Abstract
Tigecycline is the first of a new class of antibiotics named glycylcyclines and is active in vitro against a variety of gram-positive and gram-negative organisms, including nosocomial resistant pathogens such as vancomycin-resistant Enterococcus faecium, methicillin-resistant Staphylococcus aureus, extended-spectrum β-lactamase-producing Enterobacteriaceae, and multidrug-resistant-Acinetobacter spp. This medication has been approved by the US Food and Drug Administration (FDA) for the treatment of complicated intra-abdominal infections (cIAI), complicated skin and skin structure infections (cSSSI) and community-acquired bacterial pneumonia. Tigecycline's pharmacological and microbiological profile has also encouraged physicians’ to use the drug in other infections caused by resistant pathogens featuring limited therapeutics options (i.e. hospital-acquired pneumonia-HAP). In this study we publish the conclusions of an expert panel that identify and evaluate the evidence to support the use of Tigecycline in hospitalized patients with one of the following three infections: cSSSI, cIAI and HAP, including ventilator-associated pneumonia. Based on this data the panel developed an Algorithm Rational to Prescribe Tigecycline (ART) for each pathology.
Introduction
Multidrug-resistant (MDR) bacteria is found in many hospitals and contributes to increased morbidity, patient length-of-stay, and ultimately, mortality rates.1,2
In 2009, the Tigecycline Evaluation and Surveillance Trial (T.E.S.T.), which includes 133 centers from Latin America, published worrisome rates of nosocomial bacterial resistance in the region: 46.3% of vancomycin-resistant Enterococcus faecium (VRE), 46.6% methicillin-resistant Staphylococcus aureus (MRSA), 37.9% of extended-spectrum β-lactamases (ESBL)-producing Klebsiella pneumoniae, and 37.6% of Acinetobacter spp and 35.8% of Pseudomonas aeruginosa carbapenem-resistant and 43.4% of AmpC-producing Enterobacter spp. 3
Rice et al 4 recently reported these as the “ESKAPE” pathogens in order to emphasize that they currently cause the majority of worldwide hospital infections and effectively “escape” the effects of antibacterial drugs.
Tigecycline is the first of a new class of antibiotics named glycylcyclines and is active in vitro against a variety of gram-positive and gram-negative organisms, including nosocomial resistant pathogens such as VRE, MRSA, ESBLs-producing Enterobacteriaceae, and MDR-Acinetobacter spp. 5
It has been approved by the US Food and Drug Administration (FDA) for the treatment of complicated intra-abdominal infections (cIAI), complicated skin and skin structure infections (cSSSI) 5 and community-acquired bacterial pneumonia (CABP). 6
Notwithstanding this, the tigecycline's pharmacological and microbiological profile encourages physicians’ to use the drug in other infections caused by resistant pathogens with limited therapeutics options. In Argentina in 2007, Curcio et al published a report, noting that the tigecycline prescriptions were for non-approved indications in 79% of patients. 7
The objective of this study was to identify and evaluate the evidence to support the use of tigecycline in hospitalized patients in three infections: cSSSI, cIAI and hospital-acquired pneumonia (HAP), including ventilator-associated pneumonia VAP. Based on this data we developed an Algorithm Rational to Prescribe Tigecycline (ART) for each pathology.
Materials and Methods
A group of Argentinean infectious diseases (n = 12), lung (n = 1), critical care (n = 8) and microbiology specialists (n = 1), was convened to establish an expert panel for determining the appropriate use of tigecycline in hospitalized patients with cSSSI, cIAI and HAP. The standard procedures recommended by Kish 8 were followed. One of the coordinators (DC) performed a systematic searching of English published literature in PubMed and the “Literatura Latinoamericana y del Caribe en Ciencias de la Salud” (LILACS) databases on March 2009. The only term used was “tigecycline” for articles that evaluated: i-clinical trials, ii-in vitro activity, iii-pharmacodynamic profile, iv-cases series, v-individual case reports and vi-reviews.
In addition, were included tigecycline abstracts and posters from the most relevant international congresses (Interscience Conference on Antimicrobial Agents and Chemotherapy–-ICAAC-, Infectious Diseases Society of America Meeting–-IDSA- and European Congress of Clinical Microbiology and Infectious Diseases–-ECCMID-) between 2004 and 2008.
Three members (DC, LIB and WC) selected and classified the data and provided this information to the rest of the group through an ad hoc-created website, which was available one month before a one-day consensus meeting to be held in Argentina on April 22, 2009.
Eighty eight out of more than 400 articles found were selected (clinical trials n = 12, in vitro activity n = 25, pharmacodynamic profile n = 7, iv-cases series n = 28, individual case reports n = 11 and vi-reviews n = 5. Based on these articles, DC developed a questionnaire with 53 questions related with the potential role of tigecycline in the pathologies under review. Each member of the panel had to assign to each question a value within a scale between 1 (extremely inappropriate) and 9 (extremely appropriate). They were received by e-mail, by one of the coordinators (DC) 7 days before the meeting and after considering the responses, he developed the first draft of the ART for each pathology.
We then assigned three members of the panel to each present a draft ART for a plenary discussion in the meeting: PB (cSSSI), LB (cIAI) and RR (HAP).
After the consensus meeting, two members (DC and LIB) proceeded to develop the final version of the ARTs and the first version of the manuscript, which was circulated among the members for revision.
This procedure was repeated several times until all participants approved the final version. It must be noted that the definitive recommendations and ARTs arose from a uniform consensus.
Results
Main concepts of the analyzed evidence
Tigecycline in vitro activity
Surveillance studies provide invaluable information in the tracking of antimicrobial susceptibility both locally and globally.
According to the TEST Program data, tigecycline exhibits antibacterial activity against a wide spectrum of aerobic and anaerobic bacteria, including nosocomial MDR-pathogens.
As with common aerobic gram-positive pathogens, including MRSA, vancomycin-intermediate S. aureus, VRE, and penicillin-resistant Streptococcus pneumoniae, MIC90 values range from 0.12 to 0.5 μg/mL. 9 All these isolates would be considered susceptible to tigecycline at the FDA susceptibility breakpoints of 0.25 and 0.5 μg/mL for streptococci and staphylococci, respectively. 10 In addition, both tetracycline-susceptible and–-resistant strains have exhibited almost identical MIC values for tigecycline.
Community-acquired MRSA (CA-MRSA) isolates are a usually recovered from cSSSI which are associated with younger aged patients, sport team participation, recent incarceration, military recruits, snorting/smoking illegal drugs, lower comorbidity index, more frequent visits to bars, raves, and/or clubs. On a total of 1989 CA-MRSA (94.7% Panton-Valentine leukocidin positive), tigecycline susceptibility rate was 98.2% (MIC90 0.5 μg/mL). 11
Tigecycline is active against most gram-negative bacilli with exception of P. aeruginosa, Proteus spp., or Providentia spp. and many strains of Morganella morganii. Studies of these isolates suggest that these strains have constitutively overexpressed multidrug efflux pump systems (e.g. MexXY and AcrAB) for which tigecycline is a substrate. 12 More than 90% of strains of Pseudomonas aeruginosa have MIC values of ≥ 4 mg/mL and would be considered resistant to tigecycline. 10
For Enterobacteriaceae, tigecycline's MIC90 values for tetracycline-susceptible strains have ranged from 0.25 to 1 mg/mL. 9 Most studies of large numbers of isolates have shown that >95% of all Enterobacteriaceae are susceptible to tigecycline at the FDA susceptibility breakpoint of 2 μg/mL. Strains of Escherichia coli and Klebsiella spp with and without ESBLs have shown very similar MIC values and susceptibility rates to tigecycline. Similar susceptibility profiles were seen for both E. aerogenes and E. cloacae. Against these microorganisms, susceptibility rates of >90.0% were seen for tigecycline. 9
The emergence and dissemination of ESBL has compromised the use of broad-spectrum cephalosporins for empirical treatment of hospitalized patients’ infections caused by various members of the Enterobacteriaceae family. As a consequence, the therapeutic use of carbapenems has increased significantly in some hospitals, and carbapenem-resistant gramnegative bacilli have begun to emerge (P. aeruginosa, Acinetobacter spp. and carbapenemase-producing Enterobacteriaceae). 13
Tigecycline is active against Acinetobacter spp. including strains that are not susceptible to available antibiotics (MIC90 1-2 μg/mL). 9 Arroyo et al have published a report noting that tigecycline shows good activity against 46 MDR-Acinetobacter spp (colistin included), with an MIC90 of 1 μg/mL. 14 However, several authors have reported high resistance rates to tigecycline in multiple clones of MDR Acinetobacter baumannii (MIC90 ≥ 2 μg/mL).15,16 We know that the overexpression of the intrinsic multidrug efflux pump (AdeABC) may decrease the susceptibility to tigecycline in Acinetobacter spp; however, in regards to this, it must be noted that the specific test used to determined susceptibility may influence the MIC value. Sahm et al 17 have found that Acinetobacter spp. tigecycline MIC90 was 2-fold higher by E-test than by broth-based methods (BBM) and MIC90s were no lower by E-test than by BBM. Therefore, we believe that BBM should have been used, at least in the isolates with tigecycline MIC ≥ 2 mg/L, before concluding tigecycline resistance in Acinetobacter spp. Supporting this recommendation are the results found in more than 4000 isolates of Acinetobacter spp tested by BBM in the TEST Program. Only 2% of the results showed a tigecycline MIC ≥ 2 mg/L. 18
The widespread dissemination of carbapenemase-producing Enterobacteriaceae has profound implications for the clinical utility of the carbapenems. 19 Furthermore, carbapenemase-producing Enterobacteriaceae strains were generally resistant to the vast majority of antimicrobial agents available for clinical use, making the therapeutic options very limited. 20 Tigecycline is highly active against carbapenemase-producing Enterobacteriaceae isolates. Castanheira et al 21 have published that all serine or metallo-β-lactamase-producing Enterobacteriaceae isolates (n = 104) were inhibited at the tigecycline susceptibility breakpoint approved by the FDA (<2 μg/mL). This compound was the most active antimicrobial tested against this collection of multidrug-resistant strains (MIC50, 0.5 μg/mL; MIC90, 2 μg/mL).
Tigecycline is active against most anerobic bacteria, including Clostridium difficile, Fusobacterium species, Prevotella species, Poryphyromonas species, and the Bacteroides fragilis group. 10 The FDA susceptibility breakpoint to tigecycline for anerobes is 4 μg/mL, which fits the MIC distribution of anerobic organisms in the clinical trials.
Rossi et al 22 reported the four-year T.E.S.T. data (2004-2007) for Gram-positive and Gram-negative organisms collected from Latin American regional data. The tigecycline MIC90 for all the isolates of mainly MDR-pathogens were within the FDA proposed breakpoints: MRSA(n = 437)0.25 μg/mL, VRE(n = 47, E. faecium) 0.12 μg/mL, imipenem-resistant Acinetobacter spp (n = 104) 2 μg/mL, ESBL-producing K. pneumoniae (n = 280) 2 μg/mL, and ESBL-producing E. coli (n = 194) 0.5 μg/mL. In addition Casellas et al 23 have found in Argentinean isolates, tigecycline has shown excellent activity against S. aureus (n = 325, MIC90 0.25 μg/mL), Acinetobacter spp (n = 132, MIC90 1 μg/mL) and Enterobacteriaceae (n = 729, MIC90 1 μg/mL).
Tigecycline has potentially useful antimicrobial activity but may not always be effective as monotherapy (i.e. infections where P. aeruginosa is suspected or when a high serum level of antibiotic is required). Petersen et al have determined the interaction of tigecycline with other antimicrobial agents against a variety of bacterial isolates using the broth microdilution chequerboard method. 24
The combination of tigecycline and other antibiotics demonstrated either synergy (24%) or no interaction (76%) against the panel of gram-negative bacteria. Antagonism was not observed for any combination with tigecycline, against any of the strains tested. A higher percentage of synergistic combinations with tigecycline were observed with amikacin (56%), ampicillin/sulbactam (33%), piperacillin/tazobactam (50%) and rifampicin (33%). Combinations of tigecycline with amikacin also showed synergy of 40%-100% of Enterobacter spp., Klebsiella pneumoniae, Proteus spp. and Stenotrophomonas maltophilia isolates. Moreover, bactericidal synergisms occurred with tigecycline plus amikacin against MDR-Acinetobacter baumannii and Proteus vulgaris, and with colistin against K. pneumoniae.25,26
Principe et al 27 demonstrated the in vitro synergistic activity of tigecycline in combination with colistin, levofloxacin, amikacin and imipenem against five tigecycline non-susceptible A. baumannii strains, opening the way for a more rational clinical assessment of novel combination therapies to combat infections caused by MDR and pan-resistant A. baumannii. Similarly, Ozbek et al 28 have shown using the microbroth checkerboard technique that the combination of tigecycline with colistin methanesulfonate will produce synergy against carbapenem resistant A. baumannii.
With the gram-positive isolates, rifampicin displayed a synergistic effect with tigecycline for 66% of the isolates tested. The majority of the strains showing synergy were VRE and penicillin-resistant Streptococcus pneumoniae. Mercier et al 29 have found that the combination of tigecycline and gentamicin gave enhanced activity against the three Staphylococcus aureus isolates. Entenza et al 26 established that synergy occurred when tigecycline was combined with rifampicin against 64%-100% of Enterococcus spp., Streptococcus pneumoniae, Enterobacter spp. and Brucella melitensis isolates.
The main conclusion from the available data is that the interaction of tigecycline with other antimicrobials produced an indifferent response primarily and very rarely showed antagonism. Synergy detected by in vitro studies, is an encouraging outcome suggesting that tigecycline may prove to be effective in combination therapy in particular situations (i.e. in potentially problematic multiresistant species and to bacteria inadequately inhibited by tigecycline).
Table 1 shows a wide variety of gram-positive and gram-negative pathogens associated with cSSSI, cIAI and HAP including multidrug-resistant strains. Tigecycline has demonstrated activity against most of them, except P. aeruginosa and P. mirabilis.
Microorganisms isolated from patients with complicated skin and skin structure infections (cSSI), complicated intra-abdominal infections (cIAI), and hospital-acquired pneumonia (HAP).
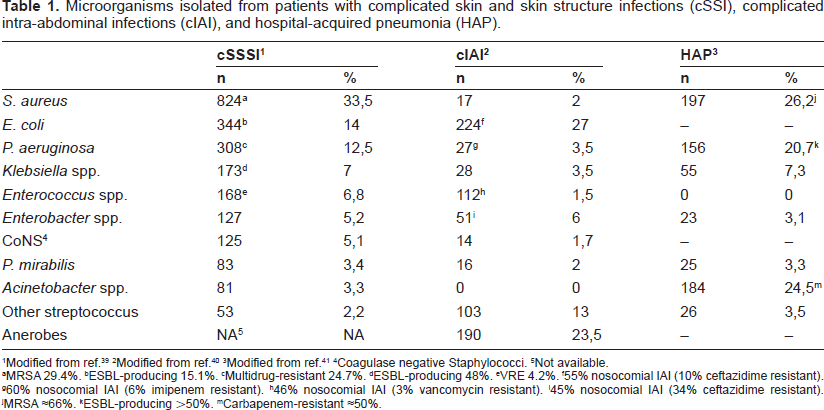
Modified from ref. 39
Modified from ref. 40
Modified from ref. 41
Coagulase negative Staphylococci.
Not available.
MRSA 29.4%.
ESBL-producing 15.1%.
Multidrug-resistant 24.7%.
ESBL-producing 48%.
VRE 4.2%.
55% nosocomial IAI (10% ceftazidime resistant).
60% nosocomial IAI (6% imipenem resistant).
46% nosocomial IAI (3% vancomycin resistant).
45% nosocomial IAI (34% ceftazidime resistant).
MRSA ≈66%.
ESBL-producing ≈50%.
Carbapenem-resistant ≈50%.
Tigecycline pharmacokinetics and pharmacodynamics
Tigecycline is available only as an intravenous (IV) formulation and is administered twice daily as a one-hour infusion. Given at the standard clinical dose of 100 mg followed by 50 mg q12h, tigecycline produced a Cmax of 0.85-1 mg/L and relatively low mean steady-state serum concentrations of 0.403 mg/L and 0.633 mg/L.30,31 The half-life (t1/2) in humans is relatively long, ranging from 37 to 67 h in healthy volunteers, probably because of its large volume of distribution. Studies with 14C in rats have confirmed that tigecycline is distributed extensively to tissues, including lung, skin, liver, heart, and bone. The concentrations of tigecycline in the skin and lungs in rats were approximately three-fold to four-fold higher than those in plasma.30,31
The mean clearance ranged from 0.2 to 0.3 L/h/kg, with ~8%-11% of the drug excreted in the urine as unchanged drug over 48 h. The major route of elimination appears to be through feces, via biliary excretion. Glucuronide conjugates of tigecycline, its epimer, and an N-acetyl-9-aminominocycline were the major metabolites found, in low concentrations, in feces, urine, and serum.30,31
The pharmacokinetic profile of tigecycline is not significantly modified by renal impairment, hemodialysis, or mild hepatic impairment. Moderate (Child Pugh B) and severe (Child Pugh C) hepatic impairment resulted in a 25% and 55% reduction in systemic clearance, respectively.30,31
When tigecycline was used in an experimental neutropenic mice thigh infection model, the time above the MIC (multiplied by a factor varying from 0.5 to 4) was the best predictor of effectiveness of this agent, compared with the Cmax or AUC indices, against E. coli and Klebsiella pneumoniae, while the AUC and the time above the MIC were the best predictors of effectiveness of tigecycline against S. pneumoniae. However, due to prolonged post-antibiotic effect against most pathogens, along with extensive volume of distribution, and prolonged elimination t1/2 in humans, the AUC/MIC ratio has been proposed as the most important pharmacodynamic index of tigecycline. 32
The AUC over 12 h (AUC0-12) of tigecycline in blister fluid after multiple doses of 50 mg every 12 h was 74% of the AUC0-12 in serum. 33
Recent pharmacodynamic analyses of clinical trial data observed an AUC24:MIC breakpoint of 12.5 in complicated skin and soft tissue infections and 7 with Enterobacteriaceae and anerobes in complicated intra-abdominal infections.34,35 Patients who had AUC24:MIC values at or greater than these values had significantly better outcomes than those with lower values. An AUC24:MIC target of 5-10 would be achieved with tigecycline for almost all of the common gram-positive pathogens and with most Enterobacteriaceae.
Tigecycline provides high intracellular concentrations in several tissues (such as lung and bowel) that exceed serum concentrations by 2- to 8-fold.30,31
The AUC0-12 of tigecycline in epithelial lining fluid (ELF) of healthy volunteers was 32% higher than the AUC0-12 value in serum. One would expect higher AUC values with pneumonia. However, tigecycline concentrations in alveolar macrophages of healthy volunteers were 78 times higher than in those concentrations found in serum. 36 Tigecycline concentrations in neutrophils have also been 20- to 30-fold higher than extracellular concentrations of the drug. While well-controlled clinical data are required to fully assess the viability of tigecycline as a therapeutic modality for pneumonia, Conte et al 36 reported that the Cmax/MIC90, AUC/MIC90, time/MIC90 and extended serum and intrapulmonary half-lives of this compound were favourable for the treatment of tigecycline-susceptible respiratory pathogens. However, Koomanachai et al 37 have studied in a murine model the in vivo bactericidal activity of tigecycline against various A. baumannii (MIC 0.25-1.0 mg/L) causing pneumonia. These studies revealed that AUC/MIC exposures of 2.17 and 8.78 were required to produce 1 and 2 log kill, respectively. Given these confounding issues, extrapolation of our current dataset to man suggests that tigecycline doses of up to 200 mg/day may be required to provide adequate exposure for A. baumannii. In concordance with Koomanachai, Burkhardt et al 38 have found that attainable extracellular ELF concentrations of tigecycline in patients with ventilator-associated pneumonia were absolutely insufficient to eradicate reliably extracellular bacteria such as S. pneumoniae, S. aureus, Escherichia coli or Klebsiella pneumoniae, based on the agent's current MIC profile, whereas the mean concentrations in alveolar cells exceed the MIC90 of all atypical bacteria through the entire dosing interval. Therefore, the current dosage of 50 mg tigecycline twice daily is probably underdosed for the treatment of pneumonia caused by typical, extracellular-acting bacteria. However, several non-controlled studies have shown that patients with VAP treated with tigecycline achieved an acceptable rate of clinical success using the standard doses (100 mg loading doses and 50 mg q12h).39–41
In summary, tigecycline has an attractive pharmacological profile to treat cSSSI, cIAI and HAP. However in the particular case of HAP, more pharmacodynamics studies are needed to optimize the dosing regimen for this particular indication.
Tygecycline clinical efficacy
Complicated skin and skin structure infections
Two phase 3, randomized, double-blind studies evaluating the efficacy of tigecycline, compared with vancomycin plus aztreonam, have been conducted in hospitalized patients with complicated skin/skin-structure infections. Both studies had the same design, and a pooled analysis of the results from these clinical trials has been reported elsewhere. 42 Patients received tigecycline (100 mg, followed by 50 mg every 12 h), or vancomycin (1 g every 12 h) plus aztreonam (2 g every 12 h) for up to 14 days. The clinically evaluable (CE) population included 833 patients–-422 treated with tigecycline, and 411 treated with vancomycin-aztreonam–-and 540 of these patients (279 tigecycline recipients and 261 vancomycin-aztreonam recipients) had a pretherapy isolate diagnosed. Most patients had extensive cellulitis or a soft-tissue infection (86% vs. 88%). Similar eradication rates were observed in the 2 treatment groups in patients with monomicrobial and polymicrobial infections.
Of note, tigecycline was as effective as vancomycin-aztreonam (78%, vs. 76%) in eradicating strains of MRSA. Of the total of 32 patients treated with tigecycline and 33 patients treated with vancomycin-aztreonam who had MRSA as a baseline isolate, 21 (32%) were CA-MRSA strains (9 in patients treated with tigecycline and 12 in patients treated with vancomycin-aztreonam). Eradication for patients with CA-MRSA was reported for 77.8% of tigecycline- and 75.0% of vancomycin-aztreonam–-treated patients
Florescu et al 43 have published a phase 3, multicenter, double-blind, randomized study which evaluated the safety and clinical efficacy of tigecycline compared with vancomycin in patients with selected serious infections caused by MRSA. In patients with cSSSIs caused by MRSA (69.2% of all patients), cure rates were similar after treatment with tigecycline (51 of 59 patients; 86.4%) or vancomycin (20 of 23 patients; 86.9%); confirmed that tigecycline is safe and effective in hospitalized patients with serious infection caused by MRSA.
Another study has assessed the predicted pharmacodynamic cumulative fraction of response (CRF) of tigecycline, compared to piperacillin/tazobactam, ceftriaxone, levofloxacin, and imipenem/cilastatin, in patients with complicated skin and skin structure infections. 44 Specifically, population pharmacokinetic data obtained in relevant clinical studies were applied in a two-compartmental intravenous infusion model using a 5.000-patient Monte Carlo simulation. Recent worldwide susceptibility data for pathogens that commonly cause complicated skin and skin structure infections were taken into consideration. Tigecycline monotherapy was associated with a high predicted pharmacodynamic cumulative fraction of response (88.2%). Similar values were achieved for the other compared agents, however only when each was assessed in combination with vancomycin.
Thus, this data demonstrated that tigecycline is efficacious in the treatment of patients with cSSSI, especially in a setting in which empirical coverage of gram-positive and gram-negative pathogens is warranted.
Complicated intra-abdominal infections
A pooled analysis of 2 phase 3, double-blind, randomized studies in patients with cIAI has also been reported by Babinchak et al. 45 In both of these trials, tigecycline (100 mg, followed by 50 mg every 12 h) was compared with imipenem-cilastatin (500 mg every 6 h). Treatment was for 5-14 days, and clinical response was evaluated at 12-42 days after therapy. The clinically evaluable population contained 1382 patients (585 received tigecycline, and 607 received imipenem-cilastatin), of whom 1262 had a pretherapy isolate recovered from their infection site. Complicated appendicitis (50%) was the most common infection diagnosis, followed by complicated cholecystitis (14%). Overall, clinical cure rates were similar (86%) in the treatment groups. Bacterial eradication rates were also similar between groups. For E. coli, the most commonly isolated pathogen, eradication rates were 86% for tigecycline and 87% for imipenem-cilastatin. Twelve (80%) of 15 patients with ESBL-producing E. coli or Klebsiella pneumoniae achieved bacterial eradication after receiving tigecycline. Two patients had isolates (K. pneumoniae and M. morganii) that were initially susceptible to tigecycline but that were subsequently found to be resistant (MIC value for each, 8 μg/mL) following therapy; these patients experienced clinical failure. Eradication rates for anaerobic bacteria were similar to those for aerobic organisms. For example, tigecycline's eradication rates for isolates of B. fragilis and Clostridium perfringens were 78% and 95%, respectively. A similar success rate against these anaerobic pathogens was observed in patients who received imipenem-cilastatin.
In a similar study, Towfigh et al 46 have developed another multicenter, open label, randomised, comparative study of tigecycline (100 mg loading dose followed by 50 mg q 12 hours) versus ceftriaxone (CTX) (2 grams once daily) plus metronidazole (MET) (1 gram to 2 grams daily) for the treatment of hospitalised subjects with cIAI. This study was conducted in 6 countries in North, Central and South America. For the CE population, clinical cure rates were 70.4% (133/189) for tigecycline versus 74.3% (139/187) for CTX plus MET (95% CI -13.1, 5.1; p = 0.009 for non-inferiority). Clinical response for subjects with Acute Physiology and Chronic Health Evaluation (APACHE) II scores >10 were 56.8% (21/37) for tigecycline versus 58.3% (21/36) for CTX plus MET. Microbiologic efficacy was similar between the two treatment arms with 68.1% (94/138) of tigecycline treated organisms and 71.5% (98/137) of CTX plus MET treated organisms considered eradicated at the test of cure.
Twenty-one patients with cIAI in which MRSA was also isolated, were included in the aforementioned Florescu et al 43 study, 15 and 6 in the tigecycline and vancomycin arm respectively. The cure rates with tigecycline in the microbiologically evaluable population (including other infections nor cSSSI) were comparable to those with vancomycin (76.4% and 80.9%).
Eagye et al 47 have calculated, using the Monte Carlo model, CFR against aerobic bacteria involved in secondary peritonitis for cefepime, ceftazidime, ceftriaxone, imipenem, levofloxacin, piperacillin/tazobactam, and tigecycline. A CFR ≥ 90% was considered microbiologic success. A CFR ≥ 90% against nonenterococcal bacteria was predicted for imipenem 500 mg q6h (96.8%), cefepime 2 and 1 g q12h (95.3% and 92.4%, respectively), ceftazidime 2 g q8h (94.2%), and piperacillin/tazobactam 3.375 g q6h (91.2%). A CFR of 84.5% was predicted for tigecycline 50 mg q12h. Ceftriaxone and levofloxacin were predicted to have a CFR < 80%. When enterococci were included in the model, the predicted CFRs for imipenem, piperacillin/tazobactam, and tigecycline were 93.4%, 88.4%, and 86.7%, respectively. Using current global data, the following regimens are adequate choices if Enterococcus is not targeted: Combination therapy (with metronidazole) using cefepime 1 g or 2 g q12h, or ceftazidime 2 g q8h; or monotherapy with imipenem 500 mg q6h or piperacillin-tazobactam 3.375 g q6h. When Enterococcus is included in the epidemiologic mix, imipenen, piperacillin/tazobactam, and tigecycline all appear to be viable monotherapeutic choices.
As happens with other new antibiotic trials, in all the tigecycline cIAI clinical trials, severely ill patients, such as those with APACHE II scores > 30 were excluded. In the studies tigecycline vs. imipenem and tigecycline vs. CTX + MET, patients with APACHE II ≥ 15 were 2.1% and 2.9% respectively.45,46
However, there are several case reports of patients with septic shock secondary to cIAI treated with tigecycline with clinical success, in monotherapy (VRE infection) 48 and in combination therapy (colistin and meropenem in MDR-Acinetobacter baumannii infection). 49
Swoboda et al 50 have published data from 70 severely ill patients with severe sepsis or septic shock (mean APACHE II of 27) who received tigecycline in the intensive care unit. Intra-abdominal infection (alone or in combination with other infection) was most frequently diagnosed (69% of patients). The majority of patients received tigecycline in combination with other broad-spectrum antibiotics (i.e. carbapenems). The authors concluded that tigecycline treatment of this group of patients appeared to result in remarkably low mortality (30%), therefore may be an important treatment option for septic patients with cIAI.
Hospital acquired-pneumonia
Tigecycline has also been evaluated in a phase III, multicenter, randomized, double-blind trial regarding the treatment of patients with hospital-acquired pneumonia, in comparison to imipenem/cilastatin (ClinicalTrials.gov Identifier: NCT00080496). Patients received tigecycline (100 mg, then 50 mg q12h) or imipenem/ cilastatin (0.5 to 1 g q8h). Tigecycline patients could receive ceftazidime and aminoglycoside to cover P. aeruginosa and imipenem/cilastatin patients could receive aminoglycoside, and vancomycin for MRSA coverage. Cure rates (CE) at test of cure–-TOC-weretigecycline67.9%, imipenem78.2%, absolute difference tigecycline-imipenem/cilastatin -10.4% (95% CI -17.8, -3.0). A significant interaction was observed between ventilator-associated pneumonia (VAP) and non-VAP patients. Tigecycline was noninferior to imipenem/cilastatin for non-VAP patients (CE cure: 75.4% vs. 81.3%). However, in the subset of patients with ventilator-associated pneumonia, tigecycline was associated with lower effectiveness compared to imipenem/cilastatin.
Two other tigecycline trials which were not specifically designed for this pathology have included patients with HAP. In one of these, which evaluated the clinical efficacy of tigecycline compared with vancomycin in MRSA infection, 9 HAP were included in the tigecycline arm. 43 Overall, clinical cure rates in the microbiologically evaluable population at the TOC were 81.4% with tigecycline. The specific outcome of the HAP group patients was not described in the published results of this trial. The other study was an open label trial to evaluate the efficacy and safety of tigecycline in patients with selected serious infections caused by resistant Gram-negative bacteria, or failures that had received prior antimicrobial therapy or were unable to tolerate other appropriate antimicrobials. Twenty three patients with HAP were included. In the microbiologically evaluable population at TOC (5 patients with HAP), the clinical cure rate was 72.2% [95% confidence interval CI: 54.8-85.8], and the microbiological eradication rate was 66.7% (95% CI: 13.7-78.8). The most commonly isolated resistant Gram-negative pathogen was Acinetobacter baumannii. In this case, again, the specific outcome of the group of HAP patients was not described. 52
Apart from clinical trials, several observational studies of patients with HAP treated with tigecycline have been reported in the biomedical literature.
A study performed in 12 institutions of Argentina 53 showed that, during the first months after its launch, 78% of the tigecycline prescriptions were for “off label” indications, mainly VAP (56%).
Schafer et al 39 evaluated the use of tigecycline alone or in combination with other antimicrobials for treatment of patients (n = 25) with VAP and/or bacteremia caused by multidrug-resistant Acinetobacter baumannii. Overall, 21 (84%) had clinical resolution. The median duration of tigecycline therapy was 20 days (range 2-56 days) and the antibiotics most frequently used for combination therapy were imipenem and colistin (intravenous or nebulized). Four had clinical failure: three with VAP and one with VAP plus bacteremia that developed resistance to tigecycline during therapy (MIC ≥ 2 μgr/mL). Microbial eradication was demonstrated in 12 (80%) of 15 patients in whom repeat cultures were obtained. Three patients with VAP had a recurrence of infection: one patient with two recurrences, and two patients had one recurrence each. All four recurrent episodes led to clinical resolution and microbial eradication.
Similarly, Curcio et al 40 have conducted a study in 7 intensive care units (ICU) in hospitals in Argentina to evaluate the clinical success rate of 73 patients with VAP caused by MDR-Acinetobacter spp. treated with tigecycline. Clinical success was 69.86% (CI = 58.65-81.07%) 51/73, without significant differences between patients with VAP due to MDR-Acinetobacter spp. carbapenem-susceptible or carbapenem-resistant and only susceptible to colistin, minocyline and tigecycline (70% 44/73 vs. 69% 29/73 respectively, p = 0.9006), and between patients who received ≤48 h of prior antibiotics (including those who did not receive any) and those who received >48 h of prior antibiotics (73.3% 22/30 vs. 67.4% 29/43 respectively, p = 0.7791). Twenty seven patients (37%) received tigecycline plus other antibiotics; in all cases with antipseudomonal activity (i.e. colistin, ceftazidime, cefepime, ciprofloxacin and amikacin). Only two of six patients with VAP and bacteremia showed clinical success (33%) and the mortality rate was 33% (24/73), which included all the patients considered treatment failures (22) and two patients meeting clinical success criteria.
Forty three patients with VAP due to MDR-Acinetobacter baumannii were included in a retrospective multicentre study conducted by Osatnik et al. 41 Tigecycline was used as a single drug during the whole treatment in 24 cases (55%) and in combination with carbapenems or piperacilin/tazobactam or cefepime in 19 (45%). Tigecycline treatment in these patients appeared to result in a remarkably low mortality (30%); therefore, they concluded that tigecycline is a feasible option for VAP in places where MDR-Acinetobacter baumannii is prevalent.
Poulakou et al 54 have published an observational retrospective study which included 18 patients with VAP (15 due to Acinetobacter baumannii) treated with tigecycline (74). Patients received combination therapy in 50% cases (colistin-7/9- or imipenem 2/9-). The global clinical success was 88% (77% and 100% in the monotherapy and combination therapy respectively). Gordon et al 55 have published a series of 16 VAP cases due to MDR-Acinetobacter baumannii and they described a clinical success of 68% but the outcome of the group of VAP patients was not described separately.
Tigecycline treatment was also associated with a positive clinical response in 4 out of 5 cases of pneumonia (nosocomial pneumonia in 4 cases) due to multidrug-resistant or carbapenem-resistant K. pneumoniae that were reported in 3 studies.56–58 In one of these cases recurrence of empyema that accompanied the pneumonia episode was noted, along with a rise in tigecycline MIC to 2 μgr/mL; re-treatment was successful. 57 In addition, tigecycline treatment was associated with a positive clinical response in 2 cases of patients with MRSA pneumonia that have been reported in the literature, one of which was after linezolid failure in a liver transplant recipient with MRSA pneumonia.59,60
In all the observational studies mentioned the authors have used tigecycline at the approved dose (100 mg, then 50 mg q12h).
A study using a pharmacokinetic/pharmacodynamic 5000-patient Monte Carlo simulation model evaluated the probability of pharmacodynamic target attainment regarding treatment with commonly used antibiotics and respective combinations for HAP. 61 The pharmacodynamic targets used for tigecycline consisted of an AUC/MIC ratio of at least 6.96, and at least 17.9, for Gram-negative, and Gram-positive pathogens, respectively. The probability of attainment of the above pharmacodynamic targets remained high as long as the tigecycline MICs of the potentially involved pathogens were not more than 0.25 mg/l, and 0.5 mg/l for Gram-negative, and Gram-positive pathogens, respectively. However, the above MIC estimates could have been higher if it was taken into account that tigecycline penetration in pulmonary endothelial lining fluid and lung tissue is about 1.3 and 2 times higher, respectively, compared to serum levels. 36 Tigecycline monotherapy was associated with a weighted probability of pharmacodynamic target attainment of 62.9% regarding all commonly involved pathogens, compared to 78.2%, 73.3%, and 62.5% for imipenem, piperacillin/tazobactam, and levofloxacin, respectively. Combining tigecycline with ceftazidime, (mainly to adjust for the poor activity of tigecycline against P. aeruginosa) was estimated to be the drug combination most probable to attain the respective pharmacodynamic targets, compared with the combinations of imipenem, piperacillin/tazobactam or levofloxacin with vancomycin. 61
We know that further controlled and randomized studies are required to properly determine the role of tigecycline in HAP including VAP. Nevertheless, while knowing its limitations, we should consider the results of the observational studies mentioned above as the therapeutic options for this group of patients are limited (i.e. VAP due to carbapenems-resistant Acinetobacter spp.).
Bacteremia in patients treated with tigecycline
Since we have already mentioned previously, tigecycline achieves (100 mg, then 50 mg q12h) very low serum concentrations (0.633 μg/mL in the steady state) at the usual dose.30,31 The consideration of the pharmacokinetic and pharmacodynamic properties of tigecycline raises some concerns regarding the effectiveness of this agent for the treatment of bloodstream infections and other infections with secondary bacteremia.
Gardiner et al 62 have analyzed pooled data from patients with bacteremia from 7 double-blind and 1 open-label trial of tigecycline compared with vancomycin/aztreonam, imipenem/cilastatin, levofloxacin, vancomycin, or linezolid as standard therapies for cSSSI, cIAI, community-acquired pneumonia (CAP), or serious infections due to VRE, MRSA, or resistant Gram-negative organisms. The primary efficacy endpoint was the clinical cure rate at the test-of-cure assessment.
A total of 190 patients with bacteremia were identified (tigecycline n = 107; comparator n = 83). Mean Acute Physiology and Chronic Health Evaluation (APACHE) II scores (8.48 vs. 7.38; p < 0.05) and body mass index (27.2 vs. 25.5; p < 0.045) were statistically significantly greater in the tigecycline than the comparator group; the groups were otherwise balanced with respect to demographic and clinical characteristics. Clinical cure rates were 76.6% and 77.1% for tigecycline and comparator, respectively (p = 1.000). Tigecycline was effective and generally well tolerated in the treatment of bacteremia associated with cSSSI, cIAI, and CAP, including MRSA infection. Cure rates were similar to those of comparative standard therapies. The high tigecycline concentration in the tissue, which is the source of these bacteremias (skin and soft tissue, peritoneal fluid and lung), may be the reason for the clinical outcome in this group of patients.
However, it should be mentioned that development of breakthrough bacteremia with Gram-negative bacteria in patients receiving tigecycline for other indications has been reported in 3 cases.16,63
Tygecycline safety profile
In phase III trials regarding complicated intraabdominal infections and complicated skin and skin structure infections, the most frequent clinical adverse events observed in patients treated with tigecycline consisted of nausea (29.5%), vomiting (19.7%), and diarrhea (12.7%) (Tygacil™, package insert). Nausea and vomiting were classified as severe in 1.1%-1.4% of patients, respectively. Of note, nausea and vomiting occurred more commonly within the first two days from the initiation of tigecycline treatment. These untoward effects are dose limiting and are not diminished by slowing the rate of drug infusion. 64 There does not appear to be a relationship between nausea produced by tigecycline and serotonin release. 65 Subjects who were fed or received anti-emetics at the time of administration had improved tolerability of tigecycline. 64 Older individuals (age, > 75 years) and men also reported less nausea than women or younger subjects. 65 One case of C. difficile–associated diarrhea was reported from a phase 2 clinical trial of patients with complicated intra-abdominal infections. 66
Transient elevations in alanine aminotransferase, aspartate aminotransferase, and alkaline phosphatase levels occurred in some test subjects. A total of 1383 patients received tigecycline in multicenter, double-blind, randomized clinical trials (phase 3). In patients treated with tigecycline for skin/skin-structure infections, no hematologic or serum chemistry abnormalities were associated with use of tigecycline. 42 Of note, activated partial thromboplastin or prothrombin time was prolonged in ~3% of patients. In patients with cIAI few clinically important or unexpected changes in any routine hematologic or serum chemistry test results, vital signs, or electrocardiogram data were associated with tigecycline treatment. Skin reactions with tetracyclines are not common but may manifest as pruritus, urticaria, and maculopapular rashes. Cross-sensitization occurs with this class of antibiotic; therefore, anyone reporting an allergy to one of these agents should be considered hypersensitive to tigecycline.
Consequences of the inappropriate initial antibiotic treatment in cSSSI, cIAI and HAP
The initial selection of antimicrobial therapy for treatment of severe infections is extremely important because an association has been shown between inappropriate empiric antimicrobial therapy and delayed clinical resolution, increased length of hospital stay, and an increased risk of mortality.67–70
In this context, Edelsberg et al 71 determined the consequences of failure of initial antibiotic therapy for patients with cSSSI through a retrospective study. From a study of 47,219 hospitalized patients, 10,782 (22.8%), showed evidence of failure of initial antibiotic therapy (defined as need of drainage, debridement, or receipt of other intravenous antibiotics, except for changes to narrower-spectrum agents). Compared with patients for whom initial treatment was successful, patients who experienced treatment failure received intravenous antibiotic therapy for a mean of 5.7 additional days, were hospitalized for a mean of 5.4 additional days, and incurred a mean of $5,285 (in 2003 dollars) in additional inpatient charges (all p < 0.01). One conjecture of the authors is that with a more severe infection in some patients, the administration of antibiotics which were not active against MRSA was an additional factor associated with the failure of initial antibiotic therapy. Unfortunately, this supposition could not be adequately addressed with the available data.
Inappropriate initial antibiotic therapy was associated with a significantly higher proportion of unsuccessful outcomes in patients with cIAI (including death, re-operation, re-hospitalization or additional parental antibiotic therapies), increased length of stay and length on therapy. Tellado et al 72 conducted a study in Spain, which included 425 patients with cIAI. Of these, 387 (91%) received appropriate initial empiric therapy. Patients on inappropriate therapy were less likely to have clinical success (79% vs. 26%, p < 0.001), more likely to require additional antibiotic therapy (40% vs. 7%, p < 0.01) and more likely to be re-hospitalized within 30 days of discharge (18% vs. 3%, p < 0.01). Similarly, in Germany, Krobot et al 73 determined over 425 patients hospitalized with cIAI, 13% of patients received inappropriate initial parenteral therapy not covering all bacteria isolated and concluded that patients were more likely to have clinical success if initial antibiotic therapy was appropriate (78.6%; 95% CI, 73.6-83.9) rather than inappropriate (53.4%; 95% CI, 41.1-69.3).
In a prospective multicenter study conducted in the medical and surgical ICUs of 30 Spanish hospitals over the course of 1 yr, mortality attributable to pneumonia was significantly higher in patients with inappropriate initial antibiotic therapy than in those who received appropriate therapy (25% vs. 16%, p = 0.034). 74 Kollef et al 75 in a retrospective, single-center, observational cohort study which included 76 patients with VAP attributed to potentially antibiotic-resistant Gram-negative bacteria (Pseudomonas aeruginosa, Acinetobacter spp, and Stenotrophomonas maltophilia), demonstrated that inappropriate initial antibiotic therapy of microbiologically confirmed VAP attributed to these pathogens is associated with greater 30-day mortality. High rates of VAP attributed to antibiotic-resistant bacteria (e.g. Acinetobacter spp) may require changes in the local empiric antibiotic treatment of VAP in order to optimize the prescription of appropriate initial therapy.
In summary, appropriateness of administration, based on susceptibility, is the most important determinant, but dosing intervals and dose probably play similarly important roles in outcomes, however this has not been exhaustively examined in humans. Other aspects of appropriate therapy that deserve attention include a shift to more ‘resistance‘-proof antibiotics in empiric therapy, which may produce better outcomes. However in most of the studies, the primary reason for administration of inappropriate antimicrobial therapy was the presence of MDR-Gram-negative and Gram-positive bacteria that were not susceptible to the prescribed antibiotic regimen, therefore it is mandatory to take into account, for each patient the individual risk factors for MDR-pathogens in order to select the best antibiotic for improvement of clinical outcomes (Tables 2 and 3).
Main risk factors for multidrug-resistant pathogens in hospitalized patients.

Risk factors for community-acquired methicillin-resistant Staphylococcus aureus.
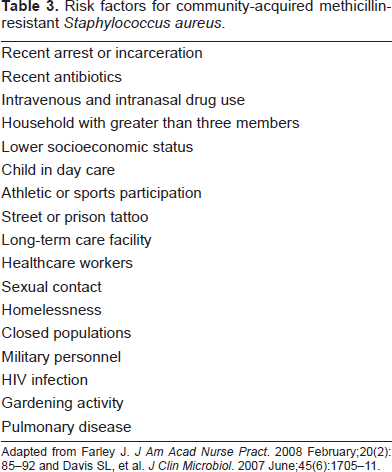
Adapted from Farley J. J Am Acad Nurse Pract. 2008 February;20(2):85-92 and Davis SL, et al. J Clin Microbiol. 2007 June;45(6):1705-11.
Proposal of the ARTs
The Figures 1, 2, and 3 represent the ARTs (Algorithms Rational to Prescribe Tigecycline) which we propose for cSSSI, cIAI and HAP respectively.
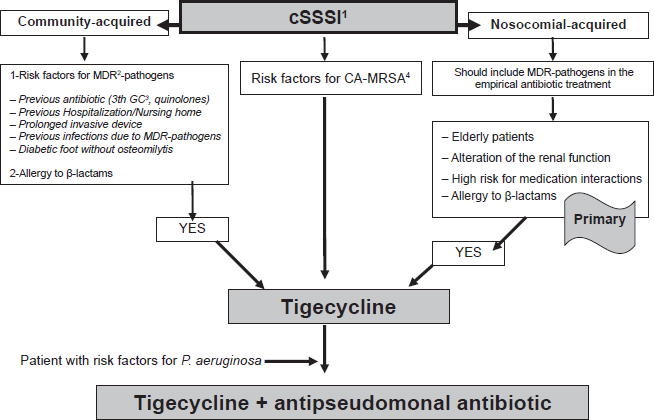
Algorithm rational to prescribe tigecycline in complicated skin and skin structures infection.
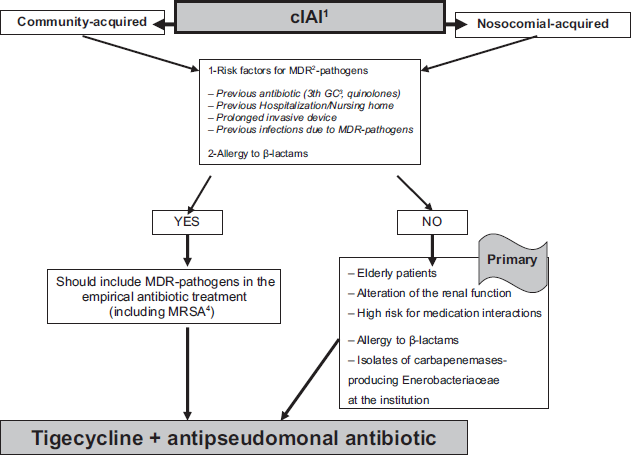
Algorithm rational to prescribe tigecycline in complicated intra-abdominal infections.
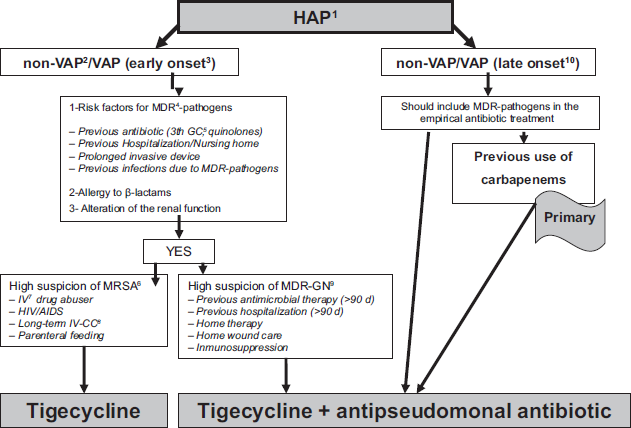
Algorithm rational to prescribe tigecycline in hospital-acquired pneumonia.
We developed the ARTs based on the data from the analyzed evidence above and focusing on the individual characteristics of the patients which include:
source of infection (community or nosocomial-acquired);
risk factors for MDR-pathogens;
need to add an antipseudomonal antibiotic;
others (i.e. older patients, alteration of the renal function, high risk of medication interactions and allergy to β-lactams).
Conclusion
In summary, tigecycline, the first in a new class, the glycylcyclines, appears to hold significant promise as a new agent that can be added to our antimicrobial armamentarium to help overcome antibiotic resistance among clinically important bacteria. Tigecycline confers broad antibiotic coverage against vancomycin-resistant enterococci, methicillin-resistant Staphylococcus aureus and penicillin-resistant Streptococcus pneumoniae. Additionally, against Gram-negative bacteria, tigecycline is highly active against many species that are MDR, including those containing ESBL and Acinetobacter species, although resistance to tigecycline by Pseudomonas aeruginosa and reduced susceptibility among Proteus species has been noted.
The ARTs were developed with an aim to aid attending physicians to find the situations in which tigecycline can be administered in hospitalized patients, and knowing what other options that could be used in the various scenarios explained.
However, we believe that beyond the ARTs, tigecycline has an attractive microbiological and pharmacological profile to be used in hospitals in an antibiotic stewardship program.
In this context, tigecycline should be taken into account for some of the challenges raised by the ESKAPE pathogens found in daily practice. These include:
the vancomycin MIC creep in MRSA which is associated with high rates of clinical failures in patients with pneumonia; 76
the emergence of carbapenemases 77 and the need to preserve the carbapenems use,
the need to use antibiotics with a lack of robust scientific evidence (i.e. colistin). 78
Finally, further evidence derived from well-designed studies on the clinical use of tigecycline for specific infections (i.e. HAP) is warranted, considering the increasing resistance rates of nosocomial pathogens to commonly used antibacterial agents.
Disclosures
The authors report no conflicts of interest.
