, virological success and hyperbilirubinemia have been associated with ATV plasma levels.9–11
Drug Resistance
The signature resistance mutation for ATV is an isoleucine to leucine substitution at amino acid residue 50 (I50L) within the HIV-1 protease gene. 12 This substitution appears to confer a unique viral phenotype in vitro, namely resistance to ATV along with increased susceptibility to other PIs. 13 It is the most frequent mutation seen at failure when failing on ATV in ART-naive patients. By contrast, in PI-experienced patients experiencing virological failure, the most common substitutions differ and are shared with other PIs. The most common changes include V32I, L33F/V/I, E35D/G, M46I/L, I50L, F53L/V, I54V, A71V//T/I, G73S/T/I, V82A/T/L, I84V, I85V, N88S and L89V/Q/M/T. 14
To ascertain if one virus isolate is resistant to ATV, a recent score has been proposed. 15 This score gives three points to changes that by their own compromise ATV response (I50L and N88S) and two or one point to other PI resistance associated mutations that reduce in a lesser extent the antiviral activity of the drug (Table 2). ATV resistance is considered when the isolate harbors 4 or more points. Of note, when L76V is present, the virus should be considered as hypersus-ceptible to ATV and one point has to be subtracted. 16
Interpretation of antiretroviral drug resistance mutations for atazanavir.
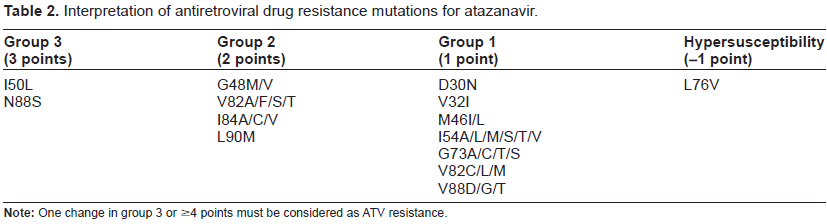
When ATV is boosted with RTV, as with other PIs, the barrier to resistance is increased and more than one drug resistance mutations are needed to impair the antiviral activity of the drug. This is particularly important for ARV-naïve patients or those who have never failed ARV therapy, as they may fail virologi-cally typically in the absence of PI resistance associated mutations.17–19 By contrast, failure to ATV without RTV boosting generally occurs with selection of primary ATV resistance associated changes. 20
As with other PIs, failure on ATV may be caused by lack of potency of the drug than by the acquisition of primary resistance mutations. 21 In contrast, in PI-experienced patients, the genotypic inhibitory quotient has been proven to be a good predictor of virological response to ATV, as it integrates both resistance mutations and drug plasma exposure. 10
Clinical Experience
Ritonavir-boosted atazanavir in antiretroviral-naïve patients
Most ART guidelines recommend the use of triple combinations including two nucleos(t)ide reverse transcriptase inhibitors (NRTI) plus a PI/r, a non-nucleoside reverse transcriptase inhibitor (NNRTI) or an integrase inhibitor (INI) as the treatment of choice for ARV-naïve patients. ATV/r is one of the preferred PI options (see Table 3).
Main trials that have tested atazanavir in antiretroviral-naïve patients.
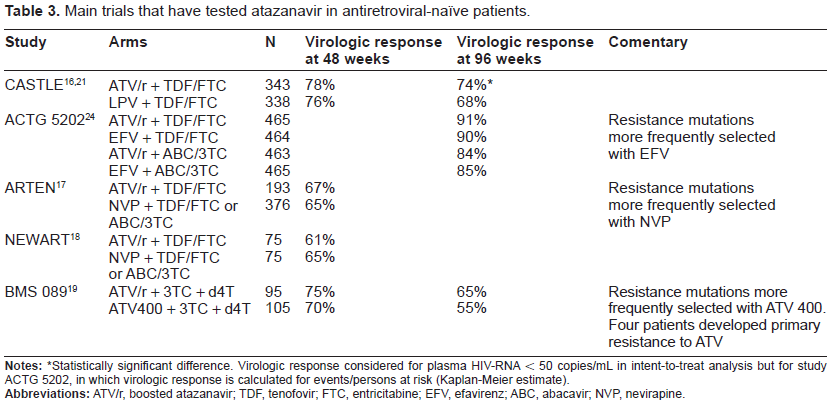
Statistically significant difference. Virologic response considered for plasma HIV-RNA < 50 copies/mL in intent-to-treat analysis but for study ACTG 5202, in which virologic response is calculated for events/persons at risk (Kaplan-Meier estimate).
CASTLE was the first study that supported the designation of ATV/r as a preferred PI-based regimen. 17 ATV/r once daily was compared with twice daily lopinavir/ritonavir (LPV/r), which was the gold standard PI at that time. Any PI was given along with TDF/FTC in 883ARV-naïve participants. This open-label, non-inferiority trial showed similar rates of virological response (<50 HIV-RNA copies/mL) at 48 weeks in the intention-to-treat analysis (78% on ATV/r vs. 76% on LPV/r) and similar CD4 gains (203 vs. 219 cells/µL, respectively). In patients with baseline viral load 100,000 HIV-RNA copies/mL there was a similar lower rate of virological response in both groups. The virological efficacy of ATV/r remained non-inferior as compared to LPV/r until week 96, 22 displaying undetectable viremia 89% and 88% of patients on treatment. However, in the intent-to-treat analysis, response rates were significantly higher with ATV/r than LPV/r (74% vs. 68%; P = 0.03). This was due to fewer discontinuations and better tolerance in the ATV/r than LPV/r arms. Moreover, in patients with more advanced HIV disease (CD4 counts <50 cells/µL), the virological response was higher for ATV/r than LPV/r.
The ACTG 5202 study provided further strong evidence for supporting the preferential use of ATV/r in combination with TDF/FTC as initial ARV therapy. 23 This large blinded study compared ABC/3TC to TDF/FTC along with either EFV or ATV/r. Due to a greater virological failure with ABC/3TC than TDF/FTC in patients with plasma HIV-RNA >100,000 copies/mL, blinded was stopped prematurely. 24 In patients with lower viral load, time to virological failure was similar among the four arms of the study. Anyway, ATV/r demonstrated similar efficacy to EFV when combined with either TDF/FTC or ABC/3TC at 96 weeks. 25 ATV/r seemed to be safer and more tolerable than EFV when combined with ABC/3TC but similar when combined with TDF/FTC. By contrast, more renal toxicity was seen in patients receiving ATV/r than EFV in the TDF/FTC arm. Another benefit of ATV/r was that primary drug resistance mutations and specifically NRTI resistance changes emerged more often in patients experiencing virological failure in the EFV arm.
More recently, ATV/r has been compared with NVP in ARV-naïve patients.18,19 At 48 weeks, the combination of these agents with TDF/FTC gave similar response rates. Outside clinical trials, ATV/r has also demonstrated durability and effectiveness in most ARV-naïve patients. For example, a multicenter retrospective study conducted in Canada that enrolled 176 patients whose first HIV combination included ATV/r showed that the mean durability of treatment was 33.5 months. Only 10% of patients discontinued treatment due to toxicity (hyperbilirubinemia), sub-optimal virological response, or any other reason. 26
Unboosted atazanavir in antiretroviral-naïve patients
The BMS 089 study randomized 200 patients to receive ATV 400 mg/day vs. ATV/r (300/100 mg/day), both combined with stavudine and lamivudine. At 48 and 96 weeks, response rates (plasma HIV-RNA < 50 copies/mL) by ITT were similar in both treatment arms with a trend in favor of ATV300/r vs. ATV400 (75 vs. 70 and 65% vs. 55%, respectively). 20 However, more patients experienced virological failure on ATV400 (20/105) than ATV300/r (5/95). Moreover, virological failure in the ATV300/r arm was not associated with selection of phenotypic resistance to the drug, whereas 4 patients failing on ATV400 developed phenotypic resistance to ATV, in all cases with the substitution I50L. The NRTI resistance associated mutation M184V also arose more frequently in patients failing on ATV400 than ATV300/r. The low barrier to resistance of ATV without RTV boosting could limit its use in patients intolerant to RTV. 1
Anyway, the BMS 089 study demonstrated the long-term efficacy and tolerability of boosted and unboosted ATV-based regimens in HIV-infected, treatment-naive patients. The immunological benefit continued to improve after the first year of therapy.
Atazanavir in patients with prior antiretroviral failure
Few studies have evaluated the use of ATV in patients with prior HAART virological failure. The AI424 045 study demonstrated the non-inferiority of ATV/r versus LPV/r in patients who had previously failed to at least two PIs. 27 At 96 weeks of therapy, the proportion of patients with plasma HIV-RNA < 400 or < 50 copies/mL were 44% and 33% with ATV/r versus 46% and 36% with LPV/r. CD4+ counts increased by a median of 160 and 142 cells/µL, respectively. 28 The relatively low proportion of patients with undetectable viremia in both arms largely reflected that nearly 90% of the study population had one primary PI resistance mutation and 50% had three or more PI resistance mutations. At the time this study was conducted, no drugs blocking new targets as raltegravir (RAL) or maraviroc (MAR) were available.
The virological efficacy of ATV/r-containing regimens as rescue therapy was also evaluated in a French cohort. At 24 weeks, 24% of patients had plasma HIV-RNA 400 copies/mL and 12% had stopped treatment. Again, most patients where heavily pretreated and received only 2 NRTI along with ATV/r. 29
Unboosted ATV is not recommended in patients with prior virological failure to another HAART regimen because it provides less virological suppression than regimens containing PI/r, including LPV/r. 30
Atazanavir as part of simplification regimens
The long-term adverse effects and the complex schedules of some ARV treatment combinations have led to explore the benefit of strategies aimed to simplify therapy. In this regard, ATV-based regimens offer several therapeutic advantages, including once-daily dosing schedule, low pill burden and lack of undesirable metabolic abnormalities. Several studies have demonstrated the efficacy of ATV in this setting (Table 4).
Main trials that have tested atazanavir in simplification strategies.
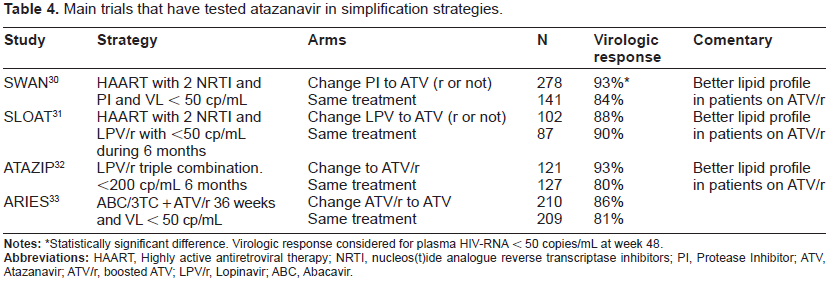
Statistically significant difference. Virologic response considered for plasma HIV-RNA < 50 copies/mL at week 48.
In patients who had virological suppression while receiving another PI-based regimen, a switch to ATV was similar to stay with the original combination in terms of keeping viral suppression.31–33
The SWAN study included 419 patients with unde-tectable viral load who were receiving different PI-based therapies. 31 Patients were randomized to keep the same treatment or switch to ATV with or without RTV (depending on whether they received also TDF). At 48 weeks, virological failure (>50 HIV-RNA copies/mL) in an ITT analysis occurred in 7% of patients with ATV and 14% of controls (P = 0.04). Patients on ATV showed greater reductions in cholesterol and triglyceride levels. Similar results were observed in ATAZIP and SLOAT studies that included only patients that were on triple therapy with LPV/r.32,33
A simplification from ATV/r to unboosted ATV has also been explored. 34 In this trial, patients who had started treatment with ABC/3TC and ATV/r and had undetectable viremia for 36 weeks were randomized to receive unboosted ATV plus ABC/3TC or continue with the same treatment. At week 84, 86% of 210 patients in the ATV group and 81% of 209 patients in the ATV/r group maintained plasma HIV-RNA < 50 copies/mL. None of the failing patients selected viruses with major PI resistance mutations.
In the real life, simplification from other PIs to ATV/r has also been proven to be efficacious and safe, allowing management of undesirable metabolic abnormalities without increasing the risk of virological failure in well controlled pretreated patients. 35
During the last few years, PI/r monotherapy as simplification strategy has been explored.36,37 Although more evidence is needed, some HIV guidelines have mentioned this possibility in special circumstances and mainly with darunavir (DRV)/RTV or LPV/r 38 while other guidelines don't recommend monotherapy in any situation. 1 Monotherapy with ATV/r has been explored in the ACTG 5201 and the ATARITMO studies.39,40 Both studies had only one arm of patients who switched from triple therapy to PI/r monotherapy. At 48 weeks, the virological response was still satisfactory and selection of resistance was low in the monotherapy arm. However, the small number of patients examined and the lack of control arms precluded drawing any conclusion. Evenmore, another potential drawback for the use of ATV/r as monotherapy is its limited penetration in cerebrospinal fluid (CSF). 41
Atazanavir in pregnancy
ATV is classified within category B by the Food and Drug Administration pregnancy score, which means that animal studies have failed to demonstrate a risk for fetuses of mothers exposed to ATV, although adequate well-controlled studies in human pregnant women are warranted. 42
A recent study performed in 41 pregnant women has noticed that plasma concentrations of ATV are similar to non-pregnant adults when using recommended doses of the drug. The safety and efficacy was also demonstrated, with no cases of vertical transmission. 43 These data have supported the recent approval of ATV/r as part of antiretroviral therapy during pregnancy.
Safety
ATV is generally well tolerated, as shown by the fact that only 5%-10% of patients discontinued the drug due to adverse events in the main registrational studies. 6 Indirect bilirubin elevation is the most frequent side effect reported with ATV therapy and is due to inhibition of the UGT1A1 enzyme. Unconjugated bilirubin elevation is frequently seen within the first months on ATV therapy and tends to slightly decline thereafter due to metabolic compensation mechanisms. Although increases in bilirubin are seen in 22% to 49% of ATV treated patients, jaundice is uncommon (10%) and leads to discontinuation in only 2% of treated patients.17,25,26 Hyperbilirubinemia seems to be associated to ATV plasma levels and genetic factors, and is more frequent when ATV is boosted with RTV. 44 Of note, hyperbilirubinemia is completely reversible after stopping ATV. With respect to direct liver toxicity, hypertransaminasemia grades 3-4 is uncommon in patients treated with ATV alone or boosted with r, occurring in less than 5% of patients.17,25,26 Even in patients coinfected with hepatitis B or C viruses, rates of hepatotoxicity attributable to ATV were rather low.45,46
PI use has been largely associated to metabolic abnormalities, including dislipidemia, lipodystrophy and insulin resistance. 47 However, the most recent PIs, such as ATV and darunavir (DRV), have showed a better metabolic profile. For example, patients in the CASTLE study on ATV/r showed greater increments in fasting total cholesterol, non-high density lipoprotein cholesterol and triglycerides at 96 weeks than patients receiving LPV/r. 48 The proportion of individuals on ATV/r with total cholesterol and LDL-c within de desirable levels (< 200 mg/dL and 130 mg/dL, respectively) was 83% and 84%. These figures in the LPV/r group were 62% and 74%, respectively.
The favorable effects of ATV on lipids compared to other PIs have been recognized in most simplification studies. Significant reductions were seen in total cholesterol, triglycerides and non-HDL-c in patients switched to boosted or unboosted ATV compared to patients remaining on their current PI regimen.31,32 Of note, the benefit in the lipid profile is only minimally affected by RTV boosting. 49
ATV does not alter fasting glucose or insulin levels significantly during 48 weeks of treatment in HIV-infected patients.20,50 In a small uncontrolled study that included 21 antiretroviral-experienced patients who had metabolic disturbances while receiving PIs, switching to unboosted ATV led to significant improvements in fasting glucose, 120-minute glucose levels, insulin and C-peptide. 51
Another well-known side effect of most classical PIs is gastrointestinal complaints. However, ATV with or without RTV has uniformly demonstrated less of these symptoms. In the CASTLE study, only 3% of patients allocated to ATV/r had grade 2-4 diarrhea, 5% had nausea and < 1% had vomiting. Not surprisingly, GI tolerance was significantly better with ATV/r than LPV/r. 47
ATV may occasionally cause atrioventricular conduction abnormalities, generally recognized as PR prolongation. 52 These effects are generally asymptomatic and limited to first-degree AV block. Anecdotal cases of second and third degree AV block have been reported. 6
Lastly, ATV has been associated sporadically with renal toxicity. First, cases of nephrolithiases have been reported. 53 This complication resembles the well-known side effect of IDV, which was able to produce kidney stones. In some instances, ATV crystals have been demonstrated in urine. However, this complication is very rare. In one retrospective study the prevalence of ATV-associated urolithiasis was 0.97%. 54 Patients with low water intake, high urinary pH, and prior history of urinary stones are at higher risk for experiencing ATV-associated urine crystallization. On the other hand, TDF associated tubulopathy may be increased in the presence of PIs boosted with RTV, including ATV. 55 By similar mechanisms, other toxicities of TDF, such as changes in bone mineral density, have been reported to be more common in patients taking ATV/r than EFV. 56
In summary, ATV/r is a good therapeutic option in antiretroviral-naive patients as well as an attractive alternative in simplification strategies, with proven long-term benefit. ATV may preserve NNRTIs and other PIs for the future due to its unique resistance profile. In patients with multiple prior antiretroviral failures, especially when including other PIs, the use of ATV/r must be cautious and only guided by resistance testing and accompanied by other active antiretroviral agents.
Author Contributions
Conceived and designed the experiments: LMC, VS. Analysed the data: LMC, VS. Wrote the first draft of the manuscript: LMC. Contributed to the writing of the manuscript: LMC, VS. Agree with manuscript results and conclusions: LMC, VS. Jointly developed the structure and arguments for the paper: LMC, VS. Made critical revisions and approved final version: LMC, VS. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
