Abstract
We reviewed the literature and found 31 adult cases and 1 newborn case of methadone-associated QTc interval prolongation and/or torsade de pointes (TdP). Parametric statistics may not be useful in studying this issue because methadone-associated TdP is a very rare event and, hence, “an extreme outlier” consistent with scalable randomness. We may have to rely upon narrative medicine in the form of case reports with all its limitations and hazards to provide our best understanding. We report risk factors for methadone-associated QTc interval prolongation and TdP based on review of published case reports. We believe both drug manufacturers and the FDA would better serve our patients and inform clinicians if they more readily reported drug-induced outliers such as methadone-associated TdP using a case report format.
Introduction
Our major aim is to identify risk factors for methadone-associated QTc interval prolongation and torsade de pointes (TdP) applying principles of narrative medicine in a case report format. Although drug-induced QTc interval prolongation may be a necessary precondition for methadone-induced/associated TdP, we argue that it is insufficient to explain TdP. We believe that the mix of biochemical/metabolic disturbances, genetic variation and structural changes combined with methadone-induced/associated QTc interval prolongation best explain TdP.
Methadone
Methadone is a synthetic opiate most commonly employed in addiction treatment (or opiate dependence) and pain management. In 2006, Drug and Alcohol Services Information System reported that in 2005, 235,836 persons received methadone either for opiate detoxification or maintenance [Drug and Alcohol Services Information System, 2006 http://www.oas.samhsa.gov/2k6/otp/otp.htm]. Methadone’s primary mechanism of action is as a mu-receptor agonist that may work as an antagonist at the NMDA receptor [Anderson et al. 2000; Roxane Laboratories, 2003]. Methadone is lipophilic and has unpredictable pharmacokinetics with large patient variability in absorption, bioavailability, peak concentration and half-life. Its bioavailability can range from 36 to 100% and its half-life from 8 to 59 hours. Its duration of action may be prolonged due to its lipophilicity and ability to remain in the liver and other tissues [Roxane Laboratories, 2003].
hERG liability of methadone
A striking feature of drug-induced QTc interval prolongation and TdP is that virtually all clinically used drugs linked with these effects share in common an ability to inhibit channels that mediate the cardiac rapid delayed rectifier current, IKr—an ionic current that is important in regulating ventricular action potential duration [Witchel et al. 2000; Hancox et al. 2008]. Human Ether-à-go-go-Related Gene (hERG) encodes a protein (hERG) that mediates IKr [Sanguinetti et al. 1995; Trudeau et al. 1995]. Due to identified structural features, hERG exhibits a high susceptibility to pharmacological blockade [Sanguinetti et al. 2005; Hancox et al. 2008]. Similar to QTc prolongation, IKr/hERG blockade is a surrogate marker of TdP rather than a direct indicator of arrhythmogenicity; it is likely that it is the downstream consequences of IKr block, such as cellular early-after-depolarisations and augmented transmural dispersion of repolarisation that contribute to the genesis of TdP [Hancox et al. 2008]. Consequently, in preclinical drug safety testing any identified hERG-blocking tendency of an investigational drug (typically assessed through the study of hERG channels expressed in mammalian cell lines) must be considered alongside other preclinical information [Gintant, 2008; Hancox et al. 2008]. Analysis of the association between hERG/IKr block and observed propensity of clinically used drugs to produce TdP has led to the recommendation of a provisional ‘safety margin’ for drugs undergoing evaluation based on the ratio of hERG/IKr half-maximal inhibitory concentration (IC50) and effective therapeutic plasma concentration (ETPC, unbound) [Redfern et al. 2003]. A 30-fold IC50/ETPC safety margin has been suggested to be sufficient for most drugs, although it is recognised that mixed ion channel block can mitigate risk [Redfern et al. 2003; Gintant, 2008; Hancox et al. 2008].
In 1986, high concentrations of methadone were reported to slow depolarization rate and prolong duration of action potentials from sheep Purkinje fibers [Mantelli et al. 1986]. Methadone and its relative L-α-acetylmethadol (LAAM) were later (in 2002) reported to inhibit hERG, with respective IC50 values of 9.8 and 2.2 µM [Katchman et al. 2002]. This study also related the observed IC50 values to maximal plasma concentrations (Cmax) following therapeutic drug administration, obtaining IC50/Cmax ratios of 2.7 and 2.2 respectively for methadone and LAAM [Katchman et al. 2002] hERG channel blockade by methadone and LAAM has been confirmed by a number of subsequent studies with reported IC50 values for methadone ranging between ~1.7 and 20 µM [Kang et al. 2003; Kornick et al. 2003; Eap et al. 2007; Lin et al. 2009; Kuryshev et al. 2010; Zunkler et al. 2010]. Examination of the effects of these agents on other cardiac ion channels has suggested preferential block of hERG [Kang et al. 2003; Kuryshev et al. 2010] with some effects of methadone evident also on hNav1.5 and hCav1.2[Kuryshev et al. 2010] and with much weaker effects on other cardiac K+ currents [Kuryshev et al. 2010] indicating that hERG/IKr K+ channels are those most likely to be linked to methadone-induced QT prolongation. This is consistent with in vitro data indicating methadone-induced prolongation of the QT interval of isolated perfused rabbit hearts[Katchman et al. 2006] and of action potentials from human stem-cell derived cardiomyocytes [Kuryshev et al. 2010]. The link between IKr/hERG and methadone-induced QT prolongation is supported by observations regarding stereoselective actions of methadone enantiomers [Eap et al. 2007, Lin et al. 2009]. Two independent studies have shown (S)-methadone to be more potent against hERG than is (R)-methadone [Eap et al. 2007; Lin et al. 2009] with IC50 values for hERG current block of 2 and 7 µM respectively [Eap et al. 2007]. This correlates with reported observations: (i) that replacing (R,S) methadone with (R) methadone in patients receiving maintenance treatment reduces the QTc interval[Ansermot et al. 2010] and (ii) that CYP2B6 slow metabolizer status (which leads to impaired (S)-methadone metabolism) correlates with a higher QTc interval in patients receiving racemic methadone than is the case for extensive metabolizers [Eap et al. 2007] IKr/hERG block by methadone may also be consistent with modestly increased QT-dispersion seen in some methadone-treated patients [Krantz et al. 2005].
The original report of hERG channel block by methadone and LAAM included comparison with other opioid agonists, with fentanyl and buprenorphine also exhibiting hERG block with IC50 values < 10 µM, while codeine and morphine had little or no effect at therapeutically relevant concentrations [Katchman et al. 2002]. However, normalization of data to IC50/Cmax values yielded only low values (<3) for methadone and LAAM, with values of >50 for other agents studied [Katchman et al. 2002]. A separate study has compared methadone and heroin effects on hERG, reporting the latter to be ~100-fold less potent than methadone [Zunkler et al. 2010]. Chlorobutanol has been suggested to potentiate the ability of methadone to inhibit hERG current [Kornick et al. 2003] and diazepam has been suggested to exacerbate hERG-blocking effects of methadone by alleviating methadone block of sodium channels and, thereby, reducing any compensatory effect of sodium channel block on hERG inhibition [Kuryshev et al. 2010].
Case report format
Rather than attempting to develop a database that can be analyzed using parametric statistics, we employed the principles of narrative medicine in a case report format as we have in our other papers assessing psychotropic drug-induced QTc interval prolongation and TdP [Vieweg, 2003; Vieweg et al. 2004; Vieweg et al. 2005; Vieweg et al. 2009; Vieweg et al. 2012]. Data from our review of case reports in the literature and analyses of those data form the body of our paper.
Methods
On January 8, 2012, we entered “methadone and QT prolongation” (81 articles), “methadone and QTc” (71 articles) and then “methadone and torsade” (38 articles) into PubMed limiting our selections to those in English. From these findings, we found 20 case reports in the literature and 12 additional case reports referenced in this literature (one was not in English).
Results
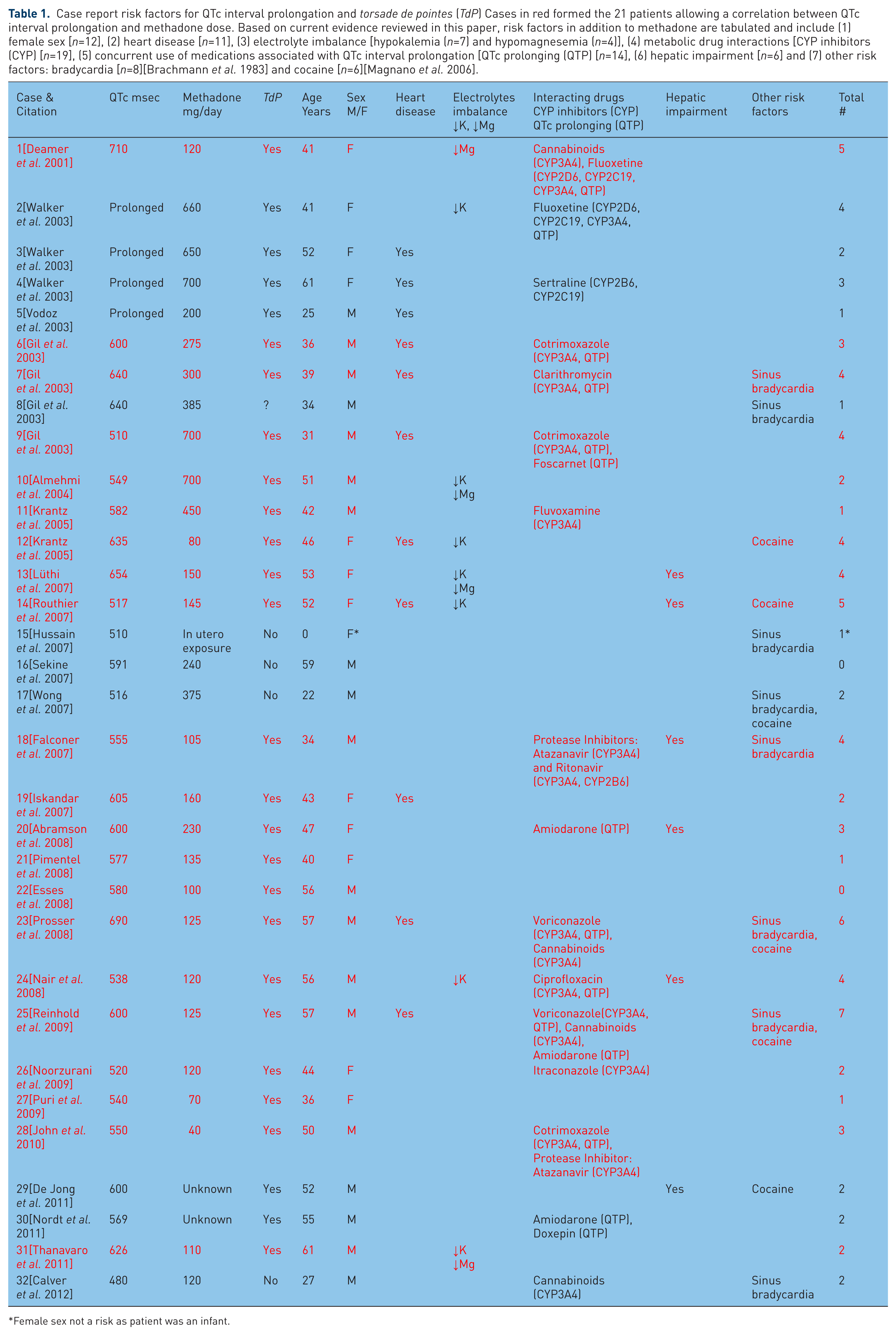
Vital information from these 32 case reports appears in Table 1. Nineteen subjects were men (age 44.4±12.9, range 22-61 years). Twelve subjects were women (age 46.3±7.0, range 36-61 years). One subject was a female newborn.
Case report risk factors for QTc interval prolongation and torsade de pointes (TdP) Cases in red formed the 21 patients allowing a correlation between QTc interval prolongation and methadone dose. Based on current evidence reviewed in this paper, risk factors in addition to methadone are tabulated and include (1) female sex [n=12], (2) heart disease [n=11], (3) electrolyte imbalance [hypokalemia (n=7) and hypomagnesemia (n=4)], (4) metabolic drug interactions [CYP inhibitors (CYP) [n=19], (5) concurrent use of medications associated with QTc interval prolongation [QTc prolonging (QTP) [n=14], (6) hepatic impairment [n=6] and (7) other risk factors: bradycardia [n=8][Brachmann et al. 1983] and cocaine [n=6][Magnano et al. 2006].
Female sex not a risk as patient was an infant.
Among the 21 subjects—nine men and 12 women—(Table 1—patients 1, 6, 7, 9-14, 18-28 and 31 cases are printed in red) (1) developing TdP, (2) having QTc interval measurements in close proximity to the time they developed this arrhythmia and (3) having daily methadone doses recorded at the time of TdP, there were insignificant correlations between QTc interval and methadone dose using both parametric and nonparametric statistics (Pearson r = −0.263, p = 0.249, Kendall’s tau_b −0.39, p = 0.808, Spearman’s rho = 0.005, p = 0.982). When we assessed the correlation between methadone dose and total risk factors (Table 1), we again found an insignificant inverse relationship using both parametric and nonparametric statistics (Pearson r = −0.084, p = 0.663, Kendall’s tau_b −0.46, p = 0.744, Spearman’s rho = −0.071, p = 0.714). (Even though all subjects received methadone and this drug constitutes a risk factor for QTc interval prolongation, we did not include methadone in our calculations of the relationship between methadone dose and total risk factors.) This is not a case control study and all statistical assertions made above have significant limitations. We discuss these limitations further when we compare our findings with those from the literature in the discussion.
Major risk factors (Table 1) for QTc interval prolongation and TdP among the 31 adult methadone users in our sample included (1) female sex (n=12), (2) heart disease (n=11), (3) electrolyte imbalance [hypokalemia (n=7) and hypomagnesemia (n=4)], (4) metabolic (CYP) drug interactions (n=19), (5) concurrent use of medications associated with QTc interval prolongation (n=14), (6) hepatic impairment (n=6), (7) and other risk factors: sinus bradycardia (n=8) and cocaine (n=6). Twenty-four of 31 adult patients (77.4%) had multiple risk factors besides methadone. This observation may add importantly to understanding our data.
Discussion
Our two main findings (Table 1) were (1) using both parametric and nonparametric statistics, no obvious relationship between methadone dose and QTc interval prolongation in patients taking methadone and developing TdP and (2) the common finding of multiple risk factors for TdP present in patients taking methadone without any obvious correlation between methadone dose and number of risk factors. The risk factors we identified (Table 1) were similar to those previously reported among methadone patients[Krantz et al. 2002; Hanon et al. 2010] and patients taking noncardiac drugs [Viskin et al. 2003]. In non-methadone psychotropic drug-induced/associated TdP, two women appear for every man. Among the elderly with this problem, women may represent up to 90% of the cases [Vieweg et al. 2009]. However, adult men appeared more commonly than women in our study (19 versus 12).
Predicting methadone-induced QTc interval prolongation and TdP
Investigators have proposed large subject sizes to predict methadone-induced QTc interval prolongation and associated TdP [Cruciani, 2008], but such recommendations assume that parametric statistics apply in this setting and this appears to be a false assumption [Taleb, 2010]. Methadone exposure may link to increased sudden cardiac death (SCD) in the community even among those with therapeutic levels of methadone [Chugh et al. 2008]. QTc interval prolongation, when it reaches 500 msec or more, predicts a population vulnerable to polymorphic ventricular tachycardia and its better known subtype TdP [Vieweg et al. 2009, Vieweg et al. 2011]. However, the rarity of these arrhythmias precludes using QTc interval prolongation alone to quantitate the risk of drug-induced SCD.
Anchersen et al. [2009] reported the prevalence of QTc interval prolongation among subjects in opioid maintenance treatment and the potential mortality associated with QTc interval prolongation in the Norwegian opioid maintenance treatment program. Among the 173 patients receiving methadone, 4.6% had QTc interval >500 msec (all taking methadone ≥120 mg), 15% had QTc interval >470 msec and 28.9% had QTc interval >450 msec. The authors found a positive dose-dependent relationship between methadone dose and QTc interval lengthening (Pearson r=0.37, p<0.01; multiple regression analysis B=0.37, p<0.01). A case control study was not performed. During the first month of methadone maintenance treatment, two deaths occurred among 3850 initiations of methadone maintenance. One patient died from intracranial bleeding and one cause of death was unknown. Anchersen et al. [2009] concluded that death due to methadone during the first month of treatment was low and did not exceed 0.06 per 100 patient-years. The authors did not report the presence or absence of risk factors associated with TdP and attendant QTc interval prolongation in this paper but did look at their patients with QTc interval >500 msec in a second paper [Anchersen et al. 2010].
Anchersen et al. [2010] provided a detailed review of their earlier subjects with QTc interval >500 msec with a particular focus on risk factors. Assessment included a detailed medical and cardiac history, genetic testing for the five most common long QT syndrome mutations, cardiac exercise testing and 24-hour Holter monitor records. The authors assessed seven subjects and found that two were heterozygous long QT syndrome mutation carriers who had both experienced cardiac complaints before and during opioid maintenance treatment. The authors detected no additional risk factor among these seven patients. In six subjects, QTc intervals fluctuated widely during 24-hour Holter monitor recording and exercise testing [Anchersen et al. 2010].
Methadone-associated QTc interval prolongation
More than 80% of patients receiving long-term methadone maintenance therapy may have QTc interval prolongation compared with reference values for patients of the same age and sex and there may be no correlation between QTc interval measurement and methadone dose [Maremmani et al. 2005]. However, others have reported a positive relationship between methadone dose and QTc interval duration [Kornick et al. 2003; Krantz et al. 2003]. Among our 21 cases of methadone-associated TdP, we failed to identify any relationship between QTc interval prolongation and methadone dose. Perhaps, multiple risk factors present may explain this finding.
A paper recently published by Roy et al. [2012] studied 180 subjects (69.1% men) in a methadone maintenance therapy program. Mean QTc interval was 420.9 ± 21.1 msec and the mean daily methadone dose was 80.4 ± 27.7 mg. There was no significant correlation between these two measurements (p = 0.33) with 8.8% of patients demonstrating QTc interval prolongation (8.3% men and 0.5% women).
In contrast to the uneven findings of the relationship between QTc interval prolongation among TdP patients and methadone dose in our and other studies, Miceli et al. [2010] found a significant positive correlation between therapeutic doses of both ziprasidone and haloperidol and QTc interval measurements in 59-medically-healthy adults with either schizophrenia or schizoaffective disorder in a well-controlled study. No patient developed QTc interval of ≥450 msec. Several factors distinguished the Miceli et al. paper from the methadone papers: (1) QTc interval measurements related positively to antipsychotic drug dose (i.e., the higher the drug dose, the greater the QTc interval), (2) neither ziprasidone nor haloperidol administrations were associated with QTc interval prolongation or TdP and (3) no risk factors for QTc interval prolongation were present such as drugs that inhibited antipsychotic drug metabolism or linked to QTc interval prolongation itself.
Case series in the literature
Krantz et al. [2002] reported 17 patients (mean QTc interval 615±77 msec on presentation) who developed TdP while taking methadone (mean daily dose 397±283 mg). Their data were tabulated but did not contain the breadth and depth of information found in a case report format. The authors concluded that very-high-dose methadone might cause TdP. The next year, Krantz et al. [2003] reported the dose-related effects of methadone on QTc interval prolongation in their 17 patients with methadone-associated TdP. Only the daily dose of methadone predicted the QTc interval (r=+0.51, p=0.03). They did not conduct a case control study.
Hanon et al. [2010] reported a retrospective case series of 12 consecutive patients (mean daily methadone dose 135 mg) hospitalized with methadone-induced QTc interval prolongation and TdP during the study period July 2007 to April 2009. Their hospital was the only referral source for 6500 methadone maintenance patients. Thus monthly (21 months), 12/21 (0.57) of 6500 (0.57/6500=0.0000879) or 8.8×10−5 patients experienced changes sufficient to produce TdP and live long enough to be hospitalized. (Daily risk would be 8.8/30×10−5 or 2.9×10−6 patients or about 3 episodes of TdP daily per 1 million methadone maintenance treatment patients.) The authors did not say how many of these 6500 patients experienced SCD outside the hospital.
Sex differences in methadone exposure and QTc interval prolongation
We found, as did others [Hanon et al. 2010], that men are more commonly involved in cases of methadone-associated QTc interval prolongation and TdP than women. This contrasts to many observations that women are more commonly involved in non-cardiac drug-associated QTc interval prolongation and TdP than are men [Vieweg et al. 2009; Vieweg et al. 2011]. With the onset of puberty, the QT interval shortens for men by about 20 msec (androgen effect) compared with women.
Chang et al. [2011] performed a cross-sectional analysis of low-dose methadone and sex effects in 283 heroin-dependent patients (229 men and 54 women) undergoing QTc interval measurement 59 days (interquartile range: 36 to 288 days) after starting methadone treatment. To assess methadone over time, a 150-patient subset (126 men and 24 women) underwent QTc interval assessment before and 37 days (interquartile range 32-44 days) after starting methadone therapy. Methadone dose-QTc interval correlation was significant for men (r=0.210, p=0.0014) but not for women (r=0.164, p=0.2363). Over six-months, 60.7% of patients developed an increase in their QTc interval compared with baseline measurements. QTc interval significantly increased in men (from 418.5 to 426.9 msec, p<0.0001) but not in women (from 437.7 to 441.1 msec, p=0.468). The authors concluded that (1) low-dose methadone treatment demonstrates dose-dependent QTc interval prolongation and links to significant QTc interval lengthening within six months of starting methadone treatment and (2) men are more susceptible than women are to low-dose methadone-associated QTc interval prolongation.
Martell et al. [2005] prospectively assessed methadone (20-200 mg/day) effects on QTc intervals in 160 patients possessing varying numbers of risk factors for QTc interval prolongation. Women had significantly longer QTc intervals than men at baseline did. At six-month follow-up, however, factors associated with greater QTc interval prolongation included male sex and methadone dose at which time 13% of men and 11% of women demonstrated QTc interval prolongation. At 12-month follow-up, methadone dose marginally linked to greater QTc interval prolongation at which time 20% of men and 2% of women showed QTc interval prolongation. No cases of TdP appeared in this study. Their work [Martell et al. 2005] coupled with others [Maremmani et al. 2005; Sticherling et al. 2005] suggest that more than 80% of patients in methadone maintenance programs have some degree of QTc interval prolongation as discussed earlier.
Men are more likely to abuse drugs than women are. Zickler [2000] traces this finding to opportunities to use drugs of abuse. This observation applies to heroin abusers and, therefore, subsequent abusers requiring methadone maintenance therapy. Among our adults, 19 of 32 case reports (59.4%) involved men (Tables 1). In the Hanon et al. [2010] series, 9 of 12 (75%) cases were men. In the Chang et al. [2011] series, 229 of 283 (80.9%) subjects were men. If methadone was the main risk factor for TdP and uniformly led to QTc interval prolongation among women and men in the cases under discussion, we would expect this drug to maintain the 2:1 F:M ratio found in non-methadone psychotropic drug-induced cases of QTc interval prolongation and TdP [Vieweg et al. 2009; Vieweg et al. 2011]. We believe the preponderance of male methadone cases is because men are much more likely than women to require methadone treatment for a variety of reasons and this findings overrides greater female vulnerability to drug-induced QTc interval prolongation and TdP [Makkar et al. 1993].
Risk factors for QTc interval prolongation and TdP
We have reviewed risk factors for QTc interval prolongation and TdP previously [Vieweg et al. 2009]. Electrolyte disturbances [particularly hypokalemia (commonly diuretic-induced) and hypomagnesemia (commonly found during hospitalization in general and intensive care units [Agus, 1999]) and among alcoholics [Backmund et al. 2003]] are among these risk factors [Arbel et al. 2007; Hanon et al. 2010].
Bednar et al. [2001] in their Table 2 identified 16 factors prolonging the QTc interval. Those risk factors included (1) congenital, (2) increasing age, (3) female sex, (4) meals, (5) sleep, (6) drugs, (7) obesity/weight gain, (8) liquid protein diet, (9) alcoholism, (10) electrolyte disturbances (hypokalemia, hypomagnesemia, hypocalcemia), (11) hypoglycemia/diabetes mellitus, (12) myocardial ischemia and infarction, cardiomyopathy, (13) hypertension, (14) hypothyroidism/pituitary insufficiency, (15) central nervous system insult: stroke, subarachnoid hemorrhage, trauma, infection, tumor and (16) cirrhosis.
Viskin et al. [2003] in their review (Figure 3 derived from 229 published cases) of the long QT syndrome caused by noncardiac drugs identified seven major risk factors in order of greatest to least as follows: (1) female sex, (2) heart disease, (3) hypokalemia, (4) toxic drug levels, (5) drug interactions involving the QT interval, (6) metabolic drug interactions and (7) familial history of long QT syndrome. The authors found that 96% of their study subjects had one or more risk factors, 72% had two or more risk factors and 39.5% had three or more risk factors. In our Table 1, we slightly modified this list to include (1) female sex, (2) heart disease, (3) hypokalemia/hypomagnesemia, (4) drug interactions involving the QT interval and metabolic drug interactions, (5) hepatic impairment and (6) others including sinus bradycardia and cocaine.
High-dose methadone (120 mg/day and above)
The United Kingdom methadone treatment guidelines [Department of Health (England) and the Devolved Administrations, 2007] recommend obtaining an EKG in persons taking greater than 100 mg of methadone per day. Anchersen et al found an association with dosages ≥120 mg and QTc interval > 500 msec [Anchersen et al. 2009]. Twenty-three of our 31 adults (74.2%) were exposed to daily methadone doses of 120 mg or greater (Table 1). In the Hanon et al. [2010] series, seven of 12 subjects (daily dose range 35 to 250 mg) received daily doses of methadone 140 mg or greater. In contrast to daily methadone doses of up to 700 mg daily (our Table 1), these patients were exposed to no more than 250 mg of methadone/day. In our sample, seven cases (25.8%) experienced QTc interval prolongation at doses less than 120 mg per day, with the lowest dosage reported being only 40 mg of methadone. Even though a methadone dose effect on QTc interval lengthening may appear [Krantz et al. 2003; Chang et al. 2011], its clinical utility is highly questionable [Cruciani, 2008].
Drug interactions
Co-administration of methadone metabolic inhibitors
Methadone is metabolized hepatically by N-demethylation via cytochrome P-450 isoforms, primarily CYP3A4, CYP2B6, CYP2C19 and to a lesser degree by CYP2C9 and CYP2D6. Coadministration of methadone with enzyme inducers may cause more rapid methadone metabolism potentially decreasing methadone effects. Coadministration of CYP inhibitors may slow metabolism thereby potentiating methadone’s effects. When coadministering methadone with drugs known to both induce and inhibit CYP enzymes, its pharmacokinetics may change unpredictably. Antiretroviral drugs such as efavirenz, nelfinavir, nevirapine, ritonavir and lopinavir + ritonavir combination will inhibit some CYPs. These drugs may also reduce methadone plasma levels due to CYP induction. Therefore, always evaluate drugs concomitantly administered with methadone for their interaction potential and evaluate individual response to drug therapy before adjusting the dose [Anderson et al. 2000; Roxane Laboratories, 2003].
Although methadone is primarily metabolized by CYP3A4, CPY2B6 and CYP2C19 are also important in methadone metabolism. CYP3A4 metabolic inhibitors administered to our 31 adult patients (Table 1) included fluoxetine (n=2), cannabinoids (n=4), clarithromycin (n=1), cotrimoxazole (sulfamethoxazole/trimethoprim) (n=3), fluvoxamine (n=1), protease inhibitors (n=3), ciprofloxacin (n=1), itraconazole (n=1) and voriconazole (n=2). CYP2B6 metabolic inhibitors administered to our 31 adult patients (Table 1) included sertraline and ritonavir, a protease inhibitor. CYP2C19 inhibitors included fluoxetine and sertraline.
Concomitant administration of drugs that may prolong the QTc interval may lead to QTc interval prolongation and TdP. Medications with the potential to prolong the QTc interval in our 31 adult patients (Table 1) included amiodarone (n=3), ciprofloxacin (n=1), cotrimoxazole (n=3), doxepin (n=1), foscarnet (n=1), fluoxetine (n=2) and voriconazole (n=2).
Multiple risk factors and TdP among subjects exposed to methadone
Among the 27 case reports involving TdP (Table 1), 22 (81.5%) had multiple risk factors for this potentially fatal cardiac arrhythmia—that is, risk factors in addition to exposure to methadone. This characteristic has been reported by others and our group previously [Zeltser et al. 2003; Vieweg et al. 2009].
Zeltser et al. [2003] reviewed risk factors for TdP among subjects taking non-cardiac drugs. They asserted that these risk factors (female sex, heart disease, electrolyte imbalances, excessive dosing, drugs interactions and family history of long QT syndrome) are easily identifiable from the medical history and/or clinical evaluation. In their review, they identified 249 subjects with TdP associated with non-cardiac drugs. Female sex was the most common risk factor (71%), almost all of their subjects had at least one risk factor and 71% had two or more risk factors. The authors concluded clinicians planning to prescribe non-cardiac drugs associated with TdP could easily identify risk factors for TdP before prescribing the culprit drug. Unfortunately, Zeltser et al. do not report how often the culprit drug is prescribed for patients with risk factors for TdP and yet it does not occur—hence, a major problem in estimating risk.
Included in risk factors for TdP may be acute structural cardiac changes in certain settings. Crotti et al. [2012] recently described TdP following acute myocardial infarction (AMI) in a genetic substrate predisposed to a time-limited development of life-threatening ventricular tachyarrhythmias. The authors studied 13 patients who developed TdP during the subacute phase of AMI. The comparison group constituted 133 ethnically-matched controls with uncomplicated AMI. They screened for long QT syndrome genes and the KCNH2-K897T polymorphism. They found that two of the 13 patients presenting with QTc interval prolongation and TdP carried long QT syndrome mutations (KCHN2-R744X and SCN5A-E446K). Nine of the remaining 11 patients carried the KCNH2-K897T polymorphism as did 35% of controls (p=0.0035). The authors concluded that AMI patients carrying the KCNH2-K897T polymorphism are eight times more likely to develop TdP than controls. Administration of methadone or any other drug associated with QTc interval prolongation in this setting likely would further increase the risk of developing TdP.
Scalable randomness and predicting drug-induced TdP
Raschi et al. [2009] recently reviewed models for predicting hERG liability (IKr blockade) and QT interval prolongation. They reported that 40 to 70% of new model entries considered as potential therapeutic drugs are abandoned early in development because they test positive for hERG blocking liability. However, lack of hERG blocking liability does not preclude a drug from linking to QTc interval prolongation and TdP.
Our study suggests that QTc interval prolongation in the setting of TdP is not linked to methadone dose, is not predictable at present and may not be Gaussian in distribution. Raschi et al. [2009] identified many risk factors underlying drug-induced TdP onset including organ impairment, drug interactions, electrolyte imbalance and genetic mutations leading to reduced repolarization reserve. Additional mechanisms and mathematical models are probably operative—especially Chaos theory [Gleick, 1987]. Minute differences in the initial conditions (genetic makeup, medication profile, electrolytes, structural abnormalities, etc.) tend to evolve into extraordinarily different outcomes (slight QT prolongation vs TdP) when exposed to methadone. We believe that any useful effort to predict methadone-associated TdP and attendant QTc interval prolongation must better understand the role of the above-identified risk factors in TdP.
Taleb [2010] discussed the concept of scalable randomness citing an example as follows. Expect a project to be finished in 79 days. If not completed, anticipate another 25 days. However if the project remains unfinished by the 90th day, it should take another 58 days to complete. On the 100th day, it should have 89 days to completion and on the 119th day, it will have another 149 days to go. If not finished by the 600th day, an additional 1590 days will be necessary. It follows that added and unanticipated complexity drives each delay. Similarly, added and unanticipated risk factors appear in patients destined to develop TdP independent of the original drug prescribed. If so, the QTc interval-prolonging drug may be a prerequisite but not sufficient without additional risk factors to best explain drug-associated TdP. It follows that multiple risk factors including the drug itself add complexity and uncertainty sufficient to negate parametric statistics as an approach to studying this problem.
What to do with our data and similar data from the literature?
Heroin dependency was the most common indication for methadone administration among our 31 adult subjects (Table 1). Twenty-four of them (77.4%) received methadone to treat heroin dependence and seven (22.6%) received methadone for pain relief. These are the two most common indications for methadone administration and simply tell us that any patient taking methadone may be at risk for TdP. The rarity of TdP among methadone users precludes knowing how many methadone users do not develop TdP and what factors separate these two groups. Therefore, we must study the published data on the subject via the case report format to learn as much as possible about TdP. Unfortunately, both the FDA and drug manufacturers tend not to make case report material available compounding the effort to better understand the relationship between psychotropic drugs and TdP in general and the relationship between methadone and TdP in particular.
Study limitations
The most obvious study limitations are data derived from case reports and the inability to provide risk estimates. Narrative medicine in the form of case reports is inherently selective and may or may not provide representative data of clinical relevance to the medical issues discussed. Studying extreme outliers (Black Swans [Taleb, 2010]) in an effort to better understand TdP may be inherently impossible.
Conclusions
At present, clinicians and oversight agencies such as the FDA must study case report material for guidance despite its limitations and hazards. We describe risk factors for methadone-associated QTc interval prolongation and TdP based on case reports. We believe both drug manufactures and the FDA would better protect our patients and better inform clinicians if they more readily reported drug-induced outliers using a case report format with a particular emphasis on multiple risk factors.
