Abstract
MMX™ mesalazine is a novel delayed release mesalazine formulation with a high-strength 1.2 g tablet for the treatment of active mild to moderate colitis and maintenance of remission. In vitro and in vivo studies show initiation of drug release in the terminal ileum and caecum with gradual release of 5-ASA as the tablet passes through the colon. Pharmacokinetic data are comparable to other 5-ASA formulations with low systemic absorption and high levels in feces and in mucosal biopsies in the left colon. Clinical trials have shown high rates of clinical and endoscopic remission in active mild to moderate colitis over 8 weeks with a 2.4 g once daily dose. There do not appear to be higher remission rates with the 4.8 g dose. Prolongation of treatment with a further 8 weeks of 2.4 g twice daily can induce remission in those failing the initial 8 weeks of therapy. A maintenance study enrolling patients who achieved remission in the acute studies showed high rates of remission maintained at one year with 1.2 g twice daily (68.5%) and 2.4 g once daily (64.4%) using the strict definition of remission (including mucosal healing) that was used in the active treatment trials. The drug is safe and effective in colitis. The high tablet strength, and once-daily dosage make this formulation a welcome addition to therapy options for patients with colitis.
Introduction
Mesalazine or 5-aminosalicylic acid (5-ASA) remains the first-line therapy for treatment of mild to moderately active ulcerative colitis, and maintenance of mucosal healing. The drug exerts a topical antiinflammatory effect on colonic mucosa. Therapeutic benefit of sulphasalazine was first described in 1942 by Svartz. 1 Recently there has been a resurgence of interest in several aspects of mesalazine use. There is new information on mechanism of action through peroxisome proliferator-activated receptor-γ (PPARγ) receptors, effect on carcinogenesis, new technology in drug delivery coatings to allow gradual release of the drug after reaching the colon, and higher dose per tablet, and a growing number of trials showing that once daily dosing is as effective as divided doses. This review discusses a new formulation of 5-ASA–-multimatrix (MMX) mesalazine: the evidence for its clinical effect, pharmacokinetics, and safety in comparison to other preparations of 5-ASA currently available.
Pharmacology
The clinical efficacy of sulphasalazine in ulcerative colitis was a serendipitous discovery. The drug was originally designed as a combination of an antibacterial agent (sulphapyridine) linked to an anti-inflammatory agent (5-ASA) by an azo bond, in order to treat rheumatoid arthritis, at a time when it was thought to have an infective as well as inflammatory aetiology. It is ironic that the initial studies failed to show the benefits of the drug in rheumatoid arthritis (that subsequently were shown in larger studies), but did show benefit in ulcerative colitis patients in the rheumatoid arthritis trial, confirmed in randomized controlled trials. 2 Later studies in distal colitis showed that the active agent was 5-ASA rather than sulphapyridine, 3 and that 5-ASA was as effective as intact sulphasalazine and exerted a topical effect on the colonic mucosa.4,5 Uncoated 5-ASA is rapidly and completely absorbed in the proximal small intestine, but poorly absorbed from the colon. 6 It is metabolised to N-acetyl 5-ASA, by N-acetyl-transferase 1 (NAT 1), present in intestinal epithelial cells and liver. N-acetyl 5-ASA had no antiinflammatory activity in two of three trials.7–9 As the sulphapyridine moiety was responsible for most of the toxicity of sulphasalazine,10,11 it was logical to develop coated forms of 5-ASA that were released in the colon. 12
Colonic release of 5-ASA
The first delayed-release 5-ASA tablet used a pH-sensitive acrylic resin (Eudragit S) and was developed and evaluated in Cardiff, and produced as Asacol®. 13 The resin dissolves at pH 7, and utilises the fact that pH gradually rises through the small intestine, to achieve pH 7 in the terminal ileum 14 (Fig. 1). Since then a range of further coatings have been developed to deliver drug to the colon using other pH-sensitive resins (Claversal®, Mesasal®), delayed-release microgranules using ethylcellulose (Pentasa®), or alternative non sulfa-containing prodrugs (balsalazide, olsalazine) (Table 1).
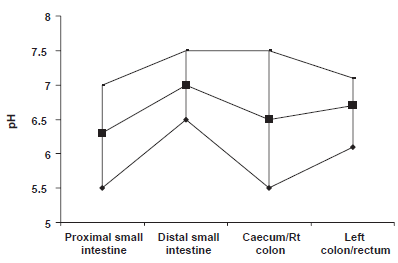
pH gradient along gastrointestinal tract in man.
Current Oral 5-ASA formulations.
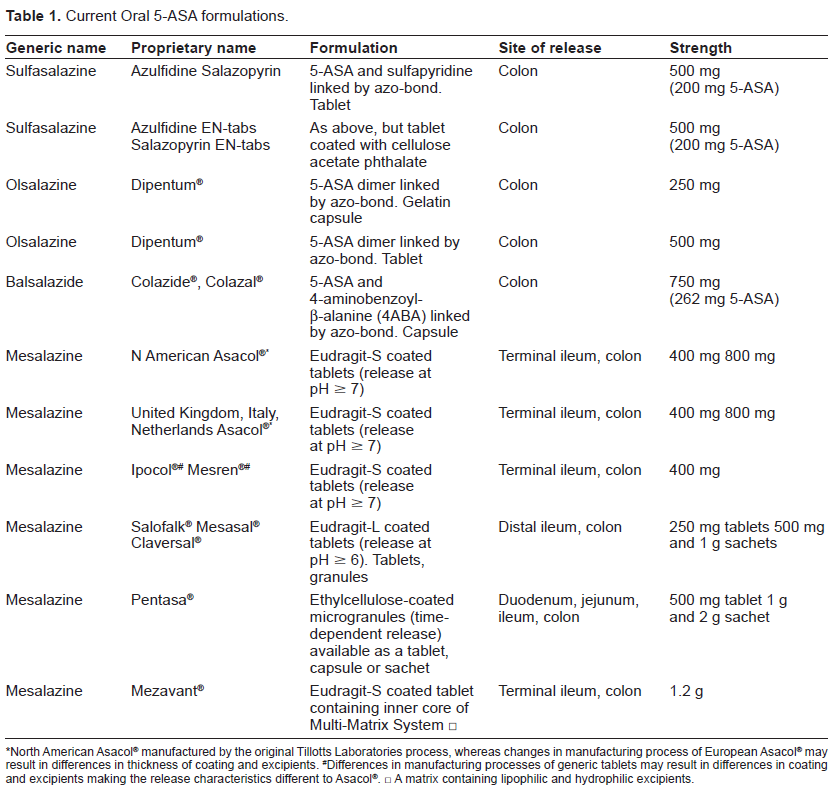
North American Asacol® manufactured by the original Tillotts Laboratories process, whereas changes in manufacturing process of European Asacol® may result in differences in thickness of coating and excipients.
Differences in manufacturing processes of generic tablets may result in differences in coating and excipients making the release characteristics different to Asacol®.
A matrix containing lipophilic and hydrophilic excipients.
Pharmacokinetics
The pharmacokinetics of mesalazine have been widely studied, 15 but it is clear that plasma levels do not necessarily reflect drug efficacy for a drug with a topical effect on colonic mucosa. It is easy to demonstrate significant variations in plasma pharmacokinetic profiles, but it is not at all clear that this correlates with drug efficacy, although there is a great dearth of adequately powered comparative studies. Plasma levels may be more important as an indicator for potential nephrotoxicity. There is however no evidence that drugs with more proximal small bowel release and higher plasma levels are any more toxic, and it seems more likely that interstitial nephritis is either an idiosyncratic or hypersensitivity reaction. 15 Most of the pharmacokinetic studies have been performed in healthy volunteers. There is also evidence to show that patients with quiescent ulcerative colitis have colonic motility and function that is similar to healthy controls.16,17 The pharmacokinetics of mesalazine should therefore be the same in quiescent ulcerative colitis as in the normal colon.
Need for novel 5-ASA delivery systems
There are a number of theoretical reasons to develop novel 5-ASA delivery systems. The ethylcellulose-coated and Eudragit-L coated preparations undoubtedly release 5-ASA higher in the small intestine than Eudragit-S coatings. 15 This should result in higher plasma levels, and thus greater urinary excretion of 5-ASA. In fact it is difficult to demonstrate this in the numerous pharmacokinetic studies, where interindividual variation is far greater, but there is some evidence of greater total urine 5-ASA for Claversal®, Salofalk® and Mesasal compared to all the others. 15 Faecal excretion is however broadly similar for all preparations. The only exception to this is the effect of administering bisacodyl to volunteers to increase colonic transit and cause diarrhoea. 18 This resulted in a marked increase in faecal excretion of the pro-drugs sulphasalazine and olsalazine. This was more significant than the increase in faecal loss of free 5-ASA when the bisacodyl-treated volunteers were given Asacol® or the other slow-release preparations. When comparing UC patients with and without diarrhoea, a similar increase in faecal loss of unsplit sulfasalazine and olsalazine is found, 19 but the amount of free 5-ASA (as a proportion of total free- and acetylated 5-ASA) rose most dramatically in diarrhoea patients receiving Asacol® from 66% to 87%. In other words, very little Asacol® was being acetylated in patients with diarrhoea, and may link with observations of patients that they pass intact or partially dissolved tablets at times. The rise in proportion of free 5-ASA in faeces was not as marked for Pentasa® or Salofalk® with diarrhoea. 19
An ideal drug delivery system would not release drug at all in the upper small bowel, but start to release the drug gradually in the ileocaecal region, and then progressively throughout the colon. In this way, unacetylated active drug would be available through the colon. It is worth noting that the right colon is generally thought to be a storage and mixing organ and chyme is likely to travel back and forth for some time in this area, as water is absorbed and the consistency of stool gets more solid. Once a mass action occurs, the stool then passes more rapidly down the left colon before evacuation. The left colon in normal patients, and certainly in active colitis, is generally relatively empty, and represents a much more challenging area for drug delivery.
Evaluation of intestinal release profiles
It is necessary therefore to evaluate the release characteristics of these formulations in the gut. There are two main techniques used to assess the rate of dissolution of slow-release tablets, and the location in the gut where this occurs. In vitro, the pH conditions of the gut can be simulated by measuring the rate of dissolution of tablet coatings in fluids at different pHs. Systems have been developed to simulate more accurately conditions in the normal gut, including pH, body temperature, peristalsis, gut secretions, nutrient and water absorption and colonic microbiota. An example is the TNO GastroIntestinal Model (TIM; TNO Quality of Life, Zeist, The Netherlands). 20 There are of course limitations to these models as they cannot completely simulate an organ whose function is still so poorly understood. It is also clear from in vivo magnetic resonance imaging of intestinal fluid along the gut, that there are fluid pockets, separated by areas of collapsed gut containing only minimal amounts of secretions. 21
The second, in vivo method, 22 involves labelling tablets with radioactive tracer (152Sm2O3) added during tablet manufacture and then neutron-irradiated to produce 153Sm2O3. Gamma scintigraphy can then follow the dissolution of individual tablets after oral ingestion, with measurement of radioactivity in regions of interest plotted out on the basis of typical anatomy (stomach, small intestine, ileocaecal, ascending, transverse, descending colon and rectum). The limitations of this technique are the complexity of the incorporation of tracer into tablets, the possibility that this may alter the release characteristics of 5-ASA, and the possibility that tracer release may not accurately correspond to drug release. Variable anatomy may also limit the accuracy of plotting regions of interest in the gut.
MMX™ multimatrix system
Mesalazine with MMX™ multimatrix system (Mezavant™, also known as Mezavant XL™ in the UK and Ireland, and Lialda™ in the USA: Shire Pharmaceuticals Inc, Wayne PA, USA, under licence from Giuliani SpA, Milan, Italy) is a novel tablet coating designed to release mesalazine gradually throughout the colon. The outer coating is a gastroresistant Eudragit-S film that dissolves above pH 7 (usually in the terminal ileum). Within this is a core of lipophilic and hydrophilic matrix. Once exposed to intestinal fluid, this absorbs water to form a sponge-like viscous gel mass, containing the drug. The lipophilic elements creates a partially hydrophobic environment in the core, and is expected to slow the penetration of aqueous fluid into the tablet, and allowing the slow diffusion out of the drug as it passes round the colon, allowing contact of the drug with a larger surface area of mucosa before conversion to n-acetyl 5-ASA. The hydrophilic elements may promote adherence to colonic mucosa. This proposed mechanism is thought to have the advantage of gradual 5-ASA release through the colon. This review will discuss the evidence that this occurs, and the clinical benefit in relation to other preparations.
In vitro, dissolution of MMX™ mesalazine has been studied in the TIM-1 and TIM-2 system (simulating stomach and small intestine, and colorectum respectively). 23 In simulated fasting and fed conditions, less than 1% of drug is released in stomach and small intestine during a mean residence time of 5 hours (fasted) and 6 hours (fed). Release of 78% (69%-87%) occurred in the colon in the simulated fasting state (over a mean 19 hours), but was 68% (61%-76%) in the fed state (mean 18 hours residence). No sign of the tablet was left in the colon residue. This model cannot indicate what level in the colon the drug is released, but measuring 5-ASA release in the colon dialysate over time indicates a sigmoidal pattern, (slow rising initially and then after a 2-3 hour lag, increasing linearly to a peak of release at 6-8 hours (fasted) and 5-6 hours (fed). There are no comparative studies with other 5-ASA preparations, but the results do suggest that MMX™ release is gradual once the colon is reached, in this in vitro model.
The in vivo release of MMX™ mesalazine has been studied using gamma-scintigraphy. Brunner et al studied 12 healthy volunteers using 153Sm2O3 labelled tablets, and plasma pharmacokinetics. 24 In fasting single-dose studies, the mean gastric emptying time was 0.4 ± 0.4 hours (hr), with appearance in the small intestine at 0.8 ± 0.5 hr, ileum at 4 ± 1 hr and colon at 5.8 ± 1.2 hr. Initial tablet disintegration started at a mean of 6.9 ± 1.1 hr, in ascending (n = 8) or transverse colon (n = 4). It was difficult to determine time to complete dissolution, as there was a tail of radioactivity behind the tablet on scintigraphic images. Plasma levels peaked (Tmax) when tablets were in the ileocaecal region. Plasma levels as area under curve (AUC) were 12.37 nmol/ml, comparable to that of other slow-release 5-ASA preparations and prodrugs. 15 19.9 ± 18.2% of drug absorption occurred in the ileal region, and 80.1 ± 18.2% in the colon. Urinary excretion was measured for 24 hours, with a mean 9% of administered dose excreted (almost all acetylated). This is less than in studies with other drugs, with mean values ranging from 11%-33% for sulfasalazine, Asacol® 10%-35%, Pentasa® 15%-53%, and up to 27%-56% for Salofalk®, Mesasal and Claversal®. 15 It should be noted however that the elimination process was not complete at 24 hours (plasma concentrations still significant at this point, and in some subjects, the start of the decay phase had still not occurred). Overall, the results suggest a low systemic availability of 5-ASA with the MMX™ mesalazine tablet, and a slower and more sustained release from the ileocaecal region onwards through the colon.
A small study has used the same scintigraphic method to compare release of MMX™ mesalazine and delayed-release mesalazine (Asacol® Giuliani, S.p.A., Milan, Italy) in eight healthy volunteers in a single-dose, two-way, cross-over design. 25 Plasma release of both tablets was very similar, with a Tmax at approximately eight hours. Scintigraphy showed initial disintegration slightly earlier for MMX™ mesalazine (4.75 ± 1.3 hours) than Asacol® (6.16 ± 1.8 hours). Complete disintegration was later however for MMX™ mesalazine (17.4 ± 8.6 hours) versus Asacol® (7.3 ± 2.1 hours). The location of complete disintegration was also more distal for MMX™ mesalazine (Fig. 2). These scintigraphic studies demonstrate the variability in location of tablet disintegration between individuals. Whether this variability is greater between different preparations is not clear.
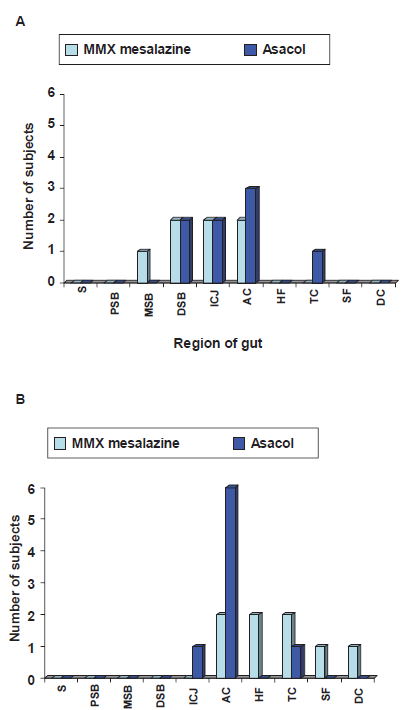
In summary, both in vitro and in vivo studies confirm that initial disintegration of MMX™ mesalazine is in the ileocaecal region but subsequent release is gradual through the colon.
Clinical trials of MMX™ mesalazine (Table 2)
A small preliminary trial was carried out in five Italian centres to evaluate the effect of MMX™ mesalazine in active left-sided disease compared to 5-ASA enemas. 26 Patients had disease extending more than 15 cm but no further than splenic flexure and had mild to moderate disease activity as determined by a clinical disease activity (Rachmilewitz score) ≥6. 27 This was an eight week double-blind double-dummy trial comparing MMX™ mesalazine 1.2 g three times daily plus placebo enema (n = 40) with Asacol® 4 g/100 ml enema with placebo tablets. The primary end-point was remission at eight weeks, defined by a Rachmilewitz score of ≤4. In the MMX™ group, 60% achieved remission, versus 50% in the enema group. This study showed that oral MMX™ mesalazine is as effective as enemas in left-sided disease, confirming delivery of drug in significant amounts to the left colon. A small dose-ranging pilot study 28 investigated the efficacy of three different doses (1.2 g, 2.4 g and 4.8 g given once daily to 13, 14 and 11 patients respectively) in patients with mild to moderate active UC (UC-DAI score 4-10) 29 extending beyond the rectum. The primary end-point was remission at eight weeks as defined by a UC-DAI score ≤ 1, with a zero score for rectal bleeding and stool frequency and at least a one-point reduction in sigmoidoscopy score from baseline. No patients in the 1.2 g group achieved remission, versus 4 (30.8%) in the 2.4 g group and 2 (18.2%) in the 4.8 g group. There was however a dose-response relationship in improvement in UC-DAI score, and also in rectal and sigmoidoscopic histology scores. Levels of 5-ASA and acetyl-5-ASA in colonic mucosa were considerably higher in the 4.8 g group (48.8 ng/mg and 29.7 ng/mg) compared to the 1.2 g group (11.2 ng/mg and 6 ng/mg) and the 2.4 g group (6.9 ng/mg and 7 ng/mg).
Active disease
A large phase III double-blind parallel-group study in 280 patients compared 1.2 g twice daily, versus 4.8 g daily versus placebo over eight weeks in mild to moderate active UC. 30 The trial used a modification of the Sutherland UC-disease activity index (UC-DAI). The index has a maximum score of 12 comprising a score of 0-3 each for rectal bleeding, increased stool frequency, sigmoidoscopic appearance and physicians global assessment. The sigmoidoscopic score was modified. In Sutherland's sigmoidoscopic score, normal scores 0, erythema, decreased vascular pattern, or mild friability scores 1, and marked friability scores 2, with ulceration and spontaneous bleeding scoring 3. This was modified to make any degree of friability give a score of 2. This has the effect of making the score somewhat more objective, reducing interobserver variability and indeed bringing it in line with the original sigmoidoscopic score devised by Baron. 31 Mild to moderate disease was defined as a score of 4-10, with the sigmoidoscopic score of 0 or more, and the physician's global assessment as 2 or less. The relapse duration had to be less than six weeks, and patients were excluded if they had relapsed using more than 2 g mesalazine. The primary endpoint was clinical and endoscopic remission at eight weeks (i.e. modified UC-DAI score ≤ 1, with a score of 0 for rectal bleeding and stool frequency, and no mucosal friability and at least a 1-point reduction in sigmoidoscopy score. Using this stringent definition, 12.9% on placebo achieved remission, with 34.1% in the 1.2 g twice daily, and 29.2% in the 4.8 g daily group (Table 2). There was no significant difference between the doses.
Trials of MMX™ mesalazine in active UC and maintenance of remission.
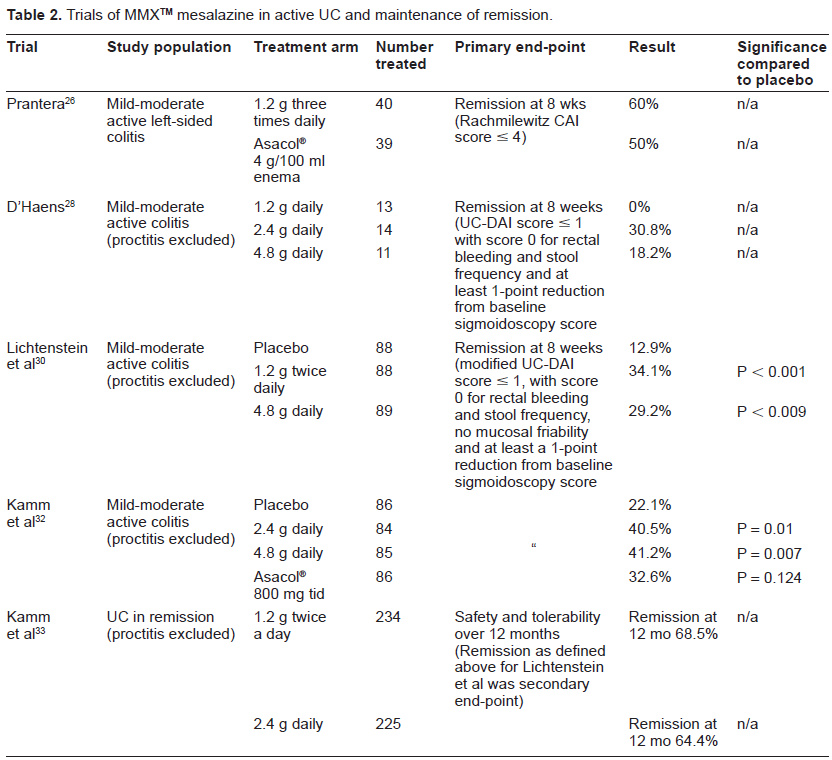
A similar trial 32 ran concurrently with this study, and had the same entry criteria, definitions and end-points. Patients were randomized to 2.4 g once daily, 4.8 g once daily, placebo, or a comparator group of Asacol® 800 mg three times daily, all for eight weeks. The latter tablets were over-capsulated with a gelatine capsule to ensure blinding. The study was not powered to detect a difference between MMX™ mesalazine and Asacol®. In spite of such similar design, the rates of clinical and endoscopic remission was higher than in the previous study at 22.1% in the placebo group, rising to 40.5% in the 2.4 g group, and 41.2% in the 4.8 g daily group. The remission rate in the Asacol® comparator group was 32.6%, which was not statistically significant compared to placebo. There were no significant safety concerns with either of these studies, and together they show convincingly that MMX™ mesalazine is an effective treatment for mild or moderate colitis in a once daily dosage of 2.4 g, with no benefit from increasing to 4.8 g daily.
Maintenance of remission
A one year maintenance trial followed on for patients in the above two trials of active colitis. This trial was designed to assess safety and tolerability of MMX™ mesalazine. 33 For those achieving remission at the end of the active treatment period, they could enrol directly (n = 246), and for others they could receive a further 8 weeks of 2.4 g MMX™ mesalazine twice daily (312 patients, of whom 213 achieved remission and were enrolled). These numbers included approximately 20% who had not achieved the strict definition of remission required in the index trials, but were deemed well enough by the investigators to enter the maintenance phase. Patients were re-randomized to either 2.4 g daily, or 1.2 g twice a day. At one year, there were 64.4% of patients in remission (using the strict definition above) in the 2.4 g daily group, versus 68.5% in the 1.2 g twice a day group. When defined as the absence of a relapse requiring additional treatment, 88.9% (once daily) and 93.2% (twice daily) remained in remission. The differences between groups were not significant. These results show high numbers remaining in remission, (in comparison to other maintenance studies), but it is worth remembering that this is a selected population, who had not relapsed previously on more than 2 g mesalazine, and who had responded well in the active treatment trial.
Dose-response with 5-ASA in Active Disease
To date, the evidence for dose-response in treatment of colitis remains contentious. In theory, a higher dose of 5-ASA might be more effective by saturating the acetylating enzyme capacity in colonic mucosa, thus increasing availability of the active free 5-ASA. 34 The trials of MMX™ mesalazine to date do not show an advantage of 4.8 g daily over 2.4 g in active disease. This is in keeping with most of the literature on 5-ASA. In the trial comparing 1.6 g Asacol® with 4.8 g, Schroeder et al 35 found the higher dose more effective. Sutherland, however, in his Cochrane review meta-analysis, 36 concluded that 1.6 g was insufficient, and at least 2 g was needed, but it was not possible to demonstrate a dose-response above this level. In the Ascend I and II trials37,38 of 2.4 g versus 4.8 Asacol® over six weeks in mild to moderate colitis, there was significant benefit of the higher dose only evident in the moderate group (72% improvement with 4.8 g versus 59% with 2.4 g in Ascend II), with no significant benefit in the mild colitis group. In the Ascend III study, 39 in spite of including 772 patients with moderately active disease, there was a numerical increase from 66% improvement with 2.4 g to 70% on 4.8 g. This benefit is of dubious clinical significance. The MMX™ mesalazine studies were not powered to demonstrate differences in the dose regimens. A pooled analysis of the two large active treatment trials do not show overall benefit for the 4.8 g dose over 2.4 g. 40 Three hundred and four patients who did not achieve remission in the two eight week MMX™ mesalazine studies,30,32 were given open-label MMX™ mesalazine 2.4 g twice daily for a further eight weeks, as reported in the maintenance study. 33 Of these, 59.5% achieved remission according to the stringent definition used in the trials. When patients were separated according to the initial treatment group they had been in, there was no difference in remission rates: the placebo group had 57% remission, 2.4 g MMX™ mesalazine 61.5%, 4.8 g MMX™ mesalazine 60.3% and the Asacol® 2.4 g group 61% remission after the eight week extension. The implication is that it is the prolongation of treatment, rather than the switch to high dose, that achieved remission. In conclusion, there is no evidence that doses of MMX™ mesalazine above 2.4 g daily have greater efficacy.
Safety and Tolerability
In the literature, 5-ASA is recognised as an extremely safe drug, with headaches and nausea being the commonest side-effects. In the trials of MMX™ mesalazine to date, no significant safety concerns have arisen. In the D'Haens 28 study, headache was the commonest adverse event, occurring in 21% of patients. This was also reported in the other trials,26,28,30,32,33 but no more commonly in the active treatment than in the placebo groups, and there was no evidence of a dose response–-i.e. no increase in frequency in the high-dose or once daily dose groups. Overall, gastrointestinal side-effects were the commonest, again no more common than in the placebo group. Two cases of pancreatitis occurred (one on 2.4 g and one on 4.8 g daily), a documented side-effect of 5-ASA. 41 Both resolved on withdrawal of medication. One patient developed a hepatitis, but a subsequent infectious mononucleosis test was positive, and glandular fever the likely cause.
No renal failure or interstitial nephritis was recorded in the 723 patients in the MMX™ mesalazine trials to date. Interstitial nephritis is recognised as a complication of mesalazine treatment and exact frequency is unknown. Renal impairment (elevated creatinine) was found in 0.2% in a large study involving 2940 patients. 42 In one study of 223 IBD patients, 43 microalbuminuria was found in up to a third of patients treated with doses of more than 3 g daily, and appeared to be dose-dependent. A UK review of reports to the Committee on Safety in Medicines (CSM) from 1991-8 (2.8 million prescriptions for mesalazine, 4.7million for sulfasalazine) found 11.1 reports per million prescriptions for mesalazine, but none for sulfasalazine. 44 Even considering the incomplete nature of adverse event notifications through this system, the disparity implies that the higher plasma levels seen with delayed-release 5-ASA 15 are relevant in causing renal damage, although they are still at least 10-fold lower than salicylate levels in patients taking aspirin. Other renal complications of untreated IBD include tubular proteinuria, renal calculi, amyloidosis and glomerulonephritis, and it has been proposed that interstitial nephritis could also be related to the underlying IBD (more severe disease likely to require higher doses of 5-ASA). However, it is well recognised that early withdrawal of 5-ASA results in resolution of the renal function, providing strong evidence that this is a drug-related hypersensitivity. If mesalazine-induced interstitial nephritis is not recognised, then permanent renal impairment is likely to result. 45 In conclusion, renal toxicity from 5-ASA does occur, but is rare, and as yet there is no strong evidence that it is dose-dependent. There is no evidence of differences in risk, either with different 5-ASA colonic-release mechanism, nor with once-daily therapy.
There is evidence that 5-ASA inhibits thiopurine methyltransferase in vitro, and so could potentially affect metabolite levels of azathioprine and 6-mercaptopurine. 46 In theory higher levels could have a more pronounced effect, with more thioguanine nucleotides, responsible for marrow suppression. A study comparing 2 g with 4 g doses of mesalazine for 4 weeks however showed no significant difference in thioguanine nucleotide levels, although 6 methylmercaptopurine levels were significantly lower on the high dose. 47 High-dose mesalazine could in theory enhance the effectiveness, and also toxicity of coprescribed thiopurines, but in reality this effect is likely to be non-clinically significant.
Once Daily Therapy with 5-ASA
In the past it was assumed that divided dosing would maximise the time that drug would spend in contact with colonic mucosa and this would improve efficacy, but this has never been shown to be true. With a growing interest in treatment adherence, once daily therapy is an important consideration, if it could be shown to be as safe and as effective as divided doses. A study in Sheffield of steady state plasma pharmacokinetics (after 7 days) showed that in normal subjects, median peak concentrations, trough concentrations, and areas under the curve were similar for once-daily and three-times-daily mesalazine dosing regimens using Asacol®. 48 Urine and fecal 5-ASA 24 hour collections were also similar, as were levels in rectal biopsies. This implies that safety is not a concern, (albeit with a maximum daily dose of only 2.4 g). The study was not able to measure peak levels in the colonic lumen, which might have therapeutic relevance. A computer simulation model of 5-ASA levels in segments of the colon predicts that both total and regional levels would be similar after once daily, and three times daily dosing with Asacol®. 49 The same model did predict that levels in sigmoid colon and rectum would be considerably lower than in the right colon with Asacol®, and this difference would be exacerbated by frequent bowel actions, as in active colitis.
A pilot feasibility study of once daily (Asacol®, using 400 mg tablets) versus conventional dosing of mesalazine for the maintenance of remission of ulcerative colitis, was the first study to investigate once daily dosing. 50 Over six months, there was one relapse in the 12 patients on daily dosing (average 2.5 g daily) versus one relapse among the 10 patients taking an average 2.7 g per day either in twice daily or three times daily administration (Table 3). The authors concluded that a larger trial was warranted.
Trials of once-daily 5-ASA therapy (MMX™x mesalazine trials shown in Table 2).
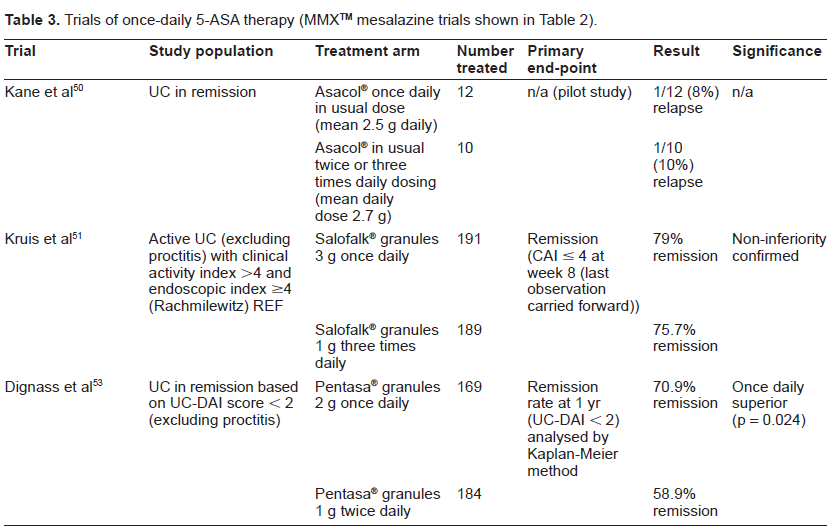
Four of the MMX™ mesalazine trials to date have included once daily arms,28,30,32,33 and have confirmed that once daily dosing is as effective as divided doses, both in active disease and maintenance of remission. The 1.2 g MMX™ tablet has made it easier to dose once daily with a small number of tablets. These studies were powered to compare each dose regimen with placebo, and were not designed to demonstrate non-inferiority of once daily compared to divided doses.
Two further studies have now been completed using 5-ASA granules that were designed as non-inferiority studies. Eudragit-L coated 5-ASA (Salofalk®) in the form of granules was investigated in active UC by Kruis et al 51 in a single-blind study. Of 381 patients enrolled, 347 completed the study, which used the Rachmilewitz CAI index 27 ≤4 as the primary end-point. 79% patients treated with 3 g once daily achieved remission, compared to 75.7% in the 1 g t.i.d. arm. The confidence interval for the difference was 3.4% [95% confidence interval (CI) -5.0%-11.8%]. Using the same end-point as in the MMX™ mesalazine trials (modified DAI score ≤ 1 with score 0 for rectal bleeding and stool frequency and at least 1-point reduction from baseline sigmoidoscopy score), 30 the once daily group achieved 37% remission, versus 39% in the t.i.d. group. These figures are virtually the same as the two MMX™ mesalazine active colitis trials.32,33 The Kruis trial results also showed that in patients treated with once daily 3 g, those with distal disease had statistically better outcomes than those with more extensive disease, and also did better than those with distal disease treated with 1 g t.i.d. This was an exploratory analysis, and is most likely to have arisen by chance, but the authors speculated that the difference could be explained by once daily therapy giving intermittent high 5-ASA levels in the distal colon.
A pharmacokinetic study in 30 healthy volunteers had shown bioequivalence for once versus twice daily administered 4 g of ethylcellulose-coated 5-ASA. 52 In the Podium trial, 53 the investigators evaluated the clinical effectiveness of a granule preparation of Pentasa® for maintenance of remission in a single-blind non-inferiority designed study. The study used the UC-DAI score, 29 and patients enrolled had to have a score of <2, with proctitis patients excluded. Patients taking 2 g Pentasa® granules once daily (n = 169) did better with 70.9% remaining in remission at 1 year, than the 184 patients taking 1 g bd. Although designed as a non-inferiority study, the outcome was in fact statistically superior for the 2 g daily dose (p = 0.024). Using Cox regression analysis, the difference related to the improved adherence in the once daily dosing group. An economic analysis showed that once daily treatment was cost-saving in this trial, mainly due to reduced relapse rate. 54
Adherence
In the controlled environment of clinical trials, with a high level of medical supervision, approximately 70%-95% of IBD patients adhere to their medication.55,56 However, this high rate of medication adherence is not reflected in normal clinical practice. A cross sectional study of US outpatients with quiescent UC found that only 40% were adherent to maintenance mesalazine therapy. 56 Several community-based studies have reported non-adherence rates ranging from 43% to 72% of IBD patients. 57 In the UK, approximately 15% of patients fail to even redeem prescriptions at the pharmacist. 58 Treatment non-adherence rates vary considerably between countries (in Europe, a survey of 203 IBD patients revealed self-reported non-adherence rates ranging from 13% to 46%). 59 The implications are extensive, with good evidence of higher relapse rates in non-adherent patients, 60 and evidence that non-continuation of long-term 5-ASA increases colorectal cancer risk. 61 Non-adherence is likely to result in a significant increase in treatment costs. 62 Poor adherence is strongly linked to complex dosage regimens. 63 In a study involving 105 hypertensive patients receiving long-term treatment, once daily (od) or twice daily (bd) dosing was associated with significant improvements in mean adherence rates compared with 3-times daily (tds) dosing, 64 and many other reviews confirm that od and bd dosing is associated with better adherence than more frequent dosing. 64 It is more difficult to show the small benefit increment from switching from bd to od dosing. 65
It is evident that non-adherence during maintenance therapy for quiescent disease is far more likely than in active disease. In the Podium trial of maintenance once daily therapy with Pentasa® granules, 53 adherence was recorded by medication returns. This was higher for the once daily (versus twice daily) group, but not statistically significant. In a validated questionnaire, patients in the twice daily group reported higher rates of forgetting medication, or carelessness about their medication. Visual analogue scores showed significantly improved acceptability for once daily. In a multivariate analysis, treatment adherence was a significant factor in improving outcome over one year, whereas the dosing regimen was no longer significant. In the Salofalk® granule study of once daily versus three times daily therapy in active UC, 51 adherence was not specifically assessed, but on questioning, 82% of patients preferred once daily dosing, compared to only 2% preferring three times daily. In the MMX™ mesalazine acute studies evaluating once daily therapy,30,32 the studies were double-blind and double-dummy, so the once daily groups were still taking multiple daily tablets and differences in adherence were not noted. In the maintenance MMX™ mesalazine trial 33 which was open-label, there was no difference in treatment adherence (recorded on pill counts as adherent if >80% of medication taken), with 93.3% adherence in the once-daily group and 99.6% in the twice-daily group. In summary, adherence is greatly improved when dosage can be given once or twice daily, but the difference between once and twice daily is not significant. It is likely that patient preference may be the most important factor, and the freedom to take medication once or twice daily is an advantage, and may be a factor in quality of life.
Quality of Life
In ulcerative colitis, disease symptoms are the main determinant of quality of life (QOL) at any one time. 66 In an eight week trial comparing 1 g, 2 g and 4 g Pentasa® or placebo in active UC, quality of life was evaluated in 374 patients, and was significantly improved compared to placebo in the 2 g and 4 g dose groups. 67 The benefit was seen in all 12 QOL parameters, including five disease-specific parameters (e.g. trips to the toilet or urgency) and seven general QOL parameters (e.g. ability to sleep, sexual relationships, or hobbies/recreations). Effective medication is thus a vital tool in improving QOL. In a qualitative study of IBD patient experiences, 68 over two thirds identified medication as necessary in controlling symptoms, in spite of being a nuisance, a concern regarding toxicity, and anti-drug feelings. Two-thirds of the patients also reported a desire to self-manage their medication. In the Podium study, 53 health-related utility was measured, and was strongly linked to disease severity, 69 and although not different between once-daily and divided dose groups, the option of once-daily dosing is likely to lead to higher quality of life if relapse rates are lower.
Conclusions
MMX™ mesalazine is a novel 5-ASA delivery tablet that starts to release the drug in the terminal ileum and caecum, and then allows the slow release of drug during passage through the colon. The theoretical advantages of this formulation are borne out in the clinical trial data. In mild and moderate colitis these show high rates of remission and mucosal healing in clinical trials with stringent end-points. Once daily dosing is as effective as divided doses, and the 2.4 g dose appears as effective as 4.8 g daily. Although the clinical trials included Asacol® as a comparator, there are no adequately powered non-inferiority or superiority trials to show that MMX™ mesalazine is more effective than other 5-ASA preparations, but the drug has the advantage of a high strength 1.2 g tablet and well-designed trials showing high clinical and endoscopic remission rates in active disease and maintenance of remission with once-daily therapy. It is becoming clear that once daily therapy is effective for other formulations of 5-ASA, and this benefit is not exclusive to MMX™ mesalazine. Other considerations, particularly cost and patient preference, must be taken into consideration in choosing a formulation for individual patients, but there is no doubt that this drug is a welcome addition to the therapeutic armamentarium.
Disclosure
Dr Hawthorne has received honoraria and participated in continuing medical educational events supported by Shire Pharmaceuticals Inc, Proctor and Gamble, Ferring Pharmaceuticals, and Dr Falk Pharma, and received research funding from Proctor and Gamble.
