Abstract
Objective
To review the efficacy and safety of armodafinil for the treatment in patients with excessive sleepiness.
Data sources
Literature was accessed via MEDLINE (1966–February 2011) and EMBASE (1980–February 2011) using the medical subject heading terms armodafinil and sleep.
Study selection and data extraction
All English-language, peer-reviewed publications were analyzed for relevance. Studies appropriate to the objective were evaluated if they prospectively assessed the effectiveness of armodafinil in patients with excessive sleepiness in a multiple-dose, comparator trial. The literature search identified published reports of 7 randomized, placebo-controlled studies.
Data synthesis
Armodafinil is a wakefulness-promoting agent used in the treatment of patients with excessive sleepiness and is the R-enantiomer of modafinil. Studies have demonstrated that armodafinil may be effective and safe when used in patients with obstructive sleep apnea, circadian rhythm disorders, and narcolepsy. Although the studies identified had limited sample sizes, they consistently demonstrated improvement on various efficacy rating scales for sleep. Study durations ranged from 3 days to 12 weeks, with doses ranging from 50 mg to 250 mg daily. The most common adverse effects associated with armodafinil therapy were headache, nausea, diarrhea, dizziness, and anxiety, although increases in systolic blood pressure and pulse rate were also reported.
Conclusion
Current evidence suggests that armodafinil significantly improves wakefulness in patients with excessive sleepiness in studies up to 12 weeks. Long-term effectiveness and safety cannot be adequately determined at this time. Although armodafinil should be considered safe for use, health-care providers should monitor for adverse effects, particularly cardiovascular complications.
Background
Excessive sleepiness (ES) is a common condition that is associated with significant morbidity and increased mortality risk to the patient and the general public.1–4 ES is characterized by an increased propensity to fall asleep when sleep is inappropriate. Because there is no clear consensus defining the point in which sleepiness becomes excessive, the reported prevalence of ES ranges from 0.3%–35.8%. 5 However, the actual prevalence may be higher since ES often goes unrecognized in clinical practice. Adverse events associated with ES are frequently serious and irreversible and dramatically affect social, familial, work, and cognitive performance. Negative health-related consequences are also associated with ES and include cardiovascular disease, metabolic dysfunction, and mood impairment. 6 Besides the potential negative effects on the patient, ES has major public health implications. 7 It has been reported that ES doubles the risk of occupational accidents and increases the risk of motor vehicle accidents.2,3,8 Given these detrimental consequences, early recognition and appropriate treatment of ES is imperative.
ES is associated with a variety of medical, neurological, and psychiatric disorders and may occur because of insufficient sleep, disrupted sleep, circadian rhythm misalignment, or pathological abnormalities of the central nervous system (CNS). Sleep apnea syndromes, including obstructive sleep apnea (OSA), are common diagnoses made in patients with ES. 7 Examples of circadian rhythm sleep disorders include shift work disorder (SWD) and jet lag disorder, while narcolepsy is a hypersomnia of central origin. 7 Other conditions associated with ES include: Parkinson's disease, fibromyalgia, multiple sclerosis, traumatic head injury, and depression. 5 In addition to disorders, medications and substances of abuse may contribute to ES including antihistamines, benzodiazepines, narcotics, and cannabis. 7
Identification of ES is critical in improving the well-being of the patient and preventing harm to the general public. Several subjective and objective measures are utilized to identify ES and assess response to therapy. The Epworth Sleepiness Scale (ESS) is a self-administered questionnaire assessing the patient's likelihood of dozing off or falling asleep in eight specific situations. Using a scale of 0 (would never doze) to 3 (high chance of dozing), the patient rates their likelihood of falling asleep in each situation. A score of 10 or more suggest some form of abnormal or pathological sleepiness. 9 The Clinical Global Impression of Change (CGI-C) and the Patient Global Impression of Severity (PGI-S) are two similarly structured subjective assessments which measure the clinician's and patient's perception of symptom severity, respectively. Each is scored on a 7-point scale, ranging from very much worse to very much improved. 10 Two objective measures of sleepiness/wakefulness are the Multiple Sleep Latency Test (MSLT) and the Maintenance of Wakefulness Test (MWT). The MSLT is a validated measure of the propensity to fall sleep, while the MWT is a validated measure of the ability to remain awake for a defined time. 11 The MSLT consists of 4–5 nap opportunities performed at two hour intervals throughout a day. The first nap opportunity begins 1.5 to 3 hours after waking up. During each nap opportunity, patients are monitored for sleep onset while lying in a dark, quiet room for 20 minutes. Mean sleep latency (MSL) or the average amount of time it takes to fall asleep is calculated from the 4 to 5 nap opportunities. A MSL of 10 to 15 minutes indicates mild or even normal daytime sleepiness, while a MSL of <5 minutes indicates severe sleepiness. 9 The MWT consists of four, forty minute trials performed at two hour intervals. Similar to the MSLT, the first trial begins 1.5 to 3 hours after waking up. During each trial, patients are monitored for the ability to remain awake while seated in bed in a dark, quiet room. A MSL of <8 minutes indicates abnormal sleepiness. 11
Behavior and pharmacologic treatment options are essential in alleviating ES, along with appropriate management of the underlying disorder. Regardless of the underlying etiology of ES, patients should be educated on proper sleep hygiene. Good sleep hygiene consists of establishing a regular sleep/ wake schedule; minimizing noise, light, and extreme temperature during sleep; and avoiding large meals, stimulants, alcohol, and exercising before sleep. 6 Patients with narcolepsy should be encouraged to take short 15 to 20 minute naps every four hours throughout the day to reduce ES. 12 Sodium oxybate is a schedule III CNS depressant indicated for the treatment of cataplexy and daytime sleepiness in patients with narcolepsy. Sodium oxybate improves nocturnal sleep, which in turn, reduces daytime sleepiness. The full effects on daytime sleepiness may take up to two to three months, thus stimulants are often added at the beginning of therapy. 12 Medications currently available to promote wakefulness and alertness in patients with ES include amphetamines, modafinil, and armodafinil. Amphetamines (dextroamphetamine, methamphetamine, and methylphenidate) are CNS stimulants approved for the treatment of ES in patients with narcolepsy. The use of these agents has been limited by their high abuse potential (schedule II) and significant adverse effect profile, most notably cardiovascular adverse events. 5 Modafinil is a wake promoting agent approved by the FDA in 1998 and is structurally and pharmacologically different from the amphetamines. It is indicated for the treatment of ES in patients with narcolepsy and SWD, and as adjunct therapy in patients with OSA receiving continuous positive airway pressure (CPAP). 13 In comparison to amphetamines, modafinil has a lower abuse potential (schedule IV) and a lower risk for cardiovascular events. For these reasons, modafinil has replaced amphetamines as preferred therapy for ES. 14 In the United States, modafinil is dosed once daily in the morning for patients with narcolepsy and OSA, and for patients with SWD, it is dosed one hour before the start of the work shift. 13 However, some patients may experience late day sleepiness and require higher doses or more frequent dosing which may increase the risk of adverse effects. 15 Recently, the European Medicines Agency (EMEA) completed a review of the safety and effectiveness of modafinil. The review was initiated because of concerns that the medicine was associated with serious psychiatric disorders and life-threatening skin reactions. Based on their review, the EMEA required all indications of modafinil be removed from the product information except for treatment of narcolepsy where the benefits continue to outweigh the risk. 16 In addition, the agency recommended that modafinil should not be used in patients with uncontrolled hypertension or irregular heart beat. 16 Armodafinil (Nuvigil®; Caphalon, Inc; Frazer, PA), approved in 2007, is the newest wafefulness-promoting agent and will be the focus of this review.
Armodafinil
Armodafinil is the longer-acting R-enantiomer of modafinil, unlike modafinil which is a racemic mixture consisting of equal amounts of R- and S-enantiomers. It is indicated for the treatment of ES associated with treated OSA, SWD, and narcolepsy. 17 Similar to modafinil, armodafinil has a lower abuse potential (schedule IV) and a lower risk of cardiovascular adverse effects compared to amphetamines. 5 The exact mechanism of action of modafinil and armodafinil is unknown. CNS activation of these agents is specific to the hypothalamus, prefrontal cortex, and anterior cingulated cortex. Armodafinil has not been shown to interact with many of the receptors and enzymes known to be involved in the regulation of sleep/ wake. 18 However, in vitro data demonstrate that armodafinil binds to the dopamine transporters and inhibit dopamine transport. 17
When absorbed after oral administration, armodafinil exhibits linear pharmacokinetics and peak plasma concentrations are reached two hours after administration in the fasted state. When administered with food, peak plasma concentrations are delayed by two to four hours. 19 The apparent volume of distribution after a single dose is 42 liters. Although protein binding data are unavailable for armodafinil, in vitro studies of modafinil have shown 60% of the drug is bound to protein, mainly to albumin. Thus drug interactions of armodafinil with highly-protein bound drugs are considered to be minimal. 17 Armodafinil is predominantly metabolized by the liver. Amide hydrolysis and sulfone formation by cytochrome P450 (CYP) 3A4/5 are the predominant metabolic pathways resulting in two primary metabolites, R-modafinil acid and modafinil sulfone. 17 The terminal half-life of armodafinil is approximately 15 hours, which is the same as modafinil. 19 Even though the half lives of each agent are the same, pharmacokinetic studies have shown that armodafinil produces a higher plasma drug concentration late in the day and is associated with less fluctuation and swing in plasma concentration across a 24-hour dosing interval compared to modafinil.20,21 Late-day armodafinil plasma concentrations were reported to be 44% higher than modafinil concentrations on a milligram-to-milligram basis. In addition, plasma concentration fluctuation and swing of armodafinil were 28% and 42% less, respectively, when compared to modafinil over a 24-hour interval. 20 A recent pharmacokinetic study reported systemic exposure of armodafinil is increased in elderly participants. 22 Systemic exposure, measured by steady-state AUC and Cmax values, was 15% greater in elderly individuals (≥65 years) compared to young men (18–45 years). The increased systemic exposure in the elderly did not result in an increase number of adverse effects. Based on these data, lower doses of armodafinil may be warranted in older patients.
In vivo studies have demonstrated that armodafinil is a moderate inducer of CYP3A4 and a moderate inhibitor of CYP2C19, thus drug-drug interactions are possible. 23 Armodafinil can increase serum concentrations of drugs that are substrates for CYP2C19 (eg, omeprazole and phenytoin), while decreasing serum concentrations of drugs that are substrates of CYP3A4 (eg, cyclosporine and ethinyl estradiol). Dosage adjustments may be warranted, however there are no formal recommendations. Since armodafinil can reduce the efficacy of birth control pills, childbearing women should be instructed to use another form of contraception while on therapy with armodafinil and for one month after discontinuation. 17
Armodafinil is available as 50 mg, 150 mg, and 250 mg tablets. Dosing for patients with OSA and narcolepsy is 150 mg or 250 mg every morning. In patients with OSA, doses up to 250 mg have not shown any additional benefit over the 150 mg dose. For treatment of ES associated with SWD, the recommended dose is 150 mg administered 1 hour before the start of the work shift. Dosage adjustments are not warranted in patients with renal insufficiency, however the dose of armodafinil should be reduced by 50% in patients with severe hepatic impairment. 17
Literature Review
A comprehensive search of MEDLINE (1966–February 2011) and EMBASE (1980–February 2011) was utilized to identify all relevant trials using the medical subject heading terms armodafinil and sleep. Results were limited to peer-reviewed studies conducted on humans and published in English. Additional articles were obtained by manually searching recent reviews for relevant articles. To strengthen the analysis of efficacy, references were evaluated only if they prospectively assessed the effectiveness of armodafinil in patients with ES in a multiple-dose, comparator trial. The literature search identified published reports of 7 randomized, placebo-controlled studies (Table 1).24–30
Summary of prospective studies of armodafinil for the treatment of excessive sleepiness.
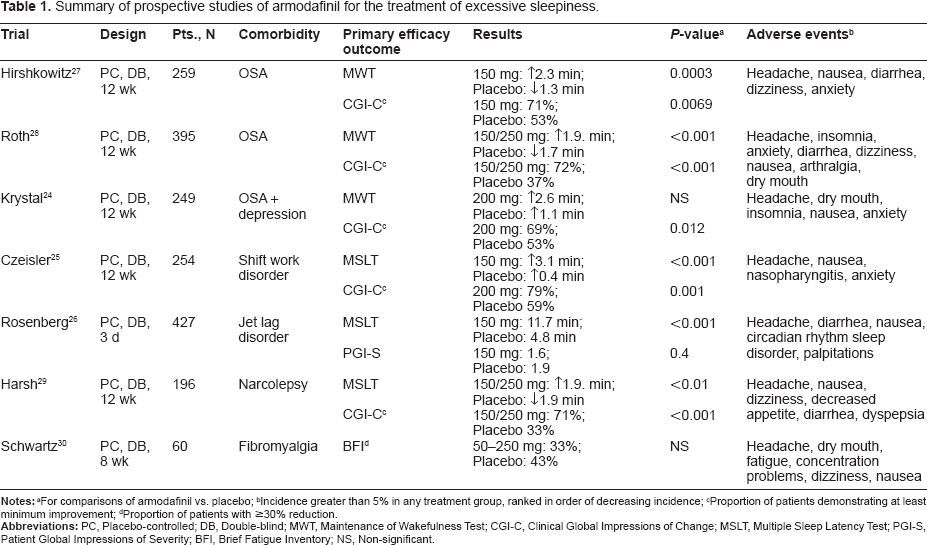
Incidence greater than 5% in any treatment group, ranked in order of decreasing incidence;
Proportion of patients demonstrating at least minimum improvement;
Proportion of patients with ≥30% reduction.
Obstructive sleep apnea
OSA is a common sleep disorder that is characterized by breathing cessation or near cessation due to recurrent episodes of partial or complete collapse of the upper airway during sleep. These events lead to recurrent arousals from sleep. 31 ES is a common symptom of patients presenting with OSA. 32 The estimated prevalence of patients with OSA with accompanying ES is 4% for adult men and 2% for adult women and risk factors include: advanced age, excess body weight, male gender, Asian descent, smoking and alcohol consumption.31,32 In addition, patients with a first-degree relative with OSA are at an increased risk for development of the disorder as well. 31
To evaluate the treatment of armodafinil in patients with ES associated with OSA, three studies were conducted, individually using a 12-week evaluation period. Each study was designed as a prospective, randomized, double-blind, placebo controlled study in patients who were currently controlled on stable nasal continuous positive airway pressure (nCPAP). Each included patients with a baseline Clinical Global Impression of Severity (CGI-S) rating of ≥4 and an ESS score of ≥10 and one study required patients to be diagnosed with comorbid major depressive disorder or dysthymic disorder and have a Hamilton Depression Rating Scale score of <17.24,27,28 Patients were randomized to armodafinil or matching placebo, initiating at 50 mg per day, taken in the morning, and titrated up every two to three days until target dosing was achieved. Primary endpoints were assessed the final visit (either week 12 or at study termination) as mean change from baseline for the MWT and the proportion of patients with at least minimal improvement on the Clinical Global Impressions of Change (CGI-C). Secondary endpoints varied for each study.24,27,28
Hirshkowitz et al studied 259 patients who were titrated to a final dose of 150 mg. 27 At 12 weeks, the primary endpoints of change from baseline for the MWT and the proportion of patients with at least minimal improvement on the CGI-C were significantly improved for the armodafinil group. With regards to the secondary endpoints, armodafinil increased episodic secondary memory (P = 0.0102), decreased unintended sleep episodes (P < 0.001) and naps (P = 0.0016), as well as improved the ESS total score (P < 0.01). At the final visit, late-day sleep latency measured by MWT across the last three time tests (1500, 1700, and 1900) did not demonstrate a significant difference compared to placebo. It was also shown that speed of memory, continuity of attention, and power of attention were not statistically significant. All adverse effects were mild or moderate in nature, with no serious adverse effects. Five patients (3.8%) discontinued due to adverse effects, compared to six (4.5%) in the placebo group, although specific causes of discontinuation were not defined. 27
Roth et al studied a total of 395 patients, with patients divided into armodafinil 150 mg, armodafinil 250 mg, or matching placebo. 28 At the final visit, MWT sleep latency was statistically more significant in the armodafinil-combined group compared with placebo and there were significantly more patients who showed at least minimal improvement on the CGI-C when compared to placebo. Additional secondary efficacy assessments demonstrated significant improvement in the ESS total score (P < 0.001), Brief Fatigue Inventory (BFI) scores (P < 0.05), unintended sleep episodes (P < 0.001), and daily naps (P < 0.01). But, the Cognitive Drug Research battery was not considered significant at the final visit, as well as late-day sleep latency across the evening time tests of 1500, 1700, and 1900. Headache was reported as the most common adverse effect, occurring in 17.6% of patients compared to 8.6% with placebo and was the most occurring adverse effect leading to discontinuation in the 250 mg dose, with nausea most commonly leading to discontinuation in the 150 mg treatment group. Overall the discontinuation rate of the armodafinil groups were 7.5% in patients randomized to150 mg and 11.5% of patients randomized to the 250 mg treatment groups. 28
In order to further assess secondary endpoints, the results of the studies by Roth and Hirshkowitz et al were pooled post-hoc. 33 When analyzed together, it was shown that armodafinil significantly improved the quality of episodic secondary memory (P < 0.05), patients’ ability to engage in activities of daily living (P < 0.0001), and was able to show a significant reduction in fatigue (P < 0.01). Armodafinil was also able to demonstrate an improvement in the patients’ ability to maintain late-day wakefulness during the evening time tests of 1500, 1700, 1900 when compared with placebo (P < 0.05). But, statistical significance was not demonstrated at the final visit for speed of memory, power of attention, or continuity of attention. As with the original studies, adverse effects were not tested for statistical significance. 33
To study the use of armodafinil in patients with obstructive sleep apnea and comorbid depression, Krystal et al enrolled 249 patients who were receiving stable monotherapy with a selective serotonin reuptake inhibitor or serotonin norepinephrine reuptake inhibitor for at least eight weeks prior to screening. 24 Patients randomized to the treatment group were titrated up to a target dose of 200 mg, but could be further increased to 250 mg if recommended by the investigator's perception of efficacy. At the end of the 12-week study period, armodafinil demonstrated a significantly greater proportion of patients with at least minimum improvement on the CGI-C, but improvement in sleep latency as measured by the MWT was not shown to be significant. The secondary endpoint of change from baseline to final visit in ESS score also demonstrated efficacy for the armodafinil group (P = 0.003). All adverse effects were generally determined to be mild to moderate in severity and the psychiatric adverse events of insomnia (7%), anxiety (5%), and worsening major depressive disorder (2%) occurred in greater than one patient. One patient in the armodafinil group reported a serious adverse event of atypical chest pain, but it was not considered by the investigator to be related to the study drug. Discontinuations due to adverse-related events, such as headache, dyspnea, dry mouth and disturbance of attention, occurred in 9.6% of the armodafinil group compared to 5.6% of the placebo group. 24
Circadian rhythm disorders
Circadian rhythm disorders are characterized by a persistent or recurrent pattern of sleep disturbances due to a misalignment between the endogenous circadian rhythm and exogenous factors that affect the timing or duration of sleep. Shift work disorder and jet lag disorder are two distinct circadian rhythm disorders recognized in the International Classification of Sleep Disorders (ICSD-2). 34 The exact prevalence of SWD is unknown, however an epidemiologic survey found that 32.1% of night shift workers and 26.1% of rotating shift workers met minimum criteria for SWD. 35 Data have indicated that female shift workers along with older shift workers may have a higher tendency to be intolerant of shift work. 34 Due to ES, workers may be at an increased risk of motor vehicle crashes during his or her morning commute.36,37 Jet lag disorder occurs from the crossing of time zones too rapidly for the circadian rhythm to adjust. The intensity and duration of the disorder depends on the number of time zones crossed, the direction of travel, and the ability to sleep while traveling. 34 Compared to westbound travel, eastbound travel places the patient at an increased risk of the disorder. Similar to SWD, the exact prevalence of jet lag disorder is unknown. Jet lag is usually self-limiting, resolving after a couple of days, but nevertheless, may negatively impact the person's life, especially those individuals involved with frequent long-distance traveling. 6
To assess the use of armodafinil in patients with shift work disorders, patients who worked 5 or more night shifts per month and had a CGI-S rating of ≥4 were included in a 12-week, randomized, double-blind, placebo controlled study. 25 Study participants in the active treatment group were randomized to armodafinil 150 mg, given prior to each night shift and no later than 11 pm. A total of 254 patients were enrolled in this study. At the conclusion of the study, armodafinil was shown to significantly improve sleep latency assessed by the MSLT, as well increased the proportion of patients who showed at least minimal improvement in the CGI-C rating when compared to placebo. As secondary efficacy endpoints, armodafinil was associated with significant improvement in unintended sleep episodes, intended sleep episodes, maximum level of sleepiness, level of sleepiness during commute home, and number of mistakes, near misses, or accidents during the night shift (P < 0.05) and armodafinil improved mean power of attention, simple reaction time, and continuity of attention (P < 0.05). It was also shown that sleep latency measured by MSLT was significantly improved at all 5 time points of 2400, 0200, 0400, 0600, and 0800 (P < 0.05). In this study, headache was the most common adverse effect and most were considered mild-to-moderate by the investigator. A total of seven patients (5.7%) in the armodafinil group and four (3.3%) in the placebo group discontinued due to adverse effects. One patient with a history of depression developed suicidal ideation, which was considered to be possibly related to the study medication. 25
In patients with ES associated with jet lag disorder, a randomized, double-blind, placebo-controlled study was conducted with 427 participants. 26 Eligible patients were flown overnight on a nonstop, privately chartered flight to their destination study center in France, creating a time zone difference of 6 hours. Participants were given no instructions regarding sleep on the flight and may or may not have chosen to sleep. At the study center, patients were randomized to armodafinil 50 mg, 150 mg, or placebo given at 8:00 am on each of the three study days. When armodafinil 150 mg was compared to placebo for the primary efficacy endpoints of MSLT and the Patient Global Impression of Severity (PGI-S), it was determined that armodafinil demonstrated a significant benefit for both efficacy measures. When armodafinil 50 mg was compared to placebo as a secondary assessment using the same efficacy measures, the smaller dose of armodafinil was able to show a significant improvement in MSLT (P < 0.001), but not for the PGI-S (P = 0.80). Both doses of armodafinil also demonstrated significant improvements in the Karolinska Sleepiness Scale (KSS) when compared to placebo (P < 0.001 for both groups). For this study, there were no serious adverse events reported and four armodafinil patients (1.4%) withdrew due to adverse effects compared to 3 patients (2.1%) in the placebo group. 26
Narcolepsy
The ICSD-2 classifies narcolepsy as a hypersomnia of central origin. Narcolepsy is a rare sleep disorder characterized by excessive daytime sleepiness and unwanted episodes of sleep attacks, with an estimated prevalence of 0.04%.12,38 These attacks can occur several times a day and can last from a few seconds to several minutes. Cataplexy is another common manifestation of narcolepsy, occurring in 60%–70% of patients. Current evidence into the pathophysiology of narcolepsy suggests loss of hypocretin-containing neurons in the hypothalamus via autoimmune-mediator cell death of hypocretin neurons. 12
To assess the efficacy and safety of armodafinil for the treatment of ES in patients with narcolepsy, a 12-week, randomized, double-blind, placebo-controlled study was conducted with 196 patients. 29 Patients were included if they exhibited a MSL of ≤6 minutes on the MSLT and a CGI-S rating of ≥4. Patients were randomized to armodafinil 150 mg, 250 mg, or placebo and titrated to target dose as previously described. At the conclusion of the study, armodafinil demonstrated a significant improvement compared to placebo in the primary efficacy endpoints of mean change in the MWT at 150 mg, 250 mg, and armodafinil-combined groups (P < 0.01 for all comparisons) and for the proportion of patients with at least minimal improvement in their CGI-C for the 150 mg, 250 mg, and combined dosage groups (P < 0.0001 for all comparisons). For the secondary efficacy endpoints, armodafinil produced statistically significant improvements in the ESS at all dosage strengths (P < 0.01), for the CDR at 150 mg and in the armodafinil-combined groups (P < 0.05), for the BFI (P < 0.01), and armodafinil reduced the mean number of unintended sleep episodes (P < 0.001) and mean number of naps (P = 0.0039), but did not significantly reduce the mean number of mistakes/near misses/accidents. Also, late-day sleep latency measured by MWT across the last three time tests (1500, 1700, and 1900), demonstrated a significant improvement for the armodafinil 150 mg and armodafinil-combined groups compared to placebo (P < 0.05). In regards to safety, 1 severe adverse effect occurred during the study, but was considered unlikely to be related to armodafinil use and 7 patients (5.3%) in the armodafinil group discontinued due to adverse effects compared to 1 patient (1.5%) in the placebo group. 29
Fibromyalgia
Fibromyalgia is a chronic pain disorder characterized by widespread pain and tenderness, along with sleep disturbances and fatigue. The exact etiology and pathophysiology is not well understood, but proposed theories include: systemic nociceptive and central pain processing abnormalities, autonomic nervous system dysfunction, hypothalamic-pituitary-adrenal axis dysfunction, neurotransmitter abnormalities, and an inability to sustain deep sleep. 39 The estimated prevalence of fibromyalgia is around 2% in the United States and women are more affected than men with prevalence rates of 3.5% and 0.5%, respectively. 40 Fatigue and sleep disturbances are common components to fibromyalgia with up to 99% of patients reporting poor sleep quality. 41
The use of armodafinil to treat patients with ES associated with fibromyalgia was described in a letter to the editor by Schwartz et al. 30 This study was designed as an 8-week, randomized, double-blind, placebo controlled study of 60 subjects with fibromyalgia. Patients were given a flexible dose of armodafinil 50–250 mg, which could be titrated based on perceived effectiveness and tolerability. The primary efficacy endpoint of this study was the proportion of patients with a 30% or greater reduction in the Brief Fatigue Inventory (BFI), which did not demonstrate a statistically significant difference compared to placebo. No secondary endpoints were described. The most common adverse effect associated with armodafinil was headache and 6 patients (22%) in the armodafinil group and 4 patients (14.3%) in the placebo group discontinued the study due to adverse effects. 30
Discussion
ES has the potential to negatively impact a person's quality of life, through effects on family, work, and social function and can result in health-related consequences.5,42 Such an impact reinforces the inherent need for appropriate evidence-based treatment of ES. Before a pharmacological intervention is considered in a patient experiencing ES, a comprehensive evaluation should be conducted to identify potential causes. Having an accurate identification of the patient's underlying cause of ES is critical in directing the therapeutic intervention, with the ultimate goal being to improve wakefulness and function. Nonpharmacologic treatments should be considered for the initial management of ES, with a particular emphasis on behavioral measures to improve sleep. In patients who have an inadequate response to these initial interventions, wake promoting pharmacologic therapy should be considered based on the patient's severity of symptoms, although it should be noted that these medications are not specific to an underlying disorder and do not replace the need for sleep. 5 In an effort to effectively treat these symptomatic patients with drug therapy, armodafinil has emerged as a reasonable treatment option with demonstrated short-term efficacy. In fact, only armodafinil and modafinil have FDA approved indications for treatment of ES related to OSA, narcolepsy, and SWD.13,17 The manufacturer of armodafinil was also seeking FDA approval for the treatment of patients with ES associated with jet lag disorder. However, the manufacturer is no longer pursuing this indication. 43 Modafinil is recognized by the American Academy of Sleep Medicine as an appropriate therapeutic option for the treatment of hypersomnias associated with central origin. 44 Since armodafinil is the R-isomer of modafinil, it would be reasonable to apply this recommendation to armodafinil as well.
The efficacy and safety of armodafinil has been investigated in seven randomized, double-blind, placebo-controlled studies, ranging from 3 days to 12 weeks in length and in these studies patients received armodafinil in doses ranging from 50 mg to 250 mg daily, with the median dose being 150 mg. Eligibility requirements were similar between each of the studies presented, yet they differed in coexisting conditions associated with ES, including OSA, circadian rhythm disorder, narcolepsy, or fibromyalgia.24–30 In these patient populations, the evaluation and determination of response to therapy can be very challenging considering the subjectivity of symptoms associated with ES. Consequently, there has been the development of numerous assessment scales in an effort to improve evaluation of these patients. Such, a variety of standardized assessment scales, both objectively and subjectively measured, were used in the studies identified in this review. Regardless of which efficacy scale was utilized, armodafinil consistently demonstrated statistical significance by improving primary efficacy measures. With the exception of the studies by Schwartz and Krystal et al, each study demonstrated significant improvements in its primary efficacy endpoints. Positive changes were also seen in the subject's cognitive functioning, specifically attention and memory, in addition to improvements in patient ratings of fatigue.24–30 When reviewing armodafinil's effects specifically on late-day or late-shift sleep latency measures, armodafinil was able to demonstrate sustained efficacy in multiple comorbidities, including SWD, narcolepsy, and OSA, although statistical significance with OSA was only demonstrated in pooled-analysis, not in the individual studies of Roth and Hirshkowitz et al.25,27–29,33 This continued efficacy reinforces the once daily dosing recommendations for armodafinil.
In reviewing the results of the Krystal et al study, it is conceivable that the high placebo response may have adversely affected its ability to detect a treatment difference. 24 This may be partly due to the comorbidities of OSA and depression, as depression studies are commonly associated with a high placebo response rate in studies specific to antidepressants. 45 The inability to demonstrate significant improvement in patients with comorbid depression is also consistent with multiple studies of modafinil.46,47 Schwartz et al also demonstrated non-significant improvement with armodafinil in its primary endpoint. Although it was stated that patients in this study were allowed to use concomitant medications, which may inherently confound the efficacy results, there was a deficiency in the study's description of the methodology used. 30 Therefore, identification of limitations is difficult to discern.
It should be noted that the studies described in this review all involved a relatively small number of subjects, ranging from 60 to 427 patients enrolled, with a maximum treatment length of 12 weeks.24–30 Therefore, long-term efficacy and safety of armodafinil in patients with ES is currently unknown and larger randomized controlled studies are necessary to validate its use for patients with chronic use longer than 12 weeks. There were two uncontrolled, open-label studies, not analyzed in this review due to weakness in their methods, which were conducted to assess the safety and efficacy of armodafinil in patients with OSA, SWD, or narcolepsy, but because no comparator was used, it is difficult to determine the true efficacy of armodafinil in these studies.48,49 This concern translates to another inherent limitation in the reviewed studies, that no study evaluates armodafinil against a comparator agent, other than placebo. Such understanding of armodafinil's use in the setting of ES would be strengthened by studies using a head-to-head design with an appropriate comparator agent such as modafinil or an amphetamine compound. Both longer and head-to-head studies would allow for stronger evidence-based management and give practitioners more support in using the most safe and efficacious medication in the treatment of patients with ES. Also, with the exception of the study by Schwartz et al, each study presented in this review was funded by, or the principal investigators were financially supported by Cephalon, Inc., the manufacturer of armodafinil.
Overall, daily dosing of armodafinil was fairly well tolerated and generally only produced mild-to-moderate adverse effects. The most common adverse effects that occurred consistently in ≥5% of study participants included headache, nausea, diarrhea, dizziness, and anxiety.24–30 Of these, headache, nausea, and insomnia have previously demonstrated a dose dependent increase in adverse effects. 17 Discontinuation rates due to adverse events generally ranged from 1.4% to 9.6%, but rising as high as 22.2% in patients with comorbid fibromyalgia. Comparatively, dropout rates due to adverse events in the placebo group ranged from 1.4% to 5.6%.24–30 When armodafinil was given to patients with OSA, SWD, or narcolepsy in two 12-month, open-label studies, discontinuation rates due to adverse effects of armodafinil were 12.7% and 12.8%.48,49
When reviewing the cardiovascular safety of armodafinil, small changes in blood pressure and pulse rate were identified. In patients with narcolepsy and OSA, armodafinil was associated with a mean increase in diastolic blood pressure of 0.4 mmHg (P = 0.029) and 0.6 mmHg (P < 0.05).28,29 But, when two studies with OSA patients were pooled together, including the study described above, no significant changes in diastolic blood pressure were identified. 33 Krystal and Rosenburg et al also reported small increases in both heart rate and systolic blood pressure, but neither study quantified this statement and both stated the increase was not considered clinically significant.24,26 When cardiovascular safety was assessed in a 12-month open-label study, systolic blood pressure was shown to rise a mean of 3.6 mmHg and pulse rate increased by 6.7 beats per minute, although statistical testing was not conducted. 48 Such increases were not demonstrated in a second 12-month open-label study. 49 Other results have demonstrated evidence of small, but consistent, increases in pulse rate, varying from an increase of 0.9 to 3.5 beats per minute. 17 Although the clinical significance of these changes cannot be determined, it does indicate the need for additional cardiovascular monitoring in patients receiving armodafinil, especially given the recent recommendations by the EMEA on the use of modafinil in patients with uncontrolled hypertension or irregular heart beat.
Conclusion
Although non-pharmacological therapies remain the first-line treatment for patients with ES, current evidence suggests that armodafinil significantly improves wakefulness in patients with OSA, narcolepsy, and circadian rhythm disorders such as SWD and jet lag disorder. But, long-term effectiveness and safety cannot be determined at this time. Historically, amphetamines have been considered the drugs of choice for ES. However, they are associated with a high potential for abuse and an increased risk of cardiovascular adverse events. For these reasons, modafinil has become the preferred treatment for ES. Since armodafinil is the R-isomer of modafinil, it possesses the same abuse potential and similar adverse effect profile as modafinil. One potential advantage of armodafinil in comparison to modafinil is that armodafinil maintains higher peak concentrations later in the day which may have clinical implications. Studies have shown that patients may need more frequent dosing or increased doses of modafinil to sustain a therapeutic response. Since armodafinil maintains higher plasma concentrations late in the day, dosing alterations may not be warranted. Although armodafinil should be considered safe for use, if the decision is made to initiate a patient on armodafinil, health-care providers should monitor for adverse effects, particularly cardiovascular complications. In addition, health-care providers should monitor for serious psychiatric disorders and skin reactions given the recent review on modafinil by the EMEA.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
