Abstract
Methadone is a synthetic opiate primarily used in the detoxification and maintenance of patients who are dependent on opiates particularly heroin. Though within last 10–15 years methadone is increasingly used to manage neuropathic and cancer pains. Unfortunately, with increased methadone use for pain is coincided with significant increase in adverse events and fatalities. Cardiotoxicity is one of the major adverse effects reported with the use of oral methadone. The purpose of this paper is to provide an overview of the cardiotoxicity of methadone, when used as an oral analgesic and protocols for safe prescribing of methadone to help physicians recognize situations where this drug offers the greatest advantage as an analgesic.
Introduction
Methadone, a synthetic long-acting opioid, has been used since 1960's for the stabilization and maintenance of patients suffering from addictive disorders. 1 Because of this lengthy association with opioid addiction, methadone carries a negative connotation with the public. 2 However, it has recently been ‘rediscovered’ as an analgesic in the treatment of chronic pain and cancer pain.3,4 Methadone is a potent μ-agonist with generally similar efficacy and side effects when compared to morphine, but it does posses several unique properties that distinguish it from other opiates. Some of these properties are advantageous in treating chronic pain, but some of its characteristics can make it a difficult drug to prescribe properly (Table 1). With increased use of methadone during the last decade, there has been an unfortunate increase in adverse events and fatalities related to the methadone.5–7 One of the challenges of using methadone (and its congeners) is its cardiotoxic adverse effects.7–9 First case report regarding its cardiotoxic adverse effects surfaced in 2001, about cardiac electrophysiologic disturbances: QT interval prolongation and/or TdP (torsade des pointes) to patients administered with LAAM (leva-cetylmethadol or Orlaam®). 8 The developed LAAM was subsequently withdrawn from European markets (EMEA 2001). In the United States, the FDA (Food and Drug Administration) strengthened warnings about this arrhythmogenic potential (FDA 2001) and screening electro cardiograms (ECGs) were recommended prior to and during LAAM titration. Finally, in late August 2003, the U.S. manufacturer of LAAM announced plans to discontinue the product.8,9
Advantages and disadvantages of methadone when used as analgesic.
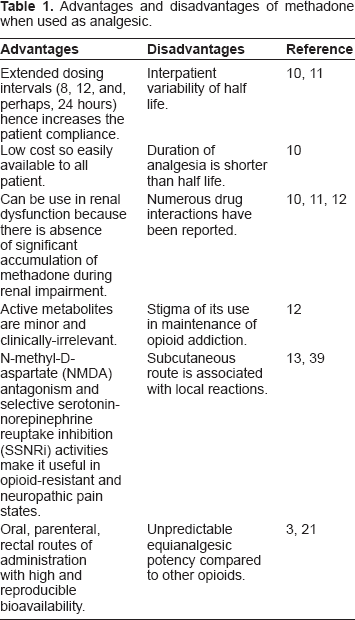
The goal of this paper is to review, the use of methadone for pain its cardiotoxicity potential and recommendations for safe prescribing methadone when used as an oral analgesic.
Use of Methadone for Pain
In many ways methadone can be considered as typical opiate. It is available in injection (10 mg/ml) and oral (5, 10, and 40 mg tablets as well as 1, 2 and 10 mg/ml oral solutions) dosage forms, and can be compounded in to suppositories. It provides effective analgesia in moderate to severe pain, and in addition it is a schedule II drug with a high potential abuse, and can be lethal in overdose. 10 However, methadone does have some differences.
One of the most distinctive qualities of the methadone relative to other opiates is its long half life. 10 This is estimated to rage from 4–130 hr, with typical steady state averages running between 22–35 hr. This property yields mixed outcomes, on one hand, it allows methadone to be doses one to three times a day in most patients without having to resort to more expensive extended release formulations. 10 At the same time, the duration of analgesic action is significantly shorter than what half life would suggest. Thus the dose interval of methadone required to maintain a therapeutic level of pain relief is usually shorter than one half life, resulting in accumulation of serum levels over several days. 10 If titrated too rapidly, these rising levels can lead toxicity and occasionally death. 5
As mentioned earlier, the half life of methadone is long, but it is highly variable also. Methadone is metabolized by both cytochrome P450 (CYP) 3A4 and 2D6, though the former can be considered the primary enzyme. Metabolism by metabolism by CYP 2D6, while somewhat less important, is not negligible and remains a source of interindividual variability. 11 Surprisingly, in spite of its high interpatient variability, and in contrast to most other opiates, metabolism of methadone appears to remains relatively stable in the elderly, in those with chronic renal failure and those with moderate liver failure. Furthermore, it metabolised to inactive compounds that are primarily excreted in faeces. 12 Thus it is one of the safer opiates to use in these patients, once the stable dose has been achieved. 12
The wide variety of drug interactions has been reported with methadone, the inhibitors and inducers of both CYP3A4 and CYP2D6 may change serum concentration. Some drugs such as verapamil or quinidine may alter methadone's absorption. 11 Other drugs, such as propranolol, tricyclic antidepressant, or certain phenothiazines, influence may influence protein binding by methadone through various mechanisms.10,11
Another highly distinctive property of methadone is its ability to antagonise the N-methyl-D- aspartate (NMDA) receptor. 13 Stimulation of the NMDA receptor is thought to play a role in lowering activation thresholds and central sensitization seen in neuropathic pain. Blocking NMDA receptor may provide methadone additional pain reducing property over typical opiates. 13 Another important property of methadone is the stigma that methadone carries as the drug of choice in the treatment of opiates dependence. 1 The lay public is well aware to take this drug out of fear of being perceived as an addict (Table 1).
Methadone is not commonly used for short term treatment of acute pain because of its potential for drug accumulation. However, methadone is good therapeutic alternative to morphine sulphate and other opiate analgesics in the treatment of severe chronic pain of both cancerous and non cancerous etiology.14–16 Placebo controlled trials have demonstrated that methadone has efficacy in the treatment of neuropathic pain. 17 However, there are no trials available that compares methadone to more standard therapies (i.e. tricyclic antidepressant, anticonvulsants) or to other opiates in this patient population. For this reason, it cannot be recommended as a routine first line agent. 17
Methadone has been tested as a part of several opiates switching trials.17,18 These were included in a recent systemic review of opiates switching.17,18 Unfortunately, none of the trials were conducted in a randomized, controlled manner. Also, the vast majority of trials included in the review were conducted in cancer patients. As a result, it can not be stated unequivocally that methadone offers clear benefits especially in non-cancerous pain. However it should be noted that vast number of trials did report opioid switching resulted in improved pain control and/or reduced opioid-related side effects.17,18
In the case of methadone prescribing in opiates naïve patients, dosing is relatively straight forward, and based on the proviso that initial dose should be low and titration should be slow. Typically recommended doses are 2.5–5 mg methadone dose every 12 hours. The total daily dose can be increased by 5 mg every 3–7 days until sufficient analgesia in achieved. One can consider shortening the dose interval to every 8 hours if symptoms allows, though a few patients will only require once daily dosing. During titration, daily contact between the patient and the physician's office is recommended and dosing should only be increased with physician advice. 19
The process of switching from another opiate to methadone is complex and numerous modalities have been published. 20 The main point of contention is the equipotent dosing of morphine and methadone. Single dose studies in opioid naïve patient suggest a 3–4:1 ration (3–4 mg morphine equal to 1 mg of methadone) to be equi-analgesic. However in opiate tolerant patients the ratio will increase with increase in morphine dose. Two of most widely used conversion tables are presented in Table 2.21,22 These conversion rates are not targeted doses they merely allow the physician to evaluate if the given patients dose is above or below the population norm. The process is further complicated by a lack of consensus between an immediate switch versus a more gradual shift. Several of these variations were reviewed by Ripamonati and Bianchi. 20
Cardiotoxicity of Oral Methadone Used as Analgesics
Several hypotheses have arisen to explain the cardiotoxicity caused by methadone and its congeners which are briefly discussed below. 23
Bradycardia
Eikenburg and Stickney in 2003, 24 investigated whether LAAM and its congeners exhibit anticholinesterase activity that could explain the induction of bradycardia. Moieties evaluated included LAAM and its two major metabolites (1-alpha-acetylnormethadol or N-LAAM and 1-alpha-acetyldinormethadol or DN-LAAM) and methadone. Study concluded that LAAM and its two metabolites (10-4 M) did exert significant anticholinesterase activity in guinea pig heart homogenate, in guinea pig plasma, against purified acetylcholinesterase, and against purified butyrlcholinesterase. 24
The activities of all of these moieties were significantly greater in plasma compared with heart homogenate and against purified butyrlcholinesterase compared with purified acetylcholinesterase (all P < 0.05 from control). LAAM and methadone were compared for their effects on cholinesterase activities in heart homogenate and plasma. At 10- 4 M, LAAM/methadone reduced heart homogenate activities to 82.11 ± 4.26/91.22% ± 5.12% of control and plasma activities to 43.43 ± 4.37/53.80% ± 9.90% of control (mean ± SEM, all P < 0.05 from control). Again, both agents were more active against plasmaenzyme activity compared with heart homogenate enzyme activity. Unfortunately, the effects of LAAM and methadone were not compared statistically. In isolated guinea pig atria studies, LAAM at 5 x 10-6 M significantly potentiated the effects of bethanecol (55.96% ± 5.75% decrease in heart rate) compared with the effect of bethanecol alone (19.78% ± 2.12% decrease, P < 0.05).
In contrast, these moieties did not reduce heart homogenate cholinesterase activity at the low concentrations (5 x 10-6 M) which produce bradycardia ex vivo which can be blocked completely by atropine. It appears that cholinesterase inhibition does not contribute to the bradycardia induced by low concentrations of LAAM, methadone, and related compounds but may contribute to bradycardia induced by high concentrations. 24
Impaired contractility
Another area of concern has been the cardiac muscle contractile response when exposed to opioids. Rendig et al in 1980, 25 evaluated the effects of six opioids (methadone, morphine, meperidine, fentanyl, pentazocine, and LAAM) at bath concentrations of 10-8 to10-4 M in the presence/absence of naloxone using an isolated cat right ventricular papillary muscle preparation. Three parameters were of interest: maximum developed tension (T), maximum rate of tension development (dT/dt), and time to peak tension (Tpk). At 10-5 M, methadone was associated with a small but significant decrease (-3%) in mean T while LAAM produced significant decreases in mean T (-9%) and mean dT/dt (-11%) and an increase in mean Tpk (+2%). At 10-4 M, methadone was associated with significant decreases in mean T (-25%), mean dT/dt (-23%), and mean Tpk (-2%). At the same concentration, LAAM was associated with significant decreases in mean T (-48%), mean dT/dt (-49%), and mean Tpk (-4%). Pretreatment of the muscle preparation with naloxone (10-4 M) did not prevent this reduction in contractile performance. It thus appears that high opioid concentrations exerted a direct myocardial depressant effect which was not prevented by naloxone and, thus, this effect was not mediated by opioid receptor agonism. The effect was felt to be due to a nonspecific local anaesthetic effect on cardiac muscle.
Prolongation of QT Interval
Most drugs which prolong the QT interval on the ECG, thus predisposing to TdP and then ventricular fibrillation and death, do so by interacting with voltage dependent HERG (human ether-a-go-go gene) potassium channels. LAAM inhibits HERG potassium channels in a dose dependent fashion, with little/no blockade at concentrations of 100 nM or less, 24% ± 3% blockade at 0.3 μM, 60% ± 3% blockade at 3 μM, and 96% ± 3% blockade at 10 μM (IC50 of 3 μM). The major metabolite N-LAAM also inhibited HERG with an IC50 estimated at 12 μM. In contrast, LAAM had little or no effect on other potassium channels such as the Kv LQT1/min K and Kv 4.3 cardiac channels. Methadone also blocks HERG channels the two major metabolites of methadone i.e. ethylidene dimethyl diphenylpyrrolidine (EDDP) and ethyl methyl diphenylpyrroline (EMDP) are extremely weak as HERG channel blockers. This blockade occurs very rapidly, with steady-state inhibition being achieved in less than 1 second at the IC50 concentration of 10 μM.26,27
In the isolated perfused rabbit heart preparation, perfusion with methadone 1–3 μM produced a 10%–12% increase in QT interval duration (P = NS). When assessing absolute QT interval values, only with perfusion at the 30 μM concentrations was a significant result compared with baseline obtained (P < 0.05). When assessing percentage change in QT interval from baseline, results with 10 μM (21% ± 5% increase, P <0.05) and 30 μM (44% ± 12% increase, P < 0.001) concentrations were significant. Trends to dose-dependency of this phenomenon were seen. 28
In addition, the electrophysiologic effects of methadone and buprenorphine have been evaluated in patients receiving opioid maintenance therapy with methadone (N = 35, 23 males, 35 ± 8 years old) or buprenorphine (N = 19, 11 males, 34 ± 6 years old) and non-dependent control subjects (N = 17, 9 males, 31 ± 8 years old). Methadone and buprenorphine recipients, having been on a stable drug dose for a minimum of four weeks, were taking maintenance doses of 69 ± 29 mg/day and 11 ± 5 mg/day, respectively. Mean ± SD QT intervals did not differ between the three groups (methadone, 407 ± 23 ms; buprenorphine, 407 ± 18 ms; controls 397 ± 21 ms). No correlation was evident between methadone dose and QT interval (P = 0.25). However, the QT interval occurring with methadone doses of 60 mg/day or greater (405 ± 29 ms) was significantly greater than that occurring with doses of less than 60 mg/day (381 ± 27 ms, P = 0.02). Two methadone patients and one control subject (all males) had prolonged QT intervals (defined in males as QTc interval ≥ 430 ms). The three groups significantly differed with respect to U wave frequencies. U waves were noted in 46%, 0%, and 13% of methadone, buprenorphine, and control subjects, respectively (odds ratio for methadone vs. other two groups pooled = 7.8). Lastly, methadone patients manifesting U waves were on a significantly higher dose than those methadone patients without U waves (89 ± 29 mg/day vs. 60 ± 24 mg/day, P = 0.004). 29
Although several other opioids are capable of blocking HERG currents, albeit in a much weaker fashion than do LAAM and methadone, the clinical relevance of HERG blockade can be assessed by dividing the IC50 value by the peak serum concentration value. For other opioids, the resulting values exceed 400 or 500, indicating a wide margin for cardiac safety. For methadone and LAAM, respective values of 2.7 and 2.2 indicate a much narrower margin for cardiac safety. 30
Short QT syndrome
Another diagnosis compatible with sudden cardiac death induced by methadone is short QT syndrome. 31 The QT interval on an ECG signifies the time required for the heart to repolarize after depolarization. Short QT syndrome is a newly described disease characterized by a shortened QT interval and by episodes of syncope, paroxysmal atrial fibrillation or life threatening cardiac arrhythmias. Short QT syndrome is caused by mutations in genes that encodes for cardiac ion channels. 32 To date, three genes including KCNH2, KCNQI, and KCNJ2, encoding different potassium ion channels involved in repolarization have been linked to the syndrome.32,33 Gussak 34 and co-worker in year 2000, first time reported short QT in three people from the same family, one of whom had several episodes of paroxysmal atrial fibrillation. The case report showed that a short QT interval could be familial and associated with atrial and ventricular arrhythmias. The definitive link between short QT syndrome and familial sudden death was described by Gaita 35 and associates in 2003, with the clinical reports of two families with short QT syndrome and high incidence of sudden cardiac death. In year 2004 Wolpert and co-worker provided the genetic and biophysical basis for the disease as well as its possible therapeutic approach. 36 Inducers of a short QT syndrome include febrile states, cocaine and methadone, sodium channel blockers (e.g. propafenone, procainamide, flecainide, bupivacaine, lidocaine, tricyclic antidepressants) and electrolyte imbalances. 35
In a recent multicenter observational trial of patients presenting with typical short QT syndrome during an acute medical event (N = 47), 16, 26, and 5 cases were due to febrile episodes, drug-related, and due to electrolyte imbalances, respectively. Fifty-one percent had malignant arrhythmias, with 38% developing (aborted) sudden cardiac death, and 6% each experiencing ventricular tachycardia and syncope. One of the victims of sudden cardiac death was a methadone recipient. 37
Recommendations for Safe Use
The US guideline to be published, on methadone use for opioid maintenance therapy and analgesia, was developed by an expert panel holding meetings in May 2003, July 2007, December 2007, and July 2008. Although methadone use as an analgesic was one target of the panel, the Center for Substance Abuse Treatment of the Substance Abuse and Mental Health Services Administration convened the meetings and the majority of participants were involved only in opioid maintenance therapy. Following recommendations from the expert panel were potentially applicable to recipients of oral methadone for analgesia:23,38
Doctors should inform patients of arrhythmia risk when they prescribe methadone.
Doctor should ask patients undergoing or considering methadone treatment about any history of history heart disease, arrhythmias and fainting spells.
Doctor should check the QT interval on the patient's ECG before starting methadone treatment within 30 days after starting treatment, and annually thereafter. If the QT interval is greater than 450 ms but less than 500 ms, the panel recommends discussing benefits and risk of methadone with the patient and frequent monitoring. If the QT interval exceeds 500 ms, the panel recommend discontinuing methadone treatment or reducing the methadone dose, eliminating possible contributing factor, or using an alternative therapy.
Follow serum electrolyte concentrations on an individualized basis.
Doctors should be aware of interactions between methadone and other drugs that prolong the QTc interval or slow the metabolism of methadone.
Methadone should be started at low dose and titrated slowly. Because of its long half life and variable pharmacokinetics methadone should not to be used to treat breakthrough pain or as an as needed medication.
Elderly people are more susceptible to the side effects of methadone therapy, it is recommended to start once daily dosing in this population i.e. 2.5 mg once daily. For frail elderly people an even smaller starting dose can be used i.e. 1 mg once daily. Dose change should not occur faster than once weekly in this group.
Conclusions
Methadone has long history of use in the treatment of opiate dependence, but only recently it has began to be used for its analgesic properties. As a result of its use today as a second-line (and, in some cases, first-line) opioid analgesic, formerly infrequent toxicities such as cardiotoxicity are becoming more prevalent. This paper has provided a concise review for the use of oral methadone as an analgesic and risk factors for methadone-associated cardiotoxicity. Recommendations have been provided for the safe use of oral methadone for analgesia in all healthcare settings. Hopefully, utilization of these recommendations will reduce the frequency of cases of cardiotoxicity associated with the use of oral methadone.
Disclosures
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author and peer reviewers of this paper report no conflicts of interest. The author confirms that they have permission to reproduce any copyrighted material.

