Abstract
Background
Patients that do not achieve therapeutic response with over the counter non-triptan medications may benefit from triptan-based treatments.
Objective
Phase I V, open-label, multi-center, prospective cohort study assessing the effectiveness of rizatriptan in the management of migraines for patients that have not responded to non-triptan treatment.
Methods
Patients were treated with one rizatriptan (MAXALT RPD®) 10 mg wafer at the onset of each migraine attack and were assessed after a minimum of one and a maximum of two consecutive headache episodes. Outcome measures included self-reported assessments (severity and duration of migraine headache) and the Migraine ACT questionnaire.
Results
A total of 369 patients were enrolled, of which 291 and 215 reported one and two attacks, respectively. For the first and second attacks, 47.2% and 53.9% of patients reported complete resolution of pain while 73.6% and 77.0% reported pain severity reduction within two hours of onset. Mean (SD) pain severity score (four-point Likert scale) during the 488 migraine episodes was reduced significantly (P < 0.001) from 2.56 (0.49) at onset to 1.91 (0.85) at 30, 1.31 (1.00) at 60 and 0.84 (1.00) at 120 minutes. Similar improvements were observed for changes in Migraine ACT questionnaire scores. No treatment-related serious adverse events were reported. The most frequently reported non-serious adverse events that were attributed to the study drug were dizziness (2.2%), chest discomfort (1.1%), nausea (1.1%), and somnolence (0.8%).
Conclusion
In a real-life setting, rizatriptan benzoate 10 mg is effective and safe in the treatment of acute migraine headaches in patients who do not respond to non-triptan treatment.
Introduction
Migraine is an under-diagnosed and under-treated chronic disabling neurological disease1–3 that affects approximately 23 million Americans 4 and more than three million Canadians. 5 In a Canadian study, only half of the 0.6 million adult females and 0.8 million adult males suffering from migraine were likely to be diagnosed by a physician. 6 Severe migraine has been classified as one of the most disabling disorders by the World Health Organization. 7 By affecting 3% to 10.6% of the population, migraine is the most common form of disabling primary headache syndromes8,9 resulting in substantial morbidity and economic burden 10 in the form of bedridden days and lost productivity. 11 Thus, migraine is an important target condition for health care interventions. 2
The goals of acute migraine treatment are to accomplish rapid pain relief, prevent recurrence, reduce the need for rescue medication, and restore functional ability. 12 Several options are available for the treatment of acute migraine including general over-the-counter analgesics such as aspirin, non-steroidal anti-inflammatory drugs (NSAIDs), acetaminophenaspirin-caffeine combination products, and prescription treatments such as ergotamine and triptans.8,13 Triptans are selective 5-hydroxytryptamine type 1B/1D (5-HT1b/1d) receptor agonists that are effective and safe for the acute treatment of migraine headaches and are indicated as first-line treatment in patients with moderate to severe migraine. 12 Rizatriptan is a triptan that is rapidly absorbed and highly bioavailable with proven efficacy for treating migraine with rapid onset of effect after oral administration.14,15
Given that less than half of moderate or severe migraine sufferers are treated with prescription medication, 16 it is suggested that most migraine sufferers are taking non-triptan medication, including over-the-counter analgesics. This observation may be partially explained by a stepwise strategy that begins with over-the-counter, non-prescription drugs as the most widely used approach. 17 Despite the availability of treatment alternatives, an important proportion of patients with migraines do not achieve adequate therapeutic response with their current treatment. At the present time there is a need to evaluate the incremental benefit, safety and tolerability of changing migraine treatment from non-triptan to triptan based regimens in a real-life setting. The aim of this study was to address this question, using a phase IV, open-label, single prospective cohort design conducted within a routine clinical practice setting in Canada.
Methods
Study design
This was a phase I V, multicentre, open-label observational study conducted by 76 investigators (mostly primary care physicians) across Canada. A total of 369 patients were enrolled from May to October 2005 and from February to June 2006. Patients were treated with rizatriptan (MAXALT RPD®, Merck Frosst Canada Ltd., Kirkland, Canada) 10 mg wafers and assessed during a minimum of one and a maximum of two consecutive acute migraine attacks. The total follow-up duration was 16 weeks in order to ensure that two consecutive migraine attacks and their possible recurrences would be captured. This assumption was based on the study inclusion criterion that required an incidence of 2 to 7 attacks per month. This study has been approved by IRB services (Aurora, Ontario), an independent institutional and ethics committee. All patients signed an informed consent form prior to initiating any study-related procedure.
Eligibility criteria
Eligible patients were aged between 18 and 65 years, had a history of migraine for at least 6 months with or without aura at a frequency of 2 to 7 attacks per month as defined by the International Headache Society (IHS) criteria, and did not use triptan medications within 3 months prior to participating in the study. In order to be included in the study, patients had to be identified by their treating physician as candidates for changing from their current non-triptan regimen and to have a Migraine-ACT 18 score of 0 to 3. The Migraine ACT questionnaire consists of four dichotomous items measuring consistency of response to current migraine treatment, pain relief within two hours of onset, ability to resume normal function within two hours of onset, and ability to plan daily activities. Higher Migraine ACT scores indicate less severity. Consequently a Migraine ACT total score of 4 indicates adequate control of the symptoms with no need for change in treatment while a score of 0 indicates poor symptom control requiring treatment modification.
Patients with history or symptoms of ischemic cardiopathy, cerebrovascular or peripheral vascular syndromes, valvular heart disease, tachycardia, uncontrolled or severe hypertension, pregnancy or nursing, hepatic impairment (Child-Pugh C) were not eligible for inclusion in the study. In addition, patients with hypersensitivity or history of serious adverse event related to prior treatment with a triptan and those treated with propranolol were excluded. Patients having difficulty in distinguishing their migraine attacks from tension or interval headaches and those suffering from hemiplegic, ophthalmoplegic or basilar migraine were also excluded from the study.
Procedure
Patients were assessed over three visits. During the screening visit (Visit 1) patients were assessed for eligibility and those qualifying (with a Migraine ACT score 0–3) were enrolled. Visits 2 and 3 took place between 1 and 7 days post-treatment following the first and second migraine attack, respectively. Each migraine episode was treated with one rizatriptan 10 mg wafer. Rescue medication including analgesics and antiemetics was allowed for non-responders whose headache had failed to resolve two hours after administration of rizatriptan. Those suffering from a headache recurrence were allowed to take a second rizatriptan 10 mg wafer, separated by at least 2 hours. Headache recurrence was defined as the return to the same original headache intensity within 24 hours after the initial dose in a patient who had responded 2 hours after treatment. Only one headache recurrence within a 24-hour period could be treated with the study drug, for a maximum of 20 mg of rizatriptan. Any further headache recurrence during the 24-period was treated with analgesics and antiemetics.
All patients were to complete a diary during two subsequent migraine attacks. They were instructed to take the study medication at the time of migraine onset and to record the severity of the migraine headache (none, mild, moderate or severe) and the presence of associated symptoms, specifically, sensitivity to light, sensitivity to noise, nausea and vomiting beginning at onset (0) and at 30, 60, and 120 minutes thereafter. At 120 minutes patients also completed a 5-point Likert scale describing their satisfaction with treatment as very satisfied, somewhat satisfied, neither satisfied nor dissatisfied, somewhat dissatisfied, and very dissatisfied. At each visit, the treating physicians administered the migraine ACT-questionnaire and collected all details regarding treatment emergent adverse events.
Outcome measures
The primary efficacy outcome measure was the proportion of patients that were headache pain-free at two hours after initiation of treatment. Secondary efficacy outcome measures included the proportion of patients with response to treatment, defined as reduced headache severity without using any rescue medication, satisfaction with treatment and the change in the distribution of responses to the individual items and in the total Migraine ACT questionnaire score during the study. The severity of pain and the presence of associated symptoms during the two hours after each attack were also assessed as secondary efficacy measures. Pain severity was assessed using a 4-point Likert scale with 0 = no pain, 1 = mild, 2 = moderate and 3 = severe. Accordingly, the pain severity rating was an integer with a range between 0 and 3 with higher scores indicating more severe pain. Safety and tolerability was assessed by the incidence of treatment-related serious and non-serious adverse events.
Statistical methods
Descriptive statistics were reported for baseline characteristics and outcome measures. The change in the distribution of the dichotomous responses of the Migraine-ACT between baseline and final follow up was assessed for statistical significance with the McNemar's Chi-Square test for paired samples. The change in the pain severity during the course of the migraine attacks was assessed with a repeated measures Analysis of Variance. The change in Total Migraine-ACT-score between study visits was assessed for statistical significance with the Student's paired t-test and linear regression analysis. Adverse events were described using the MedDRA dictionary of terms (version 9.0). No imputation methods were used for replacing missing data. All analyses were conducted with SPSS version 12.0 for Windows.
Results
Patient disposition
Patient disposition is described in Figure 1. A total of 369 eligible patients were enrolled in the study. Of those enrolled, 78 (21.1%) discontinued before experiencing a first migraine attack and did not take the study medication. Of the 291 patients who experienced a first acute migraine attack, 284 (97.6%) completed the study diary; 215 (73.9%) of the 291 patients who experienced a first episode, were followed to the second migraine episode, of which 204 (94.9%) completed the migraine diary. Reasons for discontinuation are listed in Figure 1. The current study is based on 284 patients with one attack and 204 patients with two attacks for a total of 488 migraine episodes over a 16 week follow-up period.
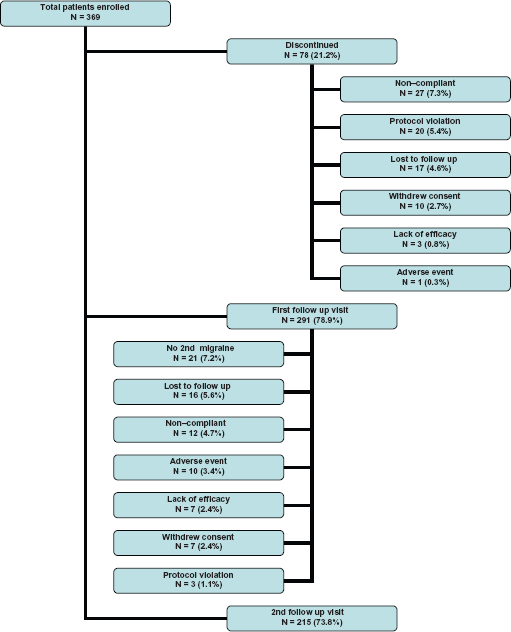
Patient disposition.
Baseline characteristics
Demographic and baseline characteristics of the 369 patients enrolled in the study are shown in Table 1. Patients’ mean (SD) age was 40.5 (11.7) years, 310 (84.0%) were female, and 317 were Caucasian (85.9%). The mean (SD) duration since the first migraine diagnosis was 11.5 (11.1) years and the mean (SD) number of episodes per month was 3.5 (1.8). For 157 (42.5%) of these patients the average duration of the migraine attacks was between 12 and 24 hours.
Demographic and baseline characteristics.

Data are based on 369 enrolled patients.
Migraine history data were missing for 2 (0.5%) patients.
Prior treatment for migraines included NSAIDs for 229 (62.1%), analgesics for 211 (57.2%) and combination of non-triptan medications for 114 (30.9%). Triptan-based treatment for the period exceeding three months prior to study entry included sumatriptan for 25 (6.8%) and zolmitriptan for 22 (6.0%) patients (Table 2).
Prior migraine therapy.
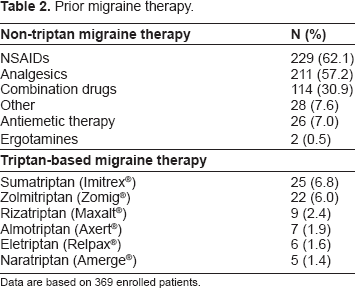
Data are based on 369 enrolled patients.
Effectiveness
For the first and second migraine attacks, 47.2% and 53.9% of the patients reported complete resolution of pain within 2 hours of treatment initiation while 22.5% and 27.0% experienced complete pain relief within 1 hour of treatment initiation. Response to treatment, defined as reduced pain severity without the use of rescue medication, was reported within 2 hours of treatment initiation by 73.6% and 77.0% of the patients for the first and second migraine episode respectively (Table 3). Rescue medication was used by 72 (25.4%) and 42 (20.6%) patients for the first and second attack, respectively. A total of 139 doses of migraine rescue therapies (88 for the first attack and 51 for the second attack) were used. The most frequently used rescue medications were acetaminophen used by 35.2% and 25.5% of the patients and ibuprofen used by 23.9% and 39.2% for the first and second attacks respectively.
Response to treatment.

284 patients completed diary following the onset of their first attack.
204 patients completed diary following the onset of their second attack.
Of the 209 and 157 patients who reported response to treatment within 2 hours following initiation of treatment for the first and second attacks respectively, 50 (23.9%) and 32 (20.4%) patients experienced a recurrence within 24 hours. Rescue medication for headache recurrence was used by 34 (68.0%) and 26 (81.3%) of the patients experiencing recurrence after the first and second episode respectively.
The majority of the patients, 61.6% after the first attack and 65.7% after the second attack, reported to be either very satisfied or somewhat satisfied with treatment. Conversely 23.2% and 22.1% of the patients were somewhat dissatisfied or very dissatisfied with treatment after the first and second episode respectively (Table 4).
Patient satisfaction with treatment.

284 patients completed diary following the onset of their first attack.
204 patients completed diary following the onset of their second attack.
The mean pain severity during the 488 migraine attack is shown in Figure 2. These results show that the mean (SD) pain severity score was significantly reduced from 2.56 (0.49) at onset to 1.91 (0.85) at 30 minutes, 1.31 (1.00) at 60 minutes and 0.84 (1.00) at 120 minutes (P < 0.001). The prevalence of associated symptoms were also significantly (P < 0.001) reduced during the first two hours after the first attack (Fig. 3). The results in Figure 4 show that the mean (SD) Migraine ACT score was significantly (P < 0.001) increased from 0.74 (1.01) at baseline to 1.88 (1.26) at visit 2 and 2.94 (1.45) at visit 3. It should be noted that the mean value for the Migraine-ACT questionnaire after the first attack (visit 1) was underestimated since the first item on the questionnaire refers to consistency of treatment response that is relevant only to patients experiencing two or more attacks. Table 5 shows that significant improvement was observed between the baseline and final visits in all Migraine ACT questionnaire items.
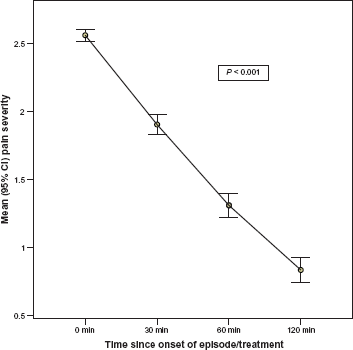
Pain severity during migraine episode. Pain Severity measured as 0 = none, 1 = mild, 2= moderate, 3 = severe.
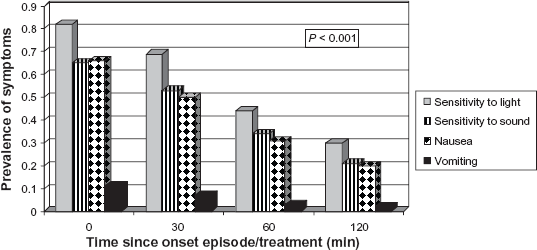
Prevalence of associated symptoms during treatment. Prevalence of all symptoms significantly reduced over time (P < 0.001). Based on 488 Migraine Episodes. Patients may have had more than one associated symptom.
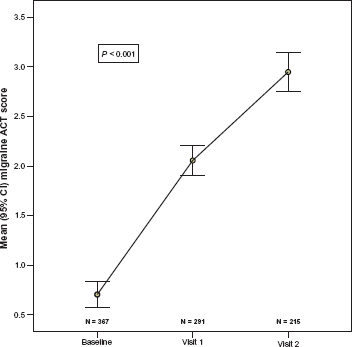
Migraine-ACT questionnaire score by visit. All between-visit changes are statistically significant (P < 0.001) based on Repeated Measures Analysis of Variance.
Distribution of the responses to the migraine-ACT questionnaire.
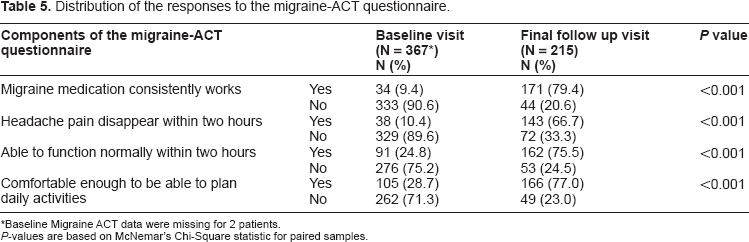
Baseline Migraine ACT data were missing for 2 patients.
P-values are based on McNemar's Chi-Square statistic for paired samples.
Safety
There were a total of 42 non-serious adverse events (NSAE) reported by 29 (7.9%) patients that were attributed to the study drug by the treating physician. The most frequently reported NSAE were dizziness (2.2%), somnolence (0.8%), chest discomfort (1.1%), and nausea (1.1%). There were no Serious Adverse Events reported during the study that were attributed to the study drug.
Discussion
The purpose of this open-label study was to assess the effectiveness and safety of rizatriptan 10 mg in the treatment of moderate and severe acute migraine headaches in patients who were requiring a change from their non-triptan based treatment regimen. This study was conducted in a real-life setting in order to assess effectiveness under less stringent conditions than those typically observed in controlled clinical trials. 19 This is based on the fact that results from randomized controlled trials cannot always be generalized to real-life routine practice, 20 in which patients have a wide spectrum of headache frequency, intensity and duration.12,21
In this study, approximately 50% and 75% of the patients reported total pain relief and headache relief to a lower severity within 2 hours following treatment initiation. These results are consistent with previously published placebo-controlled clinical trials,14,15,21,22 a pharmacy-based study 23 and other real-life setting studies,13,24–26 further supporting the effectiveness of rizatriptan 10 mg in the treatment of moderate to severe acute migraine attacks. Headache recurrence rates reported in placebo-controlled trials within 24 hours post-treatment with rizatriptan 10 mg were approximately 40.0%,15,21 which are considerably higher than those observed in our study. The difference may be due to the fact that in the current study patients were allowed the use of rescue medication. Moreover, they had failed to achieve therapeutic response with non-triptan based treatment and thus may be different from those participating in clinical trials. In the current study, treatment with rizatriptan 10 mg was equally effective in each one of the two consecutive attacks, supporting the consistency of the effect over several episodes. 27
Rapid, durable and reliable effects are important factors in treatment selection among migraine patients.28–30 The current study has shown that rizatriptan meets these requirements for patients that had not achieved the desired therapeutic effectiveness with non-triptan medications, by providing high rates of pain relief within 1–2 hours of treatment administration, low recurrence rate and consistent effect over multiple episodes. This is further confirmed by the high satisfaction rate, similar to those reported in other studies with rizatriptan,23,25,26 and a higher satisfaction rate than those reported for other oral migraine medications including analgesics, NSAIDs and ergot derivatives.31,32
Migraine attacks are frequently disabling with significant functional impairment due to associated symptoms including pulsatile pain, light sensitivity, sound sensitivity, nausea, unilateral pain, blurred vision, aura and vomiting.2,33 In the current study, the presence of these symptoms was significantly reduced within two hours of treatment initiation. Compared to baseline non-triptan therapy, we observed three-fold increases in the proportions of patients who were able to function normally within 2 hours and comfortable enough to be able to plan daily activities. Given that the severity of headache pain is a major determinant of migraine-related disability, 34 the ability to return to normal functioning within 2 hours post-treatment can also be enhanced by significantly reducing the headache pain. These results are consistent with the higher effectiveness of rizatriptan in restoring functional capacity compared to non-triptan therapy 30 and to other triptans,21,30,33,35 including sumatriptan, naratriptan and zolmitriptan.
In the current trial, rizatriptan was found to be safe and well tolerated. Its safety profile obtained in a real-life setting is consistent with previously reported studies,15,21,27 with dizziness, somnolence, chest discomfort, nausea and fatigue as the most common adverse experiences probably, possibly or definitely related to rizatriptan administration.
A potential limitation of the present study is that although patients were instructed to take the study medication at migraine onset, it was not possible to determine the exact timing of drug administration. However, previous studies have shown that whether taken at headache onset or later during the attack, rizatriptan was equally effective in relieving acute migraine headache within 2 hours post-treatment. 11 Thus, it is unlikely that the overall effectiveness results were influenced by the time of rizatriptan administration during the course of the attack. However, assessment of the time to pain relief may have been affected. The use of the stop-watch method suggested by Sunshine et al 36 to measure the exact time from medication administration to pain-relief would provide a more accurate measure that could further be analysed to estimate the rate of pain relief, using time to event methods, such as Kaplan Meier function.
In most recent clinical trials of acute migraine treatments, the standard primary outcome measure has been the percentage of patients with headache relief within 2 hours after dosing, defined as a reduction of headache pain from moderate or severe at baseline to mild or no headache during this time period. 37 In clinical trials of triptans for acute migraine therapy, although harder to achieve, 2-hour pain free as an endpoint is now preferred as it better reflects patient expectation 28 and provides more conservative effectiveness results. The Clinical trials subcommittee of the IHS has proposed that a pain-free response at 2 hours should be adopted as the primary endpoint in acute treatment trials since it is already a well-established outcome measure. 38 Thus, one of the strengths of the present study is the use of effectiveness measures in accordance with the recommendations made by the IHS Clinical Trials Subcommittee that facilitates comparison with other studies. Moreover, this study was conducted in non-controlled real-life setting, thereby enhancing generalization of the results to routine clinical practice.
It is unlikely that the results observed in this study have overestimated the effectiveness of the study treatment in the management of acute migraine. In fact, it is likely that the non-response rates observed in the current study may have been overestimated due to the possible inclusion of patients suffering from a tension-type headache. This would be the result of the less stringent criteria for patient selection that was applied in order to better emulate the real-life setting. It has been suggested that patients suffering from tension-type headache are less likely to respond to rizatriptan 10 mg. 39 Low or inconsistent absorption, and variability in individual 5HT-1 receptor profiles may also partly explain the fact that not all patients responded to treatment. 39
In conclusion, in a real-life setting, rizatriptan benzoate 10 mg is effective and safe in treatment of acute migraine attacks in patients who require a change from their current non-triptan based treatment regimen.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors confirm that they have permission to reproduce any copyrighted material. The peer reviewers of this paper report no conflicts of interest. Michel Aube has served on advisory boards for Merck Frosst and Johnson and Johnson in addition to being a member of the speaker bureau for Johnson and Johnson, TevaNeurosciences and Procter and Gamble. Fridon Chouha is a former employee in Clinical Research at Merck Frosst and currently an employee at JSS Medical Research Inc. Julie Vaillancourt and John Sampalis are employees of JSS Medical Research Inc. JSS Medical Research was contracted by Merck Frosst to conduct this study.
Footnotes
Acknowledgements
We would like to acknowledge the 369 patients and 76 investigators across Canada for their participation in this Phase IV study. Study investigators are:
